Abstract
Hatcher A, Yu K, Meyer J, Aiba I, Deneen B, Noeb JL. J Clin Invest. 2020;130(5):2286-2300. doi:10.1172/JCI133316 Seizures often herald the clinical appearance of gliomas or appear at later stages. Dissecting their precise evolution and cellular pathogenesis in brain malignancies could inform the development of staged therapies for these highly pharmacoresistant epilepsies. Studies in immunodeficient xenograft models have identified local interneuron loss and excess glial glutamate release as chief contributors to network disinhibition, but how hyperexcitability in the peritumoral microenvironment evolves in an immunocompetent brain is unclear. We generated gliomas in WT mice via in utero deletion of key tumor suppressor genes and serially monitored cortical epileptogenesis during tumor infiltration with in vivo electrophysiology and GCAMP7 calcium imaging, revealing a reproducible progression from hyperexcitability to convulsive seizures. Long before seizures, coincident with loss of inhibitory cells and their protective scaffolding, gain of glial glutamate antiporter xCT expression, and reactive astrocytosis, we detected local Iba1+ microglial inflammation that intensified and later extended far beyond tumor boundaries. Hitherto unrecognized episodes of cortical spreading depolarization that arose frequently from the peritumoral region may provide a mechanism for transient neurological deficits. Early blockade of glial xCT activity inhibited later seizures, and genomic reduction of host brain excitability by deleting microtubule-associated protein Tau suppressed molecular markers of epileptogenesis and seizures. Our studies confirmed xenograft tumor–driven pathobiology and revealed early and late components of tumor-related epileptogenesis in a genetically tractable, immunocompetent mouse model of glioma, allowing the complex dissection of tumor versus host pathogenic seizure mechanisms.
Commentary
Any conquest is an act of war. From the ancient times to the more peaceful exploration of the universe, conquering a territory means dealing with barriers. Cancer is a savage conqueror and the brain is the king to defeat. So, brain tumors have their tricks to burst it all. We now know that neuronal activity fosters the growth of cancer cells in the brain. 1 -4 Complementarily, cancer cells impact synaptic connectivity to unbalance the equilibrium between excitatory and inhibitory inputs. 5 But we still miss many details of the plan. In a recent study, Hatcher et al 6 used a glioblastoma model to follow how hyperexcitability derives from the complex interplay between microglia, astrocytes, and neurons to grow tumors from inside. The emerging field of cancer neuroscience is on fire. 7
Hatcher et al made use of a CRISPR/Cas9 strategy to induce primary brain tumors consisting of depleting three tumor suppressor genes frequently altered in patients (Tp53, Nrf1, Pten) by using in utero electroporation in mice 6,8 (Figure 1). This approach allows studying the causality between brain tumor development and its impact on neuronal connectivity in a stepwise manner to clarify the sequence of events underlying epilepsy, which is prevalent in this model. For instance, this group previously reported the impact of astrocyte heterogeneity on glioblastoma-derived seizures. 9
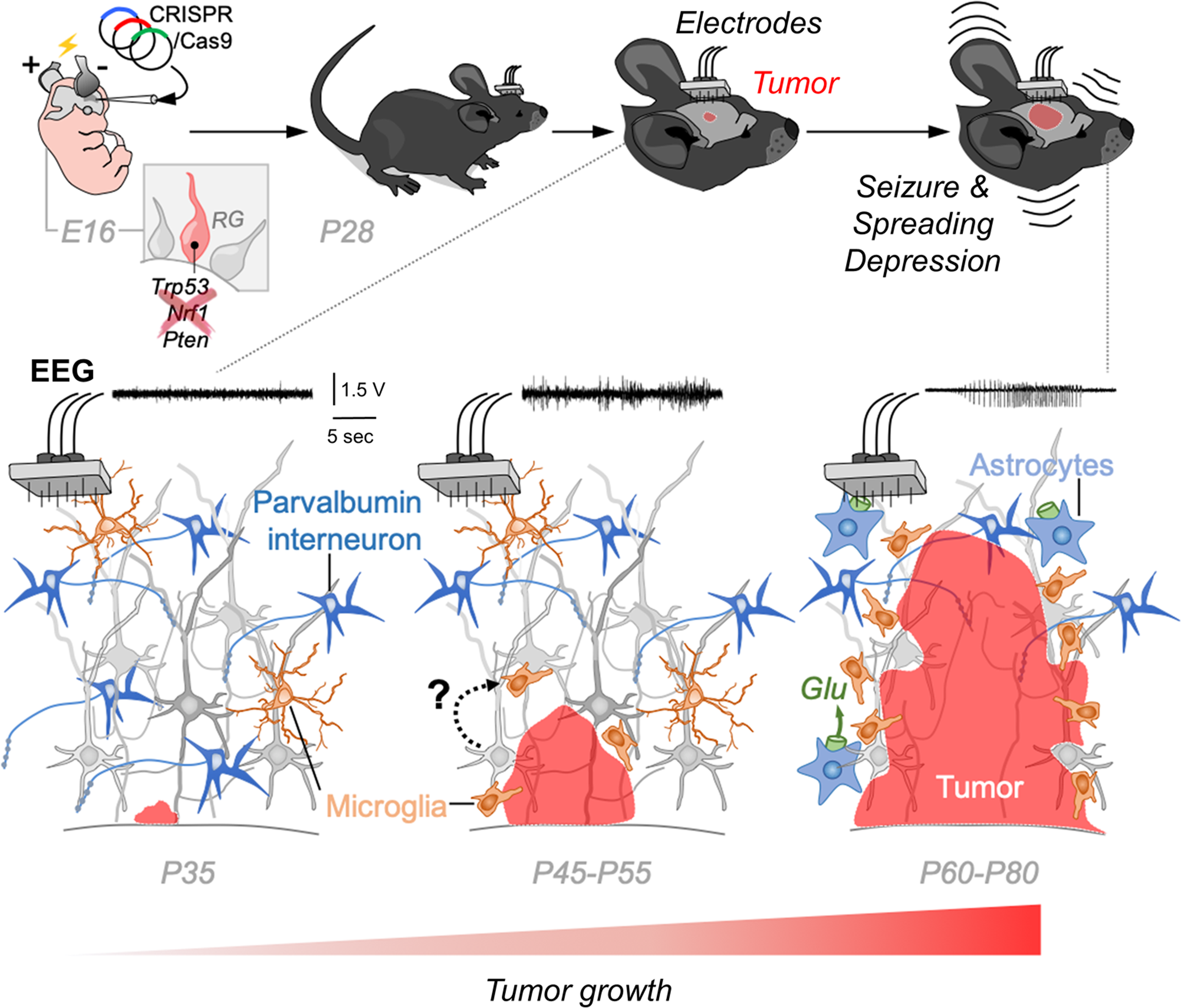
Immunocompetent CRISPR-based glioblastoma mouse model. Gliomas were generated in utero by deletion of key tumor suppressor genes such as Trp53, Nrl1, and Pten. Mice were examined along postnatal ages using longitudinal electroencephalography (EEG). Tumor-driven alterations progressed along early and late phases in association with epileptogenesis and culminating in generalized tonic-clonic seizures and spreading depression.
