Abstract
Puhahn-Schmeiser B, Kleemann T, Jabbarli R, Bock HH, Beck J, Freiman TM [published online ahead of print May 27, 2022]. Hippocampus. 2022. doi:10.1002/hipo.23447 Temporal lobe epilepsy is characterized by hippocampal neuronal death in CA1 and hilus. Dentate gyrus granule cells survive but show dispersion of the compact granule cell layer. This is associated with decrease of the glycoprotein Reelin, which regulates neuron migration and dendrite outgrow. Reelin-deficient (reeler) mice show no layering, their granule cells are dispersed throughout the dentate gyrus. We studied granule cell dendritic orientation and distribution of postsynaptic spines in reeler mice and two mouse models of temporal lobe epilepsy, namely the p35 knockout mice, which show Reelin-independent neuronal migration defects, and mice with unilateral intrahippocampal kainate injection. Granule cells were Golgi-stained and analyzed, using a computerized camera lucida system. Granule cells in naive controls exhibited a vertically oriented dendritic arbor with a small bifurcation angle if positioned proximal to the hilus and a wider dendritic bifurcation angle, if positioned distally. P35 knockout- and kainate-injected mice showed a dispersed granule cell layer, granule cells showed basal dendrites with wider bifurcation angles, which lost position-specific differences. Reeler mice lacked dendritic orientation. P35 knockout- and kainate-injected mice showed increased dendritic spine density in the granule cell layer. Molecular layer dendrites showed a reduced spine density in kainate-injected mice only, whereas in p35 knockouts no reduced spine density was seen. Reeler mice showed a homogenous high spine density. We hypothesize that granule cells migrate in temporal lobe epilepsy, develop new dendrites which show a spread of the dendritic tree, create new spines in areas proximal to mossy fiber sprouting, which is present in p35 knockout- and kainate-injected mice and loose spines on distal dendrites if mossy cell death is present, as it was in kainate-injected mice only. These results are in accordance with findings in epilepsy patients.
Anstötz M, Lee SK, Maccaferri G. Cell Rep. 2022;39(7):110822. doi:10.1016/j.celrep.2022.110822 The impact of Cajal-Retzius cells on the regulation of hippocampal circuits and related behaviors is unresolved. Here, we directly address this issue by impairing the glutamatergic output of Cajal-Retzius cells with the conditional ablation of vGluT2, which is their main vesicular glutamate transporter. Although two distinct conditional knockout lines do not reveal major alterations in hippocampal-layer organization and dendritic length of principal neurons or GABAergic cells, we find parallel deficits in specific hippocampal-dependent behaviors and in their putative underlying microcircuits. First, conditional knockout animals show increased innate anxiety and decreased feedforward GABAergic inhibition on dentate gyrus granule cells. Second, we observe impaired spatial memory processing, which is associated with decreased spine density and reduced AMPA/NMDA ratio of postsynaptic responses at the perforant- and entorhino-hippocampal pathways. We conclude that glutamate synaptically released by Cajal-Retzius cells is critical for the regulation of hippocampal microcircuits and specific types of behaviors.
Commentary
Fishing takes time. You need handling of rods and reels, making a long cast into the sea, and then…the wait. Some fishers drop bait all over and that attracts the seagulls. Banks of fish come, the rod becomes tense, and the reelin’ goes. Dropping something else into the mix has changed the game.
