Abstract
MICRO-CHEM PLUS Detergent Disinfectant Cleaner (MCP) is a commonly used disinfectant at biosafety level 4 (BSL-4) laboratories where research activities involving the most dangerous pathogens must be conducted. Using bat severe acute respiratory syndrome (SARS)–like coronavirus (CoV) WIV1 as a surrogate pathogen, we extensively evaluated the disinfection efficacy of 5% MCP in the first BSL-4 laboratory in China. WIV1 was completely inactivated in 1 minute of contact time by a 27-fold dilution of 5% MCP with a titer reduction of more than 7 lg 50% tissue culture infectious dose (TCID50)/mL, while a 243-fold dilution of 5% MCP showed very weak activity. For 81-fold dilution, WIV1 was killed by more than 6 lg TCID50/mL in 1 minute, but to get complete inactivation, the contact time should be at least 2 minutes. No loss of efficacy was detected for 5% MCP at a temperature of 0°C, ambient, or 37°C or in the solution of pH 3.2 to 12.2. No interference to disinfection efficacy was detected with up to 25% fetal bovine serum, 5% bovine serum albumin, or 8% cell debris. The decontamination of a positive pressure suit by chemical shower was tested with the filter pieces taped onto the head, chest, back, armpits, crotch, and boot soles, and no virus survived after a shower cycle of 4 minutes at 5% MCP plus 2 minutes of a soft water rinse. Keeping the ambient temperature for 79 days or in the tank for the chemical shower for 25 days, 5% MCP showed the same activity as when it was freshly prepared. Taken together, 5% MCP is a very efficient disinfectant for BSL-4 laboratories, and we recommend its application for other enveloped viruses.
Microorganisms, according to their pathogenicity, transmission, host range, and the availability of effective preventive or treatment measures, are classified into 4 groups (World Health Organization [WHO] risk groups 1, 2, 3, and 4). Risk group 4 (RG4) microorganisms are those that usually cause serious human or animal disease, which can be readily transmitted from one individual to another and for which effective treatment and preventive measures are not usually available. 1 Since RG4 microorganisms pose high individual and community risks, the scientific research on these microorganisms must be conducted in biosafety level 4 (BSL-4) laboratories.
Although BSL-4 laboratories feature the installation of Class III biological safety cabinets (BSCs) or positive pressure suits in conjunction with Class II BSCs, as well as airlock entry, chemical showers, special waste treatment systems, and double-door autoclaves, to protect personnel from exposure and to prevent infectious agents from leaking outside BSL-4 laboratories, it is also very important to disinfect infectious liquid and solid waste as much as possible in BSL-4 laboratories, even in BSCs, before autoclaving and to decontaminate the surface of positive pressure suits via a chemical shower when staff leave the BSL-4 laboratories. Therefore, having an effective disinfectant for waste treatment and the chemical shower is critical.
According to efficacy, disinfectants can be classified into sterilants and high-, intermediate-, and low-level disinfectants. Sterilants kill all organisms, including bacterial spores; high-level disinfectants kill all organisms, except high levels of bacterial spores; and intermediate-level disinfectants might be cidal for mycobacteria, vegetative bacteria, most viruses, and most fungi but do not necessarily kill bacterial spores. Low-level disinfectants can kill most vegetative bacteria, some fungi, and some viruses in a practical period of time (≤10 minutes).2,3 Widely used sterilants and high-level disinfectants, such as chlorine, formaldehyde, glutaraldehyde, hydrogen peroxide, and peracetic acid, usually are toxic, corrosive, or oxidative 2 and may not be compatible with pipelines, waste water treatment systems, and positive pressure suits in BSL-4 laboratories. Some intermediate-level disinfectants, such as disinfectants containing alcohol, iodine, or phenols, may have better compatibility but are not a good choice due either to storage safety or environment friendliness since large amounts are used during chemical showering. However, most of the RG4 agents currently classified in the United States, Europe, and China are viruses, specifically enveloped viruses, which are susceptible to the low-level detergent disinfectants that have better compatibility, storage safety, and environment friendliness.
