Abstract
Alkhachroum A, Der-Nigoghossian CA, Mathews E, Massad N, Letchinger R, Doyle K, Chiu W-T, Kromm J, Rubinos C, Velazquez A, Roh D, Agarwal S, Park S, Connolly S, Claassen J. Neurology. 2020;95(16):e2286-e2294. doi: 10.1212/WNL.0000000000010611. To test ketamine infusion efficacy in the treatment of superrefractory status epilepticus (SRSE), we studied patients with SRSE who were treated with ketamine retrospectively. We also studied the effect of high doses of ketamine on brain physiology as reflected by invasive multimodality monitoring (MMM). We studied a consecutive series of 68 patients with SRSE who were admitted between 2009 and 2018, treated with ketamine, and monitored with scalp EEG. Eleven of these patients underwent MMM at the time of ketamine administration. We compared patients who had seizure cessation after ketamine initiation to those who did not. Mean age was 53 ± 18 years and 46% of patients were female. Seizure burden decreased by at least 50% within 24 hours of starting ketamine in 55 (81%) patients, with complete cessation in 43 (63%). Average dose of ketamine infusion was 2.2 ± 1.8 mg/kg/h, with median duration of 2 (1-4) days. Average dose of midazolam was 1.0 ± 0.8 mg/kg/h at the time of ketamine initiation and was started at a median of 0.4 (0.1-1.0) days before ketamine. Using a generalized linear mixed effect model, ketamine was associated with stable mean arterial pressure (odds ratio = 1.39, 95% CI: 1.38-1.40) and with decreased vasopressor requirements over time. We found no effect on intracranial pressure, cerebral blood flow, or cerebral perfusion pressure. Ketamine treatment was associated with a decrease in seizure burden in patients with SRSE. Our data support the notion that high-dose ketamine infusions are associated with decreased vasopressor requirements without increased intracranial pressure. This study provides Class IV evidence that ketamine decreases seizures in patients with SRSE.Objective:
Methods:
Results:
Conclusion:
Classification of Evidence:
Commentary
In approximately 30% of cases, status epilepticus (SE) persists despite the administration of at least 2 appropriately selected and dosed parenteral medications (usually including a benzodiazepine). This situation is referred to as refractory SE (RSE). 1 The optimal treatment of RSE is still debated but often involves the use of continuous intravenous anesthetic drugs (CIVADs), most commonly midazolam or propofol. 2,3 In as many as a 30% of RSE cases, seizures might still persist despite anesthesia or recur while on appropriate anesthetic treatment or after withdrawal of anesthesia. This situation is referred to as superrefractory SE (SRSE). 1 There is little evidence available to guide the management of SRSE and many options are available. A common strategy is to reinstate CIVADs, either using the same molecule, switching to another molecule, or combining multiple drugs. 4 Most CIVADs, including midazolam, propofol, and barbiturates, are γ-aminobutyric acid (GABA)A receptor agonists. They are associated with sedation, cardiorespiratory depression, and hypotension and require mechanical ventilation and often the use of vasopressors. Prolonged SE and the prolonged use of GABAergic CIVADs are associated with a substantial risk of complications, mostly infections and shock, and mortality. 4 Further, evidence coming from animal studies suggests that GABAergic drugs quickly lose their ability to abort prolonged ictal discharges, mainly because of the cellular internalization of synaptic GABAA receptors while N-methyl-D-aspartate (NMDA) receptor antagonists become more efficacious because of the progressive externalization of their receptors. 5 -7 Also, animal data indicate that the concomitant use of GABAA agonists and NMDA antagonist has synergistic properties, that is, the coadministration of an NMDA antagonist decreases the dose of GABAA agonist required to abort the ictal discharge and its related toxicity. 8 Ketamine (also known as super K as a street drug) is an NMDA antagonist available for CIV administration. It differs from conventional CIVADs not only by its mechanism of action but also by its safety profile. Indeed, ketamine possesses a cardio-stimulant effect. It raises blood pressure and cardiac output and thus might provide cardiocirculatory support in critically ill patients with SRSE, partially alleviating their need for vasopressors. For all these reasons, ketamine appears as a promising therapeutic option for SRSE, potentially combining better efficacy and safety relative to other CIVADs.
