Abstract
Foot-and-mouth disease virus (FMDV) is a highly pathogenic and transmissible disease of cloven-hoofed animals with outbreaks having severe impact on livestock production. FMDV is generally studied in high- to maximum-containment laboratories to prohibit the virus from escaping into the environment. Formaldehyde is still the method of choice for fumigation of rooms and HEPA filters at FMDV facilities. However, formaldehyde, having been shown to be carcinogenic, should ideally be replaced by other, less hazardous methods. Here we show validation results for two different hydrogen peroxide fumigation methods and compare these to formaldehyde fumigation. The results identify hydrogen peroxide as a suitable alternative to formaldehyde fumigation.
Keywords
Foot-and-mouth disease virus (FMDV), a member of the family Picornaviridae, genus Aphtovirus, is a highly infectious virus affecting both domesticated and wild cloven-hoofed mammals. 1 Manipulating the virus in laboratories, vaccine production facilities, or animal facilities without adequate precautions poses a risk of environmental release and may incur severe economic losses as was the case in the United Kingdom in 2007. 2 It has been shown that as few as 10 TCID50 can be infective to cattle by the airborne route. 3 However, this is under experimental conditions and the low infective dose may relate to the relatively large size of aerosol droplets. As a consequence of the low infective dose, laboratories handling FMDV must work under high-containment conditions, in which the principal objective of the containment measures is to prevent release of virus that would give rise to animal infection outside of the laboratory (veterinary containment). Properly validated disinfection, including fumigation, is, among other containment measures, therefore crucial in these laboratories.
Formaldehyde (FA) has been the method of choice for room and equipment fumigation for decades 4 and is incidentally the only method described in the Minimum Biorisk Management Standards for Laboratories Working with Foot-and-Mouth Disease Virus by the European Commission for the Control of Foot-and-Mouth Disease (EUFMD). 5 However, FA is known to be a human sensitizer and carcinogen and can leave undesirable residues if its vapor is poorly delivered or not evacuated from the treated area within a defined period of time.6,7 In addition, decisions at the European Union (EU) level, under the Biocidal Products Directive (Directive 98/8/EC, 1998), have led to restrictions on its use. Thus, alternative methods have been developed and tested recently, including hydrogen peroxide as either vapor or aerosol.8 –13 Contrary to FA, hydrogen peroxide produces nontoxic byproducts (water and oxygen) and thus is ecologically safer, is less hazardous to personnel, and requires no postprocess neutralization or cleaning. However, the equipment used for hydrogen peroxide fumigation can be costly compared to formaldehyde.8,12 In addition, fumigation cycles have to be setup individually in sometimes lengthy processes for every room and every situation. Furthermore, technical design specifications play an important role in the success of the fumigation. 12
Two recent studies looked at the efficacy of hydrogen peroxide against FMDV.11,14 Hole et al 14 showed that activated hydrogen peroxide as a solution is an effective disinfectant against FMDV, while the results of Petit et al 11 proved that hydrogen peroxide vapor (HPV) inactivates FMDV, albeit not clearly defining to what level. With this study, we validated two different fumigation methods based on hydrogen peroxide (vaporized hydrogen peroxide [VHP] and aerosolized hydrogen peroxide (H2O2)) and compared them to FA fumigation, which we also validated. Comprehensive experiments were performed to clearly define the reduction in FMDV that may be achieved using all three methods, taking into account losses in virus titers occurring due to drying onto stainless steel discs and exposure to environmental conditions.
Materials and Methods
Fumigation Zone and Setup
Fumigations were performed in an animal room within the institute’s BSL-3Ag facility as work with FMDV may only be performed inside the IVI containment according to Swiss legislation. The fumigation zone includes the animal room with the inner wardrobe of the shower airlock containing a cupboard and bench as well as the supply and exhaust air ducts with their respective HEPA filters (Figure 1).
