Abstract
Introduction:
Bacteriophages (or phages) are viruses that infect and replicate within bacteria. Widespread use of antibiotics during World War II curtailed phage research in the western world. Unfortunately, clinical deployment of antibiotics has led to the prevalence of antibiotic resistance becoming so severe that the World Health Organization (WHO) has warned about the prospect of a postantibiotic world. The WHO is encouraging research into clinical use of bacteriophages as a means of countering antibiotic resistance.
Methods:
This review summarizes advances in phage research, particularly as it pertains to clinical use. Topics covered include considerations for research and development, genetic engineering, biosafety, One Health (clinical, veterinary, and agricultural use), as well as environmental and regulatory issues.
Conclusions:
Research and development of phage therapy is a promising avenue for combating the dangerous rise in antibiotic resistance and may eventually become a standard medical countermeasure to complement or replace antibiotics.
Keywords
Introduction
Bacteria play key roles in the health and disease of humans, animals, and plants. As part of the human microbiota, bacteria are essential for health. 1 In contrast, bacterial pathogens also caused 7.7 million human deaths worldwide in 2019, comprising nearly 14% of deaths that year. 2 This dichotomy of positive and negative effects also applies to animal health and agriculture. 3 For plants, bacteria are essential for fixing nitrogen and affecting growth, yet they cause devastating diseases resulting in financial loss and famine. 4
Fighting bacterial disease in humans was fundamentally altered in the early twentieth century with the discovery and development of antibiotics. The discovery of penicillin in 1928 led to its widescale use during World War II and launched the modern age of antibiotics. Over the next century, a wide variety of antibiotics were discovered and/or synthesized. Antibiotics are now widely utilized to treat human, animal, and plant infections.
However, under selective pressure, resistance to antibiotics inevitably followed their clinical and agricultural deployment, leading to a biological arms race. Unfortunately, bacteria develop antibiotic resistance faster than humans can discover, develop, test, and obtain regulatory approval for new antibiotics. As such, the World Health Organization (WHO) has warned for years about the prospect of a postantibiotic world.
5
Each year, approximately 700,000 deaths related to multidrug-resistant infections are reported worldwide, with one report estimating a death toll of up to 10 million people annually by 2050.
6
Notably, antibiotic-resistant infections killed more people globally than AIDS or malaria. Antimicrobial resistance (AMR) in six species of bacteria is responsible for 80% of AMR fatalities, which makes these six organisms the most important targets for alternative therapies.
7
Therefore, alternatives to antibiotics to fight bacterial diseases are needed. 8 Prevention is always best, such as vaccines, sanitation and hygiene, food safety, and so on. Chemicals from plants (phytochemicals) and manipulating the microbiota using probiotics and prebiotics can also be beneficial. Another significant element in fighting bacterial infection is bacteriophages.
Bacteriophages (or phages) are viruses that infect and replicate only within bacteria. The National Institutes of Health generally categorizes phages as Risk Group 1 organisms, because they do not directly cause disease in humans.
Phages were first observed by Frederick W. Twort in 1915, when he observed a transmissible agent that caused the destruction of bacterial colonies. 9 He noted that this agent was small enough to pass through filters that typically retained bacteria and that the agent required bacteria to grow.
In 1917, Félix d’Hérelle independently discovered a similar phenomenon. 10 Working with dysentery bacilli, he observed clear spots on agar plates where the bacteria had been killed. d’Hérelle correctly interpreted that the bacteria were killed by an “invisible microbe” or a “virus parasitic on bacteria,” and he coined the term “bacteriophage,” meaning “bacteria eater.” He went on to devise and use several phage treatments for a variety of bacterial diseases. 11
In the 1930s and 1940s, phage therapies were marketed in the United States by Eli Lilly, Squibb & Sons, and Abbott Laboratories and by others in Europe. In 1923 d’Hérelle cofounded the Eliava Institute in Tbilisi, Republic of Georgia, where extensive research and development in phage therapy continues to this day.
Unfortunately, the intricacies of phage therapy, (i.e., the exquisite specificity of phages for even particular strains of a bacterial species) were not fully understood in the first half of the 20th century. This led to the unraveling of phage therapy’s initial success. As antibiotics became widely utilized, the West lost interest in phage therapy research, although it continued and even flourished in Eastern Europe.
The AMR crisis has reinvigorated interest in phage therapy in western medicine. The WHO now encourages research into phage-based therapeutics, launching a fact sheet on the matter and publishing a report, Building the Evidence for The Use of Bacteriophage Therapy. 12 In the following discussion, we examine the dramatic increase in research on phages to improve their use in clinical trials as well as increasing numbers of clinical trials to combat bacterial disease.
Phage Biology
Phages share many characteristics with animal viruses that are familiar to those who work in the medical or veterinary fields. Table 1 is a glossary of key terms pertinent to virology and phage biology. Phages possess definite structure and are usually composed of protein capsids containing a nucleic acid genome, most often DNA. 13 Because electron microscopy preceded molecular nucleic acid techniques, phages were initially classified based on their structural appearances (Figure 1). The vast majority (∼96%) of known phages belong to the Caudovirales order, composed of phages with phage heads and tails with or without fibers (Figure 1B–D). They have double-stranded DNA genomes. The other major groups of medically relevant phages are the icosahedral (no tail or fibers, not shown) and filamentous (no defined capsid head, Figure 1E) phages. Finally, there are the pleomorphic phages (Figure 1G). The vast majority of phages are naked, but there are examples of enveloped phages (Figure 1F). Most phages have DNA genomes, single or double stranded, linear or circular; however, there are examples of RNA phages.
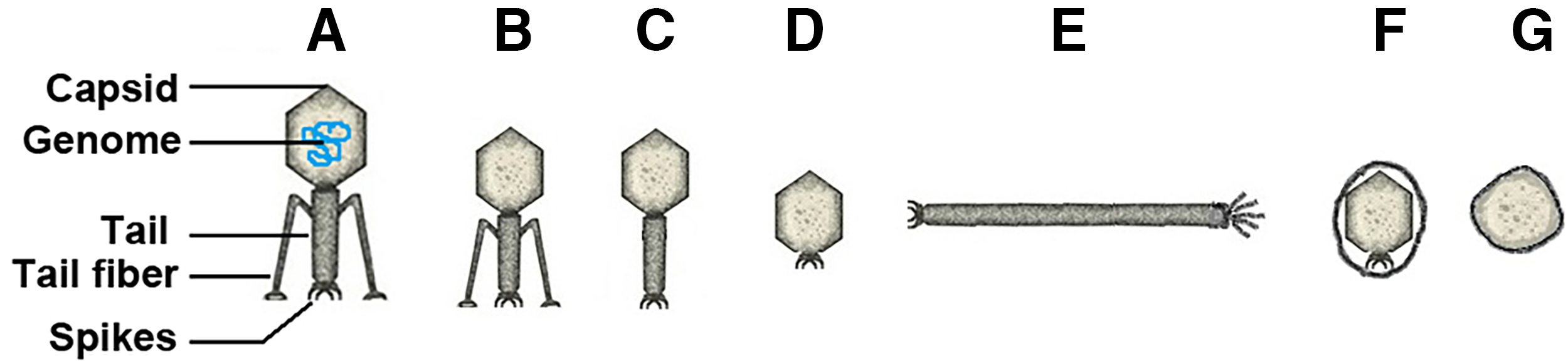
Phage structure.
