Abstract
Introduction:
Steam sterilization has been used for decades to effectively kill microbial contaminants in a variety of medical and commercial settings. One of the most critical aspects of safe operations in biosafety level 3 biocontainment laboratories (BSL-3) is the effective inactivation of biological select agents in the waste generated in these environments. The Instituto Nacional de Enfermedades Virales Humanas “Dr. Julio I. Maiztegui” (INEVH, Pergamino, Argentina) is an institute that offers epidemiological surveillance, production of biological reagents, and production of biologicals for human use and studies of reservoirs and vectors. Some of the activities need to be done in a BSL-3 that provides biocontainment, ensuring that the materials are decontaminated before they leave the facility. The objective of this study was to design and validate a decontamination procedure for biological waste from the BSL-3 facility that guarantees steam sterilization processes.
Methods:
The amount and the distribution of biological waste into the autoclave and other physical parameters were defined and evaluated by calculating lethalities.
Results:
We evaluated autoclave basic factory programmed cycles, and it was concluded that the sterilization autoclave cycle was not efficient for decontamination of waste. A new simulated load distribution had to be defined.
Discussion:
The results demonstrated that autoclave factory default settings can be inadequate for sterilizing highly infectious waste, depending of types of waste, such as animal carcass and animal bed waste.
Conclusion:
These results of the validation process can set the standard to the design of waste management protocols to ensure effective treatment of highly infectious biological waste.
Introduction
The term
Within Biosafety Level 3 (BSL-3) and Biosafety Level 4 (BSL-4) containment laboratories, it is imperative that all materials leaving the facility are effectively decontaminated to eliminate the presence of biological materials. 2 Usually, all waste is decontaminated by steam sterilization using an autoclave. Because there are no regulations for biological and biomedical waste management produced in a BSL-3 facility in Argentina, safe and reliable methods for it are not only a legal necessity but also a social responsibility. 3
The Instituto Nacional de Enfermedades Virales Humanas “Dr. Julio I. Maiztegui” (INEVH, Pergamino, Argentina) works on epidemiological surveillance and laboratory diagnosis of arbovirus, arenavirus, and hantavirus diseases. It is also responsible for the production of the live attenuated vaccine named Candid #1 against the Argentine hemorrhagic fever. Some of the activities at the INEVH are performed in BSL-3 laboratories. Trash and biological waste must be decontaminated through a double door pass through autoclave at the time of leaving the facility to minimize risks of exposure of to both the operator and the environment. In this sense, it is essential to assure that all waste is effectively decontaminated prior to its removal from the facility. 1 This may be accomplished by exposing the waste to steam at different pressures and temperatures for a certain period of time by using an autoclave. In general, this equipment has basic factory-programmed cycles that need to be validated, 2 including: variability in thermal mass and density, particularly critical when sterilizing large amounts of animal tissue or carcasses; high water content; and differences in autoclave design, among others.
To address the risk of sterilization failure, the INEVH conducted the validation of the steam sterilization procedures from the BSL-3 by using mock biological waste that included animal carcasses and animal waste. The objective of this study was to design and validate a decontamination procedure for biological waste from the BSL-3 facility that guarantees steam sterilization processes.
Materials and Methods
Equipment
The study was conducted in a double door pass through autoclave, trademark Mazden, model AVD-0432-000, serial number 222, San Martín, Buenos Aires, Argentina, serving as a barrier for the exit of material, trash, and biological waste from the BSL-3 facility. This autoclave is able to configure the loading and unloading doors according to the circulation. Contaminated waste is loaded on the BSL-3 side, and once treated, it is unloaded on the clean side and then packaged for transport and disposal. The doors cannot be opened simultaneously, which prevents cross-contamination between the autoclave’s clean and dirty sides. Steam intake and electrical and mechanical infrastructures are located on the clean side of the waste-handling area to facilitate autoclave maintenance.
Validation Parameters
To develop and validate the biological waste decontamination procedure, the following parameters were evaluated: temperature, time, and lethality (F0).
The F0 value of a saturated steam sterilization process is a unit of lethality and is a measure of the microbial inactivation of a heat sterilization process. It is defined as “the equivalent in minutes at 121ºC of all heat considered with respect to its capacity to destroy spores or vegetative cells of a particular organism” and allows the comparison of lethal effects at various temperatures.
4
The autoclave charts provide temperature and F0 values of each cycle. Thermocouples (TC) were used to measure temperatures in 14 places within the biological waste. Each of them was associated with a biological indicators (spores of
Autoclave Cycle and Sterilizations Parameters
Initially, the physical parameters were recorded. All TC used were calibrated. Different temperatures were selected, and TC measurements were compared with the standard. The BSL-3 autoclave decontamination cycle was validated using simulated loads consistent in quantity and composition with items expected from animal studies. These items included guinea pig frozen carcasses and saturated animal beds (soaked with at least 1 liter of water).
The simulated load test had a TC associated to a BI in different places into 2 double autoclave bags of approximately 14 kg. Each one contained 11.5 kg of wet sawdust with 1 liter of water (to simulate urine) from guinea pig beds and 3 frozen 0.65-kg guinea pig carcasses each. Each carcass contained 3 BI. One BI was placed in the abdominal cavity among the abdominal viscera, 1 BI was placed within a deep incision in the thigh along the shaft of the femur, and 1 BI was placed in the head cavity drilling the cranium. Each run contained 14 BI.
The load was defined by the quantity and distribution of biological waste simulating the conditions of greatest demand that could be presented in laboratory processes.
Table 1 shows the initial factory default settings tested for waste decontamination. Its parameters were 3 vacuum-vapor pulses and an exposure temperature of 121 ºC for 60 minutes.
