Abstract
Introduction:
The effectiveness of formaldehyde as a fumigant in laboratories, for equipment, and for containment barrier decontamination applications was assessed, in particular the ability to reproduce biological inactivation (6-log reduction) of commercially available rapid biological indicators in representative operational scenarios and their relative sensitivity to other biological and chemical indicators.
Objectives:
The primary objective of this study was to describe observations and results of formaldehyde fumigation efficacy in high-containment laboratories.
Results:
Biological indicators placed throughout laboratory spaces, including ventilation ductwork at distances up to 15 meters, inside pieces of equipment and in lengths of pipe, were mostly negative, demonstrating the ability of formaldehyde to reach the interior and external surfaces tested. Dwell times as short as 10 minutes were shown to be sufficient in barrier decontamination equipment for the fumigation of personal computers. Furthermore, a pipework bore:length ratio of 1:1500 was proven too great a challenge. Indicators placed after extracting HEPA filters in microbiological safety cabinets (MBSCs) were also successfully fumigated (at room temperature) relying solely on diffusion and in the cabinet workspace at 10°C to 12°C. In addition, pressures of up to 900 Pa were experienced in low-leakage laboratory spaces during fumigation.
Conclusions:
Formaldehyde fumigation was shown to be effective in a variety of scenarios representing operational activities thereby giving process assurance.
Introduction
In the UK, formaldehyde is still widely used as a microbiological laboratory fumigant due to its broad-acting microbial activity and ease of use. The formaldehyde fumigation process consists of vaporizing, dwell, and purge phases. Unlike an autoclave or effluent treatment plant that can accurately and continually control and monitor the critical parameters required for sterilization (ie, time and temperature) throughout the sterilization cycle and fail the process if these are not met, the efficacy of fumigation is a result of fumigant concentration, relative humidity (RH) temperature, and time, 1 not all of which can be accurately monitored or adjusted in real time. However, efficacy can be determined by using chemical and biological indicators. For formaldehyde fumigation to be effective, it is generally accepted that relatively high humidity is required2 -4 and that the temperature should be >20°C. Indeed, UK regulatory guidance refers to achieving 70% to 90% RH 5 , and the EU Biorisk Management Standards for Laboratories working with Foot-and-mouth disease virus (FMDV) requires a minimum 70% RH for 10 minutes. 6
Both laboratories and the equipment within them have spaces that are potentially inaccessible to formaldehyde vapor. Prior to the licensing and occupation of the facility, fumigation processes in a newly constructed and commissioned high-containment laboratory complex (designed to UK Animal Containment Level 4/US BSL3Ag) were biologically validated to ensure a reproducible kill and provide the regulators and duty holders a high degree of confidence. The processes included laboratory fumigation and barrier fumigation equipment (fumigation lobbies/hatch). In this article, we discuss the development of validated formaldehyde fumigation processes with standardized biological indicators (BIs) and the extent to which fumigant was able to penetrate and infuse into spaces to achieve a biological kill (negative BIs) in representative and worst case scenarios. Furthermore, observations acquired from operational fumigation activities are presented.
Materials and Methods
Laboratory Fumigation
Each Containment Level 4 laboratory space was served by air supplied through 1 HEPA filter and extracted through 2 HEPA filters in series and were fumigated by vaporizing 200 ml formalin (approximately 39% w/v formalin) and 900 ml water/27.3m3 (approximately 1000 ft3) using domestic rice cookers. Each rice cooker could hold up to 2.5 L; therefore, in larger laboratories, multiple cookers were used with the formalin/water mix being evenly distributed. No fans were used to circulate the fumigant. A representative number of 6 laboratories were fumigated, each being isolated by bolting a gas-tight shroud to the door frame, followed by a controlled shutdown of the space via the Building Management System (BMS) control dampers, and finally, closing the manual gas-tight isolation dampers on the supply and extract HEPA filter housings serving the space. The boundary of the fumigation zones therefore extended to the laboratory side of the closed HEPA housing dampers. Each laboratory was constructed from poured in place reinforced concrete with a multilayer, high-performance coating applied (C/S Wallglaze Armourglaze Construction Specialities Ltd., Bucks, UK) and tested to a leak tightness of <0.0091 m3/m2/hr at +200 Pa and –200 Pa. In addition, all penetrations were smoke pencil tested at –200 Pa. Self-contained BIs (3M Attest Rapid Readers; Bacillus atrophaeus at 106) were placed throughout the laboratories, including on and under benching/casework, behind microbiological safety cabinets (MBSCs), and on the floor, walls, windows, and ceiling. Between 14 and 29 BIs were placed in the representative laboratories, depending on the room size. In addition, indicators were placed in the supply and extract ventilation ductwork. The ductwork BIs were placed on the laboratory side of the closed HEPA housing isolation dampers. This was considered the worst case as fumigant would have to migrate from the space below through the ductwork in sufficient concentrations to achieve inactivation. The lengths of ductwork varied but were typically 2 to 15 meters in length and included bends.