MICRO-CHEM PLUS Detergent Disinfectant Cleaner (MCP; National Chemical Laboratories of PA) is a commonly used disinfectant in BSL-4 laboratories in the United States, Canada, and Australia (personal communication ). MCP is a mixture of nonionic and cationic detergents containing 2.25% of alkyl dimethyl benzyl ammonium chlorides, 2.25% of alkyl dimethyl ethybenzyl ammonium chlorides, and 4% to 5% nonylphenol ethoxylates and is touted to have bacteriocidal, fungicidal, and virucidal activity, including adenoviruses, influenza viruses, coronaviruses, human immunodeficiency virus type 1 (HIV-1), and hepatitis C virus. 4 Here, using bat severe acute respiratory syndrome (SARS)–like coronavirus (CoV) WIV1 as a surrogate, we report the evaluation tests of MCP at Wuhan National Biosafety Laboratory, which was designed and constructed with French cooperation and was the first BSL-4 laboratory certified by the China National Accreditation Service for Conformity Assessment (CNAS) in January 2017 5 and further approved by the National Health and Family Planning Commission of the People’s Republic of China in August 2017.
Materials and Methods
Cells and Viruses
The African green monkey kidney cell line Vero-E6 was purchased from the American Type Culture Collection, maintained in Dulbecco’s modified Eagle medium (DMEM; Gibco) supplemented with 10% fetal bovine serum (FBS; ThermoFisher, Waltham, Massachusetts) at 37°C with 5% CO2 in a humidified atmosphere.
The bat SARS-like CoV WIV1 strain was originally isolated with Vero-E6 from bat feces 6 and can replicate in human airways but lacks the virulence of epidemic SARS-CoV 7 so it can be handled in a BSL-2 laboratory. To prepare virus stocks, Vero-E6 monolayer was inoculated with WIV1, vesicular stomatitis virus (VSV), or poliovirus type I (PV-I) at the multiplicity of infection of 0.1 in DMEM + 2.5% FBS; 2 to 3 days later, the medium was collected and centrifuged at 5000 g for 10 minutes to remove cell debris. The supernatant was aliquoted and frozen at –80°C.
Neutralization and 50% Tissue Culture Infectious Dose Quantification
MicroSpin S-400 HR columns (GE Healthcare), if applicable, were used to neutralize MCP’s activity. Briefly, a MicroSpin column was precentrifuged at 700 g for 1 minute to drain the resin inside and put into a new collection tube; 100 μL of disinfection reaction solution was applied to the top center of the resin, which took about 10 seconds, followed by centrifugation at 700 g for 1 minute to elute the virus particles. The eluate was diluted 8-fold, then serially 10-fold to 7 gradients with DMEM + 2.5% FBS; 100 μL of each dilution was added to Vero-E6 cells in 96-well plates in sextuplicate and incubated for 2 hours at 37°C. The cytopathic effect (CPE) was observed 3 days later. For the chemical shower test, direct dilution of 800 times with DMEM + 2.5% FBS was used as neutralization instead.
Quantitative Suspension Disinfection Assay
MCP solution was prepared fresh with soft water, which was processed with ion exchange chromatography for the BSL-4 laboratory, but for testing the period of validity, MCP solution was prepared on the day before the test. An aliquot of virus stock was mixed with an equal volume of MCP solution. One minute later or at an indicated time, the activity of MCP was neutralized with a MicroSpin column; the residual virus was titrated with 50% tissue culture infectious dose (TCID50). To determine if the input virus was inactivated completely, the neutralization eluate was used to infect a monolayer of Vero-E6 cells in 6-well plates or 15-cm plates after dilution to 0.8 mL or 15 mL with DEME + 2.5% FBS, respectively. After incubation for 2 hours with gentle rocking, the cells were replenished with fresh medium.
Surface Disinfection with Spraying
To mimic surface disinfection, autoclaved filter paper pieces in an isosceles right triangle shape of 1 × 1 cm were put onto a plate, which was tilted about 45 degrees. The paper pieces were inoculated with 15 μL WIV1 and immediately sprayed twice with 5% MCP at a distance of about 20 cm; 1, 2, or 4 minutes later, the pieces were put into 15 mL DMEM + 2.5% FBS and infected Vero-E6 cells in 15-cm plates after being diluted to 0.8 mL or 15 mL with DEME + 2.5% FBS, respectively. Or 50 μL WIV1 was put into 96-well plates and disinfected by spraying 5% MCP twice, followed by neutralization with MicroSpin columns at 1, 2, or 4 minutes.
