Abstract
Introduction:
Existing biosafety guidelines and biosecurity policies do not adequately address the Increased deployment of mobile high-containment biological laboratories (MBSLs) to manage disease outbreak response demonstrates potentials for rapid diagnosis of risk group 3 and 4 pathogens and allows for research under the biosafety level (BSL)-4/3 environment.
Objectives:
The primary objective of this study was to evaluate limitations in current biosafety and biosecurity guidelines to address the unique operational environment and new and varied international stakeholders involved in these deployments.
Results:
Current biosafety best practices guidelines and biosecurity policy and regulatory directives are crafted mostly toward conventional high-containment biological facilities and may not be readily applicable to the MBSLs. Although the pilot MBSLs reported in published literature have adopted multiple safety precautions to reduce exposure risks and have modified process flows in the constrained space to basically meet the proximate requirements for epidemic response, there are no systematic risk assessments for these sophisticated BSL-4/3 mobile laboratory infrastructure—deployed in locations with poor public health infrastructure backup and security problems and handling highly pathogenic agents in constrained environments—with potentials for failure, the nature and extent of which are yet to be determined. The probability of potential occupational exposure and risks to the local general environment may require a reassessment of the biosafety best practice management procedures for MBSLs to deal with these contingencies.
Keywords
Mobile laboratories are invaluable resources and are deployed on a regular basis for public health surveillance, environmental monitoring, and field research studies. US military and civilian agencies such as the Environmental Protection Agency, Drug Enforcement Administration, Federal Bureau of Investigation, Department of Homeland Security, US Geological Survey, and US Department of Agriculture are all involved in the development and deployment of mobile laboratories. However, many mobile laboratories currently operational are used for environmental monitoring and involve the standard plethora of analytical instruments, climate control systems, and materials for wet chemistry laboratory work. 1 A variation of the mobile laboratories includes modular units that are transported and drop-shipped at research locations and, as needed, used as temporary facilities or integrated with existing laboratory facility. These categories of mobile laboratories are well-established field resources and do not constitute the subject of this study.
The subject of this study are the mobile high-containment biological laboratories, referred to as MBSLs, introduced in the past few years in response to the major Ebola virus disease (EVD) outbreak in 3 countries of West Africa. The MBSLs provided highly critical outbreak investigative resources in the affected region.2,3 The past few years have witnessed deployment of high-containment biological laboratories in disease outbreak investigations involving handling, storage, and field studies of highly contagious risk group 3 and 4 pathogens. This trend has introduced a general concern over the biosafety and biosecurity at these mobile research facilities as well as the potential adverse impact on public health and the environment.4,5
The European Union (EU) funded the Mobile Laboratory Capacity for Rapid Assessment of CBRN Threats located within and outside the EU (MIRACLE) consortium and developed a set of scenarios where MBSLs could add value in outbreak response. 6 The deployment of pilot MBSLs for the EVD outbreak in West Africa provided the World Health Organization (WHO) and the MIRACLE consortium with additional data on biosecurity policy implementation and international conventions. Based on a detailed scenario-based investigation, the MIRACLE consortium concluded that (a) different concepts in mobile laboratories may be needed to meet location, threat specificity, and crisis intensity; (b) MBSLs must have backup options of larger capacities as part of the deployment plan; (c) capacity development and unique training are crucial for successful operation; and (d) deployable modules must address gaps related to harmonization and standardization process, interlaboratory exercises, operational deployment risks, and sustainability. 6
The category of MBSLs considered in this assessment will consist of either laboratories that exist inside trucks, vans, or other “on-wheels” infrastructures or laboratories where modular equipment is brought to a site, hooked up, and operated. Both types of MBSLs have been used in past epidemics, such as the 2014 to 2016 EVD epidemic in West Africa, and both are able to be deployed to rural areas as needed. These mobile laboratories are used to perform field and laboratory work under biosafety level (BSL)–3/4 conditions involving exotic, emerging, or highly biohazardous indigenous pathogens. The assessment study does not include mobile laboratories that target public health work under the BSL-1/2 conditions.
MBSLs are most advantageous during an outbreak, as seen with the 2014 to 2016 EVD epidemic causing a major public health crisis in the West African countries of Guinea, Libera, Sierra Leone, Nigeria, and Mali.2,3,7 These pilot efforts involving field-deployable high-containment Ebola diagnostic laboratories were reported as cost-effective, practical alternatives for rapid deployment for training and epidemic response functions. Although these laboratories were performing clinical disease diagnostics, sample processing, diagnostic tests, and storage, they nevertheless handled large volumes of highly infectious specimens.2,3
MBSL-3/4 have aided in the rapid diagnosing and biosurveillance of many high-risk pathogens, including EVD and human immunodeficiency virus (HIV). These pilot mobile laboratories have used processes such as quantitative real-time polymerase chain reaction (PCR) assays to successfully decrease the time to diagnose a patient with Ebola. 8 One such example comes from an MBSL that was able to decrease the diagnosis time from 2 to 5 days to 3 to 5 hours. 9
Although it is less likely that an adverse incident would occur at one of these MBSLs, if one did occur, then the high-risk nature of the agents used could potentially exasperate the adversity of disease outbreak. Recognizing the attendant risks, some policy planners and researchers have suggested a more operationally oriented mobile high-containment laboratory infrastructure for specific operational deployments, pandemic response contingency operations, or mission-specific laboratories. While meeting the stringent biosafety and biosecurity requirements, this would provide tailored, targeted laboratory service capabilities for easy deployment at remote locations.2,6,10,11
According to a 2013 Government Accountability Office (GAO) assessment, although considerable expansion in the BSL-4 and BSL-3 laboratory infrastructures occurred during 2001 to 2009, no single US federal agency was responsible for an overall assessment of the requirements for a maximum-level biocontainment facility and national standards for laboratory design, construction, commissioning, operations, and maintenance. According to the report, this increased the risk of laboratory mishaps and potentials for misuse. 12 Key recommendations from this study were to determine the number, location, and mission of BSL-4 laboratories needed to meet national requirements and, in consultation with the scientific community and the industry, develop national standards for designing, constructing, commissioning, and operating high-containment laboratories, specifically including provisions for long-term maintenance. 12
Current guidelines are in place for stationary BSL-3 and BSL-4 facilities. Although public health agencies and research teams deploying and operating these units may have their own independent specifications and operational guidelines, there are no single, unified operational guidelines available to these mobile laboratories. 11 It is implicit within the existing framework that the operational guidelines apply equally to both fixed and mobile laboratories.