The fumigant dwelled for 24 hours in 5 laboratories to fulfill the requirements of the European Commission for the Control of Foot-and-Mouth Disease (EUFMD) Minimum Standards and 18 hours in a sixth laboratory operated at Containment Level 3 (BSL-3). In addition, a Containment Level 3 suite consisting of 4 laboratories and a common corridor, totaling 550 m3, was fumigated following the principles described previously. Following a purge to reduce formaldehyde concentrations <0.2 ppm, the BIs were removed and processed in a 3M Attest Auto-reader 390G for 4 hours with positive (unexposed) controls. Successful validation was no growth in all BIs from all 6 representative laboratory spaces and the Containment Level 3 suite of laboratories.
In addition, the pressure was monitored in the laboratory spaces during fumigation via the building’s secondary monitoring system (SMS; logging at 1-second intervals).
Barrier Fumigation Equipment (Fumigation Lobbies and Hatch)
The barrier fumigation equipment was bespoke units provided by PBSC Ltd. (Huddersfield, UK). Briefly, the units (5 large walk-in lobbies each approximately 12 m3 and a hatch of approximately 0.72 m3) were fabricated from stainless steel with temperature, humidity, and formaldehyde concentration (up to 2000 ppm) monitoring. The doors were of the pneumatic APR type, and the cycles were operated via a Siemens PLC HMI. Furthermore, the units, through the PLC, calculate the amount of water to be added each cycle (based on ambient humidity) to achieve a target humidity >70% RH at the start of the dwell phase. Two fans, operating during the vaporize, dwell, and purge (aeration) phases, provided thorough mixing. To comply with the EUFMD Minimum Standards, the barrier fumigation equipment (lobbies and hatch) were validated using 10 g/m3 formaldehyde. Initial validation consisted of 3 consecutive empty chamber runs for all 5 of the lobbies (each approximately 12 m3, including the supply and extract ductwork up to the gas tight dampers) and the smaller hatch (approximately 0.72 m3) with a 60-minute dwell. The 3M Attest Rapid Indicators as described previously were used and placed on the walls, floor, and ceiling and in the supply and extract ductwork, for a total of 8 BIs/lobby. A positive control, not exposed to fumigant, was incubated alongside. Generally, BIs were placed vertical with the cap uppermost; however, in a separate single run, 4 test BIs were placed vertically (cap uppermost), 4 vertically (cap downward), and 4 horizontally to determine if orientation of the BI affected the result. Successful fumigation was considered to be all exposed BIs proving negative and RH >70% for at least the first 10 minutes of the dwell phase for 3 consecutive cycles. Following empty chamber validation, representative load validation was performed in selected units (with 60-minute dwell times for the lobbies and a 10-minute dwell for the hatch); they were:
Lobby 1: Laboratory representative material (laboratory equipment and empty chemical bottles) Lobby 2: Engineering representative material (pipework/valves) Lobby 3: Line-Pickerill Helix Hatch: Personal computer Lobby 4: Coiled fumigation hoses.
In Lobby 1, BIs were placed under equipment (ie, a freezer, centrifuge, and waterbath) and in the base of an empty, upright, 1 L chemical bottle (note: a chemical indicator [CI]; Browne No. 2) was also placed on the base of the chemical bottle). In Lobby 2, indicators were placed at the midpoint inside lengths of metal pipework (150-2500 mm in length) as detailed Table 1.