In order to study the pathogenesis of epilepsy, Hatcher et al implanted mice with intracranial electrodes for longitudinal recordings over several postnatal weeks. They also used genetically encoded calcium sensors such as GCaMP7 to better resolve the spatiotemporal evolution of neuronal activity. Authors revealed a reproducible sequence of events progressing from hyperexcitability to seizures as the tumor grows along postnatal days (Figure 1). Their core finding is that changes in the tumor microenvironment (ie, anatomical changes and aberrant cortical spike discharges) and their clinical impact (ie, motor seizures) were correlated to each other, but they did not fully overlap, reflecting the intricacy of glioblastoma colonization.
In trying to solve the puzzle, authors first evaluated whether neuronal loss around the peritumoral region, and more specifically of parvalbumin expressing interneurons, could explain hyperexcitability. Counterintuitively, the tumor did not dramatically impact the viability of surrounding neurons which rather exhibit a steady reduction from the initial incidence of electrical abnormalities to the late occurrence of seizures. Instead, Iba1+ microglia cells with ameboid morphology increased dramatically in the peritumoral region and correlated better with the progression of hyperexcitability, as also seen in humans. 10 Microglial infiltration was spatially and temporally associated with degradation of perineuronal net, which potentially collude with secreting proteins from the tumoral mass to demolish the brain-protective scaffolding. Similar to human gliomas, 11 by the time seizures emerge, the expression of xCT antiporter-mediated secretion of glutamate by astrocytes has already peaked. Pharmacological treatment with a specific blocker of xCT helped to control seizures but not interictal discharges in mice with tumoral masses, suggesting these were late stages in the conquering sequence.
Defeating cancer is threatening fight. Understanding the tricks of the enemy is critical. As one of the most frequent primary brain tumors, glioblastoma multiforme is lethal. The associated high mortality is undoubtedly one of the most urgent unmet needs. However, associated morbidities such as the high incidence of neuronal deficits contribute to worsen the quality of life of affected people. Given that brain tumor-derived epilepsy is frequently refractory to drugs, even after resection, studying the underlying biology is a valuable strategy to uncover potential vulnerabilities. In addition to revealing the sequential mechanisms of glioblastoma progression, Hatcher et al, identified spontaneous episodes of prolonged electrographic seizures preceding cortical spreading depression that emerges from peritumoral regions as the mass increases. These late recurrent episodes are of clinical relevance as biomarkers of peritumoral epileptogenesis.
The work by Hatcher et al, adds on the emerging data now suggesting how cancer takes over brain mechanisms. 7 Intrinsic excitability of host neuronal networks is key for the plan. Indeed, in mice lacking the microtubule-associated protein Tau (MapT), tumorogenesis was attenuated, a result that relates to the reduced level of neuronal activity caused by Tau deficiency. These mice did not develop earlier peritumoral hyperexcitability, and seizures were delayed. Remarkably, the increase of microglia associated with tumor growth was not reproduced in the MapT knockout mouse, confirming that interaction between neurons and other resident brain cell types accelerates primary tumor progression. Although these findings suggest early crosstalk between neurons and microglial, further analysis, including the use of more specific microglia markers, is required given additional implications of Tau in brain tumor development. 12
The critical mechanistic role of microglial cells, as well as astrocytes, is gaining momentum for many brain disorders. 13,14 Their extraordinary homeostatic plasticity suggests they can be easily kidnapped by cancer to advance. Similar to neurodegenerative diseases, unique microglial cells might develop at peritumoral margins. 15 Would these disease-specific microglial phenotypes act similarly in other brain disorders? Whether glial cells could provide alternative therapeutic options for tumor-associated refractory seizures remains to be proved. Knowing the tricks could lead us to develop the best weapons. These are exciting times in the fight against brain cancer.