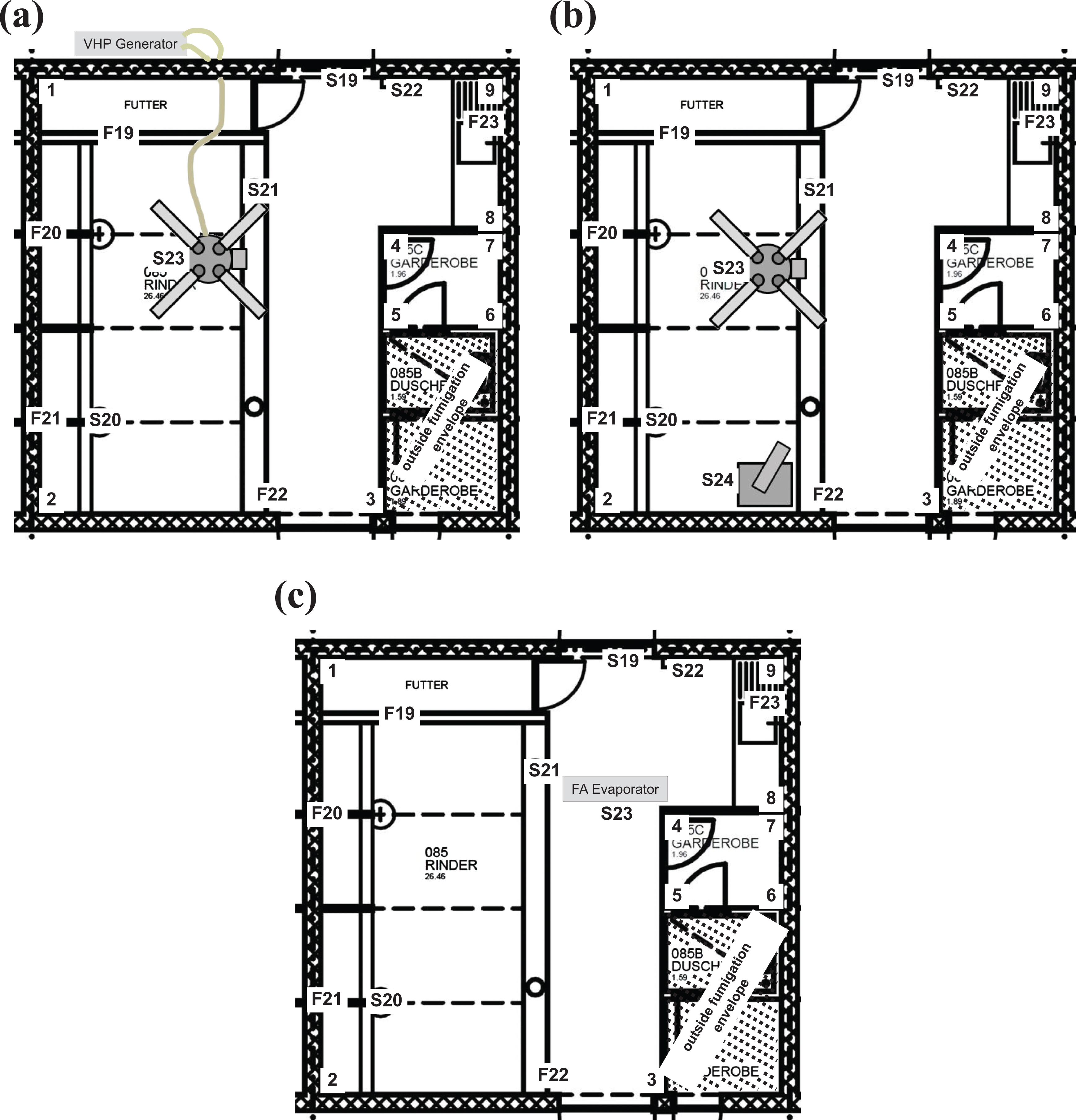
Set up of fumigation zone for VHP (a), aerosolized H2O2 (b), and FA (c). The animal room is approximately 88 m3 in volume and comprises, besides the animal holding pens, the feed and equipment stall, the sink area and the inner wardrobe of the shower airlock with its cupboard and bench. The VHP generator was located outside the fumigation zone in the waste corridor (a). The VHP was collected with a hose and led to the fumigation ventilator as depicted. Aerosolized H2O2 was generated inside the fumigation envelope and distributed using the fumigation ventilator (b). FA was also generated inside the fumigation envelope, but not further distributed using ventilators (c). Spore biological indicator (BI) pouches were placed in positions 1-9 (floor) and 10-18 (ceiling) as well as in positions S19 (window in containment door to waste corridor, S stands for spore), S20 (water supply), S21 (floor drain), S22 (hose reel), S23 (under fumigation ventilator or under FA generator, where applicable), and S24 (under aerosolized H2O2 generator, where applicable). BIs 24-27 (VHP, FA) or 25-28 (aerosolized H2O2), respectively, were positioned before and after the supply and exhaust HEPA filters (not depicted). Stainless steel discs carrying FMDV were placed in positions 1-18 as above. In addition, discs were placed at a height of approximately 1.7 m as follows: F19 (partition wall between animal pen and feed stall, F stands for FMDV), F20-F22 (on angled iron plates along wall) as well as F23 (sink).