Positive Pressure Suits Decontamination Test
Filter paper was cut into pieces of 1 × 3 cm, and a hole was punched at one end. In the BSL-4 laboratory, autoclaved paper pieces were attached to the positive pressure suits by cotton thread, which passed the hole and was taped to the positive pressure suits. The paper pieces were stuck to 8 sites: head, chest, back, left and right armpits, crotch, and left and right boot soles (Figure 1). In the chemical shower chamber, 50 μL WIV1 stock was absorbed into the paper, and then immediately a shower cycle of 4 minutes 5% MCP plus 2 minutes soft water rinse was started. During showering, as requested, arms were lifted, boots were raised backward, and the body was turned back and forth. The paper pieces were removed with forceps and put into 4 mL DMEM + 2.5% FBS, which was enough to immerse the paper pieces completely, followed by an immediate 10-fold dilution, or into 40 mL DMEM + 2.5% FBS.

Diagrammatic representation of filter paper pieces on a positive pressure suit for chemical shower decontamination tests. Filter paper was cut into pieces of 1 × 3 cm and attached to the positive pressure suit by cotton thread, which passed the hole at one end and was taped to the suit at 8 sites: head, chest, both armpits, crotch, both boot soles (solid), and back (dotted).
Data Analysis
The input virus titer was presented as the average of lg TCID50/mL of all positive controls from the same method; lg TCID50/mL is the logarithm to base 10 of TCID50/mL, which was calculated with the Reed-Muench method. 8 For example, the WIV1 input titer of 7.17 ± 0.07 lg TCID50/mL for the quantitative suspension disinfection assay was averaged from all the positive controls, while 7.53 ± 0.06 lg TCID50/mL for the positive pressure suit decontamination test was calculated from those of each day. The killing log value (KLV) was determined to be the reduction of lg TCID50 compared to that of the input virus. For most of the disinfection assay, where 5% MCP was used, no CPE was observed in any gradient dilutions, including 6-well and 15-cm plate infection, indicating that the virus was completely inactivated, so KLV was expressed as “≥input titer” for the test when 0.06% (5%/81) MCP was treated for 1 minute. CPE was observed in 6-well plates, but when no or few wells in 96-well plates showed CPE, a TCID50 was calculated assuming that all wells of the theoretical gradient below the lowest dilution showed CPE, and KLV was expressed as “≥(input titer–the TCID50) and <input titer.”
Results
Validation of MicroSpin Columns as a Neutralizer
Five percent of MCP is commonly used in BSL-4 laboratories. To test how much MCP affected cell culture, 5% MCP was serially 3-fold diluted with DMEM + 2.5% FBS and treated with Vero-E6 cells for 2 hours. The next day, the cells were checked with a microscope. Vero-E6 cells treated with more than 243-fold diluted 5%, that’s 0.02%, of MCP all died, while the cells treated with 729-fold diluted 5% MCP looked nice but had a smaller number than the control (data not shown).
MicroSpin columns contain Sephacryl gel filtration resins and can be used in spin format, which is ideal for fast separation of molecules by excluding large molecules and retarding small molecules. Five percent of MCP was spun through a MicroSpin column at 700 g for 1 minute; the eluate was serially 2-fold diluted and treated with Vero-E6 for 2 hours. Few cells treated with 4-fold dilution were alive, while those with 8-fold dilution grew well (data not shown). These results suggested that 5% MCP was very toxic to Vero-E6 cells but that MicroSpin columns could dramatically neutralize the cytotoxicity.
We then tested the feasibility of MicroSpin columns as a neutralizer against MCP. Ten percent of MCP was loaded onto a MicroSpin column, and the eluate was mixed with an equal volume of WIV1 virus for 1 minute at room temperature. As shown in Table 1, compared to the virus control (group 5), the eluate had almost no disinfection effect (group 4). To determine if MicroSpin columns had any effect on virus titer, WIV1 was mixed with an equal volume of water and then loaded onto the columns. It seemed that the MicroSpin columns slightly reduced the virus titer (group 3), but from the cumulative data tested later, MicroSpin columns retained about half of the virus, which was about a 0.36 decrease in TCID50. Therefore, we used group 3 as the control for the following tests.
Neutralization Test of MicroSpin Columns.