Approach and Outcomes
High-Containment Laboratory: Current Landscape
Figure 1 illustrates the number and distribution of BSL-4 and BSL-3 laboratories in the United States. The past 2 decades have witnessed a proliferation of high-containment biological laboratories in the United States and throughout the world. According to the latest published reports, there are a total of 8 BSL-4 and 221 BSL-3 laboratories in the United States, with most of them built and commissioned after 9/11 (GAO, 2013).
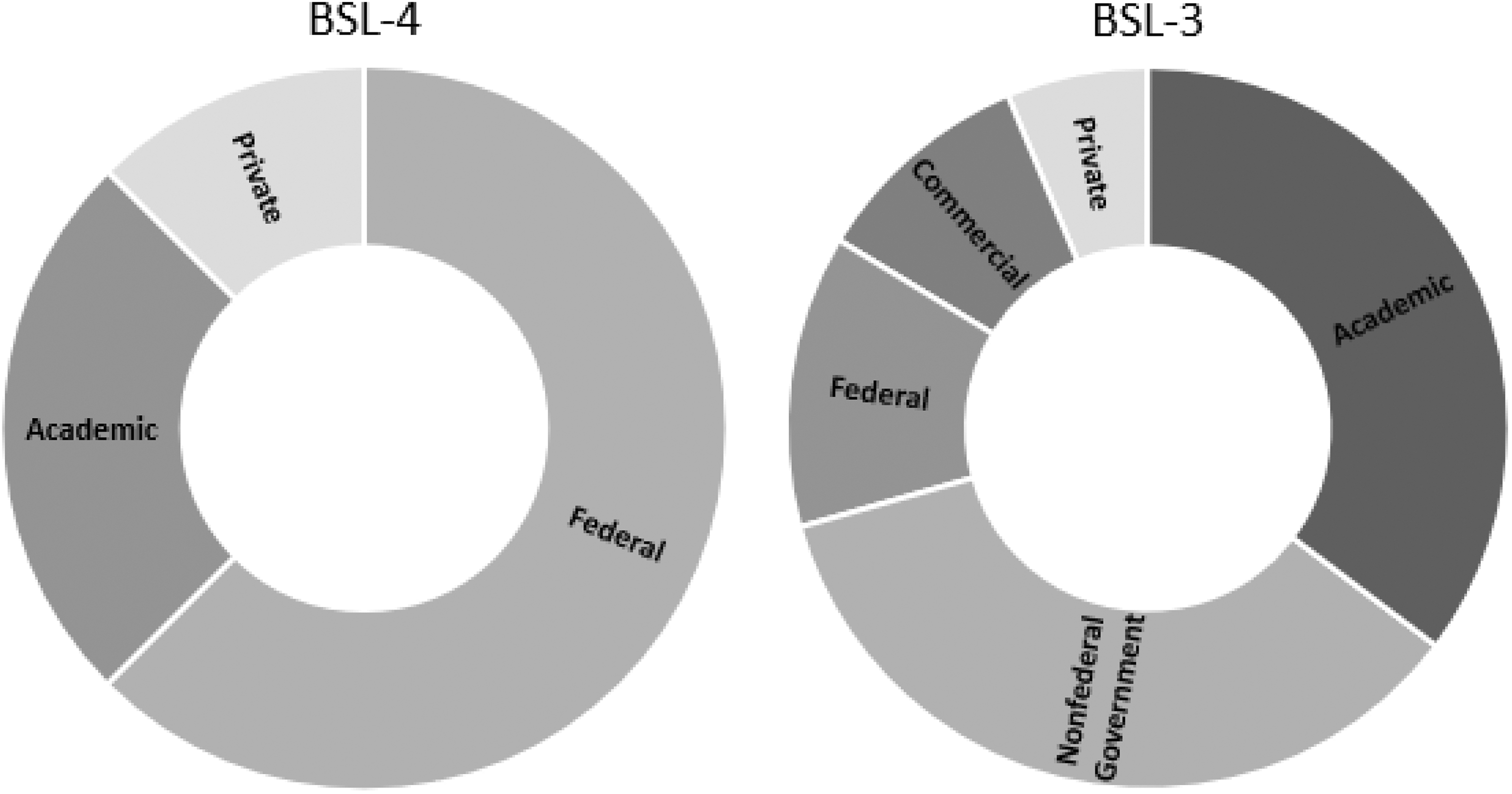
Number and distribution of the biosafety level 4 (BSL-4) and biosafety level 3 (BSL-3) laboratories by sector in the United States. BSL-4, n = 8; BSL-3, n = 221.