Biological indicators placed at midpoint of pipework open at both ends.
In addition, to provide a more challenging length:bore ratio, an additional 3 cycles were run with BIs placed in a Line-Pickerill Helix (Figure 1) in a third lobby with the helix upright and a further 3 cycles with the helix inverted. One BI was placed in the closed end of the helix, the cap tightened and taped with a second BI run in parallel taped to the outside of the helix. In addition, the helix was also placed in a facility undergoing routine fumigation with 3 g/m3 formaldehyde with an exposure time of 24 hours. In this case, chemical indicators (not BIs) were placed in the helix cap. The PC placed in the fumigation hatch had the cover removed, BIs placed within the unit, and the cover replaced. In Lobby 4, coiled hoses (for use when fumigating MBSCs) were placed horizontally and vertically with a BI placed in the midpoint to determine if the plane and the orientation of the open ends (ie, facing upward or downward) had an effect (Figure 2).

Line-Pickerill Helix (arrow indicates location of biological indicator inside cap).

Coiled fumigation hoses (arrows indicate locations of biological indicators inside hoses).
Microbiological Safety Cabinets and Half-Suit Isolators
Two 1200-mm recirculating MBSCs with single extract HEPAs were fumigated by vaporizing 20 ml 39% w/v formaldehyde and 20 ml distilled water with a dwell period of 12 hours using an all-in-one fumigation/carbon adsorption unit (Walkers Safety Cabinets, Glossop, Derbyshire, UK). After 12 hours, the fumigation unit’s internal fan was automatically energized to draw the formaldehyde through activated carbon granules. In the absence of a post-HEPA isolation damper, the extract was bagged and taped to retain fumigant. In addition to placing BIs in the workspace, additional BIs were placed after the HEPA filters to determine if formaldehyde was able to diffuse across the filter media (without the addition of the internal fans pulsing or other recirculation systems) within the 12-hour dwell time. A similar piece of work was conducted in 2 half-suit isolators (Bell Isolation Systems, Livingston, UK) using 120 ml formaldehyde and 120 ml water with the placement of BIs between the primary and secondary extract HEPAs and an 8-hour dwell. After the dwell, the extract dampers were automatically opened to initiate the purge.
In addition, an additional 5 MBSCs were fumigated in a decommissioned laboratory with the air temperature 10°C (±2°C) to determine the effect of lower temperatures on the process. Biological Indicators were placed on and under the worktray and in the top plenum (pre-extract HEPA).
Comparison to Other Biological and Chemical Indicators
Biological indicators (3M Attest 1294 Rapid Readers and Steris Spordex Strips) containing approximately 106 B. atrophaeous spores and chemical indicators (Browne: No.1 pink paper strip and No.2 Formaldehyde Control Indicator-Steris Corporation) were exposed to formaldehyde at approximately 450, 900, 1800, 3550, 6200, 8850, and 18 000 parts per million (ppm) for 18 hours in a microbiological safety cabinet. Note: These are theoretical calculated values, not directly measured. The experiments were all repeated 3 times. The Spordex strips were exposed to formaldehyde with the glassine envelope both intact and partially peeled open to directly expose the paper spore strip inside to fumigant. The biological indicators were processed as per the manufacturer’s instructions. The chemical indicators were reliant on a color change (pale pink to dark pink and blue to green, respectively).
Results
Laboratory Fumigation
Successful biological validation of formaldehyde fumigation was achieved in all 6 individual laboratories, including ventilation ductwork, and the larger Containment Level 3 laboratory suite. A dwell time of 18 hours was sufficient for 1 laboratory.
Interestingly, no growth of the test organism was recorded on BIs placed in ventilation ductwork, including in a length approximately 15 meters from the laboratory. No supplementary air circulation (ie, fans) was provided to assist the process.
The pressure within individual laboratories, as monitored by the SMS, peaked between approximately 400 and 900 Pa (relative to ambient).