Initial Factory Default Settings for Waste Decontamination.

After running the test cycle, the BI with each TC was incubated in an appropriate medium for 7 days at 56 ºC to evaluate the survival of microorganisms.
The ability of the sterilization process to reliably destroy any bio load on or within the load were evaluated by overkill destruction of a high concentration of spores of a resistant biological indicator; for example, a process with a lethality of F0BI > 12 minutes: a process that provides at least a 12 log reduction of BI microorganisms having a minimum D (the time required to inactivate 90% of the cell population or to reduce the microbial population to one-tenth of its original number) value of 1 minute.
It was defined as a validation criteria that all BI associated with each TC were negative during 3 decontamination runs performed on different days. 6
Results
At the beginning, the TCs were calibrated in order to know the thermal profile into the autoclave chamber. Two points were evaluated, 1 in the autoclave chamber and the other in the drain.
The thermal chamber profile was then performed with an empty chamber, and the heat distribution observed was homogeneous (Figure 1).
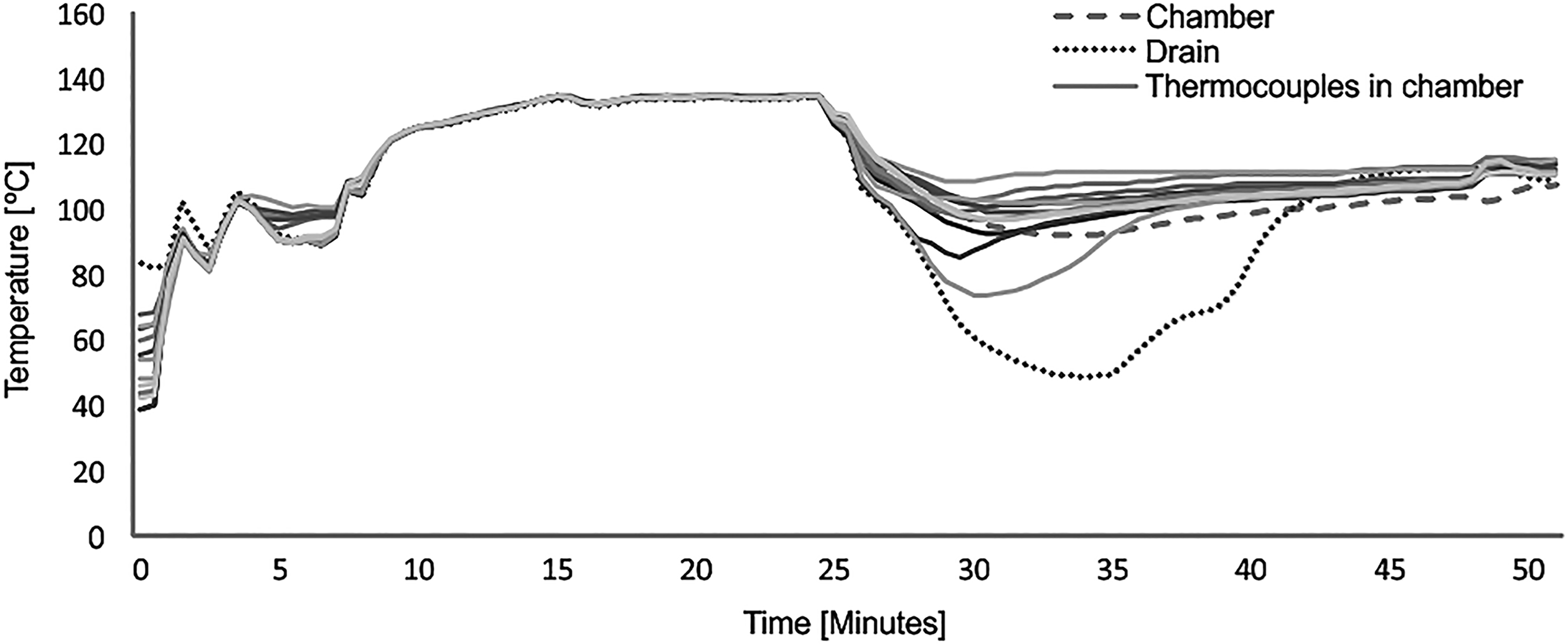
Distribution of heat in the chamber and into the drain of the empty autoclave.
