Abstract
Introduction:
Public health logic models divide interventions into three categories: inputs, activities, and outcomes. Similarly, biosafety programs can be described using this same logic model by describing inputs in the form of program staffing and budget; program activities in the form of risk assessments, routine surveillance, training, incident response, and other services; and outcomes in the form of reported exposures, injuries, compliance, finances, and client satisfaction.
Methods:
Described here is a systematic means of collecting and displaying biosafety program activity data monthly so that the organizational leadership and associated safety committees can readily grasp the scope of various activities undertaken by the biosafety program to support the broader organizational goals.
Discussion:
The simplified public health logic model affords guidance on inputs, activities, and outcomes that biosafety programs can use to their advantage. The importance of succinctly reporting the data describing activities can inform and educate upper management and other stakeholders regarding the resources needed to avoid injuries and illnesses and maintain compliance.
Conclusion:
Within the biosafety profession, on a good day, “nothing bad happens,” so conveying the message of the value of prevention can be challenging. By capturing and readily presenting data that describes the efforts of a biosafety program, individual programs can articulate their needs for staffing and resources more successfully, and the profession stands a better chance of maintaining strong administrative and stakeholder support.
Introduction
In their simplest form, public health logic models divide interventions into three categories: inputs, activities, and outcomes.1–5 Biosafety programs can be described using a similar simplified logic model by describing inputs in the form of program staffing and budget (that is, the number of full-time equivalent staff members and the total amount of money needed for the program’s budget allocation); program activities in the form of risk assessments, routine surveillance, training, incident response, and other services that are provided to the organization; and outcomes in the form of reported exposures, injuries, compliance, finances, and client satisfaction (Figure 1). While program inputs and ultimately their outcomes can often be captured by various institutionally focused data systems, such as budgets and workers’ compensation insurance claims, safety program activities become a bit more obscure because oftentimes the data are retained within the operational unit, buried in a computerized database or shadow system. Described within this article is a process developed by the authors to systematically collect and display biosafety program activity data monthly so that institutional leadership and associated safety committees (e.g., Institutional Biosafety Committee [IBC]) can readily grasp and appreciate the scope of the various activities undertaken by the biosafety program to generate the desired outcomes. Furthermore, the activities data provides a comprehensive snapshot of the proactive measures and safeguards in place to mitigate risks, prevent incidents, and protect the organization’s reputation, ultimately ensuring a safe working environment and minimizing the likelihood of accidents, exposures, and adverse media attention. The data collected have also served as the basis for the creation of a novel program prospectus that shows activities as compared to larger organizational trends such as growth in the research enterprise and facility infrastructure, which has served to maintain robust support for the biosafety program. 