Glossary of terms

The phage life cycle
Like animal viruses, the interaction of phages with host cells begins with binding to a specific receptor (Figure 2, parts 1 and 2). This step can be mediated by the phage tail, fibers, or other proteins. The specificity of the receptor binding has the greatest effect on host range and can also affect resistance of bacteria to phage infection (Figure 2, part 2). Manipulating phage receptor binding is the major element in engineering phages for practical applications, such as phage therapy. After initial binding, the phage genome is injected into the host cell, often through the tail (Figure 2, part 3). Inside the bacterial cell, the genome is expressed and replicated, resulting in production of progeny phages (Figure 2, parts 5–7). This step involves complex interactions between the phage and host genomes and components, which can contribute to host range. Consequently, merely enabling a phage to bind to a bacterial cell does not guarantee that successful, productive infection will ensue. This is important for phage therapy development.
Phage life cycles. 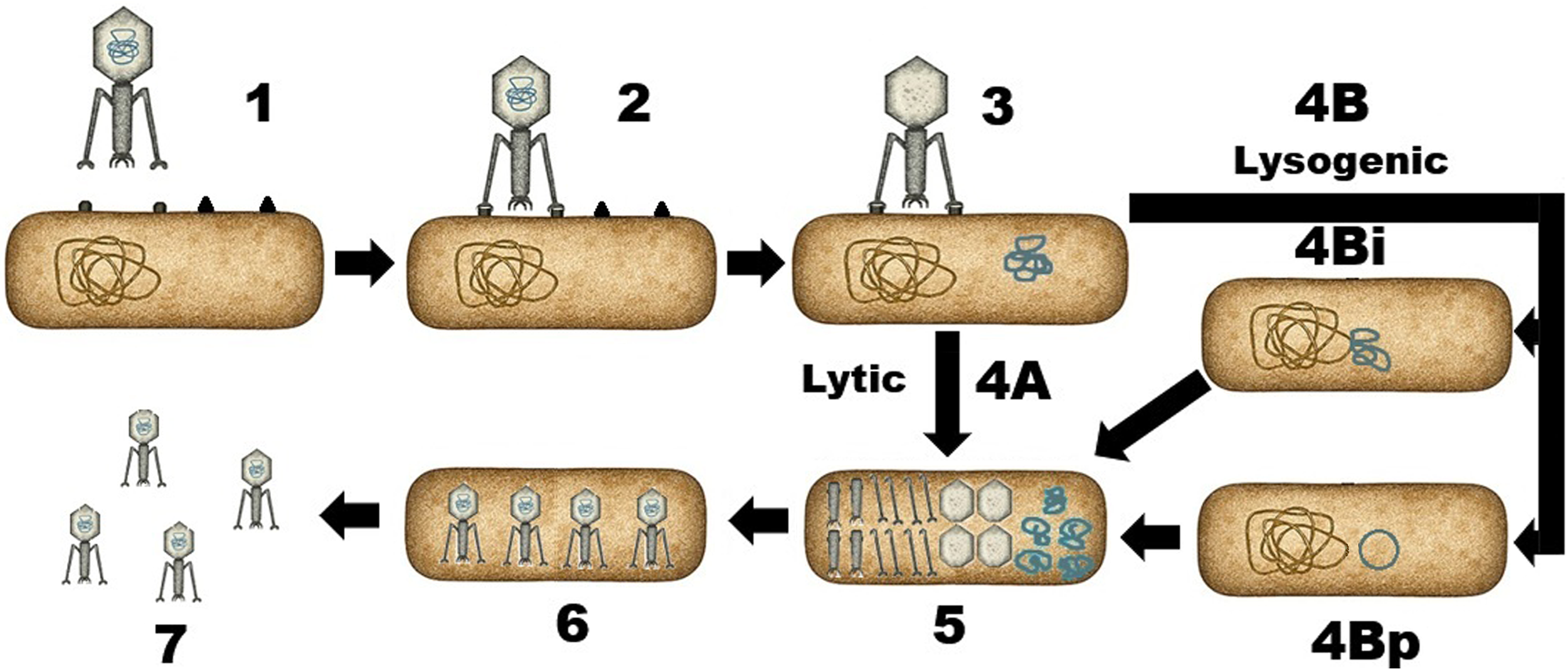
Phages are also classified by their life cycle interactions with their host cells (Figure 2):
Lytic phages always kill the target cell after infection (Figure 2, part 4A). Temperate phages can coexist with their host cells in a process called lysogeny, which bears some similarities to latency observed in animal viruses such as herpes viruses (Figure 2, part 4B). In lysogeny, the phage genome, called a prophage, either integrates into the bacterial chromosome (Figure 2, part 4Bi) or can exist outside of the bacterial chromosome as a plasmid (Figure 2, part 4Bp). At some point, lysogeny can shift to the lytic cycle causing phage replication and killing of the host cell. Chronic infection is a third form of interaction carried out by some filamentous phages. Rather than lysing the host cell or existing as a quiescent genome, the infected cells produce and secrete phage particles while remaining intact and viable (not shown).
Much like with antibiotic resistance, exposure of bacteria to phages results in selection for resistant mutants (Figure 3). Although this can render phage therapy ineffective, there can also be beneficial results. 14 For example, the most common mechanism of phage resistance is loss of the phage receptor. If the receptor has a function that is important to the organism’s virulence, the mutant may become attenuated (Figure 3, part B). Phages often use surface structures that contribute to virulence as their receptors, such as lipopolysaccharide (LPS), pili that are used for attachment to host cells, or antiphagocytic capsules. Mutants that have lost these factors are attenuated for virulence and less fit to cause disease.15,16 If a phage binds to an antibiotic efflux pump, loss of that pump renders the bacteria sensitive to the antibiotic (Figure 3, part C).17,18
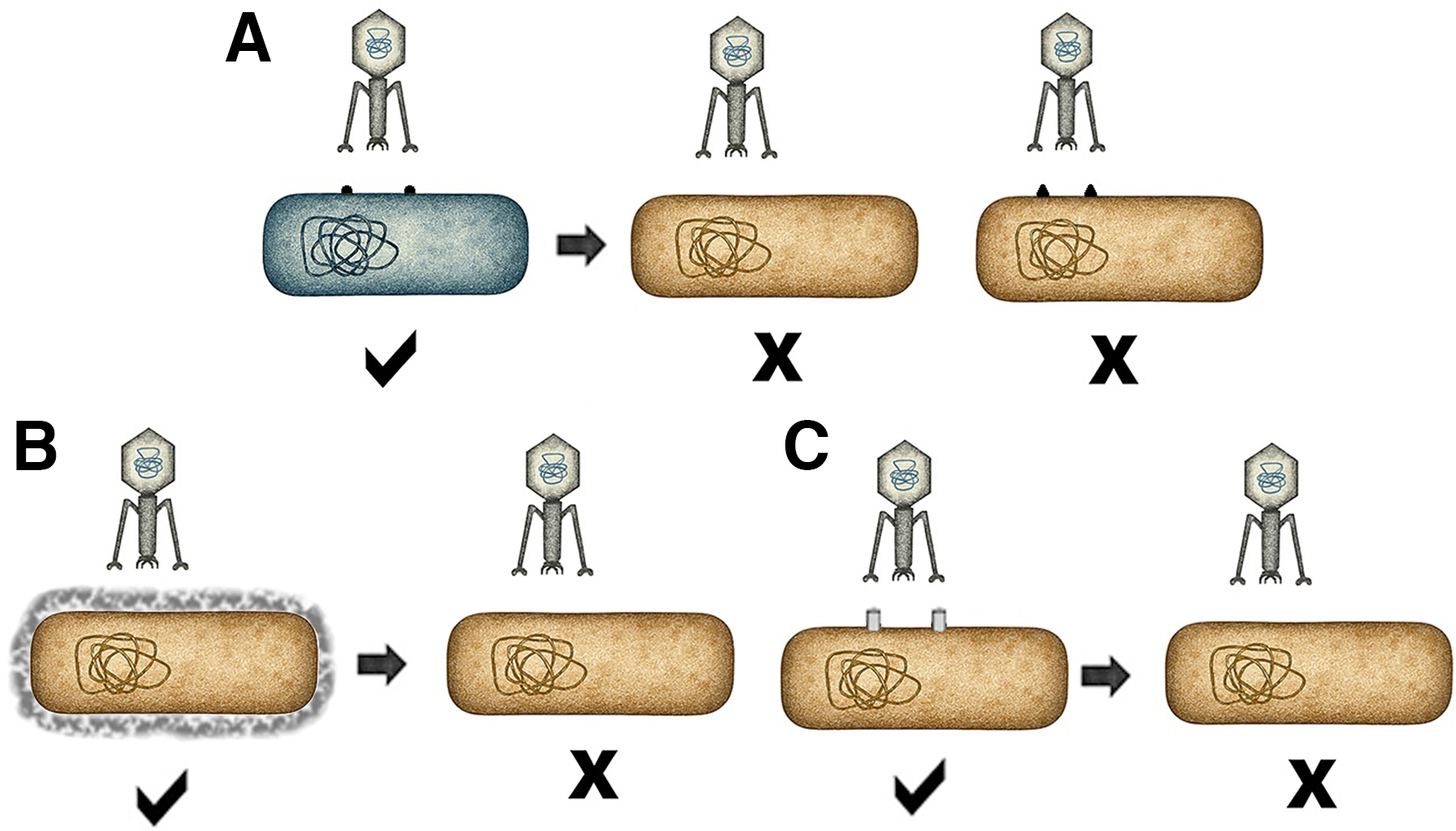
Resistance to phage infection mediated by altering the phage receptor.