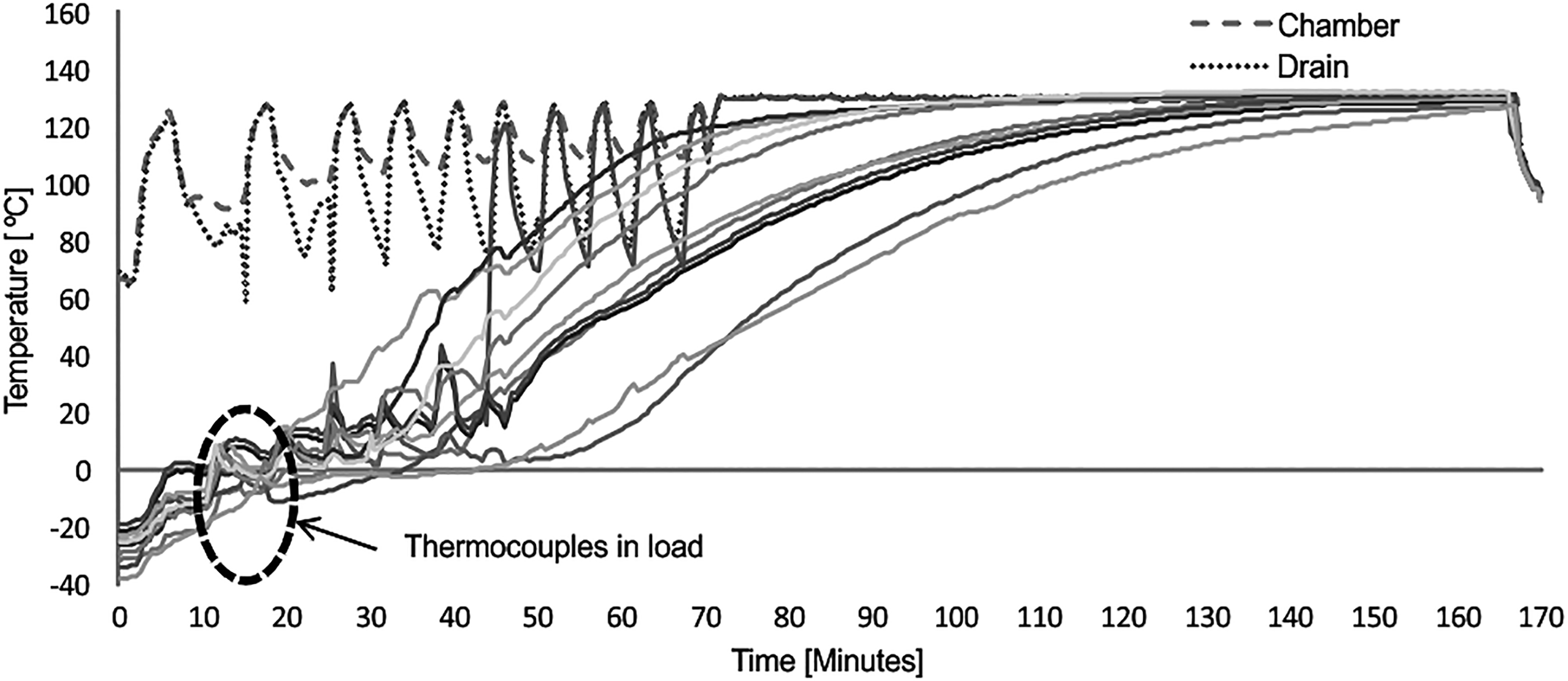
For each test run, 14 TC were added in the biological waste material that included a guinea pig carcass and bed waste to record the time/temperature profile at different locations within the load. After the evaluation of each BI, it was concluded that 6 out of 14 were positive; consequently, the decontamination could not be assured.
Heat distribution was not homogeneous, as is evident in the different temperature profiles shown in Figures 2 and 3. Although, the F0 value associate to each BI was very low, and remarkably at some point, the F0 value was zero. When we analyzed and identified which TC was associated to a BI positive, it was determined that they corresponded to those located in or close to the frozen animals. It was concluded that the sterilization autoclave cycle was not efficient for decontamination waste. A new simulated load distribution was defined: one decontamination cycle for guinea pig carcass (animal carcass cycle) and another one for the animal waste (animal bed cycle).
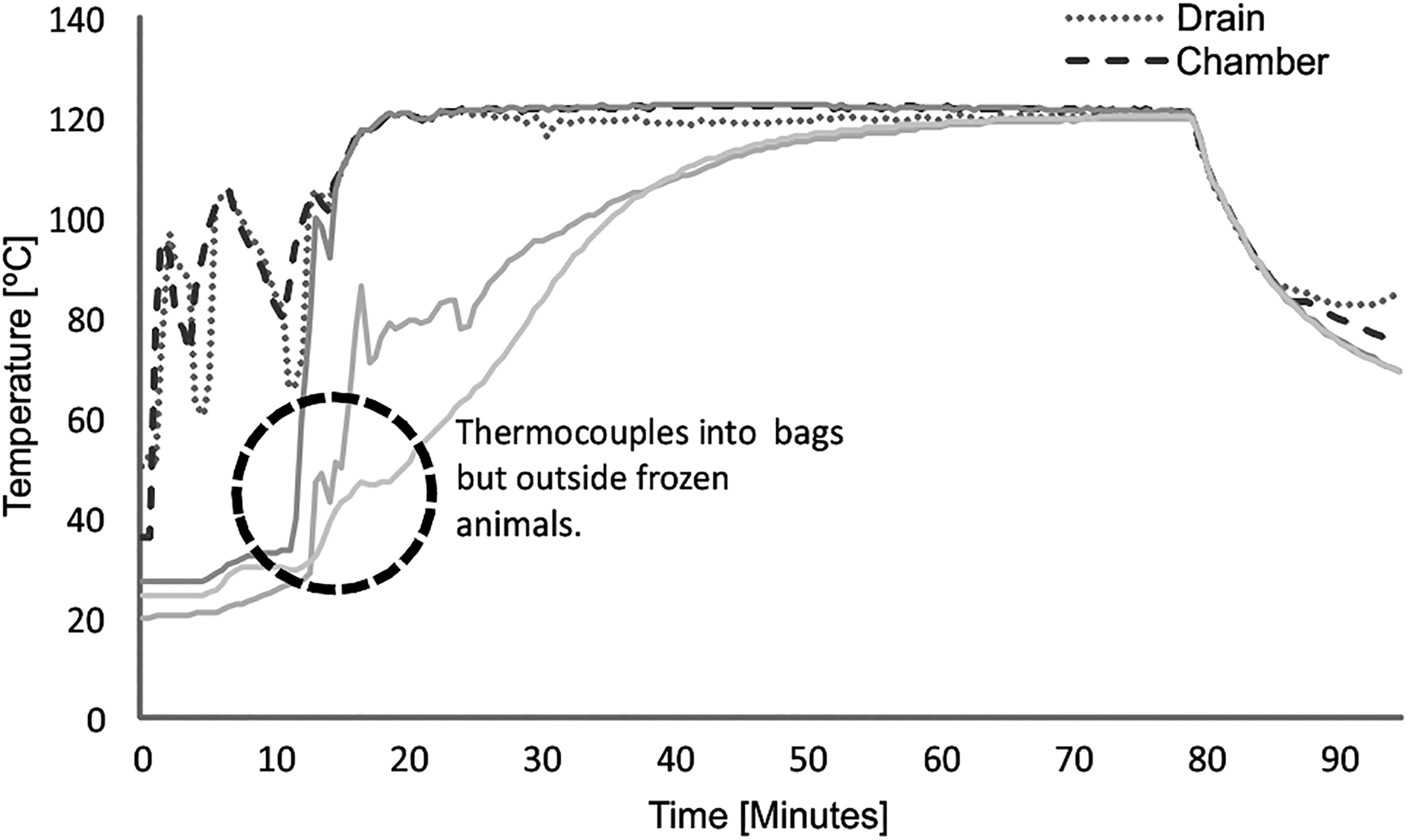
Temperature of different point outside frozen animals. It shows the profile of the temperature from the 5 Thermocouples used in the cycle (drain, chamber, and 3 inside the bags but outside frozen animals).