For VHP fumigation, the generator (Steris VHP 1000 ED-S, Steris, Mentor, OH) was located in the waste corridor outside the animal room with the VHP provided through penetrations in the wall. VHP was collected inside the animal room with a hose via camlock couplings, led to the Alpiq 6100 fumigation ventilator (Alpiq InTec Ost AG, Uster, Switzerland) and accelerated from approximately 34 m3/h supplied by the generator to a maximum of 2500 m3/h to achieve efficient and uniform distribution (see also Kümin et al 12 for an example of a fumigation ventilator based on the Alpiq 6100). Airflow was controlled by a frequency converter and directed into the animal room by a total of eight outlets using tubes (Figure 1a).
H2O2 aerosol fumigation was performed by placing the generator (Q-Jet Superior, Sanosil AG, Hombrechtikon, Switzerland) inside the animal room on a pedestal approximately 800 mm high. Uniform distribution of the aerosol was once again achieved with the Alpiq 6100 fumigation ventilator placed in the middle of the room as described for VHP fumigation with the exception that the generator was not directly coupled to the ventilator (Figure 1b).
FA fumigation was performed using an Autex 100 L evaporator (Ehret Labor- und Pharmatechnik GmbH & Co KG, Emmendingen, Germany) placed inside the animal room. No additional ventilation system was used during FA fumigation (Figure 1c).
Program Cycles
The following parameters are based on previous experiments 12 and were thus programmed into the VHP generator for the validation of room fumigation: Dehumidification to 10 mg/l absolute humidity (20 minutes), Conditioning sterilant injection rate 12 g 35% H2O2 (Vaprox, Steris, Mentor, OH) per minute (30 minutes), Decontamination injection rate 11 g/min (300 minutes), and Aeration 60 minutes. The flow rate was set at 34 m3/h for dehumidification and aeration and 32 m3/h for conditioning and decontamination. In addition, the preheater temperature was set at 60°C while the evaporator temperature was 100°C. Following aeration with the generator, supply and exhaust dampers were opened and the animal room purged using the building’s HVAC system for approximately 18 hours.
A two-step cycle was used with the Sanosil Q-Jet Superior: injection of 7.5% H2O2 (Sanosil S015, Sanosil AG, Hombrechtikon, Switzerland) for 35 minutes (end-concentration of 12 ml/m3), holding time of 60 minutes, followed by another injection of 7.5% H2O2 and a 2-hour holding time. The cycle was developed over the course of several experiments and proved to be the most efficient while giving results that could be validated. Supply and exhaust dampers were then opened to purge the room using the building’s HVAC system for approximately 18 hours.
Based on fumigation cycles developed at IVI over the years and in accordance with internationally used cycles, 1090 ml 36% FA solution (Dr. Grogg Chemie AG, Stettlen, Switzerland) together with 1040 ml deionized H2O were evaporated to achieve a calculated concentration of 4 g/m3. FA was not neutralized using ammonia as environmental exposure levels following purge were below legal threshold levels. Instead 50 ml deionized H2O was evaporated. The holding time for FA fumigation was approximately 15 hours at which time point data loggers and indicators (see below) were collected before the room was purged using the building’s HVAC system for approximately 72 hours.