A final consideration in the phage–host interaction is the mechanism of lysis of the host cell. Almost all bacteria have a cell wall that contains a peptidoglycan layer that is too rigid to enable the phages to escape the dying host cell. Therefore, most phages encode endolysins (lysins), enzymes that degrade peptidoglycan. Some of these lysins have immense antibacterial activity and are being investigated as antibiotic alternatives. 19 Manipulation of lysin activity in phages is also under study as a means of improving their utility as phage therapy agents.
Phage-mediated gene transfer
Recombination is an important aspect of phage genetics with relevance to phage therapy. Phages can be involved in three different mechanisms of recombination (Figure 4). There are two forms of transduction, in which a phage particle can transfer bacterial DNA sequences from the donor cell to a recipient cell. Lysogenic conversion involves delivery of phage genes into bacterial hosts.
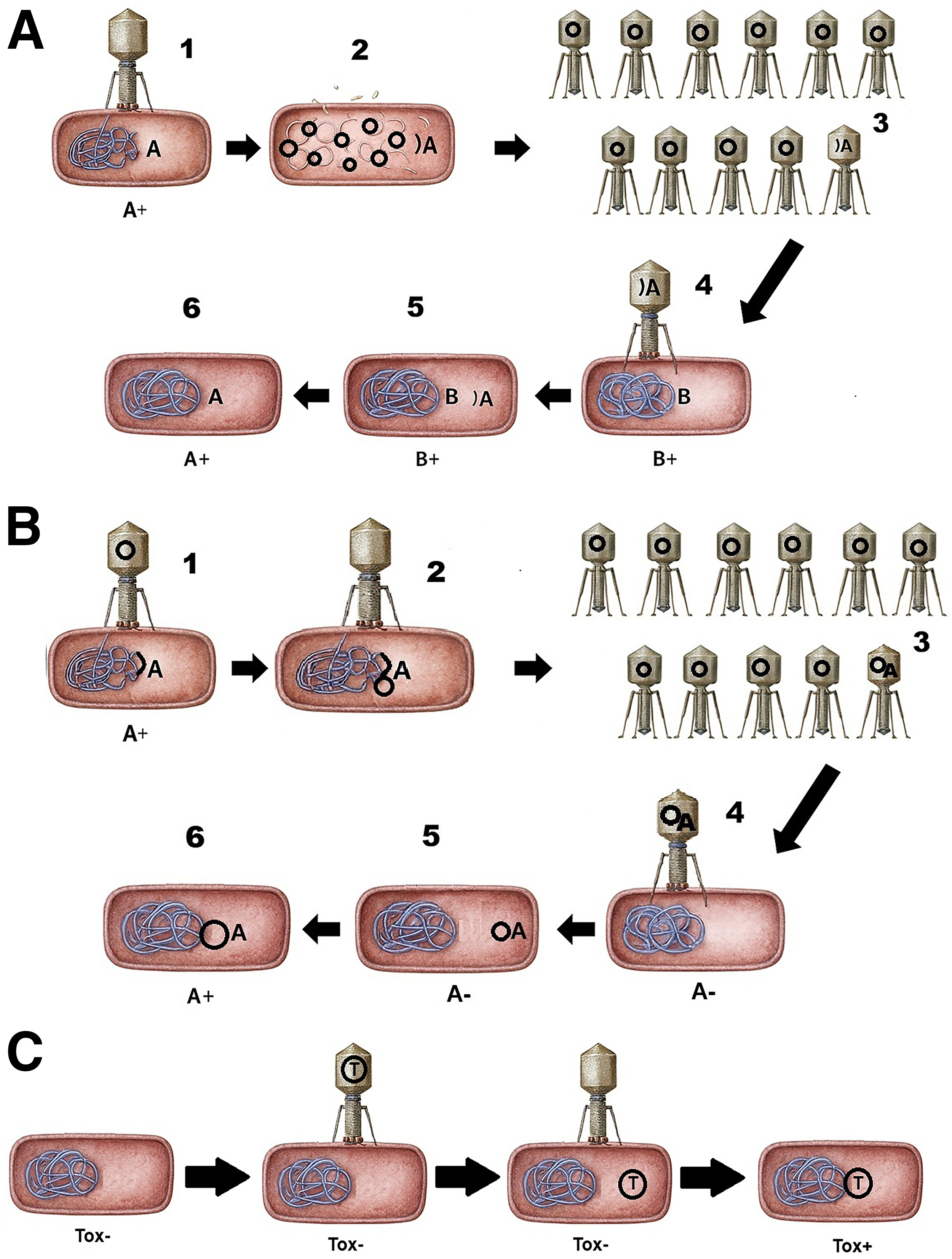
Genetic exchange mediated by phages.
In general transduction, the capsid mistakenly acquires a piece of host cell DNA instead of the phage genome (Figure 4, part A).
Specialized transduction is only possible by temperate phages whose prophages are integrated into the host chromosome. Sometimes when the prophage excises during replication, sequences of the host genome adjacent to the integration site get packaged in the progeny phage along with the phage genome. If the specialized phage transducing particle lysogenizes a recipient cell, the donor DNA sequence becomes a part of the recipient genome (Figure 4, part B).
Lysogenic conversion involves the delivery of phage genes by some temperate phages into bacterial hosts. The most notorious examples of lysogenic conversion are toxins of some major bacterial pathogens, such as those that cause cholera, botulism, and diphtheria (Figure 4, part C).
Development of Phages as Antibacterials
In the development of phages as therapy for bacterial infections, there is a critical dichotomy between using naturally occurring wild-type phages versus genetically engineered phages endowed with beneficial attributes. Although there is seemingly immeasurable diversity in naturally occurring phages, there are gaps in terms of having phages to help fight bacterial disease. Based on the critical elements of phage biology described above, filling those gaps with recombinant and synthetic biology offers promise.