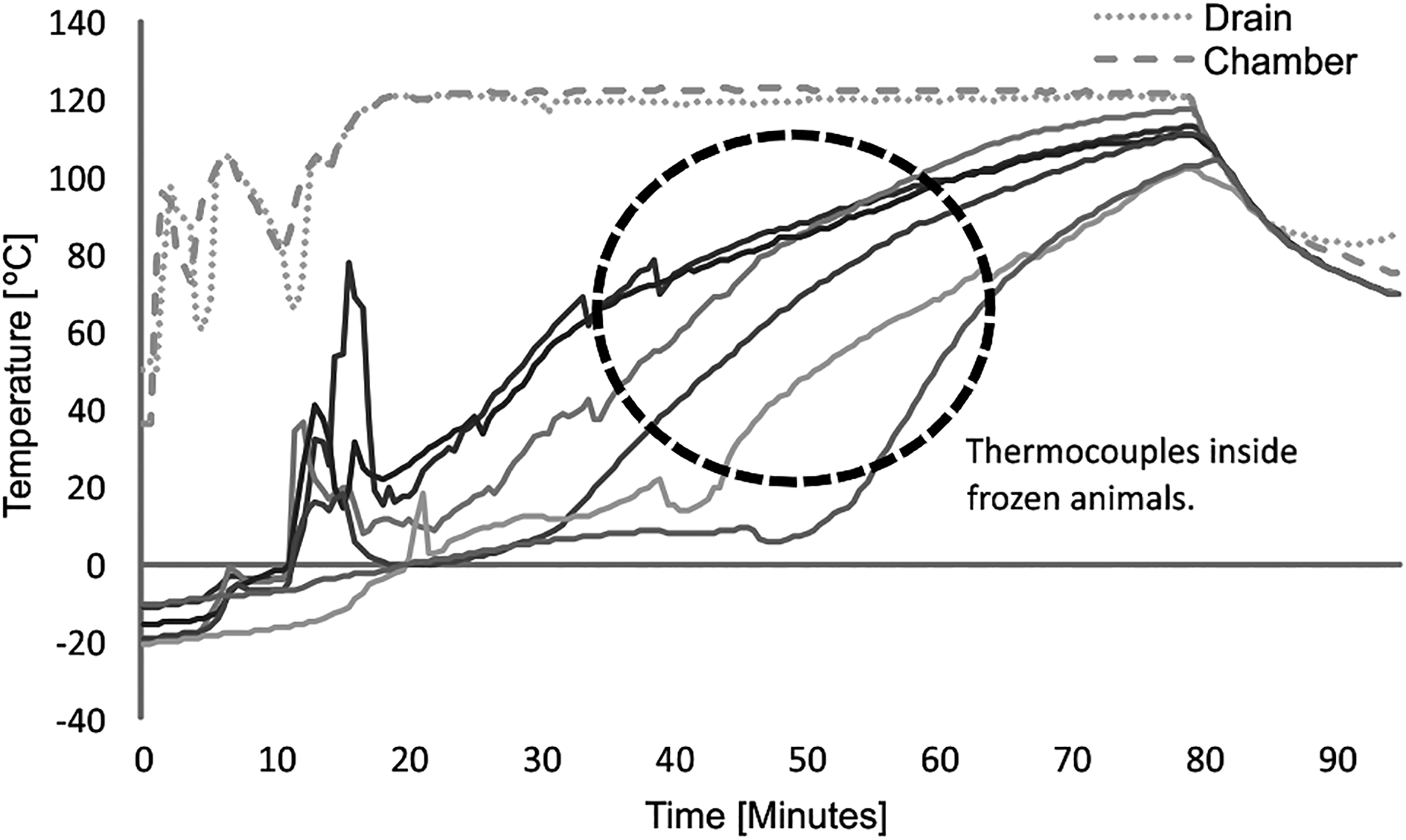
Temperature of different inside frozen animals. It shows the profile of the temperature from 11 thermocouples used in the same cycle (drain, chamber, and 9 inside the bags and frozen animals).
New runs were repeated on simulated loads multiple times with different sterilization cycle parameters (see Table 2).