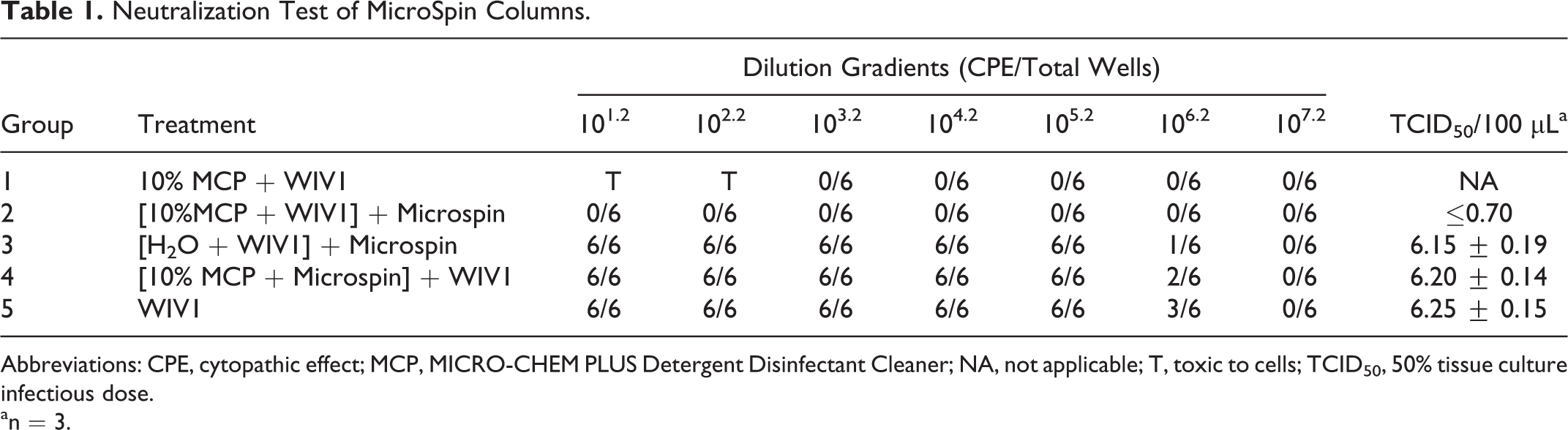
Abbreviations: CPE, cytopathic effect; MCP, MICRO-CHEM PLUS Detergent Disinfectant Cleaner; NA, not applicable; T, toxic to cells; TCID50, 50% tissue culture infectious dose.
n = 3.
Quantitative Suspension Disinfection
First, we tested the disinfection efficacy of MCP against 2 enveloped viruses, WIV1 and VSV, and the nonenveloped virus PV-I. Ten percent of MCP was mixed with the viruses, respectively, and incubated for 1 minute or 4 minutes before neutralization with MicroSpin columns. No CPE was observed for both enveloped viruses in either 96-well plates for TCID50 titration or 6-well plates for complete inactivation determination. The input titer was 7.17 and 7.04 lg TCID50/mL for WIV1 and VSV, respectively, so KLV at 1 minute of contact time was ≥7.17 and ≥7.04 lg TCID50/mL, respectively (Table 2). However, for the nonenveloped PV-I, KLV was only 1.75 lg TCID50/mL after 4 minutes of contact time. WIV1 was used for all the following experiments.
Disinfection Efficacy of MICRO-CHEM PLUS.
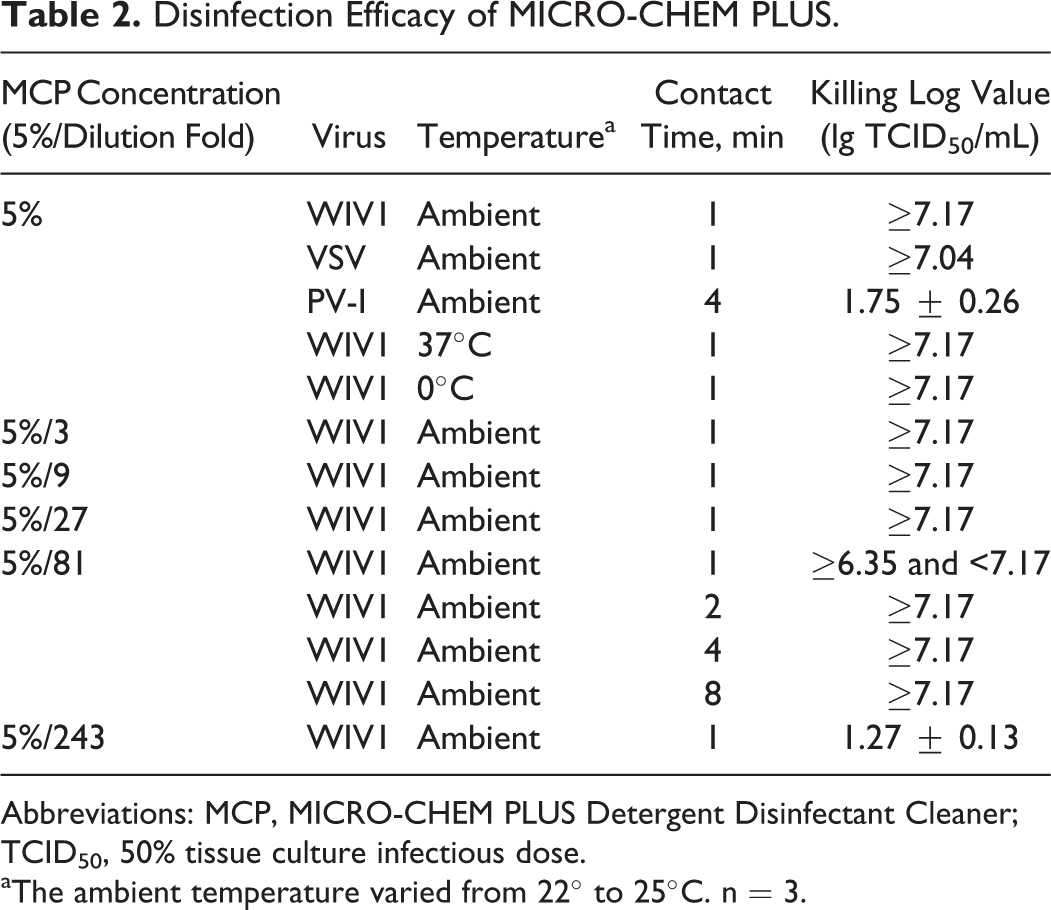
Abbreviations: MCP, MICRO-CHEM PLUS Detergent Disinfectant Cleaner; TCID50, 50% tissue culture infectious dose.
The ambient temperature varied from 22° to 25°C. n = 3.
To further investigate the disinfection efficacy, serially 3-fold diluted 5% MCP was tested. As shown in Table 2, after being diluted 27 times, which brought the final disinfection concentration to 0.19%, WIV1 was still completely inactivated. But if diluted further, to a final concentration of 0.06%, none or 1 or 2 wells for TCID50 titration showed CPE, but CPE was always observed in 6-well plates, and KLV was determined to be ≥6.35 and <7.17 lg TCID50/mL. However, if the contact time was extended to 2 minutes, the virus was completely killed. If 5% MCP was diluted 243 times to a 0.02% final concentration, KLV dramatically dropped to 1.27 at 1 minute of contact time.