One of the blessings and banes of phages as therapeutic agents is their exquisite specificity for their bacterial targets. This specificity enables phages to kill only the intended pathogen, leaving the remaining microbiota intact, as opposed to using antibiotics that may kill both the pathogen and beneficial components of the microbiota. However, the specificity of some phages results in their being able to target only a very limited set of strains of the pathogen. To overcome this specificity problem, several approaches are being undertaken.
Antibacterial approach 1: Personalized phage therapy
Should time permit in treating an infection, the identification of phages from a locally available collection that targets the patient’s infecting agent could result in effective therapy. Determining the sensitivity of a given strain of bacteria to a given phage can take as little as a few hours. This personalized medicine approach is predicated on the existence and availability of a pool of phages collectively targeting all or most strains of a given pathogenic species. The personalized medicine approach currently yields the best results in clinical trials. 20
Antibacterial approach 2: Phage collections
Pools of phages that have extensive coverage for diverse strains of a pathogenic species can address the specificity issue. An additional benefit to the pooled phage approach is that if resistance arises against one of the phages in the pool during treatment, other phages in the pool may kill the resistant bacterial mutant.
As obvious as this approach seems, there is a dearth of coordinated effort to create a catalog of phage collections. 21 However, through the latter part of the previous century, renewed interest in phages resulted in attempts at creating annotated collections. Significant hurdles remain in developing a universal phage bank for clinical use. These include intellectual property rights, standardized methodologies in characterizing phages, and sustainable interest in the venture. Finally, the production of broadly active phage pools is predicated on the isolation of phages with the requisite specificities. Of course, gaps can exist where a phage with the desired specificity does not exist or is yet to be isolated, categorized, and made available.
Antibacterial approach 3: Genetic engineering of phages
Genetic engineering of phages allows for the creation of phages with desired specificity, especially for cases where no naturally occurring phages target the isolated strain of the pathogenic species.
Targets for genetic engineering: Receptor binding
Much like with animal viruses, the ability of a phage to bind to a specific host cell governs its tropism or host range. In tailed phages, receptor binding is performed by tail fibers. Ando et al.
22
switched tail fibers that govern host specificity among phages that infect various species of Enterobacteriaceae. The genetic engineering was accomplished using the yeast
To circumvent the limited repertoire of phage specificities existing in nature, generation of new binding specificities has been undertaken. 23 For example, random mutagenesis was used to generate numerous new binding sequences in the host range-determining regions of phage T3. 24 Genetically engineering tail fiber specificity should extend host range to bacteria other than the original host strain, possibly even other species.
Targets for genetic engineering: Enhanced bacterial killing
As detailed above, one caveat to such phage engineering is that the ability of a phage to infect, propagate in, and kill a bacterial cell involves more than just binding to the surface. To overcome this barrier, phages have been engineered with mechanisms that enable them to cleave specific DNA sequences in target bacteria, which could either broaden or limit their activity. For example, Bikard et al.
25
created phagemids (plasmids that are packaged and delivered by phages) specific to
Following the phagemid development, Gencay et al.
27
created recombinant phages that encoded CRISPR-Cas systems, named CRISPR-Armed Phages, that targeted
Human phage therapy studies
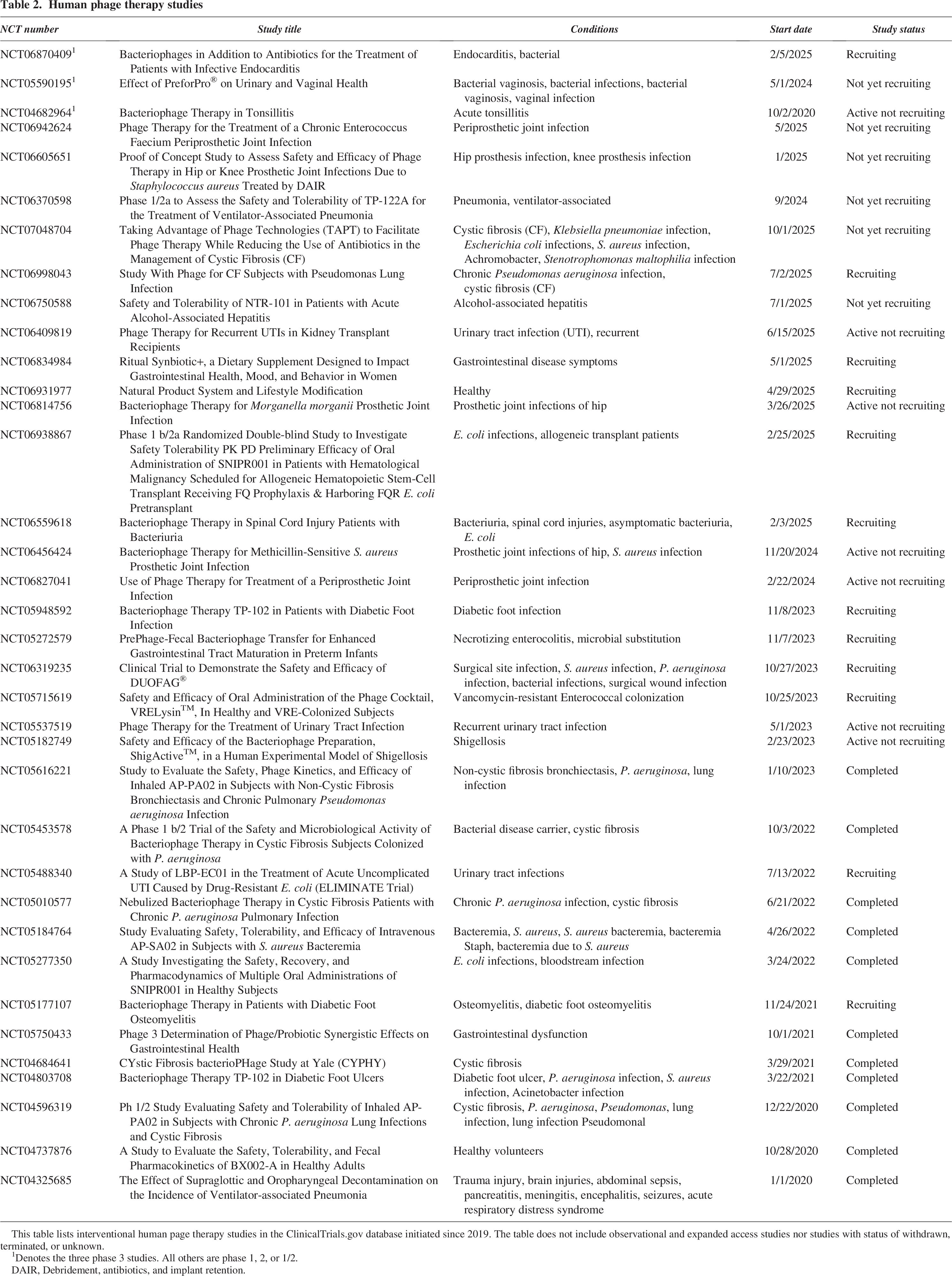
This table lists interventional human page therapy studies in the ClinicalTrials.gov database initiated since 2019. The table does not include observational and expanded access studies nor studies with status of withdrawn, terminated, or unknown.
1Denotes the three phase 3 studies. All others are phase 1, 2, or 1/2.
DAIR, Debridement, antibiotics, and implant retention.
In the development of therapeutic phages, the most promising techniques revolve around engineering new phages from components of existing phages or utilizing synthetic biology. Mitsunaka et al.
28
developed a system where phage genomes could be assembled by cloning or synthesizing large overlapping segments. Building phages from scratch is called rebooting. They rebooted several wild-type phages from a variety of gram-negative bacteria and even an acid-fast mycobacterium. They created a constitutively lytic P22 phage from
In the realm of engineering expanded phage host specificity, Dunne et al.