Parameters Set After Cycle’s Optimization for the Adequate Waste Decontamination.a
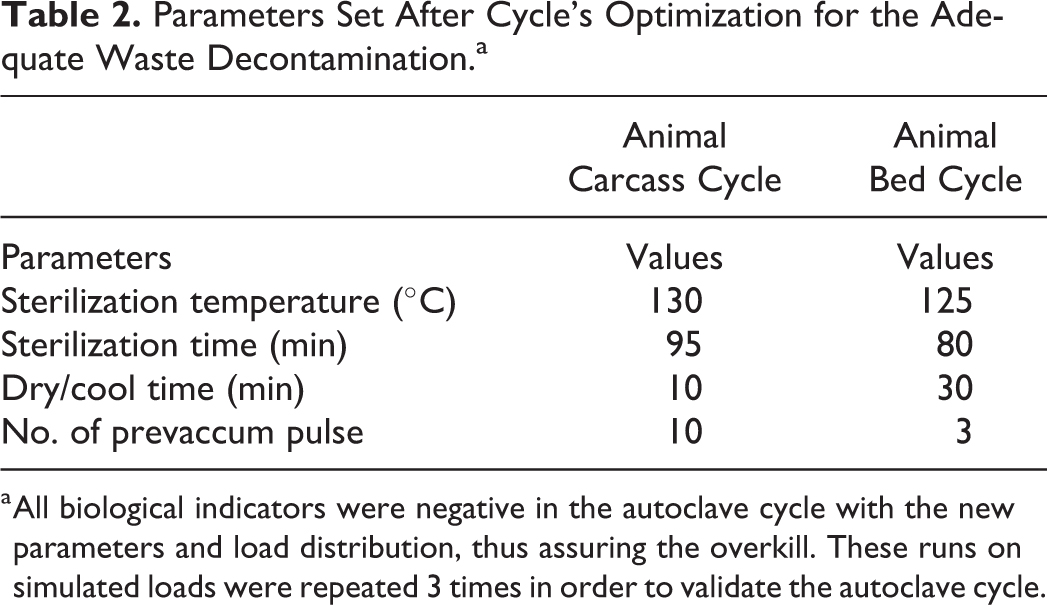
a All biological indicators were negative in the autoclave cycle with the new parameters and load distribution, thus assuring the overkill. These runs on simulated loads were repeated 3 times in order to validate the autoclave cycle.
Discussion
Reliable lethality of sterilization cycles used for all types of waste from BSL-3 and BSL-4 laboratories is a critical component for safe operation and is crucial to protect public, occupational, and environmental health. A number of challenges must be resolved when using steam sterilization to process these unique types of waste, water content, and initial temperature, particularly in carcasses and animal waste. 2,3
A preliminary risk assessment identified 1 main risk associated with waste disposal and autoclave use: failure to effectively sterilize waste. In this regard, we conducted a study to design and validate a decontamination procedure for biological waste from the INEVH BSL-3 facility that guarantees the steam sterilization process.
The results of the experiments demonstrated that autoclave factory default settings can be inadequate for sterilizing highly infectious waste, depending on types of waste such as animal carcass and animal bed waste, the way that waste loads are packaged prior to treatment, quantities, and initial temperature.
As result for the initial decontamination cycle (Table 1, Figures 2 –4), it was observed that the lethality calculated at each point shows a big difference between the different points in the load. These demonstrated great variability of temperatures despite having a properly functioning autoclave that showed uniformity with an empty chamber (Figure 1), which proves that factory default settings are inadequate for sterilizing waste. In this case, it was not enough to ensure overkill. F0s were zero, especially in those TCs that were in the animal carcass (peritonea and cranial cavity; Figure 4).
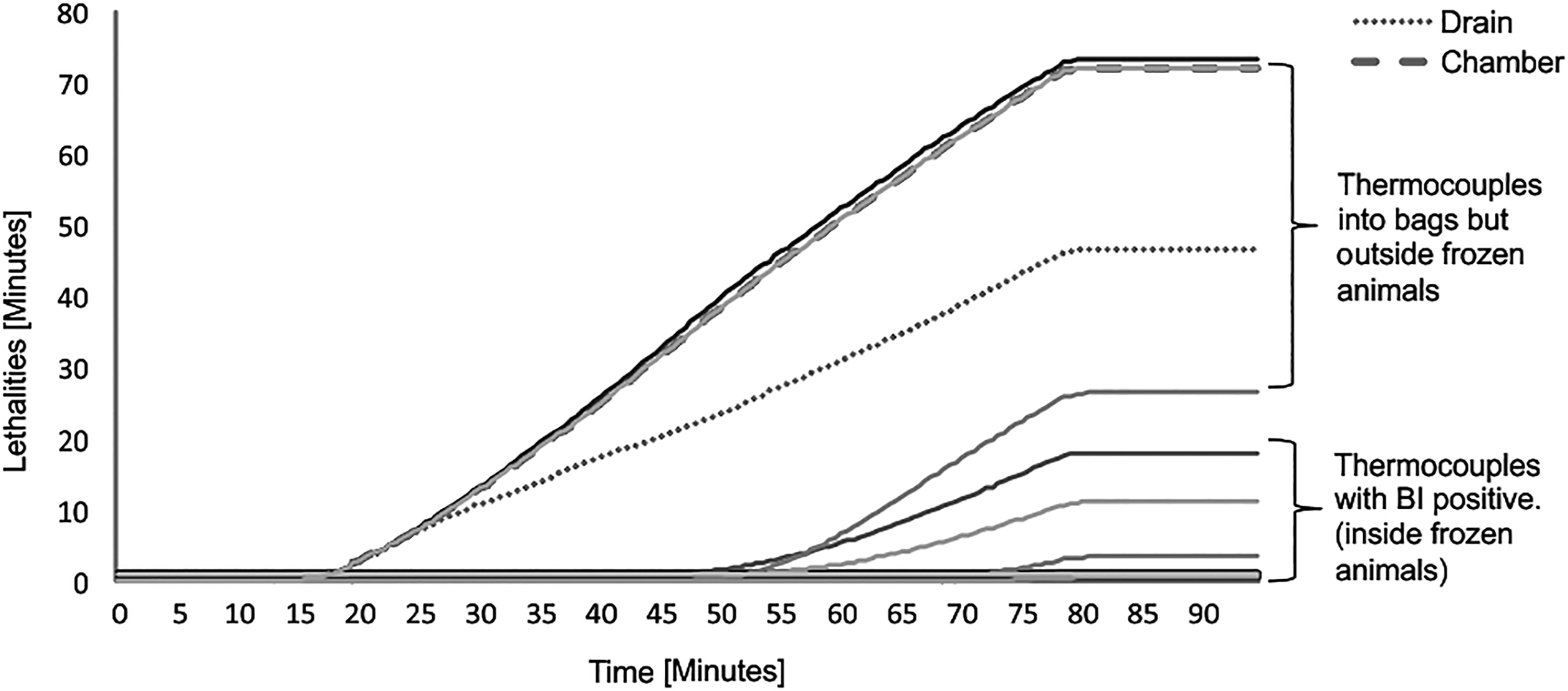
F0 value calculated for each biological indicator on different points into the load. It shows the values of F0 from each thermocouples containing the biological indicator.