Figure 2 illustrates the number and distribution of BSL-4 laboratories worldwide with the EU. European community-funded European Network of Biosafety Level 4 laboratories (Euronet-P4) were established in 2005 to basically enhance the laboratory capacity in the EU, which accounts for 45% of the BSL-4 capacity in the world. More recent additions from Asian and African counties now account for 17% of the total global capacity. 13
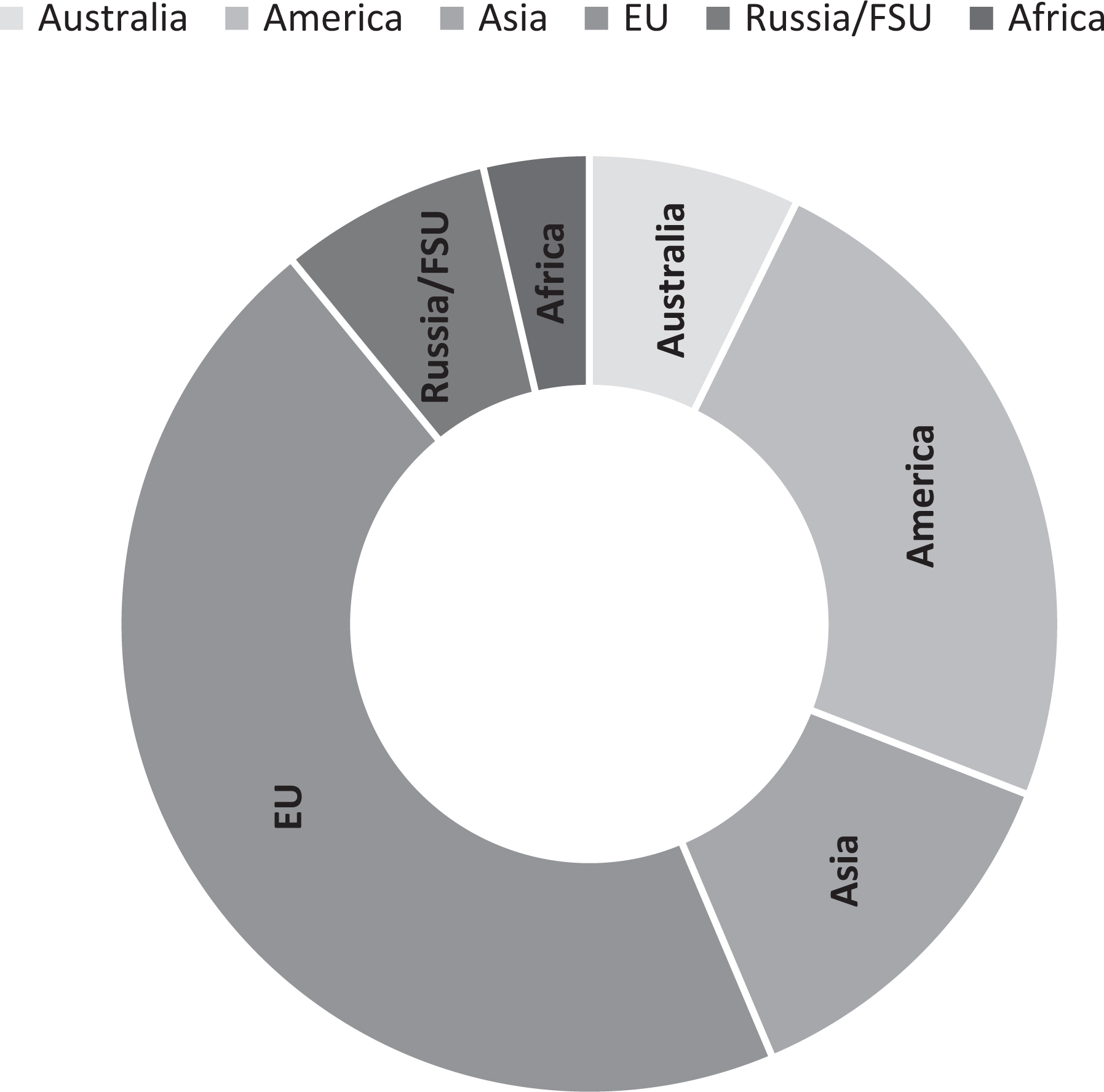
Number and global distribution of the biosafety level 4 laboratories (n = 55).
Figure 3 illustrates increases in BSL-4 laboratories worldwide, with a significant increase in the post-9/11 period. As shown in the figure, the post-9/11 period has had a nearly 300% increase in the number of BSL-4 laboratories. It is noteworthy that the proliferation of high-containment biological laboratories in these regions is increasingly found near urban and high-population areas. It is estimated that 1.8% of the world population lives within the commuting belt of a BSL-4 laboratory. This is a 4-fold increase from 1990, when only 0.57% of the world population resided within the commuting belt of a BSL-4 laboratory. Potential reasons for this dramatic increase are due to the increasing number of BSL-4 laboratories and that new facilities are mostly established in high-density urban areas.4,5
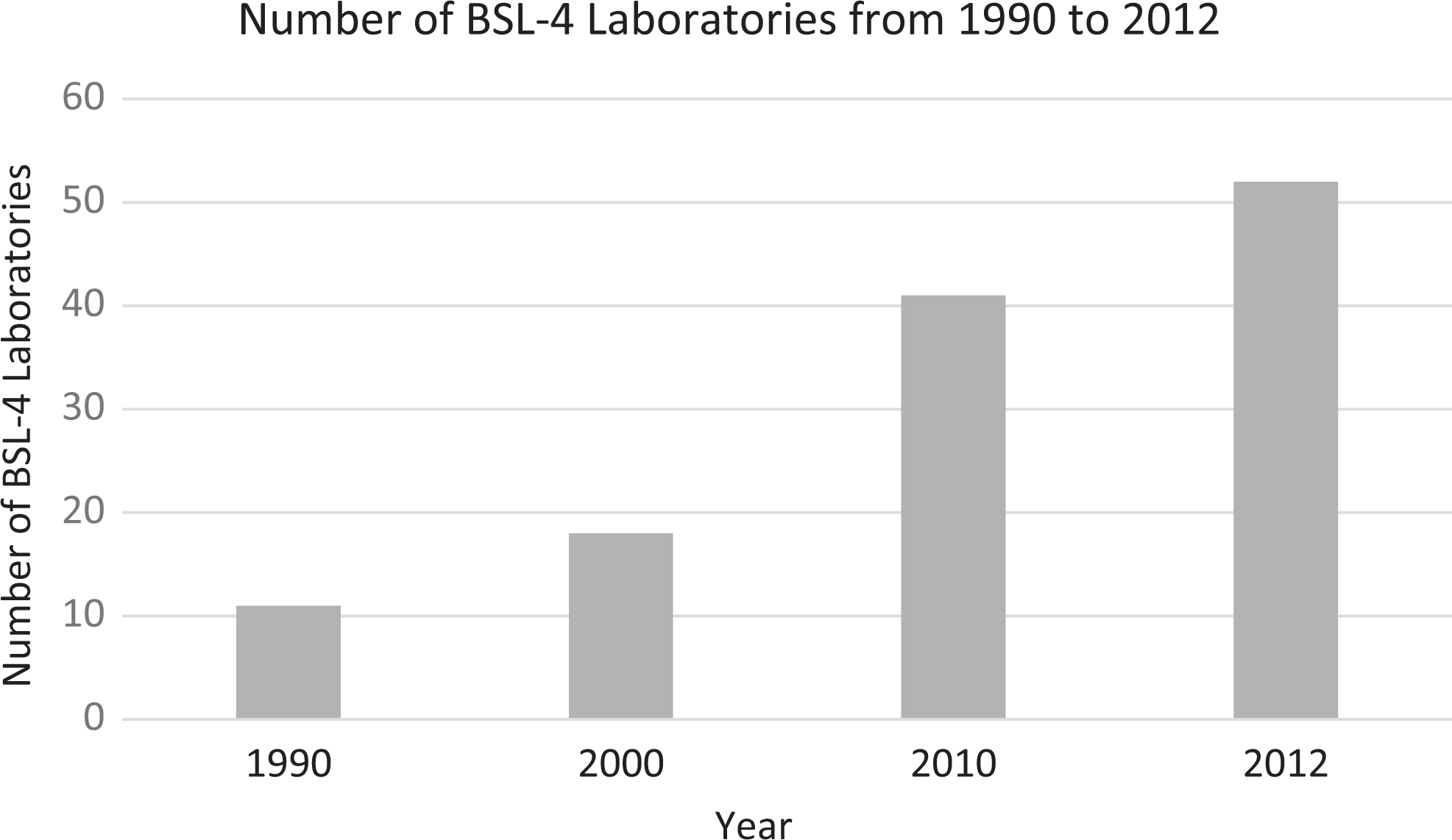
Increase in biosafety level 4 laboratories worldwide from 1990 to 2012.
The 2004 Homeland Security Presidential Directive 9 (HSPD-9) directed the creation of secure, state-of-the-art biocontainment laboratories for the purposes of research and development of exotic disease diagnostics capabilities, including zoonotic diseases of concern. The past decade has witnessed a proliferation of high-containment laboratories, particularly expansion of BSL-3 laboratories widespread across the country. With funding from federal government, most state governments now have BSL-3 capacity for diagnostic and analytical services. The rapid increase in the number of high-containment laboratories has introduced an overarching concern for the biosafety and biosecurity at these facilities as well as the potential public health and environmental impact they could have as many of these laboratories are found in urban settings. 