6 The approach described also holds the potential to serve as a possible common data collection mechanism across the biosafety profession to facilitate meaningful comparison and benchmarking. We have strong anecdotal evidence from invited peer reviews of biosafety programs that support the importance of capturing and articulating the program’s information as well.
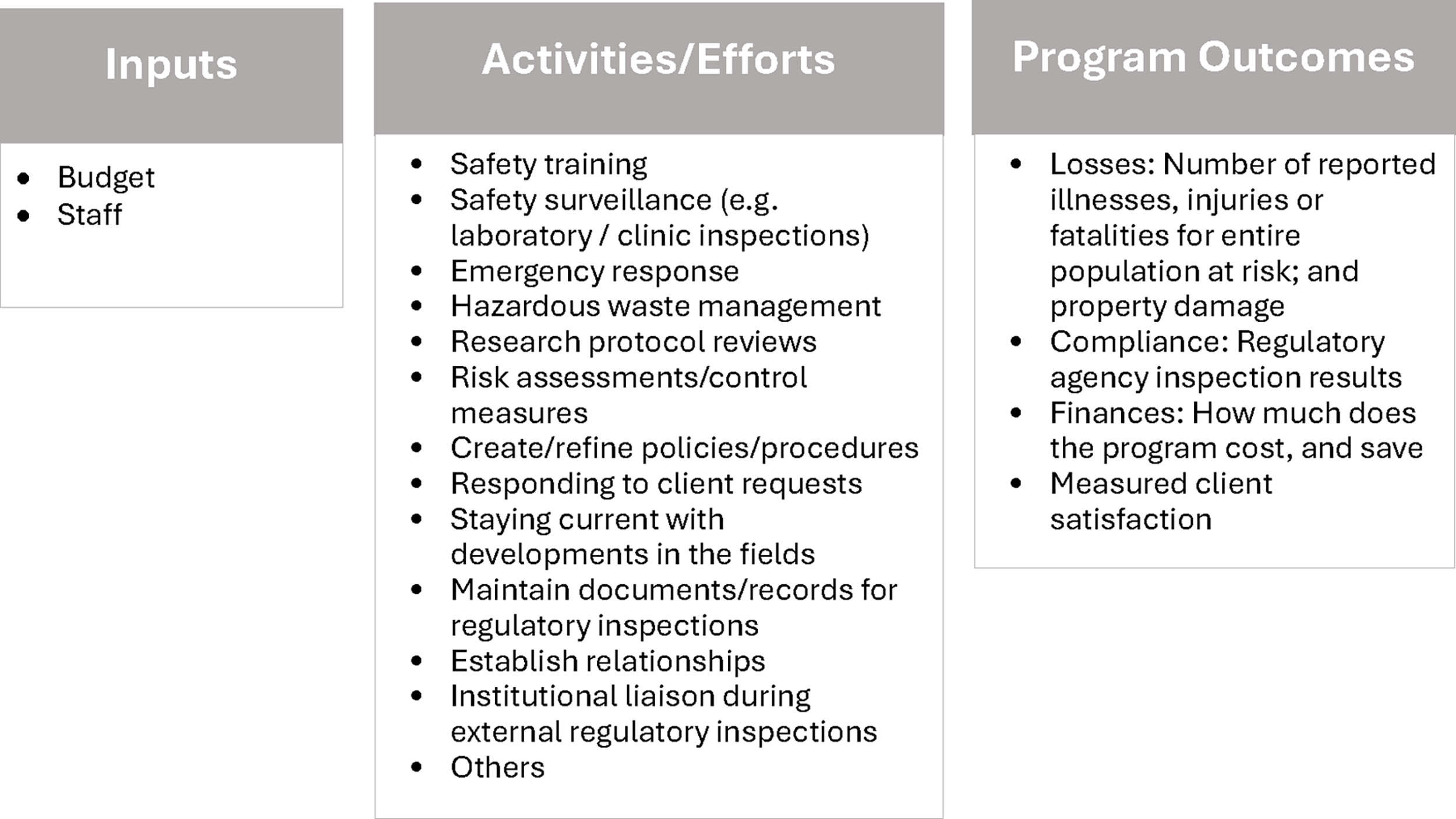
Simplified Biological Safety Program Logic Model: Inputs and Outcomes.
Methods
To begin to succinctly capture the day-to-day activities that are carried out by the (The University of Texas Health Science Center at Houston) biosafety program, the initial efforts involved a series of unit-focused discussion sessions with the biosafety staff wherein a comprehensive list of routine work activities was assembled. Once this master list was created, a set of general categories became readily evident. Equally important were discussions regarding which activities could be objectively quantified and would resonate with stakeholders outside of the program (e.g., risk management, legal affairs, compliance).
From these initial focus group discussions, six broad categories of activities were identified: surveillance activities, occupational and environmental monitoring, investigations (e.g., incidents, injuries, illnesses, exposures), incident (emergency) response, administrative reviews (e.g., risk assessments for work with biological materials), and training. These groupings were then shared with the IBC membership during a convened meeting, where they were defined and explained, and the committee members’ endorsement was obtained for these reporting measures. Following endorsement of the IBC, these groupings subsequently became the foundation for developing the biosafety program’s monthly activity report.
There are a variety of data management software systems currently available on the market that target health and safety program operations, which can be used to help collect and organize program data; however, some organizations have opted to develop their own homegrown data management systems. At (The University of Texas Health Science Center at Houston), the decision was made several years ago to implement a commercially available software system. Within this web-based data management system, staff members can collect and record a myriad of routine activity data, including laboratory safety surveys and associated reports, biological agent risk assessment protocols, biological agent inventories, laboratory equipment inventories, and safety training records. In addition, all reported incidents, including near misses, injuries, illnesses, and exposure events, and the incident investigation summary report communicating investigation findings, corrective actions, and preventative measure recommendations, are also documented and tracked within the database system. Reports can be queried from the system to provide a summary, monthly or during other desired time periods, of the cumulative activities and efforts of the entire program staff.
