Abstract
Artificial intelligence (AI) is increasingly used in medical image analysis and has accelerated scientific discoveries across fields of medicine. In this review, we highlight how AI has been applied to neuroimaging in patients with epilepsy to enhance classification of clinical diagnosis, prediction of treatment outcomes, and the understanding of cognitive comorbidities. We outline the strengths and shortcomings of current AI research and the need for future studies using large datasets that test the reproducibility and generalizability of current findings, as well as studies that test the clinical utility of AI approaches.
Keywords
Summary
Big data neuroimaging studies in epilepsy using artificial intelligence (AI) and its sub-fields machine learning (ML) and deep learning (DL) are increasing substantially. AI applications are powerful tools for classification and predictions using multimodal imaging data with varying degrees of direct human interaction. ML and DL using different imaging modalities have shown early promise for automatic classification of patients with focal and generalized epilepsies from healthy controls, detecting lesions, and predicting seizure outcomes. However, the reproducibility and generalizability of these preliminary results and their clinical validity are still to be determined. Further neuroimaging AI studies are needed to predict cognitive outcomes following epilepsy surgery and determine the risk for cognitive decline over time.
Introduction
An incomplete but straightforward definition of artificial intelligence (AI) has combined computer science and data science that enables big data problem-solving.
1
AI includes the sub-fields of machine learning (ML) and deep learning (DL), terms that are often used interchangeably but have a different meaning.1,2 While ML is more data-driven, involves statistical models, and depends on human intervention, DL uses multiple layers of neural networks, eliminating some human intervention (Figure 1).2,3 Machine learning (ML) and deep learning (DL) are sub-fields of artificial intelligence (AI) that seek algorithms to create expert systems that make predictions or classifications based on input data. Basic concepts about artificial intelligence, machine learning, and deep learning.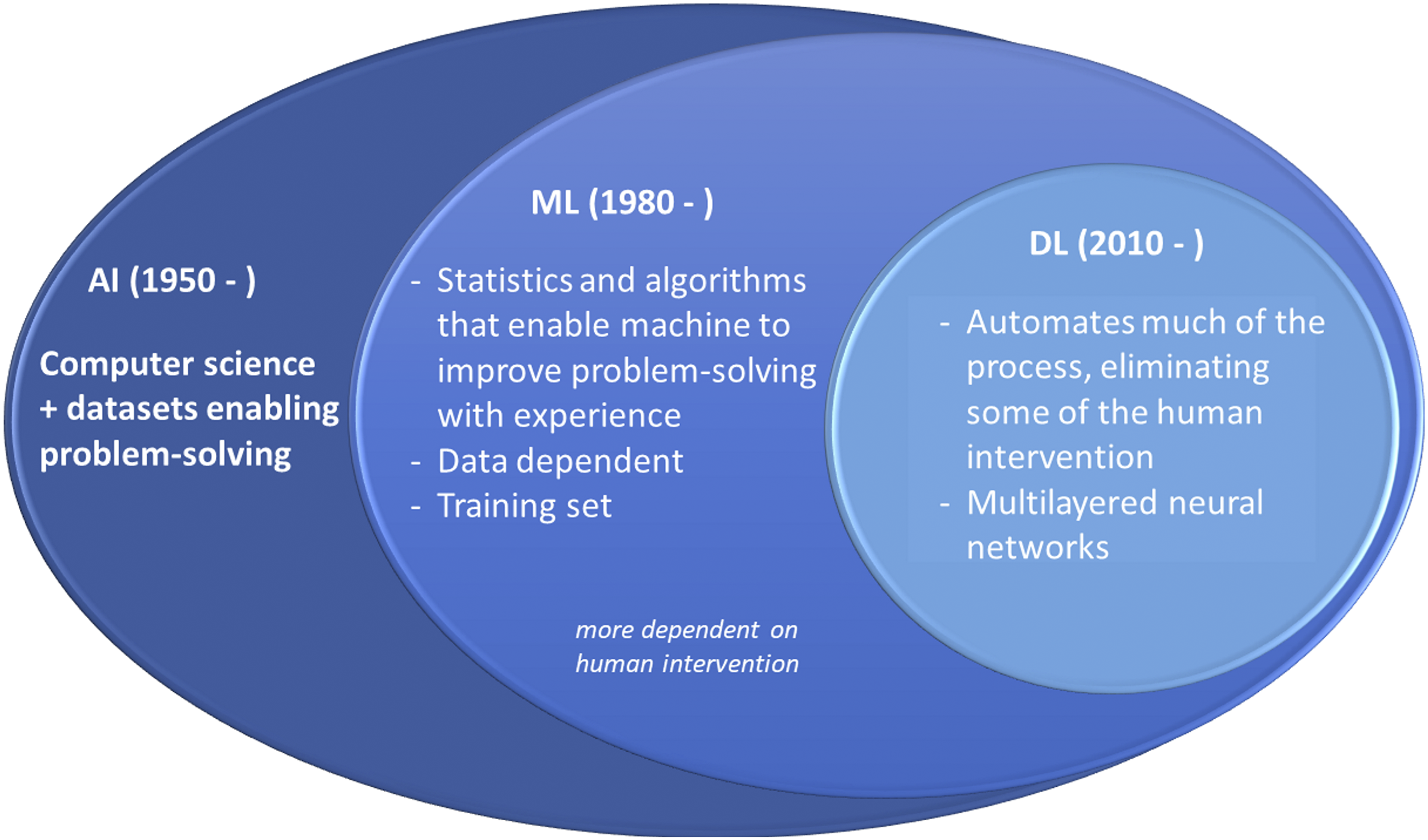
Although conventional statistics and ML may be used for both prediction and inference, ML methods are better suited for predictions. 4 These approaches can also be described along a continuum of data analyses in two axes: in one axis, they extend from an entirely human-dependent to a fully human-independent interaction. The other axis extends from data-training dependent to data-training independent.2,5 For example, AI applications can now automatically segment and calculate the volume or shape of brain structures on MRI with little or no human intervention. 6
Advances in computer science and faster hardware enabling big data analyses has transformed modern life in many ways, including search engines, marketing, entertainment, and how science is conducted. 7 Many complex questions about diseases and brain function can now be investigated using big data sets.2,8,9 However, attention should be taken to avoid the overuse of AI methods simply because they are new and powerful. 10 As with any new technique, AI, ML, and DL are sometimes used for applications where they not entirely justified, overlooking methodological limitations and overemphasizing results easily obtained by straightforward statistical analyses.10,11 However, when performed correctly, well-powered studies using ML have led to important advances in medicine, which could be translated to epilepsy in the near future.