5 Furthermore, proliferation of biocontainment laboratories has introduced additional biosecurity-related policy questions on the optimal size, type of the containment laboratories, increase in the number of laboratories resulting in enhanced risk of accidents, and/or opportunities for misuse. Although the likelihood is low for an adverse event at one of these facilities, if one did occur, then the high-risk nature of the agents used could cause devastating damage to the urban surroundings and associated ecosystem. 14
MBSL: Biosafety and Biosecurity Policy Implications
The prevailing US and international biosafety guidelines,
Wide acceptance and adoption of the
Regarding governance, it is unclear what modifications are necessary to ensure that safety officers and the Institutional Biosafety Committee oversight are effective. It is unclear whether the structural and functional integrity and resilience of MBSL pilot laboratories deployed globally at low-conflict locations with a disease pandemic are assessed using maximum credible event scenarios and biorisk assessment. One such scenario involves compromise of MBSLs’ structural integrity due to an asymmetric threat or natural disaster, posing additional public health risk and enhancing the potential for an adverse impact on the general environment. MBSLs involved in highly infectious biological sample processing, testing, storage, and safe disposal should undergo thorough biorisk assessments to ensure structural integrity and functional resilience while operating in low-conflict regions with a disease pandemic.
Prevalent US biosecurity policies and guidelines do not provide any guidance on the MBSL and, in particular, what specific modifications may be necessary to ensure biosafety and biosecurity of MBSLs. There is no US initiative similar to the EU’s MIRACLE consortium to assess biosafety and biosecurity of MBSLs. 6
Current US biosecurity guidelines remain fragmented as various government agencies attempt to formulate agency-level policies to prevent potential misuse of biological pathogens, emerging technologies, and products derived from such research and applications in national security programs. 15 Existing policy formulations regarding biosafety and biosecurity do not address the infrastructure and functional flow process constraints in MBSLs, positing limitations on throughput, which could potentially introduce a serious biosafety/biosecurity risk to the staff and immediate general environment.