Epilepsy research has proven a hard game to crack. Temporal lobe epilepsy (TLE) is a neurological disorder characterized histologically by hippocampal cell death, dyslamination, and layer-specific changes in spine and dendrite density. The accompanying aberrant, pro-convulsive circuitry is thought to be responsible for cognitive comorbidities affecting spatial memory and emotion. For years, hippocampal dyslamination was in part blamed on a deficit of extracellular Reelin, a glycoprotein secreted by Cajal-Retzius (CR) cells. Absence of Reelin or not enough Reelin leads to cell dispersion, aberrant spinogenesis, and seizure susceptibility. 1 Are CR cells all about Reelin? Two recent papers now suggest we may be missing something else; another player in the game. 2,3
Cajal-Retzius cells regulate neuronal migration, lamination, and synapse formation during development. They are among the first-born cells in the mammalian brain, and mostly disappear postnatally via programmed cell death. However, in the healthy hippocampus, 15% to 20% of CRs persist after circuit maturation is completed. Critically, in many human epileptic syndromes, CRs overcrowd the hippocampus and their persistence correlates with seizures. 4
In the first paper, Puhahn-Schmeiser et al compare granule cell (GC) dispersion in the hippocampal dentate gyrus and dendritic changes in mouse models of TLE. They used the very same methods they previously applied to a series of human hippocampal surgical specimens of TLE patients. As models of human TLE, the authors compare Reelin-deficient (reeler) mice, Reelin-independent p35 knockout (p35 KO) mice, and healthy mice treated with unilateral intrahippocampal kainate (KA) injection, which compromises proper Reelin release due to its abnormal proteolytic processing. 1
All mouse models studied by Puhahn-Schmeiser et al show dyslamination, with p35 KO mice exhibiting milder alterations, KA-injected mice having a near 3-fold dispersion, and reeler mice taking the lead with the largest deviation from a laminar, structured GC layer (Figure 1A). Apical dendrites were visibly less vertically oriented and bifurcated with wider angles in p35 KO and KA-injected mice as compared with healthy mice. In reeler mice, they lacked any orientation. While GC spine density in healthy mice is typically low close to the soma and increases distally in the dendritic tree, in all 3 models, proximal spinogenesis was remarkable. In KA-injected animals, a reduction in distal spines was also evident.
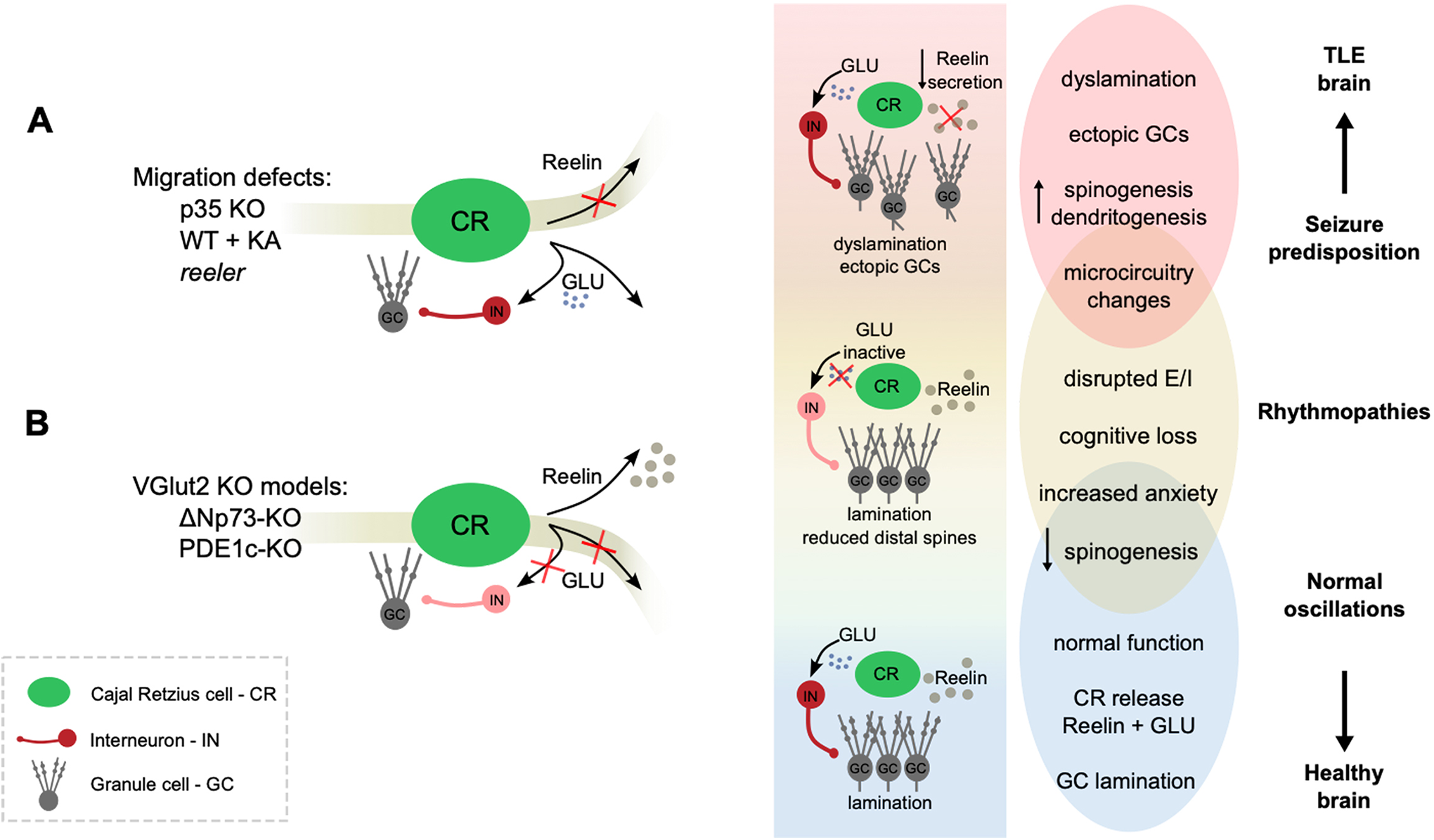
Schematic representation of the potential role of CRs in TLE. A, Puhnahn-Schmeiser et al (2022) investigate 3 mouse models that present histological defects in line with human TLE. P35 KO, KA-injected, and reeler mice all show dyslamination and altered spino- and dendritogenesis; the latter two, in a Reelin-dependent manner. The 3 models exhibit different propensity to seizures and associated cognitive impairment. The shadowed stripe indicates that Reeling release is altered in 2 of these models. B, Anstötz et al (2022) study the effects of selectively abolishing glutamate release of Reelin-secreting CR cells in the hippocampus and see no dyslamination (shadowed stripe). Some altered spinogenesis seen in TLE models is recapitulated. VGlut2 KO models present with impaired spatial memory and anxiety. Overall, the above schematic illustrates the potential role of CR cells in health and disease via 2 potential different signaling pathways: the Reelin-mediated function, with key roles in controlling lamination; and a microcircuit function mediated by glutamate. By regulating the structure and function of hippocampal networks, CR cells may be key in the transition from a healthy to a pro-epileptic brain. CR indicates Cajal-Retzius; KA, kainate; TLE, temporal lobe epilepsy.
