Abstract
Introduction:
The number of institutions engaging in research with potentially biohazardous materials has increased, indicating a need for newly formed Institutional Biosafety Committees (IBCs) in the United States and for similar biorisk management committees located outside the United States. Our institution identified the need for an IBC due to the growth of pertinent activities on campus.
Objectives:
This article shares our experiences creating a new IBC at our institution from September 2017 to April 2019. Our lessons learned and approaches to the challenges faced may be helpful to others finding themselves with similar needs.
Methods:
In this case study, we outline IBC membership, documents, relationships with federal agencies and within the institution, creation of registration forms, and the review process. Along with our account, we have included links to helpful resources from federal agencies.
Results:
At the time of the submission of this article, we have established our IBC and reviewed two registrations.
Conclusion:
This case report demonstrates the successful creation of an IBC that works for our current institutional needs.
Introduction
Reasons for Creating an IBC
At its inception in 1893 as a teacher-training school, Southern Connecticut State University (SCSU) was primarily a teaching institution. SCSU remains a primarily undergraduate institution (PUI) serving approximately 10 000 students and 450 faculty. However, SCSU’s faculty have recently increased the quantity and scope of their research, attracting increased external federal funding. Our institution already had an established Institutional Animal Care and Use Committee (IACUC) and Institutional Review Board (IRB) but did not have an official Institutional Biosafety Committee (IBC) in place (Table 1 details all acronyms). Oversight for the use of hazardous materials was provided by the University’s Environmental Health and Safety (EHS) office. An official IBC is mandated if anyone at the institution is receiving federal funding or collaborating with investigators receiving federal funds at other institutions for work with recombinant/synthetic nucleic acid (r/sNA). As an increasingly Public Health Service (PHS)–funded institution, therefore, SCSU identified a need for new research infrastructure to maintain important government-mandated standards of compliance.
Abbreviations.
Why Your Institution Needs an IBC
A review of the current edition of the NIH Guidelines for Research Involving Recombinant or Synthetic Nucleic Acid Molecules 1 (April 2019) provides the regulatory requirements for those institutions that have received NIH funding. Section I-C-1-a-(1) notes that anyone receiving funding for this research must be at a location that is in conformity with the NIH Guidelines. 1 Section I-D clarifies this requirement further to ensure that institutions are aware that the requirement to comply with the NIH Guidelines applies to all research, including research that is funded by the NIH and research that is not. The NIH Guidelines follow up on their compliance requirements in the following 2 sections to ensure that there is no confusion as to the applicability of its requirements for institutions receiving funding (Sections 1-D-1 and 1-D-2). 1 Many grant agencies require institutional assurance that the research activities will be conducted in compliance with all federal and local regulations. An IBC can provide verification to grant agencies and journals that the research described has been reviewed and approved by an IBC.
Beyond federal funding requirements, the paramount purpose of an IBC is to help the institution ensure a safe environment for employees, students, and the general public. An IBC and associated policies require a written risk assessment and risk management plan to obtain authorization to perform research with biohazards or regulated biological materials. This provides an opportunity for the IBC subject matter experts to confirm the adequacy and suitability of the proposed protective measures. The IBC, through the requirement to report incidents or issues and periodic renewal or reapproval of registrations, establishes and maintains a relationship with the Principal Investigator (PI) conducting the research. EHS, in conjunction with the IBC, may also assist in the process by conducting site visits before and after the initiation of the project to assess the facility and work practices and ensure proper use of personal protective clothing and containment control. The IBC review process can also ensure documentation of appropriate training and competency for new laboratory personnel.