The current biosecurity landscape is split into 2 main domain areas: (1) biosecurity, which specifically focuses on preventing theft, diversion, and/or deliberate malicious use of biological sciences knowledge, skills, and technologies to cause harm, and (2) biodefense, which involves the development of capabilities and a knowledge base to assess, detect, monitor, treat (or vaccinate against), and respond to biological threats. These 2 domain areas often affect the same stakeholders, which may result in mutual benefits among defense-oriented policies or present barriers to achieving either defense or security objectives. For instance, the Department of Health and Human Services issued in 2017 a policy framework for research involving Enhanced Potential Pandemic Pathogens (P3CO) to raise awareness on mutual benefits to biosecurity and biodefense efforts. However, lack of consistency of agency-level policies has introduced gaps and barriers to evaluate broader consequences of investments and regulations as stakeholders fail to understand their roles in achieving biosecurity and biodefense objectives. Complicating the public policy and implementation to counter biological threats is the rapidly changing biotechnology landscape, which simultaneously presents new opportunities for building technological capabilities for defending against biological threats and for enabling new and often unknown security risks and vulnerabilities with potentially serious downstream national security consequences. 16 As the US government finalizes its new National Biodefense Strategy and begins preparing its Global Health Security Strategy, understanding the current policy landscape and the potential ability or inability of policies to achieve biodefense objectives is crucial to ensuring that the new strategies address longstanding gaps and challenges.
Current policy does not explicitly address the biosecurity policy implementation for mobile biocontainment laboratories, even though mobile biocontainment laboratories and clinical diagnostic laboratories in response to pandemics are becoming more acceptable and commonplace. This increase in acceptability is due to the immediate deployment of needed resources and relatively lower costs involved in creating fixed facilities and minimizing the risk of transport of samples containing dangerous pathogens.
Even the US government is generally moving away from large investments in fixed laboratory infrastructure to flexible, contingency operations in threat reduction, threat elimination, and surveillance sorts of missions involving chemical and biological threats. Edgewood Chemical Biological Center, a Department of Defense technical organization for nonmedical biological and chemical defense, has its own line of mobile laboratories that range from fully mobile to semipermanent, from modular to transportable. 17
MBSL: Generic Components and Processes Affected
As a maximum-containment facility, institutions with MBSLs are required to establish and maintain a highly stringent facility infrastructure and strictly ensure that material and personnel flow processes meet the highest standards of biosafety. This includes moving packages and samples from areas with no containment requirements to isolated sections of the facility that have increasing levels of biocontainment, as well as moving from BSL-2 unpackaging rooms that are attached to the MBSL-3 suite and connected through interlocks to the maximum-containment parts of the laboratory. 18 Likewise, all materials leaving the MBSL-3/4 laboratory must pass extensive testing that includes going through to sets of autoclaves and 2 sets of cross-linked glutaraldehyde dunk baths for chemical sterilization of the outer surface to ensure that only inactivated material enters level 2 areas. 18 Demanding material management protocols and personnel safety at BSL-3/4 comes with a very high construction, operation, and maintenance cost.
Two independent research teams recently reported rapid deployment of a MBSL-3 unit in Sierra Leone during the 2014 EVD epidemic, providing considerable detail on the mobile unit design features, supportive equipment, processes, and decontamination-related operating protocols.2,3 Using this unit, which was composed of 3 container vehicles that could be driven, the research team tested a total 1635 suspected cases of EVD over a 2-month period. The MBSL in these cases operated as a public health laboratory testing blood and oral swab specimens. Test samples were stored in –20°C and –80°C freezers within the laboratory and one –80°C freezer outside the MBSL-3 unit. Biorisk assessment involved the collection of air samples to assess potential inhalational exposure to the aerosols, as well as swab samples collected from various hard surfaces and protective clothing, that were tested for the presence of Ebola virus. 3 The South African team established a modular field Ebola diagnostic laboratory (FEDL), an MBSL-3 in a remote area of Sierra Leone, and performed EVD detection in over 11 250 samples over a 22-month period during the Ebola epidemic. 2 Both teams attempted to provide additional diagnostic capabilities in difficult-to-reach locations to assess the nature and extent of the EVD pandemic in remote populations.
The engineering design for high-containment operations and process flow varied considerably between the prototype MBSLs. The main 2 modified measures put in place in the prototype MBSLs were the use of sealed gloveboxes or the wearing of personal protective equipment (PPE). The glovebox approach was generally considered the optimal way to decrease the risk of exposure to workers, especially when working in a hot and humid climate as the glovebox serves as PPE. However, additional PPE may still be needed in case of glovebox failure, when handling and unpacking samples that are too large for the glovebox, or in instances where the inside of the mobile laboratory is contaminated. 19
Within these 2 main measures, the type of sterilization, the setup of the MBSLs, and how the samples were collected differed in approach and were inconsistent with each other. These inconsistencies in structural design and functional processes require a more detailed technology alternatives assessment from the point of biosafety processes and best practices.
From a functional process standpoint, the prototype MBSL units in published literature were fitted with supportive infrastructure and a reasonably good layout for personnel, material, and air and water flows. However, there was no maximum credible event analysis to demonstrate that the unit would withstand a worst-case scenario involving a likely mishap scenario during routine operations or a terrorism event, as MBSLs are by design to be deployed internationally in low-conflict zones. Maximum credible event (MCE) analyses are analogous to worst-case scenarios involving a likely mishap scenario in biotechnology bioprocessing operations, biological products testing laboratories, and biological specimen repository facilities, leading to release of particulate/aerosolized etiologic agents into the environment. 20
MCE analyses are performed to estimate the effectiveness of existing safeguards such as the engineering controls, administrative procedures, and the attributes of facility design that, in combination, prevent the probability of release of potentially pathogenic or toxic material from the test facility to the external environment. As part of our support to the US Chemical Biological Defense Program, we have developed a unique set of realistic MCE worst-case scenarios for fixed BSL facilities involved in biodefense-related laboratory and clinical investigations as they relate to the biologics product development process. 20 These MCE analyses did not consider MBSL units that are, by design, not equipped to withstand worst-case incidents, including potentials for terrorism in low-conflict zones.