Process Controls and Monitoring
A PortaSense II Gas Leak Detector (Analytical Technology, Inc., Collegeville, PA) fitted with an H2O2 electro-chemical sensor (0-2000 ppm) was placed in the animal room on the Alpiq 6100 fumigation ventilator facing the door to the waste corridor to monitor real-time and log H2O2 concentrations during VHP and aerosol fumigation. FA concentrations were not monitored due to lack of suitable equipment. Temperature and relative humidity in the animal room were also monitored and logged (Ebro Electronic GmbH & Co KG, Ingolstadt, Germany). A total of five loggers were placed at different positions throughout the animal room, including the box with the climate controls (see below). H2O2 chemical indicators (PCC051, Steris, Mentor, OH) were placed at different locations (N = 28 or 29) in the animal room and around the HEPA filters to visualize the extent of H2O2 distribution during VHP and aerosol fumigation. FA distribution was visualized placing chemical indicators (2401, Albert Browne, Ltd., Leister, UK) at various locations (N = 28).
Sterility Validation with Bacterial Spores
To validate the extent and efficacy of the VHP and aerosol fumigation process, biological indicator (BI) pouches containing >106 spores of Geobacillus stearothermophilus 12980 dried on stainless steel metal discs sealed in Tyvek (HMV-091, MesaLabs, Bozeman, MT) were placed, together with the chemical indicators, at different locations within the animal room and around the HEPA filters (N = 27 or 28; Figures 1a and 1b). One batch of biological indicators was used; its lot number and D-value (in 2 mg/l gaseous H2O2) were H0727 and 1.1 minutes. Upon completion of the fumigation cycle, indicators were retrieved and opened, and the discs were transferred into either Spordex culture medium (Steris, Mentor, OH) or Purple Releasat Culture Medium (MesaLabs, Bozeman, MT) and incubated at 56°C for up to 7 days.
Since no dedicated spore formulations for FA fumigations were available, FA fumigation was validated with triplicate biological indicator pouches containing >104, 105, or 106, respectively, spores of Geobacillus stearothermophilus 12980 dried on stainless steel metal discs sealed in Tyvek (LOG-456, MesaLabs, Bozeman, MT) which were placed together with the chemical indicators, at different locations within the animal room and around the HEPA filters (N=30; Figure 1c). Two batches of biological indicators were used; their lot numbers and D-values (in 2 mg/l gaseous H2O2) were P2287 (0.3, 0.4, 1.0 minute, respectively for the triplicate amounts of spores used) and P1737 (0.3, 0.3, 1.2 minutes, respectively). In addition, Attest biological indicators for autoclave validation (1262, 3M, Maplewood, MN) containing >105 spores of Geobacillus stearothermophilus 7953 on filter paper were placed at the same locations as the other indicators. Two batches of biological indicators were used; their lot numbers and D-values (at 121°C) were 2018-09JD (2.0 minutes) and 2018-12JE (2.0 minutes). Following the holding time and before aeration of the animal room, the animal room was accessed in full PPE consisting of a DuPont Tychem C suit, full facial mask and nitrile gloves, indicators were retrieved and opened, and the discs were transferred into Purple Releasat Culture Medium (MesaLabs, Bozeman, MT) and incubated at 56°C for up to 7 days. The Attest indicators were incubated in their supplied medium for up to 3 days.
Sterility Validation with Foot-and-Mouth Disease Virus
Foot-and-mouth disease virus strain O Lausanne was used for the validation of the fumigation methods. The tested virus suspension had a titer of 108.16 TCID50/ml and was frozen in aliquots.
Based on the guidelines of the German association for the control of virus diseases, 15 stainless steel discs were used as carrier material (20 mm diameter, GK Formblech GmbH, Berlin). Before use, the carrier discs were decontaminated in 1% VirkonS for 1 hour. Afterward, the discs were rinsed twice with deionized water and then placed in 70% ethanol for 15 minutes. Finally, the discs were dried under a biosafety cabinet.