Barrier Fumigation Equipment
Three successful, consecutive runs for each empty fumigation lobby (5 in total) and the hatch (1 in total) was achieved. Most BI placements returned negative (no growth) results, indicating successful decontamination. The exceptions were in Lobby 4 when a 5-m coiled hose was placed in the vertical plane but with the open ends uppermost (see Figure 1; a positive BI was returned from the hose in 1 run; the other 2 runs were successful). In addition, all 3 runs with the Line-Pickerill Helix were unsuccessful. All other locations in all lobbies were successful for 3 consecutive runs. A 60-minute dwell time for the lobbies was adequate to achieve the desired performance for the empty chamber and most representative loads. However, a 10-minute dwell time for the hatch achieved the performance requirements. The CI placed in the base of the chemical bottle only partially changed color.
Microbiological Safety Cabinets and Isolators
In the first experiment, in addition to the BIs placed in the workspace, BIs placed downstream the extract HEPA filter following fumigation were negative. For the half-suit isolators, the BIs placed after the primary extract HEPA were also negative. In the second study investigating the effect of lower temperatures, all BIs placed on/under the worktray or in the exhaust plenum of the 5 MBSCs at ambient temperatures of approximately 10°C were also negative.
Comparison to Other Biological and Chemical Indicators
There was a significant difference in the sensitivity of the indicators tested. The Browne No.1 indicators all showed a color change from pale to dark pink at the lowest concentration tested (450 ppm). The results for the Browne No. 2 indicators were less defined: at 3550 and 6200 ppm, 2 of 3 experiments did not result in the required color change, and 1 of 3 showed only a partial, inconclusive color change. At 8850 ppm, 2 of 3 demonstrated successful color change, with 1 of 3 inconclusive. At 18 000 ppm, all indicators changed color. Total kill was achieved in the 3M Attest BIs at 900 ppm and above but not at 450 ppm. The Spordex BIs were far more resistant, with total kill only being achieved at 18 000 ppm. There were no differences between the sealed and partially open strips. In summary, the relative sensitivities were as given in Table 2.

At 8850 ppm, 2 of 3 indicators showed a color change, and 1 of 3 showed a partial color change, suggesting concentrations as high as 18 000 ppm are not required.
Discussion
Fumigation of laboratories and of equipment leaving high-containment areas is necessary to protect personnel and the environment from the pathogens deliberately worked on in the facility. Although, and where possible, it is good practice for high-containment laboratories to keep dedicated tools and equipment within the contained space, there remains a requirement to be able to decontaminate visiting engineers’ tools for release to the noncontained space. Furthermore, periodically defunct laboratory equipment requires decontaminating prior to safe disposal. Given the criticality of the process, assurance must be provided that a suitable reduction of viable pathogenic agents is achieved. For microbiological applications, a 6-log reduction is often considered to fulfill this requirement, although 4-log reductions have been considered adequate in certain applications,7,8 and many disinfectants are tested to achieve a 3- to 4-log reduction. It is generally accepted that bacterial spores are more resistant than viruses 9 ; therefore, substitute test organisms of greater resistance (ie, bacterial spores) to those worked on in the facility were used as the test organism. Furthermore, it can prove problematic validating the efficacy of fumigation in real-life scenarios with actual high-consequence pathogens in new buildings not licensed or approved to work on those organisms.