Case Report
Previous Attempts to Form an IBC at SCSU
In recent years, when new faculty arrived on campus to continue their research with biological material at Biosafety Level 2 (BSL-2), the EHS office had little experience with biosafety, and no IBC existed. This situation was challenging since faculty need assistance from a compliance standpoint when setting up a new laboratory and acquiring Connecticut Department of Public Health (DPH) certification, according to the State Regulations (Section 19a-36-A25 to 19a-36-A33). 2 While the institution provided facilities for the researcher, there was no IBC or biosafety officer (BSO). EHS had 1 chemical safety officer, who led the attempt to establish an IBC along with support from the department hosting the new faculty member, but this committee was unable to gain traction: membership was comprised primarily of nonspecialists, and meetings were not consistent enough to establish a charter or registration workflow. Additionally, the EHS employee driving the committee left the institution. While this attempt was unsuccessful, the need for an IBC had been identified by the institution. Technically, an institution is not required to have a BSO unless at least 1 laboratory performs higher risk work at BSL-3 or works with culture volumes greater than 10 liters (NIH Guidelines section IV-B-1-c). 1 With limited resources and few faculty working with biohazardous material, it was not prudent to hire a dedicated BSO. At our institution, some of the responsibilities of the BSO are addressed by the new director of EHS, who is experienced in chemical safety and risk assessment. In addition, we invited external IBC members with extensive biosafety knowledge and training. With this plan in place, the time was now optimal to initiate the establishment of an IBC. The result was made possible by a close collaboration between EHS and the Sponsored Programs and Research (SPAR) Office within a newly established Division of Research and Innovation (DRI).
Where to Start: Logistics
Selection of IBC Members
Deciding the appropriate membership composition and number is important when creating an IBC. One needs to balance the varied expertise in the group and the potentially unwieldy size that leads to difficulty in scheduling meetings, which prohibitively slows turnaround time for registration/proposals. According to NIH Guidelines, an IBC must have at least 5 members, including 2 members who are not affiliated with the institution (NIH Guidelines section IV-B-2-a-(1)). 1 Membership composition in terms of area of expertise was considered to cover the STEM (science, technology, engineering, and math) campus departments that might work with potentially infectious materials. This allows for committee insight into the inner workings of their respective STEM departments, and vice versa allows for departments to feel connected to the IBC as they have a colleague representing them and their respective challenges, contributions, tools, and facilities. For example, if the institution conducts recombinant work with plants, an expert in plant research would need to be appointed as a member of the IBC as outlined in the NIH Guidelines section IV-B-1-d. 1 Experts in specific areas pertaining to research needs can always be added ad hoc to the committee as consultants for specific registrations. If an investigator were proposing work involving vertebrate animals, a trained veterinarian would be added ad hoc to guide the PI and IBC as animal work can often present a significant biosafety risk. Importantly, note that student or occupational health should be consulted if a research protocol involves a risk that could be mitigated by immunizations or postexposure prophylaxis.
One should examine the needs and research areas of his or her institution when thinking about the composition of the IBC. When considering membership, you must ask, “What materials and techniques are our faculty working with?” The answers will guide you in your selection of members for your IBC. The NIH Office of Science Policy (OSP) online registration page for NIH-funded IBCs guides institutions through the process, helps identify the various categories of research conducted, and ensures that relevant experts are on your committee (https://www.ibc-rms.od.nih.gov). 3
Originally, our strategy for membership roster was to include many faculty and administrative members (17 in total). It quickly became clear at our first meeting that reorganization of this Leviathan was necessary due to scheduling difficulties and the length of time required to obtain valuable feedback from every member. After the chairperson was elected, she immediately reached out to 3 other IBC chairs at universities around the country to survey best practice. In addition to garnering valuable advice on IBC documents and procedures, other IBC chairs offered advice on membership composition and size. The first order of business was to decrease the committee’s membership to a more manageable size. Next was to find 2 qualified external members. Luckily, SCSU shares its broader community—the Greater New Haven region—with many universities doing excellent research, and along with these resources come experts in biosafety management. One external member is a faculty member in biomedical science and the chair of their university’s IBC. The other external member is a biosafety professional working within EHS with a track record in microbiology at a large research-intensive institution (and an author of this article: Dr Schniederberend). This member also resides in the county where SCSU is located and therefore serves additionally to represent the local community. These external members are aware of their responsibilities to represent the public around our institution and serve its interests. A community member can be anybody residing in the local community (e.g., members of a public health department, science teachers at secondary schools, or even real estate agents). Our committee is currently comprised of 4 faculty members in science departments at SCSU, 2 external members, and 2 nonvoting members (Table 2). Adding our 2 nonvoting members for administrative, compliance, and guidance purposes was important. The director of our SPAR office assists with federal grant guidelines and opportunities available for proposal workflow and training offered by the institution as well as recruiting administrative support from the SPAR office such as booking meeting space, recording and reporting meeting minutes, and helping to move our paper-based registration system to an online database. Our institution’s EHS officer is our other nonvoting member, who provides valuable advice and guidance to faculty preparing their registration forms. Importantly, no IBC member may be involved in review/approval of a project in which the member has a financial interest, as outlined in the NIH Guidelines section IV-B-2-a-(4). 1 This size, 8 in total, has worked well as we have ease in scheduling meetings and reviewing registrations in a timely manner.