For BSL-2 or BSL-3 laboratories, the disinfectant solution may be prepared daily or weekly. To determine how long the disinfectant could maintain its efficacy, 10% MCP was prepared every few days and kept at the ambient temperature. As shown in Table 3, WIV1 was completely killed by the disinfectant solution prepared all days up to 79.
The Effect of Storage Time to Disinfection Potency.
Abbreviation: TCID50, 50% tissue culture infectious dose.
One of 3 experiments was shown.
Effect of Temperature, pH, and Organic Components on Disinfection Efficacy
To test the effect of temperature, 10% MCP and WIV1 were equilibrated in ice water at an ambient temperature (22°–25°C) or 37°C, respectively, and then mixed equally and incubated for 1 minute at the same condition before neutralization with MicroSpin columns. KLV was determined to be ≥7.17 lg TCID50/mL as above (Table 2). The ambient temperature in our BSL-4 laboratory was 21° ± 1°C, and the liquid temperature in the tanks for the chemical shower was 18°C to 22°C; both were close to the ambient temperature in the BSL-2 laboratory.
It is possible that infectious liquid with a different pH would be disinfected with MCP, so the effect of pH was investigated as well. The pH of freshly prepared 10% MCP with soft water was pH 11.2, which was then adjusted to pH 12.2, pH 9.2, pH 7.2, pH 5.2, and pH 3.2 with NaOH or HCl. No difference was detected from pH 12.2 to pH 3.2 with complete killing of WIV1 (Table 4).
The Effect of pH to Disinfection Efficacy.

Abbreviation: TCID50, 50% tissue culture infectious dose.
The original pH of 10% MICRO-CHEM PLUS with soft water. n = 3.