The second paper suggests some of these changes may actually be caused by another player. In their work, Anstötz et al specifically focus on a relatively ignored facet of adult CRs: apart from Reelin they also release glutamate. 3 This might change the whole game.
In healthy brains, adult-born GCs have to reach their laminar position and eventually form synaptic connections within the local microcircuit. Recent data show that CRs not only stabilize GC structure and lamination by releasing Reelin but they also facilitate their integration via interposed GABAergic cells. 5,6 Thus, CRs are not merely a Reelin factory, but a major circuit player. 7 This later role, however, is not yet contemplated in epilepsy research.
In their work, Anstötz et al use CR-specific conditional knockout lines to block glutamate signaling without affecting Reelin secretion (VGlut2-KO models, Figure 1B). Interestingly, impairing CR glutamate release was not sufficient for dentate gyrus dyslamination, confirming the Reelin-dependent changes observed in the reeler and KA-injected mice. Instead, feedforward inhibition onto GC and maturation of excitatory entorhinal inputs were severely impaired, and this correlated with anxiogenic behavior and spatial memory deficits.
An appropriate excitatory/inhibitory (E/I) balance is crucial for keeping physiological levels of activity away from pro-epileptic regimes. Hippocampal rhythms such as theta (4-12 Hz) and gamma (40-80 Hz) depend on well-tuned E/I balance and are critical for memory function. 8 By directly controlling disynaptic inhibition, CR-mediated glutamate release may also contribute to physiological oscillatory activity. Their impaired glutamatergic function may thus result in hippocampal rhythmopathies and the associated cognitive deficits. 9,10
In VGlut2-KO mice, the distal dendritic spines of GCs and CA1 pyramidal cells decreased by 30% to 40% relative to controls. This is reminiscent of the spine loss seen by Puhahn-Schmeiser et al in KA-injected mice. Interestingly, entorhinal inputs critical for memory function target the distal dendritic trees of CA1 and GCs, and recent work suggests that postnatal ablation of CRs leads to maturation defects of entorhinal-hippocampal connections. 11 The hippocampal-entorhinal loop is especially suited to sustain and propagate seizures in TLE, making these connections of special interest here. Thus, the study by Anstötz et al suggests other roles for CR cells in shaping epileptogenic microcircuits beyond lamination defects.
Although the balance between synaptic versus structural effects of CR activity is not yet clear, one can hypothesize that its Reelin-mediated function outweighs its glutamatergic function during development. In adult stages, on the other hand, its role in the local microcircuitry may take precedence over Reelin. Hence, the balance between the glutamatergic activity, circuit integration, and Reelin secretion of CR cells may provide novel multifocal targets for epilepsy research. Investigating CR cells becomes exciting: a potential game changer after all.