Driven in part by the promise of ML, collaborative studies using multicenter neuroimaging big data have increased substantially. 12 One successful example is the Enhancing Neuroimaging Genetics through Meta-analysis (ENIGMA) brain imaging consortium that has revealed associations between brain imaging, genetic variations, and cognition in both healthy subjects and different diseases, including epilepsy.13–16 Another is the multicenter epilepsy lesion detection (MELD) project. 17 These datasets include hundreds to thousands of MRI and clinical data in patients with epilepsy and are ripe for AI applications.
Artificial intelligence Applications for Epilepsy Diagnosis and Management
In recent years, ML algorithms using multimodal MRI have been shown to lateralize hippocampal pathology in patients with temporal lobe epilepsy (TLE) and hippocampal sclerosis (HS).18,19 A recent ENIGMA-Epilepsy study investigated the performance of ML and DL algorithms using structural MRI and diffusion MRI (dMRI) to classify controls vs patients with TLE with HS and MRI-negative TLE. 13 This study revealed that structural MRI and dMRI-based models had similar accuracy and that models for TLE-HS were more accurate than for MRI-negative TLE. 13 While the ability of automatic quantification methods may not currently exceed visual inspection of MRIs by imaging experts in all situations, AI algorithms and tools provide important support tools and may become of great importance when such expertise is not available. Some AI tools have already been integrated into clinical care for epilepsy, 20 but it will remain important for clinicians and AI-experts to remain in close dialog for newer ML and DL approaches to be adapted for clinical use. 2
Focal cortical dysplasia (FCD) is one of the most common causes of pharmacoresistant focal epilepsy. However, FCDs are often undetected on conventional MRI and the pre-surgical diagnosis depends heavily on the expertise of the examiner. 21 Several MRI post-processing techniques have been used to improve the detection of subtle FCDs.22–25 A recent multicenter-validated study showed that DL using multimodal MRI data could reliably identify previous MRI-negative FCD lesions, suggesting that DL shows promise for assisting non-expert clinicians in this challenging diagnosis. 26
AI methods can also combine imaging and clinical data to build models for predicting clinical outcomes in patients with epilepsy. 27 For example, automated volumetric MRI measurements incorporated into statistical models help to predict postoperative seizure outcomes in TLE and frontal lobe epilepsy (FLE), revealing that subtle cortical atrophy beyond the surgical resection influences seizure outcome.28,29 DL applied to whole-brain connectomes can also help to predict postoperative seizure control in patients with TLE.30,31
Another application of AI has been implemented by the MELD project—a retrospective multicenter cohort of 580 patients with FCD. 17 Here, AI was not used for lesion detection. Rather, the MELD team trained logistic regression models to test for associations between clinical data and the location of FCD lesions that were delineated on T1-weighted MRI scans by imaging experts. Their data-driven atlas validated smaller independent studies which showed a non-uniform distribution of FCDs with higher concentrations in the superior frontal sulcus, frontal pole, temporal pole, and superior temporal gyrus. Lesions in primary sensory areas were associated with earlier age of epilepsy onset whereas lesions in association cortices were associated with a later epilepsy onset. The likelihood of seizure freedom decreased with a longer duration of epilepsy. 17