When working on virus cultures or animal experiments, infectious liquid waste with different amounts of serum, protein, or cell debris may be produced, so the effect of these organic components was investigated. WIV1 was mixed in an equal volume with serially 2-fold diluted FBS, bovine serum albumin (BSA; Merck Millipore), or wet cell debris for which cells were collected with centrifuge and frozen, thawed, and then disinfected by twice the volume of 10% MCP. Initially, the neutralization eluate was diluted 8 times and infected Vero-E6 cells in 6-well plates as above, but the cells with BSA or a high concentration of FBS and cell debris died of cytotoxicity. So, the eluate was diluted 150 times and infected Vero-E6 cells in 15-cm plates. The cells showed no sign of CPE even 15 days postinfection with KLV estimated to be ≥7.19 lg TCID50/mL, indicating that 5% MCP still kept its efficacy against the liquid with up to 25% FBS, 5% BSA, or 8% cell debris.
Surface Disinfection with Spraying
Besides infectious liquid disinfection, 5% MCP would be used for surface disinfection with spraying. Two kinds of experiments were conducted to test the surface disinfection efficacy. WIV1-absorbing filter paper pieces were put onto a tilting plate to mimic surface contamination and were disinfected with two 5% MCP sprayings, followed by neutralization with direct dilution. Another method was that WIV1 was put into 96-well plates and disinfected with 2 sprayings of 5% MCP, followed by neutralization with MicroSpin columns. Again, for both methods, WIV1 was inactivated completely in 1 minute (data not shown).
Positive Pressure Suit Surface Disinfection with Chemical Shower
For most BSL-4 laboratories, including ours, scientists are protected with positive pressure suits when working with high-risk pathogens and must have a chemical shower with the suits on in an air-locked shower chamber before exiting the lab. Therefore, the chemical shower is a critical disinfection procedure to prevent pathogen leakage. The shower cycle of our BSL-4 laboratory was set to be 4 minutes of chemical plus 2 minutes of a soft water rinse. According to the regulation CNAS-CL53 issued by the China National Accreditation Service for Conformity Assessment, 9 8 sites were selected to stick on the filter pieces: head, chest, back, both armpits, crotch, and both boot soles. After showering, instead of a MicroSpin column, which was not applicable as a neutralizer, the filter pieces were immersed into DMEM + 2.5% FBS, resulting in about 800- to 1000-fold dilutions, which led to at least a 2-log decrease in test sensibility. As shown in Table 6, no residual infectious virus was detected after the chemical cycles with 5%, 1.67% (5%/3), or 0.56% (5%/9) of MCP with KLV determined to be ≥5.13 lg TCID50/mL. However, to make sure that the virus was completely killed, after showering with 0.56% MCP, 15 mL of the 40-mL immersing dilution of the filter pieces under the right boot sole was applied to infect Vero-E6 cells in 15-cm plates with the result of no CPE as well. KLV was further determined to be ≥7.10 lg TCID50/mL.
The Effect of Organic Component to Disinfection Efficacy.
Abbreviations: BSA, bovine serum albumin; FBS, fetal bovine serum; TCID50, 50% tissue culture infectious dose.
Final concentration; FBS in V/V, BSA in W/V, and cell debris in wet weight/V. n = 2.
Killing Log Value (lg TCID50/mL) with MICRO-CHEM PLUS Chemical Shower.
Abbreviation: MCP, MICRO-CHEM PLUS Detergent Disinfectant Cleaner.
n = 3.
n = 2.
After chemical shower, the filter papers were put into 40 mL Dulbecco’s modified Eagle’s medium + 2.5% fetal bovine serum for neutralization, and 15 mL was used to infect Vero-E6 cells in 15-cm plates with the result of no cytopathic effect. The kill log value was calculated to be ≥7.10 lg 50% tissue culture infectious dose (TCID50)/mL, while the input virus was 7.53 lg TCID50/mL.
The tanks for the chemical shower could hold about 1000 L of liquid, which could provide multiple chemical cycles, so it was necessary to test how long the disinfectant liquid could be kept in the tanks without any loss of its efficacy. A large volume of 5% MCP was prepared, and the chemical cycles were run every 3 or 4 days. The results of the last 3 tests are shown in Table 6. Up to 25 days later, no loss of disinfection efficacy was found.