Current IBC Roster at SCSU.
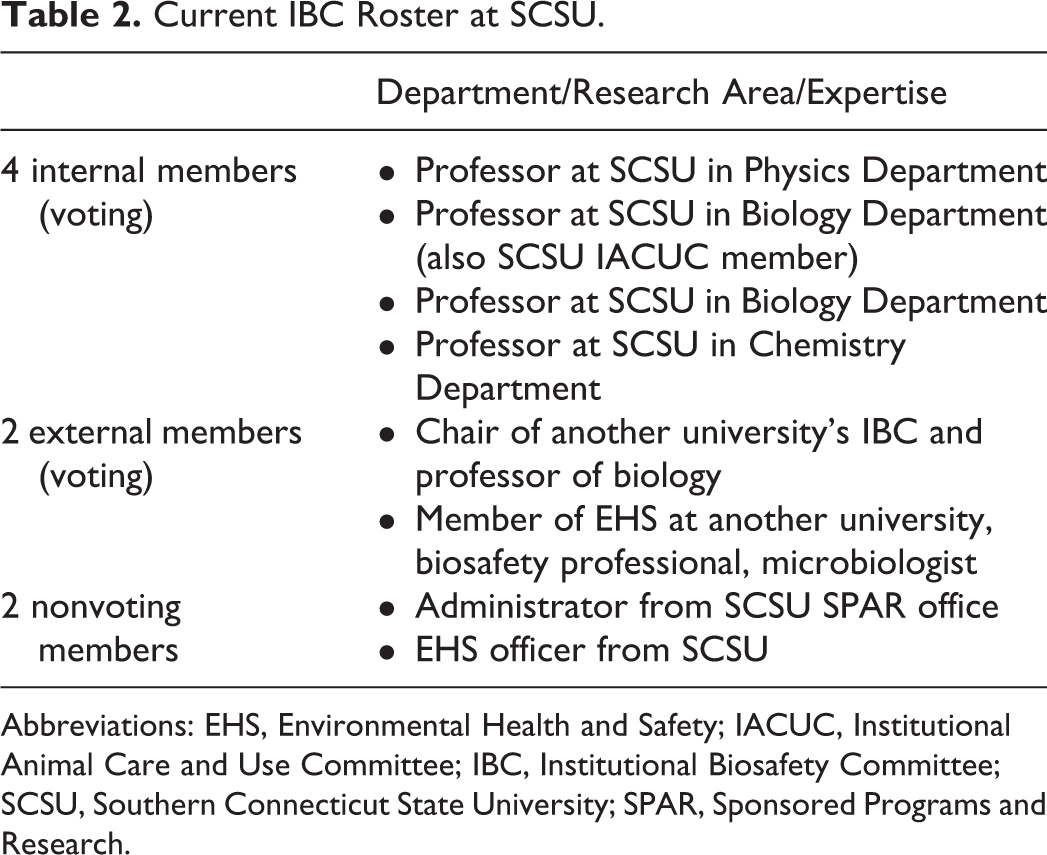
Abbreviations: EHS, Environmental Health and Safety; IACUC, Institutional Animal Care and Use Committee; IBC, Institutional Biosafety Committee; SCSU, Southern Connecticut State University; SPAR, Sponsored Programs and Research.