It is thus not surprising that it has attracted increasing interest in this setting over the last decade. Its use in RSE, and mostly SRSE, has been reported in several case reports and case series, now totaling over 300 patients (summarized in Table 1). Ketamine has been tried in a wide range of causes of SRSE, from anoxic brain injury to new-onset refractory SE (NORSE; a probable autoimmune condition) through stroke and CNS infection. Its reported efficacy (ie, ability to permanently control SE) ranges from 32% to 91%. This variability likely owes to the differences in case mix, intrinsic severity of SE, latency to initiation, and definition of SE control, between studies. Adverse events imputed to the drug have been relatively few and led to its interruption in only a small minority of cases (Table 1). However, the association with adverse events might have been obscured by the overall critical state of the patients and the prior and concomitant use of several other anti-seizure medications and CIVADs. A recent study drew attention to the occurrence of hypothermia in patients receiving a combination of propofol and ketamine for SRSE, especially with between higher doses and longer duration of exposure to ketamine. 9 Somewhat disappointingly, the anticipated beneficial effect on blood pressure and requirement was not obvious in most of these studies. However, this might have been blurred by their retrospective design and the lack of detailed hemodynamic data. Finally, one concern remains regarding the administration of ketamine to brain-injured patients, due to its possible effect on intracranial pressure (ICP) and cerebral perfusion. Although there is reassuring evidence coming from anesthetized patients undergoing neurosurgery for a variety of reasons, including traumatic brain injury, little is known of its risk when administered for a prolonged period of time in critically ill patients. 10,11
Summary of Case Series of Ketamine in Refractory or Superrefractory SE.a
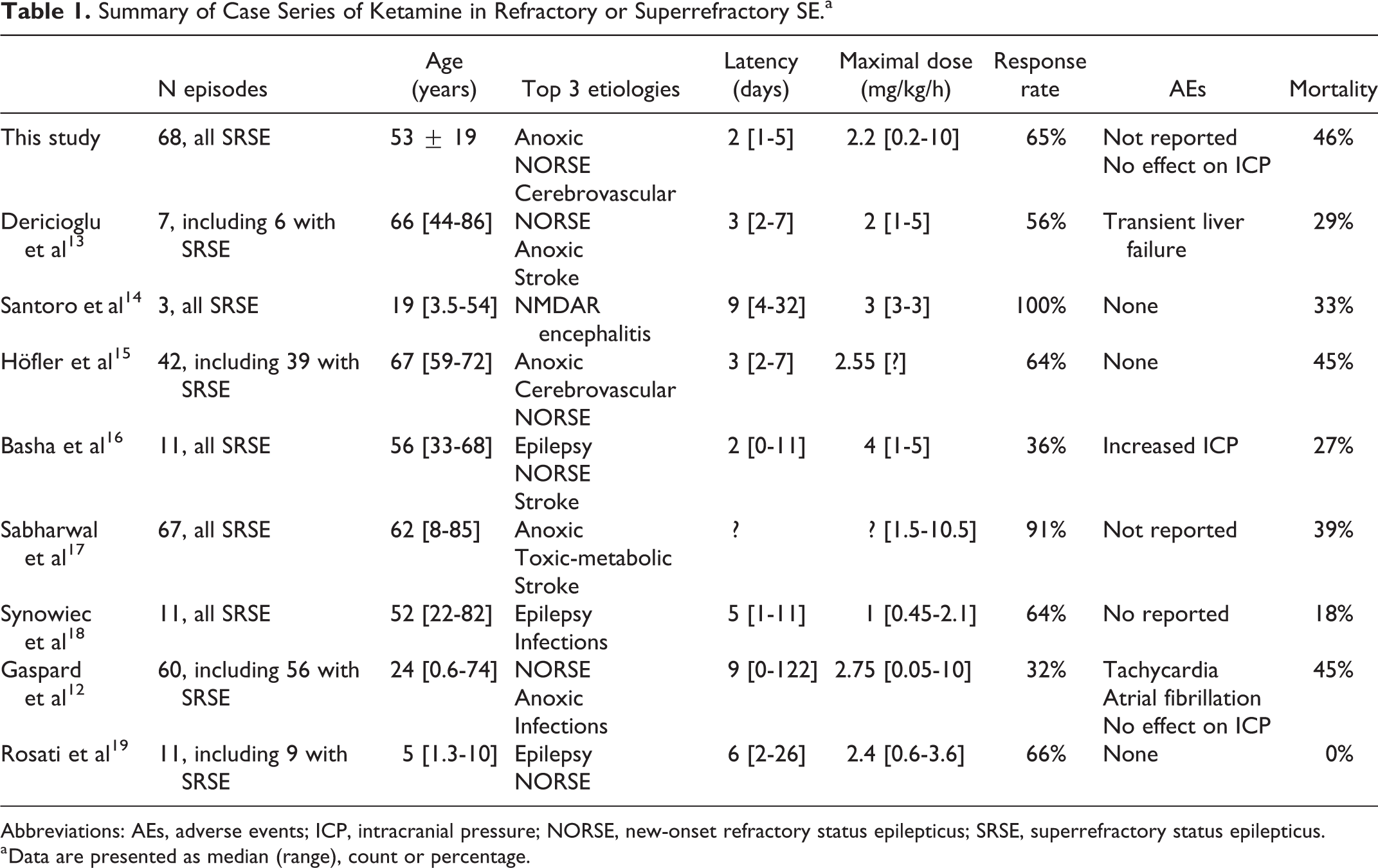
Abbreviations: AEs, adverse events; ICP, intracranial pressure; NORSE, new-onset refractory status epilepticus; SRSE, superrefractory status epilepticus.