29
used elegant structural and molecular biology with a
Further engineering of phages has endowed them with qualities that enhance utility in fighting infection and industrial contamination involving biofilms. Biofilms are dense communities of bacteria adherent to surfaces that are characterized by matrices that impede antibacterial activities of phages, antibiotics, and other antibacterial modalities, including immune responses. Lu and Collins
30
engineered
Phages as Drug Delivery Vehicles
Phages are also being developed to aid in treatment of noninfectious diseases, such as cancer (reviewed by Li et al. 34 ). Phages are being engineered to deliver drugs and peptides to tumors in a specific manner. For example, Li et al. 35 engineered a filamentous fd phage to target breast cancer in a mouse model by binding to and neutralizing an angiogenic protein, angiogenin, that is important for tumor viability. The tip of the phage was genetically engineered to express a breast cancer antigen-binding peptide that was itself identified via phage display technology. The coat protein of the phage, present in over 3,000 copies, was genetically engineered to express a fusion with an angiogenin-binding peptide, also identified with phage display. By homing to tumors, the antiangiogenin activity would have greater efficacy while not having unwanted systemic effects. This so-called nanofiber treatment of mice containing human breast cancer cells was effective in reducing tumor size and stimulating necrosis of tumor cells.
Two related articles demonstrated the potential utility of phages in diagnosing and treating Alzheimer’s disease. Frenkel and Solomon 36 showed that a filamentous phage expressing a recombinant antibody that recognizes Amyloid β that is present in Alzheimer’s disease lesions could be intranasally administered to mice and could bind to Amyloid β plaques in the brain. Solomon and Frenkel 37 continued this line of investigation using phage display technology to create a filamentous phage expressing a peptide sequence that disrupts Amyloid β plaques. In theory, this phage could be used therapeutically in Alzheimer’s disease.
A powerful recombinant system with broad applicability, named SpyPhage, was recently developed by Liyanagedera et al. 38 They split a bacterial fibronectin binding protein (FbaB) into two domains that retained attraction to each other. They genetically inserted the receiving domain into the phage coat protein (SpyTag). Proteins to be attached to the phage capsid were genetically fused with the corresponding SpyCatcher domain from the FbaB protein. Proteins with the SpyCatcher domain are covalently fused to the SpyTag domain on the capsid on contact, thereby enabling the decorating of the phage with essentially any protein. Fusion proteins with human epidermal growth factor or Salmonella Rck enabled the phages to be taken up by human cells, rendering the phages intracellular delivery vehicles.
McClary et al. 39 similarly developed a Lambda phage system in which capsid proteins were genetically modified to enable the covalent coupling of proteins, such as tumor-targeting antibodies, and even chemotherapeutic drugs. This dual-acting phage-based designer nanoparticle was effective at delivering the breast cancer drug UbV.7.2 into breast cancer cells via the anti-Human epidermal growth factor 2 monoclonal antibody trastuzumab and inhibiting cancer cell growth in vitro.
An interesting study was recently published in which phages exerted dual activities of killing pathogenic bacteria while delivering a drug to the target site. Zheng et al.
40
isolated a phage against
Uses of Phage Other than Medical
Over and above the use of phages to prevent and treat bacterial infections and to aid in other diseases such as cancer, phages have wide use in other realms. They can be used to prevent or treat bacterial infections in animals in agriculture or in veterinary practice. They can clear food animals and food products of bacteria during preparation, packaging, and storage. Phages can be used to prevent or treat bacterial infections of plants and even to disrupt the flora of harmful insects.
One Health
One Health is an integrated consideration of health across people, animals, and ecosystems. Such an all-encompassing approach is aimed at exerting higher efficiency at preventing disease than targeted interventions in only one realm, for example, medicine.
Agriculture—plants
Bacterial diseases are major problems in agriculture worldwide, threatening crops and resulting in food scarcity. Some bacterial infections have been treated with antibiotics; however, resistance has emerged. Therefore, phages have gained acceptance as tools in fighting bacterial diseases of plants of agricultural significance. 41 Czajkowski et al. 42 recently reviewed the status of phage therapy in treating or preventing plant diseases. There are several phage products approved for use in preventing bacterial diseases of plants in the United States and Europe. In the United States, Omnilytics, Inc. produces several phage products targeting tomato bacterial canker, citrus canker, and fire blight for apples and pears, among others. Most recently, A&P Inphatec obtained E.P.A. approval for XylPhi-PD for the reduction of Pierce’s disease in vineyards. These products not only kill the targeted pathogen but also modify the local microbiota to improve plant health. There are other phage products approved for use in the United Kingdom and elsewhere in Europe. Moving forward, the development of naturally occurring phages for prevention and treatment of bacterial infections of plants represents the most expedient avenue for phage technology.
Agriculture and veterinary—animals
The history of using phages to fight bacterial infections in animals is as old as that for medical use. Felix d’Herelle, one of the discoverers of phages, used phages to fight
Aquaculture
There are numerous bacterial diseases of fish, shellfish, and crustaceans, for example, shrimp, with great economic impact. Although significant research on the potential use of phages to prevent or treat these infections is ongoing, there are currently no approved products. 43
Insects
Although phages will not exhibit direct activity against insects, many insects depend on symbiosis with specific bacteria for their health and even viability. Therefore, targeting critical bacteria in the environment could aid in suppression of harmful insect populations. For example, Tikhe and Dimopoulos
44
demonstrated under laboratory conditions that phages targeting
Food preparation
The FDA considers phages as Generally Regarded as Safe for use in food.
41
The first FDA-approved phage for use in food was ListShield produced by Intralytix in 2006. There are now numerous phage products that are approved in the United States, Canada, Israel, United Kingdom, China, and elsewhere.
45
The most commonly targeted bacteria are
Clinical Trials
With regard to regulatory and biosafety considerations of phage, the primary, but not sole, ongoing activity is in clinical trials. It took nearly a century for phage therapy to gain enough attention in western medicine for serious basic science studies and eventual clinical trials to be performed.
It is important to note the unique aspects of phage therapy that affect the difficulty of conducting such trials in the current regulatory environment. This has been recently reviewed.46,47 The first problem is the nature of phages, themselves. As viruses of bacteria, as opposed to chemical drugs, their interactions with target bacterial cells, production, and purification can be extremely variable, depending on the specific bacterial targets and the sourcing of the stocks for a clinical trial. Therefore, a phage preparation for a given infectious disease might be highly effective in one trial at one site, but then fail miserably at another similar trial at a different site. For example, if phages in the treatment stock are not confirmed to exhibit activity against the target(s), the trial is doomed to failure. It would therefore be ideal if therapeutic phage stocks were sourced from common entities that had thoroughly characterized the phage activity. There are some issues in common with antibiotic studies, for example, the nature of the infection and complexity of the infecting agents. Superficial infections might be easier to treat than systemic infections, and single agent infections would be easier to treat than polymicrobial infections. Like antibiotics, the route of administration can have an immense effect on efficacy. Unlike antibiotics, the nature of the phage preparation can affect success. For example, phages might benefit from being coated in a matrix that improves stability or resists being cleared by the host innate immune system. Another major problem is trial design. When treating serious bacterial infections, it would be unethical to withhold antibiotic treatment. Therefore, most trials involve adding phage therapy to antibiotics. This makes interpretation of the results difficult. Furthermore, it is possible that coadministration of antibiotics could hinder the effectiveness of phage therapy if the target bacteria are impeded from being able to serve as productive hosts for phage replication, for example, by use of protein synthesis inhibitors. There have been many compassionate use studies where there were no effective antibiotics. However, such studies cannot be scrutinized in relation to controls or by rigorous statistical analysis because they are most often single subject studies.