Details on IBC Membership
One consideration is length of term for both the chair and the members. Initially our members, both internal and external, were perhaps wary of signing up for this new committee that was yet untested. The IBC supported an initial 1- to 3-year term for each member (length of appointment selected by initial members) and then a staggered 3-year term for any new members appointed such that longer-standing members would always be present on the committee. The chair is elected annually by members of the IBC and can be reelected as chair for subsequent years. Our current model is that the provost formally appoints members (selected by the chair in consultation with DRI). We have already modified this 1- to 3-year staggered term of membership as our first year and a half included an incredibly steep learning curve and we wanted to reach a certain level of functionality before adding new members to the committee. We have also already considered changing the term for chair as switching too often can lead to a lack of cohesion, dysfunction, and loss of institutional memory. Chairs can serve long terms, and this may prove valuable, especially in the transition from the nascent phase of the IBC at the institution.
IBC Meetings
For meetings to officially proceed, there must be quorum, which we define as 50% of the total committee plus 1. Thus, we require at least 5 members to be present since the complete IBC roster is composed of 8 members. In addition, we ensure that at least 1 nonaffiliated (external) member is present for our meetings. For a vote to proceed, there must be 5 voting members present (Table 2). Email as a form of communication is not permitted for any meetings, registration commenting, or voting. All meetings are live in person or by other electronic meeting platforms that allow for real-time interaction. Our IBC currently meets 4 times per year. We have many items on the long-term agenda that we are addressing, and the need for timely review of nascent registrations dictates a more frequent meeting schedule.
Documents Needed for Your IBC
Charter
We created a charter that outlines the responsibilities of the IBC at SCSU, along with details of membership and meeting procedures. The charter defines the length of membership and specifics about the selection of the chairperson. The charter also contains details on the approved timeline for a new registration as well as a timeline for the renewal of a registration.
Registration Form
When faculty want to submit a new registration, they must complete a form indicating the nature of their proposed work along with proof of relevant training. Faculty are strongly advised to work with EHS while developing the Safe Operating Procedures (SOPs) for their registration form, especially regarding risk assessment and proper safety precautions. The most important items to include in the registration are organism, volume/amount and source, procedures adding additional risk such as centrifugation or the use of sharps, location of the work, and training plan. The training plan can utilize online-training modules located on electronic learning portals and must be completed by the PI as well as anyone whom the PI supervises, either in the research laboratory or the classroom, who will be using the proposed material. The PI is then responsible for ensuring that all personnel affiliated complete required training modules as outlined in the registration. While that is our current model, our IBC is considering adding an optional amendment to registration document where each individual working on the proposed research (including undergraduates) would be included along with their proof of training from both biosafety modules and the PI. The needs of the specific institution’s research and teaching should be considered when creating registration documents.
At our institution, we have two separate registration forms (to view these documents, go to SCSU’s website) 4 —one for r/sNA and the other for all other biohazards and potentially regulated biological materials (eg, materials infectious to plants, humans, and animals). Investigators can consult the NIH Guidelines to see if their work with r/sNA is exempt from oversight (NIH Guidelines section III-F). 1 If human pathogens are involved, PIs should review Appendix B of the NIH Guidelines to identify the starting Risk Group for the proposed human pathogen they plan to utilize. 1 PIs must be made aware that Risk Groups are only the starting point of the risk assessment process (Table 3). Consideration of the proposed procedures, equipment, and even the personnel performing the work is part of the process required to select the final biosafety level (BSL) for the proposed research.
What Needs to Be Registered with an IBC.

Abbreviations: IACUC, Institutional Animal Care and Use Committee; IBC, Institutional Biosafety Committee.
How to Run an IBC
Registration Review Process: Current and Future
Currently at our institution, registrations submitted to the IBC are paper-based or electronically submitted via email to the chair. The chair then logs the registration in a spreadsheet, assigns a code, and distributes the registration documents to the rest of the committee. This system leaves a lot to be desired as it is time-consuming moving paperwork (virtual or physical) from office to office and office to committee members after photocopying and printing; moreover, investigators submitting registrations through these means are not able to access the real-time status of their registrations as they move through the evaluation process. We are now moving all institutional research compliance to an online platform, which provides IACUC, IRB, and in the coming months, IBC registration submission. Anyone affiliated with the institution can create and submit protocols/registrations using this system. Additionally, at any point, investigators can see the status of their registrations. As we evolve in our workflow of proposals, we have been pleased how online registration management systems facilitate transparent submission and review for research compliance. This system also identifies errors and omissions in submissions before PIs submit to oversight committees, which saves time for both the PI and the committee. Further, online management systems can be configured easily to include more detailed drop-down menus in decision trees so that PIs with simple, straightforward applications and those with more complex registrations are able to use the same registration document.