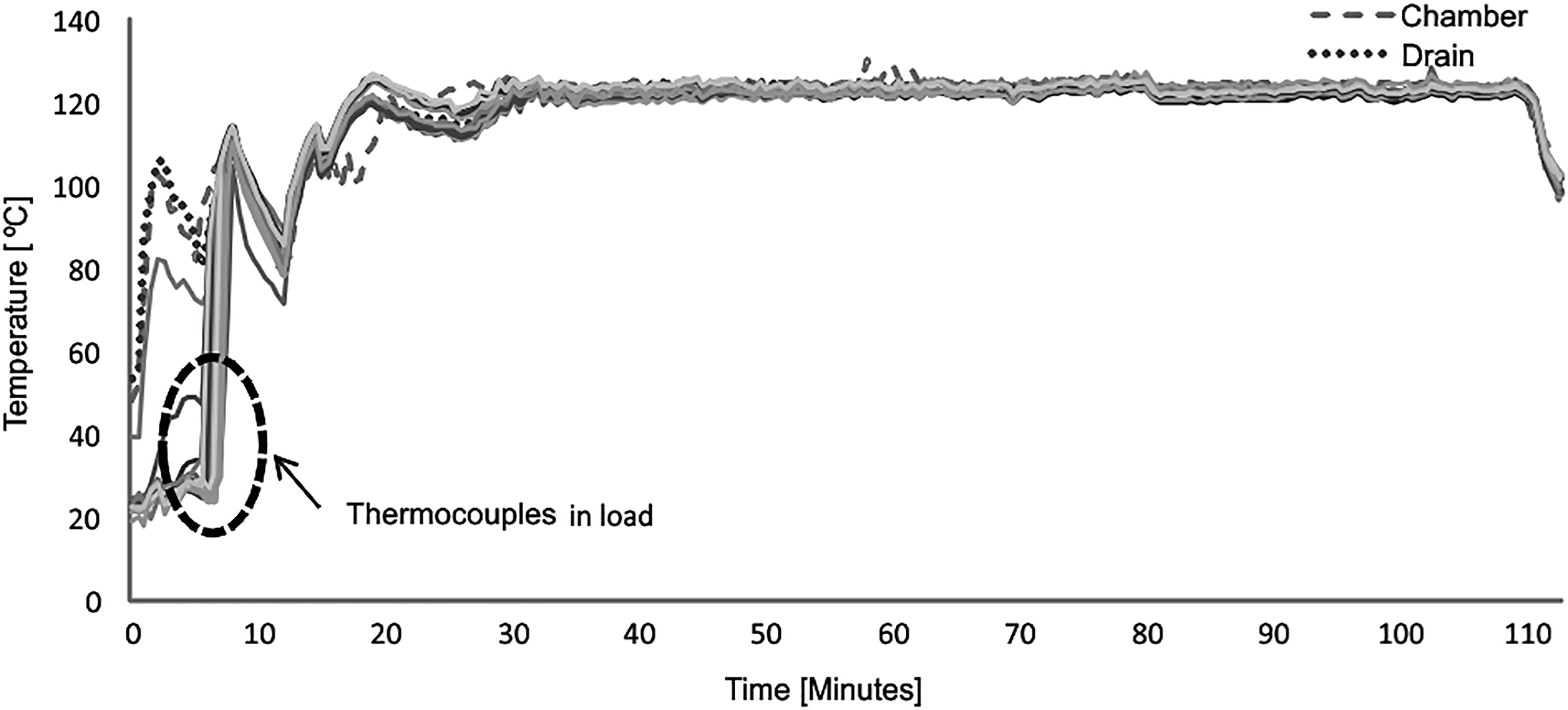
Consequently, new autoclave cycle procedures were developed in which waste was divided in 2 different runs: 1 of them only with animal carcass and the other 1 with animal bed (Table 2). In both cases, the temperature, the sterilization time, and the number of prevacuum pulse were increased. The optimized cycles and parameters for sterilizing waste provided that the conditions were sufficient to guarantee a safe decontamination of the waste. As seen in Figure 5, the TC show different slopes until the required temperatures are reached. This uneven increase is due to the characteristics of the load. Due to the locations of the TC in regions of the carcasses with greater difficulty of steam penetration, the sterilization temperature was reached later. Figure 6 shows that the lethalities in the load, as described for temperatures, are also variable. In any case, all have exceeded the overkill sterilization conditions.