The current
MBSL: Oversight of Select Agent Program
The Federal Select Agent Regulation (FSAP) governs the possession, use, and transfer of biological select agents and toxins (BSATs). BSATs are identified as pathogens and toxins that can cause significant damage to public health and safety if accidentally or deliberately released. Some examples of select agents include ricin toxin, the reconstructed 1918 influenza virus, and the Marburg virus. 21 In the aftermath of 9/11, the USA Patriot Act of 2001 defined restricted persons and illegitimate uses of BSATs between approved institutions. Table 1 is a summary of key changes included in the 2012 and 2017 updates to the select agent program. As summarized in the table, key changes/updates in 2012 and 2017 are as follows:
Selected Regulatory Changes in the 2012 and 2017 Updated BSAT Regulations.a

This table does not include all changes issued in the 2012 and 2017 updates to the suspicious activity report, but rather highlights those changes that led to opportunity costs for affected stakeholders.
Enhanced security requirements for Tier 1 agents, issued in 2012 Baseline security requirements for non–Tier 1 agents, as required after the 2012 Final Rule and which apply to laboratories that conduct severe acute respiratory syndrome coronavirus (SARS-CoV) work Policy elements that apply to both Tier 1 and non–Tier 1 agents, such as the new inactivation guidelines issued in the 2017 FSAP Final Rule
A key requirement under the 2012 and 2017 changes to the FSAP involved enhanced physical security measures to the fixed laboratory infrastructure and to upgrade the physical security systems of their Tier 1 laboratories to meet these enhanced requirements issued in the 2012 suspicious activity report (SAR) final rule and the cost to retrofit or move laboratories when new agents are added to the select agent list. 22 The direct costs and financial burden associated with physical security infrastructure improvements are primarily for fixed facilities. Notably, US agency-supported MBSLs have proven to be invaluable resources, typically performing diagnostic and surveillance support, and do not possess or conduct research on BSATs at these mobile facilities. International entities deploying pilot MBSL-3 for outbreak investigative work on risk group 3 and 4 pathogens are not required to comply with the FSAP, thereby rendering critical biosafety-biosecurity issues unaddressed.2,3 The EU-funded MIRACLE consortium identified the need for international guidelines for MBSL units, harmonization, and training for researchers and public health teams engaged or planning to field prototype units. 6
Prevailing regulatory and policy directives have no references to how the revised 2012 and 2017 changes to the FSAP apply to MBSL-3 and MBSL-4 units. To remain compliant, research institutions have incurred considerable financial costs in establishing and upgrading physical security barriers, which includes upgrading the physical security systems of Tier 1 laboratories, requiring considerable upfront investments. It is noteworthy that the costs of maintaining and upgrading physical security infrastructure to remain compliant with FSAP were a key factor in the decision of many academic institutions and public health laboratories to shut down their select agent programs. Most laboratories let their select agent registration expire after the 2005 FSAP updates. Given these high direct costs to remain complaint with FSAP, it is unclear how MBSL units can remain compliant with the stringent physical security requirements, particularly when they are deployed in low-conflict zones and international locations with minimal or no security and support infrastructure backup resources. 22
MBSL: Logistics Planning and Risk Analysis Framework
Logistics for expeditionary deployment of MBSLs requires systematic planning to deliver just-in-time diagnostic and medical countermeasure services as part of the epidemic response. MBSLs provide a much-needed resource at locations far removed from centralized laboratory facilities and are more flexible to locate and relocate as necessary during an outbreak. However, recent deployment of modular, high-biosafety MBSLs by various non-US international agencies and entities during the 2014 Ebola outbreak in West Africa brings focus on the mission and likely risks associated with these MBSL-3 facilities operating at locations with civil conflict and security risks.
MBSLs used as diagnostic mobile facilities at remote locations during a disease outbreak may have difficulty meeting the US FSAP registered laboratory requirements such as compliance with training for access to BSATs, biocontainment, incidence response, and biosafety and security awareness. MBSLs deployed in international locations and operated by non-US entities and agencies are not governed by the FSAP and may work with Tier 1 BSATs alongside diagnostic functions. In some cases, deployed mobile laboratories could end up operating as fixed, centralized laboratories.
Considerable upfront planning is the first step in the field deployment of MBSLs. Inglis 23 outlines planning, development, and modifications for a flexible diagnostic and microbiology laboratory response, including limitations to logistics, operational considerations for a mobile laboratory unit that must cope with a range of unexpected problems, limited access to high-level maintenance, troubleshooting support, data interpretation, and reagent resupply. Sustainability of MBSLs is subject to availability of maintenance and repairs while deployed. Lack of easy-to-conduct diagnostic tests, modified storage units, and test protocols could introduce additional limitations.
Figure 4 outlines the logistics planning and a risk analysis framework for deployment of MBSLs in response to disease epidemics. Any MBSL-3 deployed as part of an outbreak response must first develop a clear mission-level statement of objective, an intended role for the mobile unit, and functional areas to be covered such as diagnostics and medical countermeasure delivery. The next step, defining the course of action, must take into account the cost affordability, functional areas covered, feasibility of the planned operation, and risk-benefit analysis. Although deployed as mobile diagnostic facility, MBSLs would have to remain compliant with FSAP registered laboratory requirements such as the security plan, threat awareness training, and record maintenance security in terms of risk of theft, diversion, and deliberate and malicious use of the materials and/or pathogens and toxins. More frequent use of MBSLs as part of a mobile diagnostic and epidemic response may introduce certain limitations with regard to compliance with the FSAP registered laboratory requirements.