PIs can consult with EHS regarding the submission of their IBC registrations. We work closely with EHS to help ensure that a very thorough prereview of proposals is conducted prior to submission and that all required information is provided (Table 4). Once the PI has completed a preliminary review process with EHS, his or her proposal is submitted to the IBC chair. The chair then distributes the registration form to the other members of the IBC, and it is discussed at the next scheduled IBC meeting (Figure 1). The PI is welcome to attend the IBC meeting discussing his or her registration, offer clarification for any questions the committee may have, and obtain feedback from the committee, except during the actual vote. After the meeting, the IBC chair drafts a letter summarizing the result of the IBC’s review and informs the PI about the decision on the proposed registration, which is delivered by email and hardcopy. Notification of the PI is outlined in the NIH Guidelines section IV-B-2-b-(2). 1 The PI then can revise his or her registration form, if necessary, and the IBC chair, on behalf of the committee, will then ensure that all recommendations are met.
Which Information Needs to Be Provided for Registration with an Institutional Biosafety Committee.

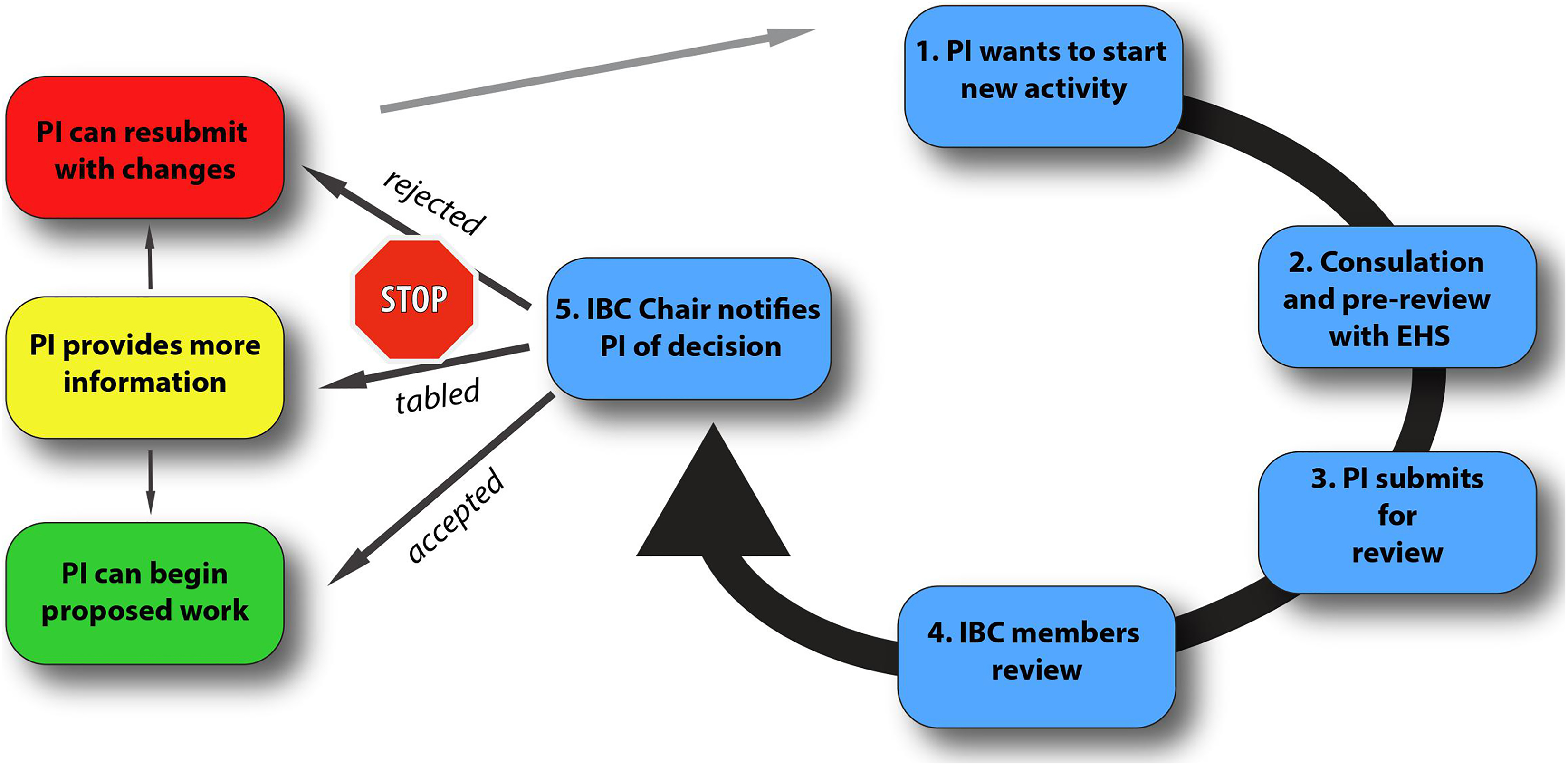
Institutional Biosafety Committee review process.
Working with Faculty
Informing faculty when an IBC has been formed at their institution and how it is easily accessible is important. The most effective way to do this is to bring your program out in the field, ideally through face-to-face communication. Either the IBC chair or EHS can hand-deliver registration documents to faculty. We are implementing faculty outreach on how to submit a registration form for research and classroom work involving potentially biohazardous material on a targeted level. From the IBC chair’s desk, she is meeting with chairs of STEM departments to help target specific faculty who need to submit registrations. This is important as many faculty members have not submitted these documents previously (so have limited experience), and we want to work with faculty to assist in their compliance toward a safe and effective environment for their research. Additionally, emails to the campus community from our provost, DRI executive director, and research integrity officer explain the need and direct people to training and oversight committee chairs. Our EHS office also meets with all new faculty, regardless of department, and this outreach when arriving on campus helps a great deal with education and compliance. Now that we have created a registration form for PIs for their research, the next step is to work with faculty leading teaching laboratories to assist them with