Temperature of different points into the load for animal carcass cycle. It shows heat distribution in each thermocouple using the new parameters for animal carcass cycle.
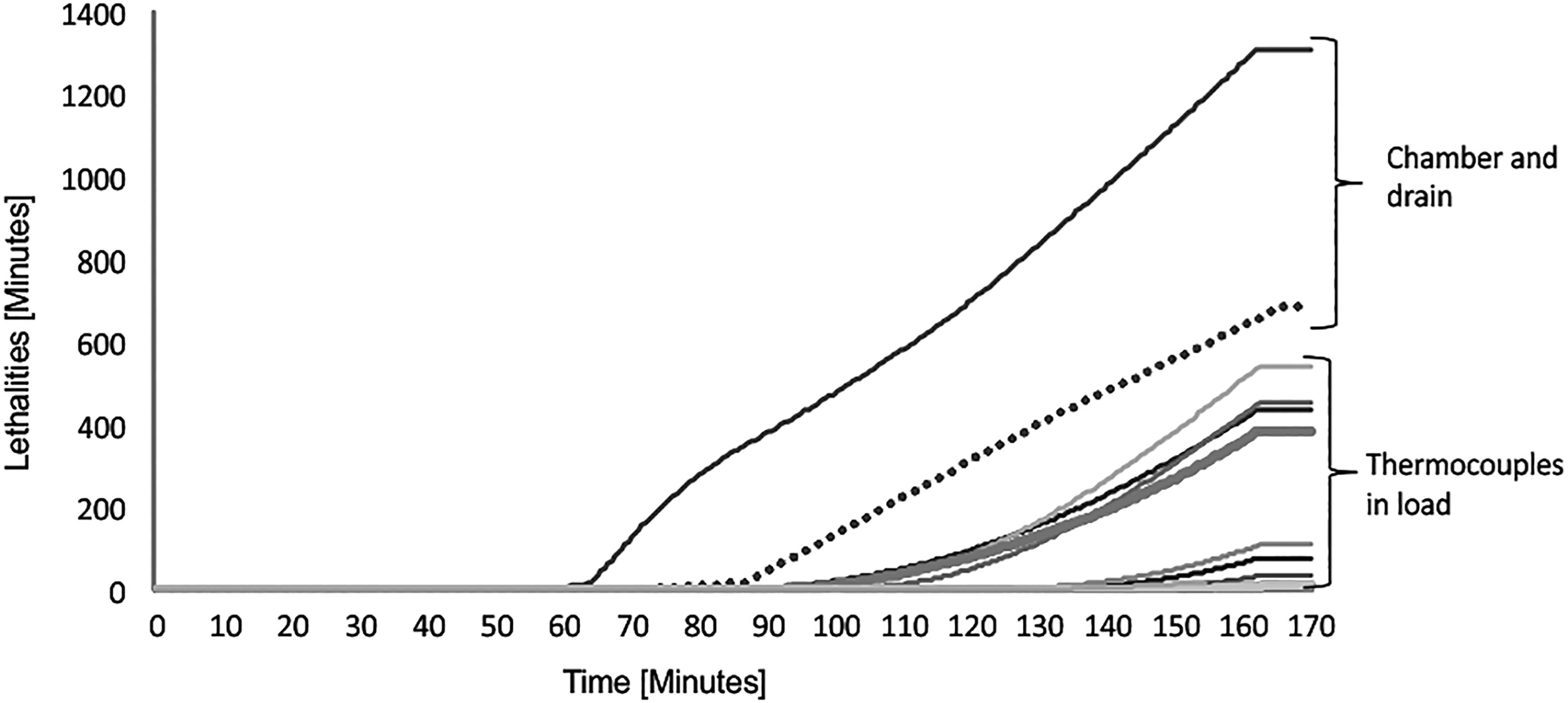
F0 value calculated for each biological indicator on different points into the load for animal carcass cycle. It shows the different lethalities calculated at different points of the load, the chamber, and drain.
Figures 7 and 8 show that sterilization conditions are reached with the animal bed cycle. Due to the homogeneous configuration of the load of animal bed, the TC show values that increase evenly in both temperature and lethality.
Temperature of different point into the load for animal bed cycle.