After thawing, 50 µl of the virus suspension was pipetted onto the middle of the stainless steel disc and dried in the biosafety cabinet for approximately 1 hour. Afterward, the discs were transferred into the stable and placed on the allocated spots for fumigation. One disc was prepared in the same way with medium only (G-MEM BHK-21, Gibco, with 2% FBS and 5% Tryptose Phosphate Broth Solution, Sigma) for the control of the cytopathogenic effect of the fumigant. Following fumigation, the discs were placed into sterile screw top containers (Carl Roth GmbH, Art.-No. A553.1) containing 0.5 g of sterile glass beads (0.25-0.50 mm diameter, Carl Roth GmbH). One ml of cold medium was added to the discs and the containers were placed on ice. Directly before titration the dried virus suspension was resuspended by vortexing and titrated onto BHK21 cells in 96-well plates. Every disc was titrated in triplicate. After 72 hours the cytopathogenic effect of the FMDV was read and titers were calculated according to Spearman and Kärber.16,17
Since the formaldehyde residues on the carrier discs after formaldehyde fumigation was toxic for the BHK21 cells, the resuspended virus solution was purified by loading onto small Sephadex G-25 gel filtration columns (PD MiniTrap G-25 columns, GE Healthcare 28-9180-07) before titration. According to the manufacturer, there is a recovery rate of up to 95% of virus infectivity.
Results and Discussion
VHP cycles were setup based on previous experiments (12) and inactivation confirmed by using spore biological indicators (BIs; data not shown). However, even though all spore biological indicators showed a six-log reduction, FMDV could not be completely inactivated at all spots tested as shown by virus titration (Figure 1a and data not shown). Especially the indicators placed at the ceiling level of 3 m proved to be hard to inactivate. VHP is heavier than air and sinks to the floor. Concentrations at floor level were thus higher than at the ceiling with the indicators placed at the ceiling level requiring further VHP input. In fact, to show complete inactivation of FMDV in all tested spots, more than twice the amount of H2O2 (Vaprox) was required compared to the 6log reduction of the BIs (data not shown). This clearly indicates that commercially available bacterial spore formulations may not be the best choice as surrogate biological indicators for nonenveloped viruses. This notion may even be extended to other organisms including enveloped viruses. However, further experiments are required and are currently being developed by our group.
Eventually, 25 out of 27 BIs (Table 1) could be completely inactivated. The two positive BIs were placed before and after the supply HEPA filter of the animal room and thus on the edge of the fumigation envelope. VHP never reached the supply HEPA filter in sufficient quantities to inactivate the spores, as it could not be pushed into the supply air ducts during fumigation while supply and exhaust air was shut off. This is, however, no cause for concern as the supply air ducts would very likely not be contaminated during an animal experiment while the HVAC system is running. In contrast, the BIs placed around the exhaust HEPA filters showed no growth. Apparently, the amount of VHP passing by these BIs during aeration of the animal room was sufficient to inactivate the spores.
Number of Spore Biological Indicators Inactivated Out of the Total Number of Indicators Placed in the Animal Room and Around the HEPA Filters.