Finally, AI, including ML and DL, has been applied to neuroimaging data for predicting clinical diagnosis, that is, clinical phenotyping from imaging,32,33 and predicting response to antiseizure medications. 34 For example, dMRI measures and connectome profiles may identify patients with juvenile myoclonic epilepsy 32 and distinguish patients with focal epilepsy vs healthy controls. 33 However, these studies are few in number and larger studies that are validated in external datasets are needed to determine the reproducibility and generalizability of current findings.
Artificial intelligence Applications for Understanding Cognitive Comorbidities
The application of AI to neuroimaging data for understanding cognitive comorbidities in epilepsy is in its infancy, with only a handful of studies published to date. Most studies have focused on classifying cognitive impairment based on whole-brain dMRI or resting-state functional MRI (rsfMRI), with only one study incorporating features from multimodal imaging. Four studies have employed machine learning to classify or predict language impairments,35–38 three to predict IQ,39–41 two to predict memory,42,43 and one to predict embodied cognition (i.e., action-concept deficits) in epilepsy. 44 Although most have focused on adults with TLE, studies are also emerging in children with epilepsy and patients with other epilepsy syndromes. These studies are briefly summarized, along with the challenges and opportunities that lie ahead for AI in the future.
In summary, studies using AI to characterize the neural substrates of cognitive impairment in epilepsy have provided new insights into the vast extent of network dysfunction underlying cognitive comorbidities and align with research challenging region-specific theories of cognitive dysfunction. 45 These studies have leveraged the power of whole-brain analyses, employing feature selection strategies to reduce the dimensionality of imaging data and improve prediction accuracy. However, no studies have used large, external datasets to test the reproducibility and generalizability of the results. In addition, no studies have used AI to predict cognitive outcomes following epilepsy surgery or to determine the risk for cognitive decline over time. Thus, the application of AI to cognitive networks in epilepsy remains in the discovery phase, with its clinical utility yet to be established.
Conclusion
The application of AI to medical image analyses has exponentially increased over the past decade and has the potential to reshape our approach to clinical diagnosis, prediction of treatment outcomes, and management of cognitive comorbidities in epilepsy.9,46–48 However, despite the promise of AI, the majority of studies applying ML to epilepsy have been modest in sample size, raising concerns for overfitting and limiting the application of DL models that require thousands of patient samples (e.g., convolutional neural networks). Such barriers will hopefully be lifted in the future as more powerful AI methods are developed and imaging and cognitive data are aggregated across centers and harmonized in the context of big data efforts.
Footnotes
Declaration of conflicts of interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FC: disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Grants from the São Paulo Research Foundation (FAPESP) grant #2013/07559-3, and from the National Council for Scientific and Technological Development (CNPq) Brazil, grants #311231/2019-5 and #465346/2014-6.CRM: Funding from the National Institutes of Health NINDS R01NS120976; R01NS122827; R01NS065838; and R21NS107739.