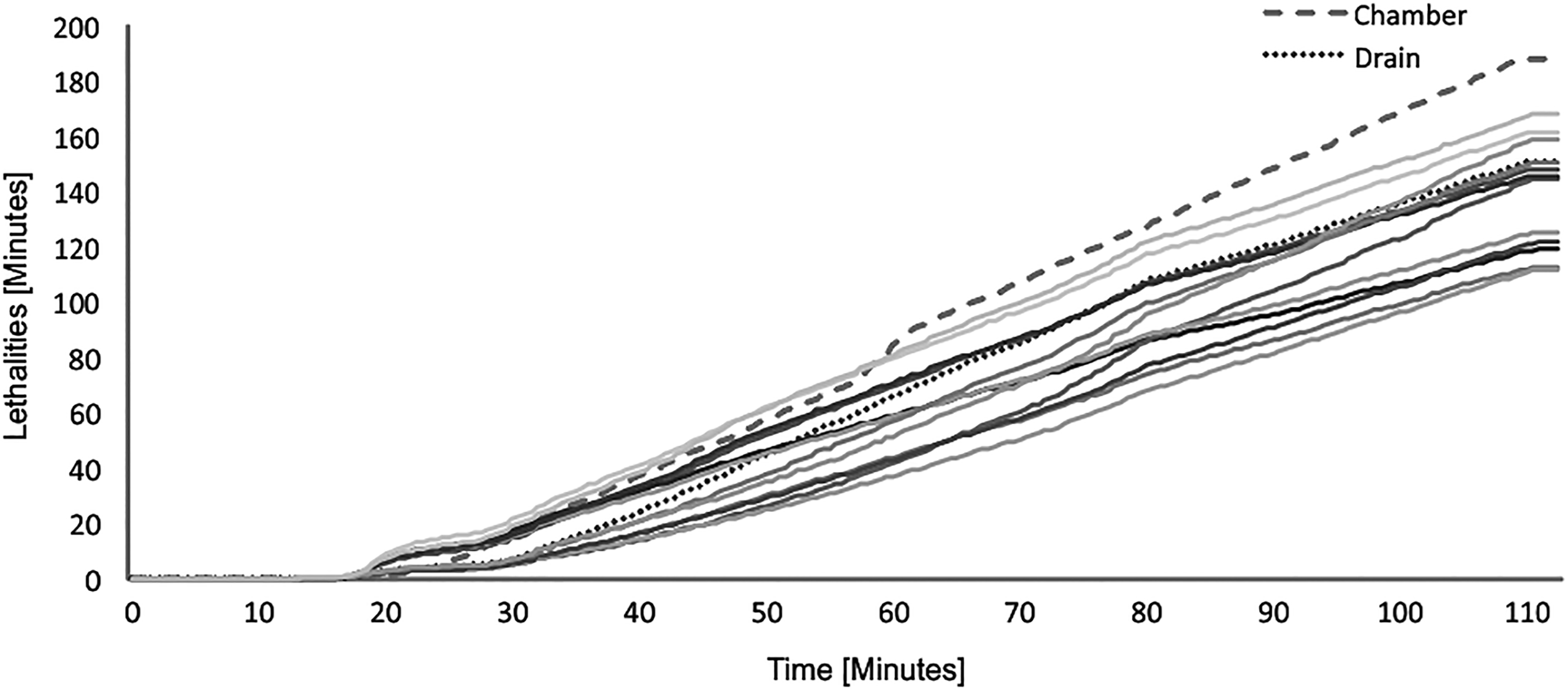
F0 value calculated for each biological indicator on different points into the load for animal bed cycle.
As mentioned before, BSL-3 labs require that materials, trash, and waste must be decontaminated through autoclave before disposal to minimize risks of exposure to both the operator and the environment. 3 Our results demonstrate that the waste from the INEVH BSL-3 facility is safe for handling after decontamination; consequently, it provides protection for the personnel who manipulate it and for the environment.
The methodology used and the results obtained could serve as a basis and guide for other laboratories with similar characteristics.
Conclusion
One of the most important findings of this study is that autoclave factory default settings are potentially inadequate for sterilizing highly infectious waste. Individual facilities will need to develop their own protocols to handle waste safely. Although time-consuming, this steam sterilization validation process is essential to maintain the public trust in our ability to safely work with biological select agents. In this article, we described an empirical study to evaluate and validate a decontamination procedure for biological waste from the INEVH BSL-3 facility that guarantees the steam sterilization process. Using animal waste, we found that animal carcasses require a different treatment in order to guarantee proper decontamination. The time to achieve sterilization temperatures inside the carcasses was considerably longer than for typical laboratory autoclavable goods. Moreover, the heat was not distributed homogeneously throughout the material. Hence, we designed 2 different cycle: 1 for animal waste and another 1 for animal carcass.
It is important to highlight that decontamination cycles must be designed and validated in situ with the particularities of each laboratory, meaning validation to verify that the specified requirements are adequate for an intended use. 7 Autoclave parameters may need to be adjusted, with particular attention paid to the way that waste loads are packaged prior to treatment. Each facility utilizing autoclaves for the treatment of infectious biological waste should validate their waste management protocols with simulated waste loads and within-bag biological indicators to ensure that the waste is properly decontaminated.
The validation of a decontamination process, specifically, should be seen as a continuous process that begins at the development and engineering stages and continues in the operation and maintenance of the validated operating conditions.
Footnotes
Acknowledgments
We thank the anonymous reviewers for critical suggestions and advice that helped to improve previous versions of this article. We are grateful for the collaboration of the staff of Virology, Quality Control, Engineering and Quality Assurance services. We also thank to Diego Bonano for helping in the figures design and Miguel A. Grimaldo for constructive comments and editorial corrections. The results of the following work were present in the article: (1) Cascardo E., Goenaga S., Fassio R., Fossa S., Bottale A., Levis S. y Riera L. Desarrollo de un ciclo de descontaminación de animales por calor húmedo en un laboratorio de bioseguridad de nivel 3 (BSL-3). IX Congreso Brasileiro de Biosseguranca IX Simpósio Latino Americano de Biosseguranca de Produtos Transgénicos. Trabalho científico SD03. Porto Alegre, Rio Grande do Sul, Brasil. Del 29 de septiembre al 1 de octubre de 2015. (2) Cascardo E., Goenaga S., Fassio R., Fossa S., Bottale A., Levis S. y Riera L. Desarrollo y validación de ciclo de descontaminación de residuos por calor húmedo en un BSL-3. IX Congreso Brasileiro de Biosseguranca IX Simpósio Latino Americano de Biosseguranca de Produtos Transgénicos. Trabalho científico SD04. Porto Alegre, Rio Grande do Sul, Brasil. Del 29 de septiembre al 1 de octubre de 2015.
Ethical Approval Statement
The Ethics Committee of the INEVH waived the need for approval for this study.
Statement of Human and Animal Rights
Not applicable to this study.
Statement of Informed Consent
Not applicable to this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partially by Fondos Concursables Asociación Nacional de Laboratorios e Institutos de Salud 2014 (FOCANLIS 2014).