the completion of the registration forms to identify what biohazards or regulated biological materials may be used. To help PIs correctly classify their proposed research according to the NIH Guidelines, PIs can access the SCSU IBC charter electronically, which contains a link to the NIH Guidelines. The NIH Guidelines recommend BSL for different sections. In addition, the authors generated a help guide for PIs at SCSU to help classify NIH sections and BSL levels for their proposed research. The help guide can be accessed on SCSU’s IBC’s website. 4
As SCSU has a strong base in teaching students interested in entering health professions and biotech fields, many of our courses could potentially be using Risk Group 1 and Risk Group 2 research materials. Although the work with r/sNA is likely exempt, the IBC and EHS office work together to review the research materials utilized and ensure that the laboratory work is conducted safely according to federal regulations. The registration and approval of potentially biohazardous material in the classroom are good ways to maintain a safe environment for students, employees, and the general public.
Working with the Administration
Support from high-level administration personnel is important so that no faculty feels that he or she is alone in policing peers. At our institution, SPAR (as an office currently of DRI) offers some previously described administrative support. Currently, the chair of the IBC handles all documentation; this includes distribution of the submitted registration forms to other IBC members and drafting and sending approval letters as well as reporting annually to the NIH OSP. If an incident (eg, noncompliance, accident, or exposure) occurs, the chair of the IBC informs the OSP. These are all things that could be facilitated through SPAR or similar administrative support. The constant open line of communication that exists between the DRI, director or SPAR, and the provost is incredibly valuable at our institution.
Another huge way that the administration can support an IBC is to allow the chairperson to receive course release time to work on the duties of the IBC. For smaller institutions and PUIs, this early form of organization support is critical to create the IBC. Especially in the first few years when all documents and registrations must be created from scratch, a tremendous amount of time can be spent on this committee. The chair of the IBC currently receives 3 credits (1 course per semester) release time per semester, which is the same amount given to other chairpersons of regulatory committees (IACUC, IRB) on campus. This is eventually assuming similar number of registrations as each of these committees; however, this can vary significantly at different institutions.