Importantly, general definitions and guidance for each of the IBC-endorsed categories were developed to provide clarity and consistency of data collection and reporting between program staff members. Each biosafety staff member was then asked to maintain a simple monthly log, wherein data about their routine work activity was recorded. (Note: to this day, the notion of the time frame for each activity is still being debated and hopefully will be resolved in the future without becoming an overburdening data collection exercise—more on this topic in the Discussion section).
At the end of each month, the biosafety program manager compiles data from each biosafety program staff member and enters the information into a succinct one-page report. Collectively, this activity report essentially “tells the story” of the activities performed by the biosafety program each month. This report is then shared with departmental leadership and the IBC each month to serve as a program summary. In addition, the report has also been shared with various regulatory agencies during inspections and site visits involving biosafety aspects. The Biological Safety Program monthly activity summary has also been shared in Biological Safety Officer training courses offered by (The University of Texas Health Science Center at Houston) and during ABSA International-sponsored pre-conference courses at the annual Biosafety and Biosecurity Conference administered by the institution’s biological safety program staff.
Over the 25 years of use, the six categories have persisted. However, the subcategories have changed as the program grows, and responsibilities shift to or from other departments for better alignment with changing stakeholder needs and expectations. Lab surveillance activities were documented in the first few years of data collection. Still, it was not until 6 years after implementation that non-compliance items were documented, including the department responsible for correction.
In 2011, protocol amendments were added to administrative review and later separated further to indicate administrative and significant amendments. The latter involved changes to the research, which increased safety precautions and required review and approval at the IBC meeting. In 2017, protocol reviews were subcategorized as screenings (exempt from IBC review), full review (including human gene therapy, recombinant/synthetic DNA, and infectious agent work), and conditionally exempt, which applied mostly to clinical research at a lower biosafety level. These changes recognize the various levels of complexity of research being conducted, which forecasts the breadth of knowledge required for biological safety professionals. The 25-year prospectus will further illustrate the complexity of protocols and provide new data sorting protocols by biosafety level, NIH Guidelines categories, and cross-reference with animal welfare and human subject protection committees.
In 2018, as the clinical enterprise of our institution grew, primary responsibility for clinical surveillance and training activities was reassigned to the newly formed Hospital and Clinic Safety Program. We realized that, out of necessity based on the incredible growth of the enterprise within our institution, a stand-alone program was needed to oversee the health and safety of personnel working and learning in this high-risk environment.
Results
Example criteria contained within the monthly activities and efforts data collected by the (The University of Texas Health Science Center at Houston) biosafety program are displayed in Figure 2, which is shown within the actual spreadsheet used by each Biosafety Program staff member to track these activities and efforts over the course of time during the fiscal year. We also recognized and appreciated that biological safety programs can vary greatly inside and outside of academic institutions. For example, some biological safety programs may be solely focused on containment laboratories and management of Select Agents and Toxins. Likewise, some biological safety programs may include biosafety cabinet certification within their portfolio of services. Despite the targeted focus of the biological safety program, a monthly activities summary is modified for any program with tracking of the efforts taken by the program during the year. The department possesses approximately 25 years of these data and assembled a subset of the data to depict a 10-year prospectus that highlighted trends affecting the program within the context of institutional dynamics. 6 A 25-year prospectus is in the process of being assembled and will be shared in the future. Of special note is that all the authors of this work were previously, or are currently, involved with the oversight of the (The University of Texas Health Science Center at Houston) biosafety program and contributed actively to the creation and evolution of the monthly activity report and this article. In addition, all have completed the requirements to be designated as a Certified Biological Safety Professional (CBSP ABSA) by the American Biological Safety Association—International (ABSA). Furthermore, during courses taught at various conferences and training events, the activity report described here is commonly the most sought-after document by attendees.