Discussion
MCP is commonly used to disinfect the infectious liquid waste in BSL-4 laboratories before heat-sterilization of the waste water treatment system.10,11 Here, using bat SARS-like CoV WIV1 as a surrogate for BSL-4 pathogens, we extensively evaluated the disinfection efficacy of MCP in our brand-new BSL-4 laboratory, including not only suspension liquid disinfection, surface disinfection with spraying, and positive pressure suits decontamination but also the storage time of the working solution and the effect of temperature and pH, as well as the presence of FBS, BSA, and cell debris. MCP completely inactivated WIV1 at 5% concentration in 1 minute of contact time, even at 0°C; pH and the presence of interfering components, such as 25% FBS, 5% BSA, or 8% cell debris, did not affect the disinfection efficacy, and 5% MCP could be kept at least 79 days for liquid disinfection and 25 days for a chemical shower. Our data demonstrated again that 5% MCP is an effective disinfectant for a BSL-4 laboratory.
We found that although 81-fold dilution of 5% MCP (ie, 0.06%) caused virus titer reduction by more than 6.35 lg TCID50/mL, it could not completely inactivate WIV1 in 1 minute of contact time. Taking the effectiveness of interfering components into consideration, the real disinfection limit of 5% MCP should be far beyond the titer of WIV1, which was 7.17 to 7.35 lg TCID50/mL in this study.
Uddowla et al 10 and Klaponski et al 11 used VSV as a surrogate to test the disinfection efficacy of MCP. Since we had committed to apply for SARS research activities in our BSL-4 laboratory before this study started, bat SARS-like CoV WIV1 was chosen as the test virus. However, VSV was also tested in the study and was inactivated as well as WIV1. However, another virus, PV-I, which is nonenveloped, was poorly inactivated by MCP.
Dey-Engley (D/E) neutralization solution was used in the studies by Uddowla et al 10 and Klaponski et al. 11 However, as Klaponski et al 11 reported, we found that the mixture of D/E solution and 5% MCP caused severe cytotoxicity to Vero-E6 cells. Although 243-fold diluted 5% MCP showed little disinfection ability in 1 minute of contact time but killed all cells if put onto Vero-E6 for 2 hours, the border of no disinfection ability and that of no cytotoxicity should be close, since 729-fold diluted 5% MCP led to little cytotoxicity and the envelope of viruses is from host cells. Therefore, D/E neutralization solution may not be a good neutralizer for MCP. We then turned to MicroSpin S-400 HR columns, which contain Sephacryl resin of different pore sizes. They allow separation of virus particles from MCP by the process of gel filtration. WIV1 particles more than 80 nm would be excluded from the resin and elute first, while MCP would penetrate into and be retarded in the matrix. But if the infectious liquid with interfering components was disinfected with 5% MCP, cytotoxicity was observed after neutralization with the columns. A large complex of MCP and BSA may be formed and elute with virus particles since alkyl dimethyl benyl ammonium chlorides have been reported to bind with BSA. 12
To test the efficacy of MCP for a chemical shower, Klaponski et al 11 used brushed stainless-steel disk carriers onto which VSV was dried. However, when WIV1 with soil load was dried on disk carriers, the recovery dose was less than 1 × 106 TCID50/carrier. According to the technical standard for disinfection issued in 2002 by the Ministry of Health of China, steel disk, glass, filter paper, or cotton cloth could be chosen for different subjects to be disinfected. 3 Therefore, we used filter paper taped onto positive pressure suits for the chemical shower tests. Klaponski et al 11 selected the areas of back, arm, chest, thigh, hip, and boot cuff on the positive pressure suits, but as requested by the Chinese regulation CNAS-CL53, 9 we tested hidden sites, such as armpits, crotch, and boot soles with the same results as the open sites.
For a virucidal disinfectant or a good disinfection procedure, a 3- to 4-lg reduction in virus titer 13 is required by the Environmental Protection Agency (EPA) and, in China, at least a 4-lg reduction for liquid disinfection 3 and at least a 3-lg reduction for positive pressure suit decontamination at all sites, including head, chest, back, armpits, crotch, and boot soles. 9 Our results and others’ demonstrated that the disinfection efficacy of 5% MCP exceeded these requirements. Therefore, 5% MCP is a very efficient disinfectant for BSL-4 laboratories, and we recommend its application for other enveloped viruses, including RG2 and RG3 viruses.
Footnotes
Acknowledgments
We thank Jia Wu, Ge Gao, and Yun Peng for participating in the chemical shower test; the facility maintenance team; and the biosecurity team and biosafety team for keeping the BSL-4 laboratory running.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the Science and Technology Planning Project of Guangdong Province (2015A020213007).