a Data are presented as median (range), count or percentage.
The study highlighted in this commentary attempted to address some of these issues. The authors retrospectively included 68 adult patients (46 females; age: 53 ± 19 years) who received CIV ketamine for SRSE. The most common etiologies were anoxic brain injury (n = 18), NORSE (n = 12), and stroke (n = 11). The infusion was started a median of 2 days after SE onset and after only a mean of 2 AEDs had been used. This is in line with the most recent series published since 2015 (Table 1) and indicates a progressive shift of ketamine toward earlier use in RSE and perhaps wider adoption of the drug, at least in some centers. As expected from a retrospective observational study in which the doses and duration of treatment are left to the discretion of the treating physicians, those varied widely (median [range] dose: 2.4 [0.2-10] mg/kg/h; median [range] duration: 2 [1-4] days), likely reflecting the heterogeneous severity and refractoriness of the episode of SE. Overall, 43 (63%) patients had complete cessation of seizures within 24 hours after the initiation of ketamine, and 44 (65%) remained SE free after ketamine was stopped (but other interventions were concomitantly performed in some patients). These figures are in the upper range of prior studies and might perhaps be explained by the early initiation of ketamine, as suggested by the authors and supported by at least 2 previous studies. 12-13 This does not necessarily imply that earlier initiation is associated with greater intrinsic efficacy of the drug but might also result from a selection bias. Episodes of SE requiring more rounds of treatment are possibly more severe and thus simply less likely to respond to any drug in general. None of the investigated clinical or demographic variables, including etiology, dose, and duration of ketamine infusion, were associated with the response rate.
One of the key strengths of the study lies in the detailed analysis of daily hemodynamic monitoring and vasopressors use. By doing so, the authors were able to demonstrate that ketamine infusion allowed some patients to be weaned off vasopressors while maintaining a stable mean arterial pressure (MAP). Even more important is that this association was dose and duration-dependent, further indicating a supportive circulatory effect of ketamine.
Another interesting piece of information provided by the study comes from the subgroup of patients (n = 11) who received ketamine while undergoing invasive brain multimodality monitoring (MMM). By reviewing the concomitant recordings of ICP, cerebral perfusion pressure (the differential between ICP and MAP), and cerebral blood flow, the authors could demonstrate that ketamine infusion was not associated with increased ICP and did not alter cerebral perfusion, even when administered at a high dose (>3.5 mg/kg/h) and for a prolonged period of time. This was of course shown only in a small subset of patients, likely those with an etiology for which MMM was indicated due to the risk of secondary brain injury (subarachnoid hemorrhage, traumatic brain injury, etc) and who might have also received additional treatment for ICP control. Whether or not these observations can be generalized to all patients is thus unclear, but they at least add to the growing body of evidence that ketamine infusion does not lead to unmanageable ICP issues in patients with SRSE and acute brain injury.
A small regret I have is the relative lack of information on any serious adverse events. The literature so far has been rather optimistic, with very few reported adverse events (Table 1), but information on cardiac arrhythmias, confirming, or disproving prior studies, for instance, would have been welcome. Another aspect I would have liked to see investigated in this study is the potential sparing effect of ketamine on the concomitant dose of CIV midazolam required to achieve SE control, as suggested by animal studies.
Overall, the study suffers from all the limitations of its retrospective uncontrolled design. Whether or not ketamine represents a good option for SRSE will only be settled by a randomized controlled trial. These are however difficult, albeit not impossible, to perform in such an uncommon and complex situation. In the meanwhile, this study further expands the existing body of literature on the potential efficacy and the good safety profile of ketamine in SRSE. It also provides undisputable evidence of its hemodynamic supportive effect and should mitigate any worries that one might have to use it in brain-injured patients. To the best of my knowledge, with well over 300 cases in the literature, ketamine is the drug for which there is the most published (low-quality) evidence in this indication and should be considered by every team facing such difficult and complex cases.