Working with EHS
Our EHS is housed within SCSU’s facilities department. Through laboratory inspections, EHS ensures that facilities meet the specified containment criteria, yielding a safe work environment for the ongoing research. Conformity with Appendix G of the NIH Guidelines 1 and with BSL-1 and BSL-2 containment practices from the CDC/NIH Biosafety in Microbiological Laboratories, fifth edition 5 is verified. The visit also includes inspecting emergency equipment such as eye washes, emergency showers, and fire extinguishers. Another aspect is the distance and accessibility of emergency equipment from their point of use. EHS also ensures the proper functioning of chemical fume hoods and biosafety cabinets by verifying that they have been tested and certified at least annually after initial certification. Physical safety aspects are on the list of things EHS is checking as well, including ensuring safe egress, basic electrical safety, eliminating trip hazards, and proper and secure storage of gas cylinders. Any safety issues that EHS observes are addressed at the time of the laboratory visit wherever possible. For certain registrations, and especially if directed by the IBC, EHS will perform a work observation using mock research materials when high-risk research will be performed. This assessment provides guidance for the IBC in making its final assessment of the proposed research. Additional work practice training may be required before the registration can be approved. Institutional manuals provide information on safe operating procedures. For examples, Chemical Hygiene Plan, Hazardous Material Communication, and Biosafety Manual can be viewed under SCSU’s and Yale’s Biosafety Manual (Table 5).
List of Helpful Resources.

We cannot stress enough the importance of having the EHS group meet with new faculty as part of a welcome or orientation meeting to describe IBC policies and other institutional requirements pertinent to biosafety. The BSO or EHS staff member is in a unique position to establish strong relationships early on with PIs and new personnel. Members of the EHS department and/or the BSO are usually the personnel who respond to biohazard emergencies or other incidents and should be cognizant of the NIH reporting requirements (NIH Guidelines section IV-B-2-b-(7)). 1 As a follow-up to this response, these emergency responders will partner with researchers to identify corrective measures to reduce the opportunity for reoccurrence.
Registering Your IBC with the NIH
Once our IBC was finalized, we registered the IBC with the NIH’s Registration Management System (NIH-RMS). 3 We found this to be efficient and straightforward. Either the chairperson or his or her designated administrative support can register the new IBC. In your registration, you must include the current IBC roster denoting the role and a 2-page CV of each member. Reregistration is required each year after the initial registration for the required annual update.
Training Requirements
IBC members as well as researchers, students, and facilities employees working with potentially biohazardous materials should have easy access to training specific to their needs. For example, if a PI wants to work with human pathogens or any human material (this includes human cell lines), he or she must complete annual bloodborne pathogens training. If a PI wants to work with clinical specimens, he or she must complete Health Insurance Portability and Accountability Act (HIPAA) training. We provide these trainings through our online learning portal. Many institutions already host online training portals through their human resources office or the EHS department. PIs, students, and facilities employees can simply print or include a screen shot of their successful completion of required training modules to accompany their registration. The IBC, alongside EHS, can also recommend additional training that should be completed.
Web Space for Your IBC
We have a website, hosted by the institution, located under the DRI Research Protection Program, 4 which includes links to other institutional committees like IRB and IACUC. Our IBC webpage includes a brief descriptive paragraph about the mission of the IBC, along with 4 links—FAQ, Charter, Registration Form for Experiments with r/sNA, and Registration Form for Experiments with Material Infectious to Humans, Plants, or Animals. Since the NIH recommends that IBCs open their meetings to the public (NIH Guidelines section IV-B-2-a-(6)), 1 having the schedule of meetings on our website is one way to inform the public of the meetings.