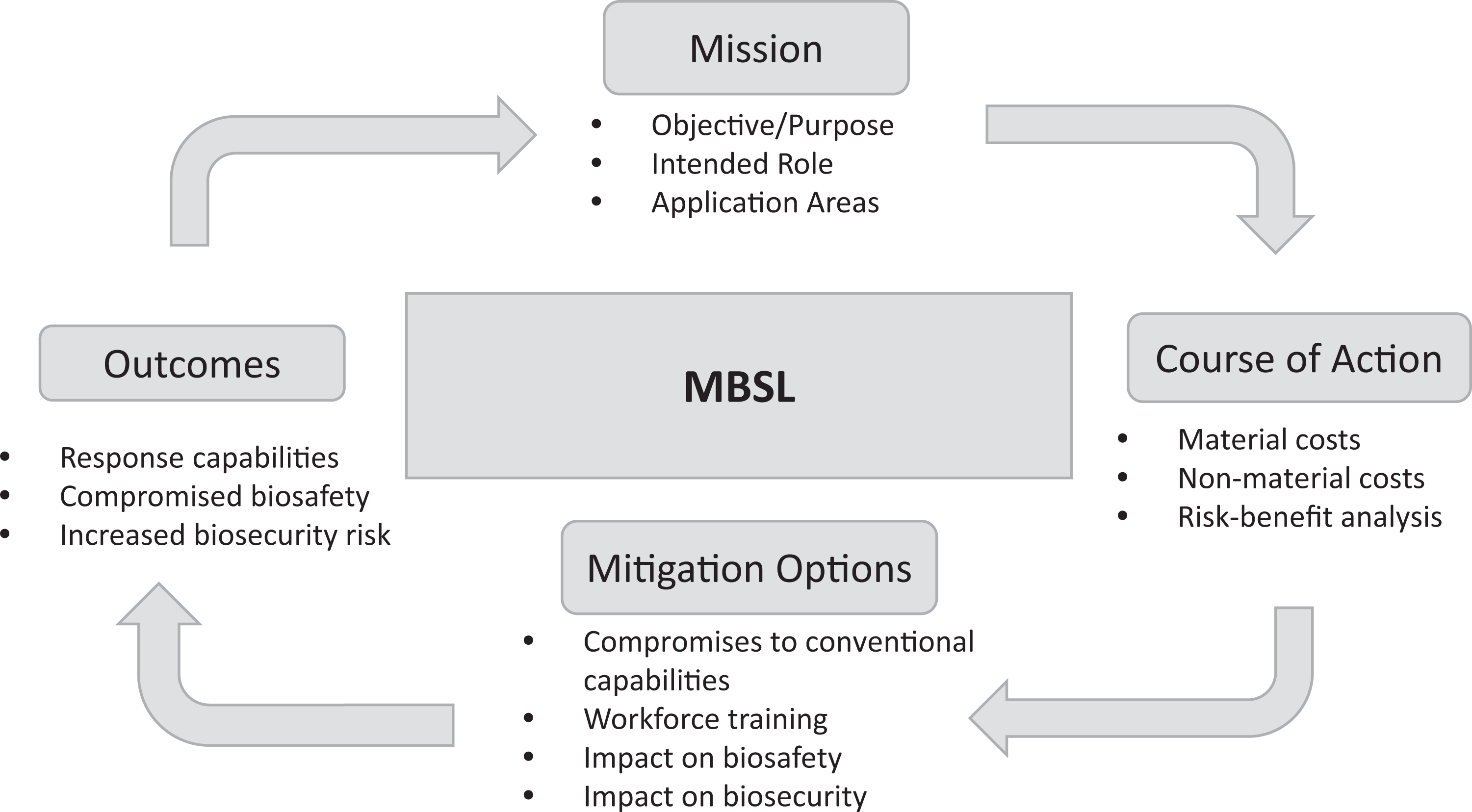
Logistics planning and risk analysis framework for deployment and use of mobile biocontainment laboratories for epidemic response.
These limitations identified as part of the planning process might affect feasibility of the proposed action, risks to operational sustainability, and workforce safety issues. The underlying principle using this breakdown is to identify the operational limitations imposed on mobile laboratories when deployed at international locations, taking into account the prevailing local conditions and baseline threats. Civil unrest in low-conflict zones, along with a dangerous disease epidemic, introduces a security threat and a potential loss of trained operators and specialists, thus rendering MBSLs deployed in these zones unworkable.
In some instances, the initial contingency operational demands requiring deployment of MBSLs as mobile diagnostic laboratories may require that they end up operating as Tier 1 BSATs, beyond the initial objectives. Therefore, it is prudent to capture the overall feasibility of what could and could not be accomplished with the MBSLs as part of biorisk assessment to identify gaps, as well as assign risk levels to biological materials, chemicals and equipment, biocontainment and the likelihood of an adverse event, the nature and extent of consequences of workforce safety, and the general environment. In general, operational feasibility and scaleup beyond the designated functions for MBSLs would entail additional time and resources to comply with the revised Concept of Operations (CONOP) and biosafety requirements.
Transportable or containerized MBSLs for small-scale operations as part of public health response have demonstrated a valuable role in response measures. However, scaleup operations with smaller units are not generally sustainable, which is a key limitation for planners of well-organized, large public health agencies to deploy MBSLs in the epidemic zones. Lack of upfront planning and demand for services during an outbreak leading to deviation from the initially planned role primarily as a diagnostic laboratory could introduce unacceptable levels of risk. In particular, these mobile units are equipped for Tier 1 BSAT activities. Available biosafety guidelines do not address workforce safety, standard operating procedures, biosafety, and security measures for MBSLs.
Finally, upfront planning for MBSL deployment must identify contingency measures such as long-term sustainability, local public health capacity, security situation, access to trained and reliable local staff during a surge, and availability of a larger field laboratory to meet scaled-up operational demand during an outbreak response.
Discussion
Published literature is replete recognizing a key role for mobile laboratories in the rapid diagnosis and biosurveillance of dangerous pathogens, as exemplified by the benefits of prototype MBSL-4 laboratories deployed during the 2014 to 2016 EVD epidemic, as well as incident-free biosafety practices employed to protect laboratory workers and the general local environment.8,19,24 -29 Nevertheless, the existing policy landscape regarding biosafety and biosecurity remains inconsistent among various stakeholders and offers no clear guidance on the specific biosafety and biosecurity requirements for MBSLs deployed at international locations in response to a disease epidemic.