Formaldehyde fumigation of personal computers has previously been demonstrated with a risk-based 4-log reduction of Geobacillus stearothermophilus. 8 The results of this article demonstrate a 6-log reduction of B. atrophaeus in equally inaccessible spaces within a PC, with the casing being replaced after the BI placements. This approach has given a high level of assurance as to the validity of the process and supplements the work by Abraham et al. 8 Similarly, BIs were placed in locations considered the worst case in laboratory equipment and engineering components with biological validation being achieved. Of particular interest were the results obtained from placing BIs in lengths of metal pipework. It was found that a 6-log reduction was achievable at the midpoint up to a length to bore ratio of 62.5:1 in a double open-ended piece of tubing (effectively a 32.25:1 ratio in a closed end pipe). This study was taken further to determine the length to bore ratio in piping open at one end at which fumigant was able to penetrate. The efficacy of ethylene oxide sterilization and low temperature sterilization and fumigation (LTS&F) cycles in vacuum-assisted autoclaves may be demonstrated using a coiled piece of tubing with a length to bore ratio of 1500:1, commonly referred to as a Line-Pickerill helix. This piece of standardized test equipment provides a worst case scenario representative of medical devices with lumens open at one end. In an LTS&F autoclave cycle, a vacuum is pulled within the chamber before formaldehyde is introduced, thereby allowing greater penetration into the helix. The tests performed within this study were all performed in equipment not assisted by pulling a partial vacuum. Although this study did not determine a precise endpoint at which kill was not achievable within pipework or quantify survival rates, it did demonstrate that adequate fumigant penetration was occurring up to 31.25:1 pipe diameters in representative loads but not 1500× pipe diameters using the Line-Pickerill helix with 10g/m3 formaldehyde with a 60-minute dwell time. However, a sensitive chemical indicator (Browne No.1) placed in the cap of the helix did show a slight color change following a longer dwell time of 24 hours. This study has shown that the Browne No. 1 CI totally changes color at a formaldehyde concentration of at least 450 ppm; therefore, low levels of formaldehyde were present in the cap of the helix at some point during the 24-hour dwell. Not surprisingly, previous studies have shown that the percentage of surviving test microorganisms increases with the length of tubing therefore, there comes a point at which fumigant is unable to penetrate far enough into pipework or tubing at sufficient concentrations to achieve satisfactory decontamination within a given time. In previous studies, lengths of tubing were sealed at one end. 10 In this study, both single and double open-ended scenarios were investigated as the latter is a realistic real case load type (eg, copper piping and scaffold tubes).
Adequate fumigant dispersal into ventilation ductwork was also demonstrated with a 6-log reduction up to 15 meters distant in ductwork 200 to 300 mm in diameter, including 3 to 4 bends up to 90°, without supplementary air dispersal (ie, fans) during laboratory fumigation.
The placement orientation of the BIs used in this study did not appear to affect performance. Paper strips in glassine envelopes and other similar biological indicators have a complete permeable surface over which fumigant is able to penetrate. The rapid indicators used in this study are composed of an impermeable plastic vial where formaldehyde can only enter and then come into contact with the biological indicator organism through the cap. Placing the indicators in different orientations did not prevent achieving a satisfactory kill. Interestingly, microbiological kill was achieved in an empty, upright chemical bottle, but the extent of color change of a parallel chemical indicator suggested lower levels of formaldehyde inside the bottle relative to outside the bottle as indicated by the extent of color change of the indicator. This conclusion is partially qualitative as chemical indicators are intended to be straight pass or fail; however, the intensity of the color change appears to be relative to formaldehyde concentration or exposure time (author’s personal observations). This may align with the observation in the present study of a fail in a coiled pipe placed in the upright; that is, formaldehyde vapor in these relatively short dwell times may not penetrate fully in spaces only open at the top.
Although extended dwell times up to 24 hours are used for laboratory and animal facility fumigation (author’s personal observations) and it is convenient to fumigate MBSCs overnight, more rapid turnaround times are desirable for barrier transfer applications, particularly when decontaminating engineers’ and contractors’ tools. A dwell time of 60 minutes was achieved for the fumigation lobbies, but the smaller fumigation hatch was validated with a dwell phase as short as 10 minutes (giving an overall cycle time <60 minutes allowing for a purge to reduce formaldehyde concentrations to <0.2 ppm). Although not presented in this article, dwell times in laboratories of as little as 2 hours (following a 3-hour vaporize phase) and stopping the cycle in a fumigation lobby immediately at the end of the vaporize phase (ie, no dwell) have been shown to be effective when using the rapid biological indicators used in the present study (author’s personal observations).
Relatively high pressures of between 400 and 900 Pa were recorded by the building’s secondary pressure monitoring system during fumigation of laboratories. The laboratory spaces were all designed and constructed to meet an air leakage rate of <0.0091 m3/m2/hr at 200 Pa (positive and negative), and therefore significant increases in pressure during vaporization in low leakage spaces were not unexpected if the vaporized formalin is assumed to behave as an ideal gas:
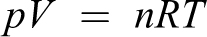
An increase in the number of gas molecules (n) as formalin is vaporized, where V is fixed (assumption as a result of the low air leakage), results in an increase in pressure (p). There would also be an increase in temperature (T) as a result of heat generated by the vaporizer’s heating element.