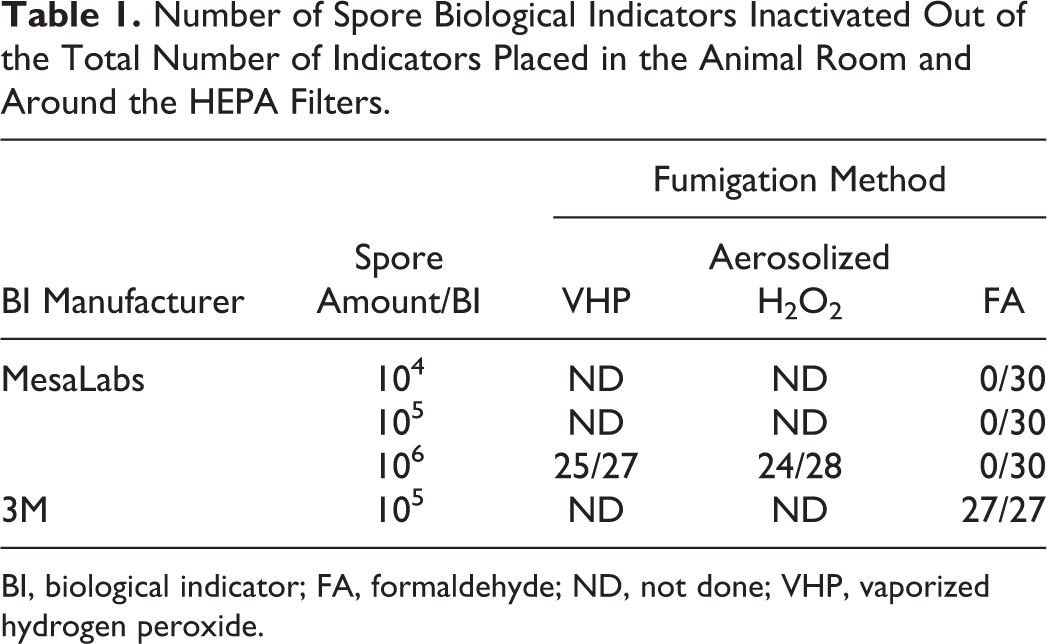
BI, biological indicator; FA, formaldehyde; ND, not done; VHP, vaporized hydrogen peroxide.
Using this cycle, all FMDV samples were completely inactivated (Table 2). Overall, a reduction of at least 4 logs could be shown for FMDV. We generally lost approximately 1 log by drying FMDV onto the stainless steel discs and an additional log by exposing the stainless steel discs to ambient temperature for 24 hours, that is, the course of the fumigation. The loss of FMDV due to drying is similar to the loss for other viruses, as shown in other studies. 18 This confirms the data published by Petit et al 11 who demonstrated that FMDV is inactivated by H2O2 fumigation. Contrary to Petit et al, however, we could not achieve a six-log reduction. The difference may be explained by the fact, that Petit et al kept their controls in the fridge while we exposed them to the environmental conditions during the course of the fumigation. This is further supported by initial experiments performed with the VHP fumigation system in a 500 x 500 x 800 mm Plexiglas box (data not shown). The ambient temperature inside the box increased to levels above 40°C over the course of the fumigation caused by the hot vapor released by the VHP generator. FMDV controls kept inside an additional plastic box and thus away from the VHP were inactivated as efficiently as samples exposed to VHP. Comparable results were obtained when FMDV dried onto stainless steel discs was exposed to vapor from deionized water instead of VHP at the temperatures recorded, that is, approximately 2 hours at 40°C or above (data not shown). Clearly, inactivation of FMDV can be achieved by increasing temperature alone, as the effect of water vapor must be negligible. Similar losses due to high temperatures have also been shown for other viruses. 10 Thus, our data more closely reflect the inactivation of FMDV by H2O2 as losses due to environmental conditions are taken into account.
FMDV Titers Before and After Exposure to Fumigants.
BI, biological indicator; FA, formaldehyde FMDV, foot-and-mouth disease virus; VHP, vaporized hydrogen peroxide.
Titer expressed as log10 TCID50.
Mean value of three BIs.
In sealed box in animal room during fumigation (no exposure to fumigant).
In sealed box in laboratory (no exposure to fumigant).
Original virus titer (24 hours at 4°C following thawing).
Mean value and standard deviation of three validation runs expressed as log10 TCID50 reduction based on the climate controls which reflect all losses due to drying onto the discs and exposure to the environmental conditions.
Following purification over Sephadex columns.