Reflections
Relationships and Interactions with Related Oversight Committees
Open communication among the IBC and other research compliance committees such as IRB and IACUC is important. When researchers want to initiate a study that requires approval from multiple compliance committees, our IBC wants to be able to communicate any concerns identified in projects that involve biohazards or other potentially infectious materials. The proposed research must be approved by each committee. When one institutional committee approves the registration, only a conditional approval should be given. No research should be allowed until all required committees have provided approval. On our website, we have created a flowchart to help investigators determine which committees’ approvals are required for their research. One suggestion nicely modeled at Yale University is to require a research “registration start-up meeting,” especially when more than one approval from multiple institutional committees is required (eg, IBC and IACUC). This requirement in an approval letter will inform a PI that as a condition of authorization, a meeting involving representatives from all relevant groups must be held to review the unique aspects of the research. This meeting could include a representative from the IBC or EHS, a representative from the IACUC or a veterinary representative if applicable, the PI or his or her designee (lead researcher), and an IRB member or clinical trial coordinator if applicable. This not only ensures that all applicable groups meet to verify that the conditions of their approvals are enacted, but it also helps the PI with any questions that he or she may have for each of the approval groups.
One way to facilitate open communication among institutional committees is to encourage cross-membership whereby a member of the IBC might also serve on the IACUC or a member of IRB might also serve on the IBC. Another way to encourage this interaction is to have a research oversight committee, as we do, comprised of chairs or members from IRB, IACUC, and the IBC, along with SPAR and DRI. If (in addition to IBC approval) a federal agency needs to approve the proposed research, the IBC helps the PI to submit the appropriate application. This is the case for Dual Use Research of Concern (DURC) 6 and Select Agents (SA) 7 (Table 3).
What Happens if Researchers Do Not Comply?
The IBC has the authority to suspend research if needed (eg, if activity is noncompliant with the NIH Guidelines 1 ). EHS assists with monitoring for compliance with periodic inspections to ensure a safe work environment. Any significant findings or issues are reported to the IBC along with a warning delivered to the PI. The IBC reviews each report on a case-by-case basis. The IBC then works with the PI and EHS to resolve issues of noncompliance so that once the violation has been corrected, research can continue. The IBC follows up with a report to the NIH OSP, where applicable, as detailed in the NIH Guidelines section IV-B-2-b-(7). 1 The initiation of nonexempt r/sNA research without IBC approval is an example of a reportable incident to the NIH OSP. Any significant research accidents or illnesses involving r/sNA research materials are also examples of a reportable incident.
Our First Registration Review
The chair worked with a faculty member to create and submit the first registration that our IBC reviewed. This registration was for classroom activities in an introductory microbiology classroom. Going through the process from start to finish with an actual registration was extremely helpful as we identified many important areas that needed to be addressed on the levels of facilities and state regulations as well as the registration document itself. We encourage everyone to review a registration as soon as possible, even if it takes longer to gain compliance across the board, as this will help move the committee and the institution forward in their goals to have a safe and effective laboratory environment.
Conclusion
Where We Are Now
Our role was to establish an IBC and create a system for addressing r/sNA and other biohazard registrations at the research and classroom levels. We have successfully accomplished this. We have set up a system that meets our current needs. At the time of the submission of this article, we have reviewed 2 registrations. Our goal is to have all SCSU labs registered with the IBC by fall 2020.
At SCSU, we consider a successful IBC to be one that has a transparent process that is clearly communicated to the university community, responsible and excited committee members, an efficient yet thorough turnaround process, and a healthy relationship between IBC and EHS as well as PIs and EHS and PIs and the IBC. Our goal is to allow for freedom of research while providing a safe workplace at SCSU. Additionally, we want cost-efficient and streamlined waste management with the appropriate level of containment of potentially infectious material to ensure the health of the general public. We hope that the future of our IBC will establish justification for facility upgrades as needed for safety and efficacy of research and teaching at our institution.
Helpful Resources
Table 5 provides a list of references for further details regarding specific biosafety concerns and regulations (Table 5).
Footnotes
Acknowledgments
Thanks to SCSU Executive Director, Research & Innovation, Dr Christine Broadbridge; Amy Taylor, SPAR director; and Julianne Fowler for careful reading and thoughtful insight in the preparation of this article.
Ethical Approval Statement
Not applicable to this study.
Human and Animal Rights Statement
Not applicable to this study.
Informed Consent Statement
Not applicable to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Jeffrey is funded through a Connecticut State University AAUP grant for her research.