As is usually the case, science is advancing faster than the associated regulations or regulatory guidance. There has been an explosion in PubMed listed publications involving phage therapy with 5,726 of the 8,394 publications published between 2015 and 2025. Similarly, the overwhelming majority of ClinicalTrials.gov entries for clinical trials involving bacteriophages have taken place within the last 6 years. A search of the ClinicalTrials.gov database for the search term “bacteriophage” yielded 115 studies, of which 85 were interventional phage therapy, as opposed to observational. Among interventional studies not represented in the ClnicalTrials.gov database, 16 multisubject studies were reviewed by Stacey et al. 48 and 41 individual subject case studies were summarized by Uyttebroek et al. 49 (Figure 5). Figure 5 shows that more than half of the clinical trials involving phages are multisubject interventional. These are the most effective clinical trials for gaining meaningful data. Observational and individual case studies and expanded access trials can contribute some evidence, but they lack some of the components that are integral to controlled clinical trials. Of the 115 studies in ClinicalTrials.gov, 72 were initiated between 2019 and 2025. Select interventional studies from this group are listed in Table 2. As a general trend, the number of clinical trials utilizing phage-based investigational products is increasing, as is the percentage of trials demonstrating clinical efficacy.49,50 Sites of infection under study vary widely from implanted devices to disseminated infections and infections localized to specific sites or organs. Similarly, various methods for phage delivery vary widely with oral administration being most common (30%), followed by topical (16%), intrarectal (15%), injection (13%), transdermal (10%), and inhalation (3%). 41
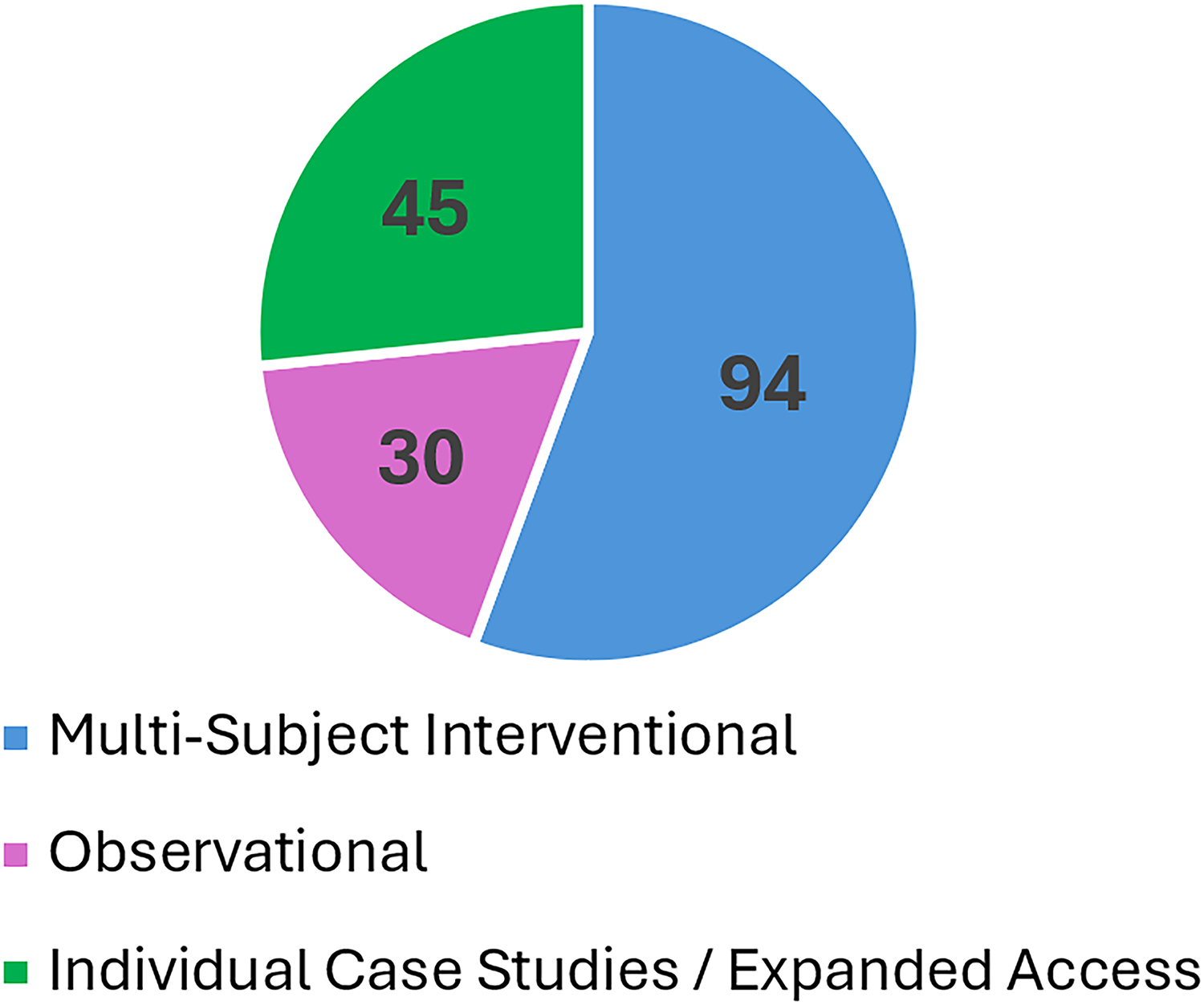
Phage therapy studies reported in ClinicalTrials.gov or select published review. A search of the ClinicalTrials.gov database for the term “bacteriophage” yielded 115 studies of various types and status. Studies represented that are not documented in the ClinicalTrials.gov database include 16 controlled, multisubject studies reviewed by Stacey et al. and 41 individual case studies summarized by Uyttebroek et al. 49
Before reviewing recent clinical trials from the United States, the extensive literature from Eastern Europe, particularly Poland and the former Soviet Union, cannot be ignored. Sulakvelidze et al.
51
reviewed the literature and early phage therapy history, especially from the non-English literature that was mostly ignored by western scientists moving into phage therapy. They noted that after the brief commercial use of phage therapy in the West was stopped for several reasons, it was never halted in Eastern Europe. Therefore, conducting clinical trials on established treatment modalities was not considered essential. A notable study was conducted in Tbilisi, GA in the early 1960s involving over 30,000 children.
52
Half received a phage cocktail against
Phage therapy clinical trials are rapidly advancing in western medicine (reviewed by Palma et al. 55 and Liu et al.). 56 Although there are numerous ongoing clinical trials in the United States, there are very limited peer-reviewed publications describing the results of completed trials. Ibrahim et al. 46 reported the results of 11 recent phage therapy clinical trials that met rigorous criteria. It is clear that none of the phage therapies caused significant adverse events in study subjects. Unfortunately, data on efficacy are extremely limited. Most of the trials are phase 1 and/or 2, and so, they consist of very small sample sizes and the primary objectives are safety, not efficacy. Pirnay et al. 20 summarized the results of phage therapy in 100 patients treated in Belgium. Commercially available phage preparations were used to treat a variety of complex bacterial infections. Although the ongoing study has demonstrated both safety and efficacy, it was important to confirm that the causative bacterial agents were sensitive to the phage products using a personalized medicine approach. One other noted that phage therapy success was a recent study on phage treatment of hip joint replacement infection. 57 Combination of phages and antibiotics outperformed antibiotics alone. Ibrahim et al. 46 also discussed failures in phage therapy, which could not always be explained. Liu et al. 56 described results of numerous clinical trials and focused on the nature of the phage therapy: noncustomized to the patient’s infecting agent, where the phages are blindly obtained from existing sources; customized selection, where the phages are confirmed to kill the patient’s infecting agent; and customized production, where the phages are propagated on the patient’s infecting agent. Customized production was optimal, but it also had the most severe practical limitations in terms of obtaining the necessary phages in a timely manner, if at all. Their meta-analysis confirmed the safety and efficacy of phage therapy. Other recent reviews of phage therapy clinical trials have reported similar efficacy and uniform safety. 48
We conclude this discussion of phage therapy clinical trials by discussing in detail a model study that is ongoing, but whose preliminary results were recently published.