As future biological threat elimination missions may increasingly rely on novel strategies and technical expertise to deploy modularized and mobile technologies such as MBSLs, risks involved in these deployments are yet to be clearly understood. These mobile platforms are extremely complex engineering structures supported by a modified workflow process design coupled with complex logistics coordination to ensure minimal in-country footprints, leaving behind little or no legacy wastes in challenging global work environments. Hence, it is imperative that a more focused review of the applicable biosafety, biosecurity, and concept of operation is undertaken to evaluate applicability of the prevailing guidelines, developed mostly for a fixed-facility infrastructure, to MBSLs and make modifications as necessary.
A detailed and comprehensive review of the current biosafety and biosecurity policy landscape regarding MBSLs is needed, especially as it becomes a more acceptable platform for global deployment in response to disease pandemics. The review process would have to entail a detailed biorisk assessment of MBSLs based on MCE-based scenarios unique to the emerging disease epidemic and threats uniquely related to the location of deployment. Our recent study on the current biosecurity policy landscape together with extensive engagement with the national stakeholders involved in the formulation, implementation, and evaluation of the biosecurity policy indicated no specific policy and programmatic initiatives regarding MBSLs and related deployment risks. 22 It is essential that the hazards and associated risks of deploying MBSLs are evaluated together with security measures in place to protect the workers, local population, and surrounding environment.
Existing biosafety guidelines are inadequate to address the unique operational environment and modified laboratory processes for the MBSL. A thorough risk assessment should guide modifications to the biosafety guidelines to address MBSLs, taking into account their unique operational environment and modified laboratory processes. These modified guidelines should also address physical security systems specifically for MBSLs. Existing physical system requirements for fixed laboratory facilities have expensive upfront capital investments, as well as maintenance and operational costs, which are often the deciding factor for many public health laboratories and academic institutions to let their select agent registration expire. Public-sector investments in MBSLs will serve as an additional, specialized, and deployable resource during public health emergencies and/or response to bioterrorism events.
International regions where MBSLs are likely to be deployed tend to augment serious shortcomings in local public health infrastructure, shortage of trained workforce, laboratory surveillance, and technologies. However, these deployments should ensure that clinical and environmental samples are secure, a compromise of which could potentially exasperate an epidemic outbreak, as seen during the 2014 to 2016 EVD outbreak. 30 Contingency operations involving MBSLs must undertake an overall assessment of the available public health infrastructure, reach-back resources, and overall knowledge and awareness of the threat among the general population as part of deployment and operational plans.
Pilot MBSLs deployed at faraway regions of the world have introduced lack of trust and a rise in suspicion from a local population, posing considerable personal risk to the scientific team and health workers. Given the sporadic nature of the personal safety and security challenges for the deployed medical and health workers in West Africa during the 2014 Ebola epidemic, it is crucial that an Ebola awareness team deployed in response to an epidemic includes staff who are familiar with MBSLs for biosafety- and biosecurity-related training and education. 31 Hence, future contingency operations responding to disease pandemics in difficult-to-work locations should include sufficient physical security measures and biosecurity-related training to ensure the integrity of the deployed MBSL and supportive systems.
As illustrated in Figure 4, opportunity costs of using MBSLs in risk-benefit terms must take into account the overall benefits of flexibility and operational readiness with the attendant direct costs of compliance with biosafety and biosecurity guidelines and potential downstream risks. Policy makers should undertake a detailed opportunity cost analysis as part of the overall review and applicability of current biosafety and biosecurity policy and guidelines to the unique MBSL infrastructure and functional process. This study must ensure that none of the regulatory processes required to comply with biosafety and biosecurity are omitted, and any modifications to guidelines specific to MBSLs must retain the biosafety best practices observed at BSL-4/3 laboratories.
For instance, the most recent EVD outbreak in the Democratic Republic of Congo has been declared to be largely contained by the World Health Organization after the first cases were confirmed in August 2018, despite EVD reaching Mbandaka, a river port with a population of over 1 million people. Among the actions taken to contain this outbreak was the deployment of MBSLs. 32 By deploying MBSLs, diagnosis sped up and supported effective epidemiological investigations. 33 Four MBSLs were deployed: one to Bikoro Reference Hospital on May 12, 2018, which was fully operating by May 16, 2018; one to Mbandaka; one to Itipo, which had been functional since May 30, 2018; and one to Iboko, which was run by the Institute National de Recherche Biomédicale and virologists from the Institut Pasteur in Dakar. 34 While there are currently no definitive data to show to what extent MBSLs helped to cull the outbreak, the speeding up of diagnosis time by MBSLs and its impact on effective intervention measures were reported as valuable despite potentials for hazards in the deployed areas.
Conclusion
The new paradigm in MBSLs, which have a rapidly deployable, sophisticated infrastructure and laboratory capability, offers strategic advantages to respond quickly and effectively by offering timely resources and capabilities for disease pandemics, having a minimal in-country footprint, and capitalizing on transportable technologies. Key characteristics of the new paradigm require (a) understanding and adapting to the complex global environment, to successfully support global health threat reduction efforts; (b) a global network of strategic partners resources and experienced personnel with mobile technology capabilities, ready to deploy worldwide; and (c) biosecurity and biosafety mechanisms fully in place and considering possible “surprise factors.” Additional policy and regulatory guidance regarding biosafety and biosecurity is crucial for new and transportable technologies with flexible options to cover a broad array of clinical laboratory and more sophisticated MBSL-4/3 requirements.
Footnotes
Ethical Statement
Authors declared the manuscript has not been published or under consideration for publication whole or in part elsewhere. Authors have been personally and actively involved in substantive work and hold themselves jointly responsible for its content.
Statement of Human Rights
Authors declare no work was performed on human subjects during the course of the study.
Statement of Informed Consent
Authors declare no experiments on human subjects were performed, or the need for Informed Consent during the course of the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