Having shown that VHP inactivates FMDV, we proceeded to test another H2O2-based method. Aerosolized H2O2 uses much lower H2O2 concentrations than VHP (7.5% compared to 35%). There is no heat input into the fumigation envelope and the risk of condensation and thus problems with material compatibility due to high concentrations of H2O2 following condensation is much reduced. Similar to VHP, aerosolized H2O2 inactivated the bacterial spores more efficiently than FMDV (data not shown). Ultimately though, all bacterial spores inside the animal room were inactivated (Table 1). Inactivation of spores around the supply and exhaust HEPA filters proved more difficult. The aerosolized H2O2 was even more difficult to get into the air ducts than VHP. Once again, all FMDV samples were inactivated showing a four-log reduction just as for VHP (Table 2). Overall, it could be shown that, regardless of what H2O2 method was used, FMDV could be successfully inactivated.
Finally, we wanted to compare the two H2O2-based methods with FA fumigation which is still regarded as the international standard. 5 We thus proceeded to repeat the above experiments, this time using the in-house FA fumigation cycle. Once again, we could show that FMDV is successfully inactivated with at least a four-log reduction (Table 2). All three methods show similar effects on FMDV and may thus be used interchangeably to fumigate rooms that have been exposed to FMDV.
Interestingly, FA fumigation showed very different results with bacterial spores (Table 1). Spores placed on stainless steel discs could not be inactivated regardless of whether the discs were placed inside their Tyvek pouch or not (discs without Tyvek were placed at positions 2, F19 and F23; Figure 1c). Spores on filter paper, however, could always be inactivated. It has been shown previously that spores on filter paper may give false positive results and are unsuitable for validation of FA fumigation. 19 Also, Macellaro et al 20 showed that, regardless of whether FA or VHP was used for fumigation, the choice of the correct indicator to show inactivation of the desired agents is of outmost importance. Seeing the difference in the efficacy of inactivating bacterial spores and FMDV with H2O2 as well as with FA, we support the notion to test fumigation methods with the agents to be inactivated rather than simply using surrogate indicators, whenever possible.
Overall, we were able to validate three different fumigation methods for FMDV showing that all three methods gave at least a four-log reduction in three consecutive runs each. One could argue that the stainless steel discs carrying FMDV were prepared with tissue culture supernatant and that the indicators lacked additional organic loads that may interfere with the fumigation. Therefore, BSA was added to the FMDV supernatant to a final concentration of 0.3 g/l, 15 and these carriers were fumigated with VHP as described above. The same reduction was achieved as for the experiments without BSA, confirming our notion that the effect of organic loads is minuscule (data not shown). Furthermore, we fumigate animal rooms only once they have been thoroughly washed down and chemically surface disinfected. Thus, any contamination left would hardly contain any organic load. In addition, it is fair to assume, that contamination would hardly be above 104 TCID50 at any one spot in the animal room following the cleanup prior to fumigation. Furthermore, in another animal trial with porcine epidemic diarrhea virus swab tests never showed any residual virus prior to fumigation unlike the samples before cleaning and disinfection which were positive (data not shown).Therefore, a four-log reduction, as shown here, will be sufficient for our purposes.
We were able to validate two different H2O2 methods and show that H2O2 is a valuable alternative to FA fumigation. Not only did we achieve the same reduction rates with H2O2 and FA, but could also show that H2O2 fumigation reduces stand-down times as animal rooms were ready for other experiments 24 hours following the start of the fumigation while FA fumigation would take 4 days until animal rooms could be handed back over to staff. Also, depending on what H2O2 method is used, H2O2 fumigation may be as economical as FA fumigation, thus obliterating the long held notion that H2O2 is too expensive as a fumigation method.
Conclusion
We validated two different hydrogen peroxide based fumigation methods (vapor and aerosol) against FMDV and were able to show that FMDV can be efficiently and, depending on the method employed, economically inactivated. Also, both methods were compared to formaldehyde fumigation and showed similar results. However, we were also able to show that the use of bacterial indicator spores may not always reflect the inactivating capacity of a fumigant against other agents, particularly viruses, and thus recommend the use of the actual target agent when validating fumigation methods.
Footnotes
Acknowledgments
We would like to thank Linda Hüsser and Urs Pauli for thoroughly reading the manuscript and providing valuable input.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partly funded by the Federal Office for Construction and Logistics BBL. The Q-Jet Superior for the validation study was provided by Sanosil AG (Hombrechtikon, Switzerland).