Despite the positive pressurization of the spaces, formaldehyde levels in adjacent spaces, some operating at pressures negative relative to atmospheric and thereby creating pressure differentials of up to 1 KPa with surrounding spaces, remained <0.2 ppm, with most sample points reading no more than background. This result gave confidence in the ability to fumigate individual laboratories while adjacent spaces remain operational.
Previous validation studies involving placing BIs post both the primary and secondary filters in double-HEPA-extracted MBSCs programmed with intermittent pulsing of the internal fans, or circulation devices (ie, attaching an external fan to ports pre- and post-HEPA), were successful (author’s personal observations). In work reported here, BIs were placed after a single-extract HEPA with fumigant diffusing through the filter media for 12 hours without supplementary motive force to assist the process. Because these were recirculating MBSCs with carbon absorption to remove formaldehyde, fumigant would not have been actively drawn across the filters at any time (ie, during purging). Previous studies have shown this location to be most challenging yet demonstrate the ability to achieve a level of kill with both Bacillus spp and poliovirus 4 and complete inactivation of lower spore challenge counts. 11 Although formaldehyde fumigation is considered more effective at temperatures around 40° to 45°C, 12 performing fumigation in this study at much lower temperatures (∼10°C) than normally found in laboratories (∼20°C) did not prevent a successful biological validation of the process when indicators were placed in the workspace, under the worktray, and in the top plenum adjacent to the extract fan (pre-exhaust HEPA).
The significant variation in the sensitivity of biological and chemical indicators gives an indication of the importance of using the correct surrogates when validating fumigation. Previous studies have shown that commercial BIs could be unreliable as general indicators and encouraged careful consideration in the selection of commercial indicators. 13 Interestingly, there was a notable difference in the levels of formaldehyde required to achieve full kill (106) in 3M Attest Rapid Readers and Steris Spordex spore strips, particularly as both indicators utilize the same test organism (B. atrophaeous), although the demonstration of inactivation differs. The former required 900 ppm, yet the spore strips required as much as 18 000 ppm, although total inactivation was almost achieved at 8850 ppm. The rapid readers rely on the inactivation of beta-glucosidase, a naturally occurring enzyme of the bacterial spore involved in spore growth and cell function that catalyzes the reaction of a fluorescent product. The presence of the fluorescent product indicates viable spores. Spore strips rely on the inactivation of spores evident as turbidity and/or color change following incubation in culture media. The advantage of rapid readers is they deliver a result in 4 hours, whereas spore strips are normally incubated for up to 7 days. Rapid readers certainly have their uses, particularly where quick results are required. Formaldehyde concentrations as low as 600 ppm have been shown to give up to 6-log reductions experimentally 14 ; therefore, the rapid readers could be an adequate surrogate. Their apparent ease of inactivation compared to spore strips is only an issue if the target organism is not as readily inactivated, hence the importance of performing parallel studies, wherever possible, to generate baseline data as to the suitability of rapid readers or spore strips for ongoing process validation.
Conclusion
In conclusion, this study was able to demonstrate effective formaldehyde fumigation of ductwork serving laboratories up to 15 meters distant, in open ended pipework with a length:bore ratio of 62.5:1, in personal computers with as little as 10 minutes of exposure time, across HEPA filters by diffusion, and that even at lower temperatures, formaldehyde fumigation can still be effective. Furthermore, the report demonstrates significant differences in the sensitivities of chemical and biological indicators and that care should be taken when considering such indicators for process validation. In addition, increases in pressure within laboratories of almost 1 KPa were achieved.
Footnotes
Ethical Approval Statement
The authors declared that no animals were used in the study.
Statement of Human and Animal Rights
The authors confirmed that no human subjects or animals were involved in generating data for this article.
Statement of Informed Consent
The authors confirmed that no patient information was included in the data presented in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Biotechnology and Biological Sciences Research Council BBS/E/I/00007037.