58
This is a phase 1/2a trial examining a phage cocktail named BX004-A to treat
Biosafety, Biocontainment, and Regulatory Matters
As noted above, the unique aspects of phages compared with antibiotics and other drugs raise important considerations moving forward with their development in medicine and elsewhere. The regulatory environment in dealing with these issues has not always kept up with the science.
Desirable properties for phage-based investigational products
Ideally, phage-based therapeutics would possess the following attributes:
59
The ability to efficiently kill the intended pathogen in vitro and in vivo without a large proportion of bacterial survival or damage to the indigenous microbiota. This means that the phage should be virulent, not temperate. The phage should be easy to propagate, produce in high-titer preparations, and purify (e.g., >1010 plaque-forming units [PFU]/mL). The final preparation should be free of harmful contaminants, including bacterial endotoxins. The phage should be stable over a range of concentrations, such that storage for extended time periods at refrigerator temperatures is not associated with substantial loss of infectivity. Samples diluted down to 106 PFU/mL may rapidly lose viability.
60
The phage genome should not include any genes that are toxic, oncogenic, or otherwise encode virulence factors. The phage should not have the capability to function as a generalized transducing phage, unless it is intended to do so to carry out its therapeutic effect. When using a phage cocktail, the phages should not utilize the same cellular receptor, as loss of that receptor would allow the host to develop resistance to the entire cocktail.
Biosafety considerations
Lytic phages are best for combating bacterial infections. 59 Temperate phages can be utilized if the genes involved in lysogeny (i.e., repressor of lytic phase and integrase) are deleted. As noted above, this is being done in developing recombinant phages with improved qualities. Lysogenic conversion can enable phages to confer deleterious properties, such as toxins, to lysogenized bacterial hosts (Figure 4C). Therefore, all phages to be used in clinical trials must be genomically sequenced. If dangerous genes are identified, they can be deleted using recombinant means. Papudeshi et al. 61 recently devised an automated phage sequencing and analysis tool named Sphae that scans phage genomes for virulence factors, antibiotic resistance genes, and genes involved with lysogeny.
Phages are species-, and sometimes, strain-specific, meaning that they will only replicate in their natural host. Most of the phages being considered for phage therapy will therefore have bacterial pathogens as their hosts and propagating the phages would require large-scale cultures of these pathogens. Therefore, to avoid these complications, when possible, avirulent species or attenuated strains should be used for phage production. As noted above, cell-free extracts allow for phage replication without utilizing live bacterial host cells.28,38,62–64 It is possible to create cell-free extracts from a virulent host in containment, validate the lack of live virulent bacteria, and then transfer the extract to a lower biosafety level for phage production. Of course, this strategy is dependent on the risk assessment, validation of the methodology, and approval of the Institutional Biosafety Committee (IBC) and any applicable local/Federal regulatory agencies.
Environmental release
Biosafety professionals are trained to focus on containment, with little to no tolerance for environmental release of active biological hazards, especially those that contain recombinant DNA. The FDA currently does not consider the environmental release of a replicating gene therapy vector until after the dose expansion phase of a clinical trial, typically phase II.65,66 The FDA normally does not consider biosafety and environmental issues until deliberating on whether to issue marketing approval for a biologics license application, typically at the conclusion of a phase III clinical trial. If the clinical trial design involves replication-competent phages, it is a reasonable assumption that phages will be shed in bodily fluids, especially in urine and feces.
Hospital wastewater contains a diverse array of microorganisms, including human pathogens.67–70 Large healthcare facilities within the United States must comply with the EPA Clean Water Act and are typically regulated by local municipalities. Large medical centers are typically considered industrial users and are required to develop industrial pretreatment programs for their wastewater before it may be released to the sanitary sewer for final treatment. Pretreatment decreases the microbial burden, but it does not completely eliminate biohazards. Healthcare facility wastewater quality is an area of research and debate with several groups calling for regulatory requirements to mitigate biological hazards posed by healthcare facility wastewater.
Wastewater treatment at a treatment plant proceeds through several steps with different capabilities for inactivating biohazards. Large particles are removed through screening, grit removal, and sedimentary clarification. Secondary treatment involves aeration and introduction of bacteria to break down biological waste, resulting in activated sludge. This process is slow to break down phages as MS2, a surrogate for nonenveloped enteric viruses, was observed for up to 35 days after introduction in activated sludge, whereas Φ−6 bacteriophage, a surrogate for lipid enveloped viruses, was detected for no more than 4 days after its introduction. 71 The final steps in wastewater treatment involve filtration and disinfection with chlorine, ozone, or UV light, which have the greatest efficacy in phage inactivation. 72
Biocontainment
If phages are shed into the sanitary sewer, biocontainment can be achieved through the creation of replication-deficient phages in a manner that bears similarities to replication-deficient viral vectors (Figure 6). A gene essential to phage replication is deleted, rendering the phage replication defective (Figure 6, part 2). The deleted gene is cloned into host bacterial cells, which function akin to packaging cells for viral vectors enabling the propagation of the phage (Figure 6, part 3). Mitsunaka et al. rendered T7 phage replication deficient by deleting either the packaging signal sequence (Pac) necessary to package phage genome into the head particle or the
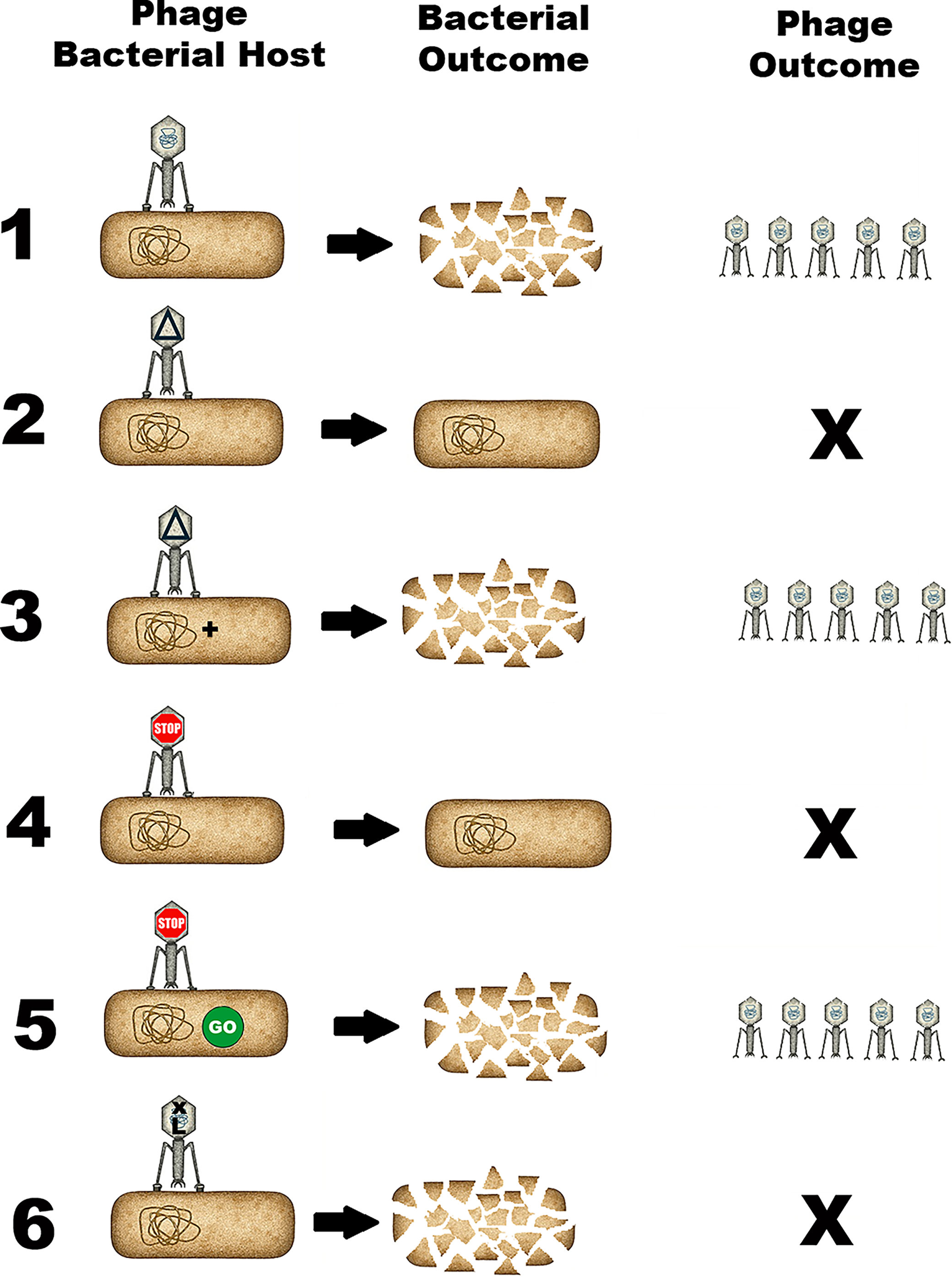
Conditionally replicative phages.