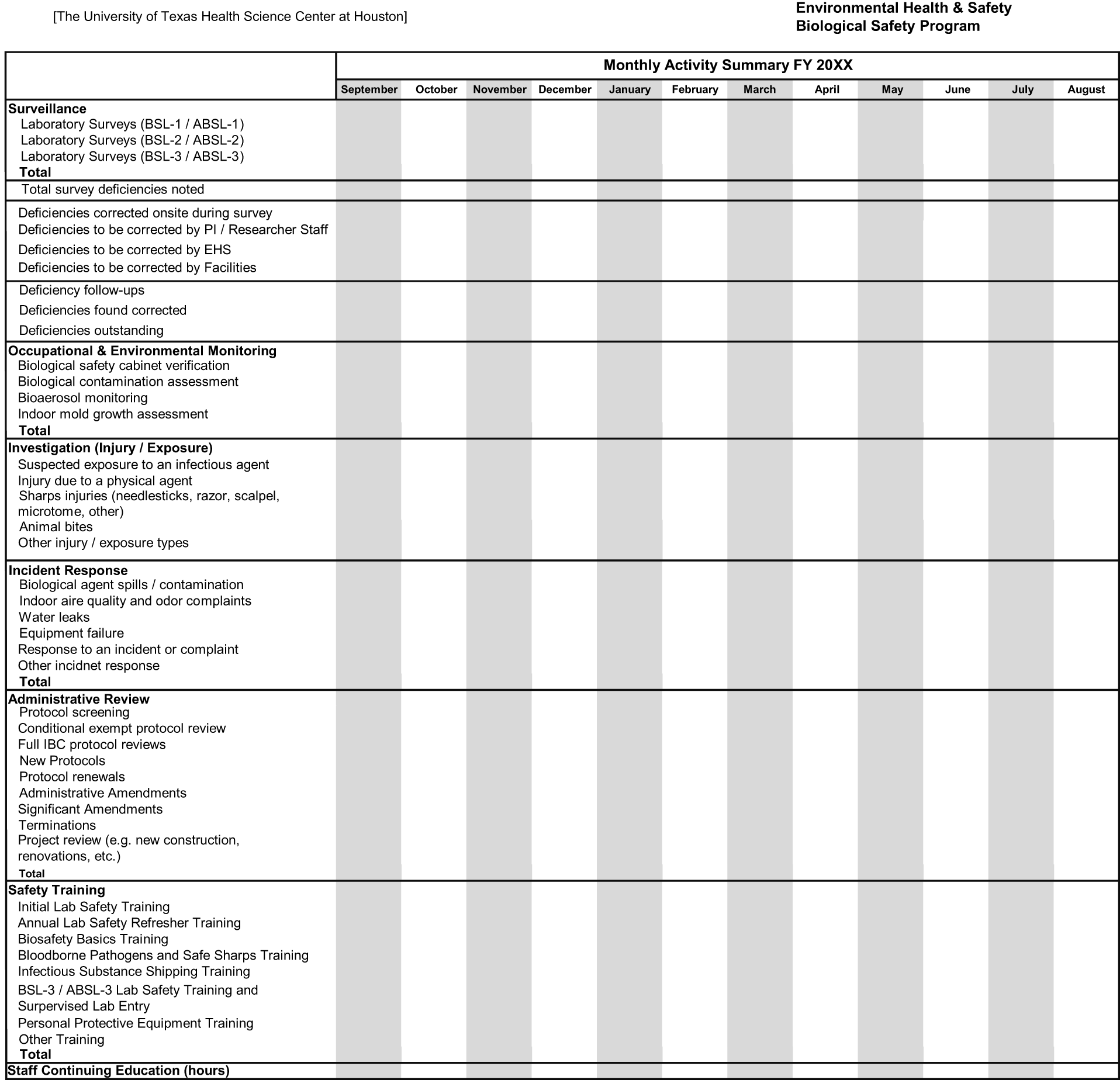
Example Biosafety Program Activities Report Data Collection Worksheet provided by The University of Texas Health Science Center at Houston. Each biosafety specialist records their activities on this worksheet, which the program director compiles on a monthly basis into a concise report.
A review of the monthly program activity summary is a standing agenda item for each IBC meeting. This presents a chance for the committee members to grasp what the program does day-to-day and to ask about trends or outliers. It also allows the biosafety program to obtain endorsement and support for priorities and initiatives based on data-driven decision-making.
Ultimately, the program data collected over the entire course of the fiscal year is summarized and folded into a larger departmental annual report that depicts the activities carried out by all the departmental units. In addition to the biosafety program activities, the annual report contains information about related programs in chemical safety, radiation safety, occupational and fire safety, environmental protection, hospital and clinic safety, risk management and insurance, and employee health (also referred to as occupational health) activities within the organization. 7 While this organization is specific to our institution, we recognize that many biological safety programs are separate programs outside of Environmental Health and Safety (EHS). While some biosafety programs may exist as stand-along units within certain organizations, it has been our experience, based on 49 university peer reviews and over 15 years of professional development training courses for ABSA International, that the vast majority of biosafety programs within the academic, research, and biotech environment exist within some form of an EHS paradigm or must necessarily coordinate with other safety specialties regularly, as rarely does a biosafety issue reside solely within the purview of the potentially infectious agent itself. Hence, the data collection mechanism described in this article is germane to the biosafety profession regardless of their organizational alignment.
Discussion
Successful biosafety programs function largely in the realm of prevention; hence, conveying the story of what amount of work is needed to help prevent negative outcomes or “make nothing bad happen” is a daunting challenge. Based on the professional experience of the authors, as well as feedback from a long-standing professional development course that has been provided each year since 2008 at ABSA-International’s annual biosafety and biosecurity conference, biosafety programs typically struggle with telling this story of prevention through data. The simplified public health logic model affords guidance on inputs, activities, and outcomes that biosafety programs can use to their advantage. The importance of succinctly reporting the data describing activities can inform and educate upper management and other stakeholders regarding the resources needed to avoid injuries and illnesses and maintain compliance.
A recurrent question that arises amongst various administrators is “What does Biological Safety actually do?” which is reflective of the challenge of telling the story of “making nothing bad happen.” So, in a parallel effort, we began capturing data systematically that described the staff’s daily activities. In this pilot study, the authors utilized the ecological momentary assessment (EMA) research technique to record the work activities being undertaken by biological safety personnel during a typical 8-hour workday to augment the Brown et al. models 8 (which focused on staffing and resourcing needs compared to statistically significant variables such as net assignable square footage) by addressing worker productivity. Practicing biosafety professionals were evaluated one day per week over a 5-week period (for a total of 5 workdays, all weekdays) and asked to report the type of work conducted during the normal 8-h work shift. The activities were predefined and agreed upon in the biological safety program. The top five activities for the biological safety program were attaining professional knowledge (21%), safety surveillance activities (18%), protocol and plan review (16%), delivering training (10%), and responding to client service requests (10%). 9 This data provides insights to explain exactly what biosafety program staff do.
A common issue encountered by biosafety programs that represents a tremendous opportunity to demonstrate goodwill value is when the typical administrative question arises: “what does the biosafety program cost?”, however, what we want to be able to show is “what is the biosafety program worth?” Goodwill value is an accounting term used to quantify the monetary value of intangible assets of an organization. 10 Goodwill value can be illustrated by our metrics and institutional data by determining the percentage of extramural research expenditures that involve risk assessment by the Biological Safety Program and IBC review. When presented with this compelling statistic, the question about the cost of program operation was put into perspective, and the reality of the value of the program became evident. 11
A major challenge in attempting to capture the value of prevention is the peril of “overmeasurement,” where staff are overburdened with data collection, so much so that the necessary work is not being accomplished. So, some consensus regarding which measures are meaningful and resonate with upper management is critical.
Over the years, the ability to instantly respond to administrative inquiries and challenges with data describing what the biosafety program does has been valuable and has resulted in strong and continuous support of the program. The collection of this data can also identify tasks that need to be completed but are not being done due to a lack of resources. These productivity gaps can also be a liability to the university. Biosafety peers with whom we have shared this technique have reported similar success.12,13 This data has also informed the development of a program prospectus, which illustrates the value of biosafety initiatives by comparing program activities to broader organizational trends, such as research growth and infrastructure expansion, thereby demonstrating the program’s necessity and strategic importance.
Conclusion
Biosafety programs exist to assist individuals in going home as healthy and as safe as they arrived at work. Within the profession, on a good day, “nothing bad happens,” so conveying the message of the value of prevention can be challenging. By capturing and readily presenting data that describes the efforts of a biosafety program, individual programs can articulate their needs for staffing and resources more successfully, and the profession stands a better chance of maintaining strong administrative and stakeholder support.
Authors’ Contributions
Each author contributed equally to the creation, writing, and editing of this article.
Footnotes
Acknowledgments
The authors, all of whom served in a leadership capacity over biosafety program operations at The University of Texas Health Science Center at Houston over the past 25 years and contributed to the assemblage of this program activity data, wish to express their sincere appreciation to all the staff of the The University of Texas Health Science Center at Houston biosafety program, current and past, for their contribution to the datasets described here.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No financial support was provided for this article.