Another method of generating a conditionally replicating phage is engineering Amber stop mutations in critical replication genes (Figure 6, part 4). 73 These mutations render the replication proteins nonfunctional because their translation is prematurely terminated. However, when propagated in a bacterial host that encodes an Amber-suppressor mutation in a tRNA gene, the proteins are completely translated and the phage can reproduce (Figure 6, part 5). These strategies allow the phage to propagate in the complementing/suppressing host cells. It is possible to use nonreplicating phages for therapy (Figure 6, part 6). If such a phage can infect a target bacterial cell and particularly if it has been engineered with a super lysin, it can still lyse and kill the cell without replicating itself. The downside of these biocontained phages is that they do not replicate in the treated patient. As noted above, such phages have demonstrated efficacy in animal models.
Regulatory Considerations
As noted above, regulation of the development of the use of phages in medicine, agriculture, and elsewhere has lagged behind the scientific development. In June 2025, the United Kingdom became the first country to publish guidance on regulatory considerations for use of phage-based therapeutics. 74 Such guidance is necessary to standardize expectations for the preclinical research needed to obtain permission to conduct clinical trials, manufacture bacteriophage-based investigational products for clinical trials, and write clinical protocols.
In the United States, phage therapy is regulated by the FDA under the expanded access IND program for single-patient investigational new drug applications. 75 This requires several quality assurance steps such as characterization of the phage and sterility of the final stock. Wurstle et al. 76 developed a pipeline for rapid identification and characterization of phages for use in patients in less than 3 weeks. This study highlights many essential practical components of development and ultimate use of phages in the clinic.
For phage therapy to advance through good laboratory practice compliant preclinical animal studies and good manufacturing practice (GMP) compliant clinical use, the phage preparation must achieve an adequate level of purity, particularly the absence of bacterial host cell-derived molecules. Scalable purification methods that can adequately remove bacterial host cell endotoxins, proteins, and DNA must be implemented. For example, endotoxins are pro-inflammatory LPSs found in the outer membrane of gram-negative bacteria and can cause toxic effects such as fever, multiorgan failure, septic shock, and sepsis. To circumvent these problems, phages could be propagated on strains with decreased or less toxic components.59,77–79 Ideally, such hosts should be nonpathogenic, well characterized, and devoid of endogenous prophages that can cause lysogenic phage contamination. 63 Purification methods are critically important in generating safe phage preparations. 80
Another approach to control LPS contamination is production of phage in cell-free extracts.38,62–64 Although it may seem like a technically difficult problem, production of phages in bacterial cells is much more straightforward than for animal viruses, which navigate intracellular membranes and compartments. Cell-free in vitro transcription and translation kits have been used for decades to produce bacterial proteins in vitro. Extending these systems to production of whole, infectious phages has found remarkable success. As noted above, they have been useful in rebooting phages from synthesized genomes.28,62,63
In the United Kingdom, biologics are regulated by the Medicines and Healthcare products Regulatory Agency (MHRA). Thus far, GMP-compliant manufacturing to ensure consistent production and quality control. Controlling contamination risks, especially from adventitious agents, during the production of bacterial cultures and subsequent purification of the medicinal product. This includes measures to prevent contamination with other bacteria, viruses, or cell lines. A robust quality control strategy for seed and cell banks as well as for the final product. The MHRA encourages the development and use of real-time, nondestructive methods for monitoring contamination during the manufacturing process.
Future Directions
With all of the advancements in the development of phages as tools in medicine, agriculture, and food safety, what lies in the future? Turner et al. 81 propose a spectrum of possibilities. On the very practical side, they note that production and characterization of phage repositories would greatly help effective treatment. On the more futuristic side, they propose that phages could be produced on-site as needed. This would involve existing technologies such as cell-free production. They propose a phage printer that would synthesize the genomes of the needed phages, produce the phages in a cell-free system, and then purify and characterize the phages. Given the challenges of hospitals to keep up with developing diagnostic technologies today, on-site phage production does not seem very likely.
A more science fiction-like musing on the future of phage therapy was published by Pirnay. 82 He proposed that DNA from an infected site would be quickly isolated and sequenced to identify the causative agents and their relevant characteristics. Artificial Intelligence algorithms would predict which phages would be best suited to treat the infectious agent(s). As proposed by Turner et al., 81 the therapeutic phages would be produced on-site.
Conclusion
For phage therapy and broader uses of phages to be successful, several changes in thought and understanding at many levels are required. The scientific community has already embraced phages for their usefulness and is rapidly advancing knowledge and technology. Governments are moving toward developing regulatory frameworks to enable phage research to be conducted and products to be implemented. Industries must buy in to the potential of phages to be profitable entities or they will not invest in producing them. Finally, the public must understand and accept phages as beneficial tools. All of this requires effective communication of scientific advancements and education of all stakeholders.
Research and development of phage therapy is a promising avenue for combating the dangerous rise in antibiotic resistance and may eventually become a standard medical countermeasure to complement or replace antibiotics. Biosafety professionals and Institutional Biosafety Committees should become familiar with the technology, risks, and associated safety/regulatory considerations to safely oversee the research.
Authors’ Contributions
P.G. wrote the majority of the article. D.E. wrote the abstract as well as portions of the introduction and sections pertaining to biological safety and environmental and regulatory considerations. D.E. originally conceived of the article and served as project leader, editor, and corresponding author. S.S. created the tables and participated in thoughtful discussion and review of the article.
Footnotes
Acknowledgments
The authors wish to thank Stephanie Pyle and Mark Fields for assistance with article preparation.
Funding Information
No funding was received for this work.
Authors’ Disclosure Statement
P.G. works as a part-time contractor and IBC Vice Chair for Advarra. S.S. and D.E. are employed by Advarra, a for-profit clinical research compliance company. Advarra did not provide any funding for this work.
