Abstract
The utilisation of the CRISPR/Cas9 technology has sparked a renewed interest in gene drive mechanisms. These mechanisms of biased inheritance may yield promising applications in the fields of vector control and nature conservation. However, the same properties that will enable these applications may also pose a risk if organisms that are equipped with gene drive cassettes are unintentionally released into the environment. Although several groups of scientists and regulators have started to address these safety concerns, there are currently no dedicated guidelines published on the required risk assessment and minimal control measures applicable to gene drive organisms in contained use. To fill this gap, this paper describes a fundamental approach to assessing the risks of these organisms while handled in a contained laboratory environment. Based on the likelihood that an adverse effect will arise from the handling of a gene drive organism and the severity of this effect, three risk classes for contained use activities are presented. Finally, specific minimum requirements regarding physical measures and working practices are proposed according to the presented risk classes and tailored to activities with rodents, insects, and fungi, which are most likely to be used for gene drive applications in the near future.
Introduction
Promising applications of CRISPR-enabled gene drive technology are foreseen for several applications, such as the elimination or suppression of insect vectors1,2 transmitting (plant) diseases or nature conservation by eradicating invasive species.3,4 This technology enables the alteration of almost any DNA sequence in sexually reproducing organisms and its spread through wild populations over generations. Several reports of expert meetings5 -7 and publications8,9 have already addressed the environmental risks associated with gene drives as well as ethical and societal questions to consider and knowledge gaps in risk assessment to fill before the technology could be utilized in the environment. 10 Nevertheless, at this stage of development of the technology, additional research on the efficiency of gene drives, the effect of a gene drive on a (sub)population, and its ecological consequences is needed before the technology can be applied in a wild population in the environment. To generate these data, gene drive organisms (GDOs) first must be constructed and studied under contained conditions. However, due to their inherent properties, which are linked to the eventual purpose of becoming established in the environment to alter, suppress, or even eliminate a wild population, any unintentional release must be avoided in this preliminary research phase by employing adequate containment and control measures.
During this phase of initial research in a contained environment, controls need to be proportionate to the estimated risk of an establishment of the GDO in the environment in case of an accidental release. For instance, if a GDO cannot spread its gene drive cassette into a wild population, less strict containment and control measures would be acceptable compared to a case where a GDO can survive and establish itself outside of the laboratory. To assess the adverse effects of an activity with a GDO, a risk assessment needs to be performed to identify the minimum necessary containment and control measures to prevent unintentional release and potential subsequent harm to the environment. This poses unique challenges to both regulators and users. 11 Furthermore, the assignment of adequate control measures needs to occur in a context of insufficient experimental data regarding the likely behavior of a particular GDO and hence in a situation of uncertainty.
An organism able to initiate a gene drive, and thus subject to the risk assessment as outlined here, is any organism that is capable of sexual reproduction and genetically modified with a DNA sequence that encodes a site-specific endonuclease and in the case of the CRISPR/Cas9 gene drive technology, a guide RNA (gRNA) that integrates at a genome position within the cutting sequence of the endonuclease. 12 These features force the integration of the DNA sequence that is thus passed to future generations.
To assess the adverse effects of GDOs while still in contained use, European users and regulators look toward the risk assessment principles of the European Union’s Directive on the contained use of genetically modified microorganisms (GMMs). 13 This directive is restricted to the assessment of GMMs and is therefore primarily concerned with the identification of harmful effects on human, animal, and plant health due to potential pathogenic properties of microorganisms. However, the general principles of the risk assessment procedure are also used by several European countries to assess and manage other genetically modified organisms (GMOs), such as plants or animals. The long-term experience with this risk assessment procedure and its application to organisms other than microorganisms made it a logical choice to analyze its use for GDOs as well.
As a GDO is typically nonpathogenic, its hazard potential arises from its designed property to transfer a genetic modification at a higher frequency to its offspring than is expected from normal Mendelian inheritance. 14 Should any incident involving an unintentional release of a GDO occur, adverse effects to target and nontarget organisms in the environment, immediate or delayed, may result. This environmental impact is, however, also accounted for in the directive 13 as an element of assessment, namely, in the form of considering deleterious effects due to an establishment or dissemination in the environment or the natural transfer of the inserted genetic material to other organisms. Given this provision, the general principles stipulated in the directive seem to provide a useful framework for risk assessment by identifying potentially adverse effects of GDOs and their likelihood of occurrence as well as assigning risk classes to a contained use activity with a GDO.
Here we describe a risk assessment procedure for GDOs based on the principles of the directive. Risk management measures are presented for different classes of risks as well as different types of organisms.
Principles of a Biological Risk Assessment
The aim of a biological risk assessment is to assess the potential adverse effects that may arise from a biological hazard—namely, the (genetically modified) organism and the intended activities—with respect to the environment and health. Widely adopted principles, at least in Europe, to assess these adverse effects are described in Annex III of the directive 13 and presented in Figure 1. The principles consist of the procedure to be followed and the elements to be taken into account. These elements are (1) the identification and nature of the potential adverse effects on human, animal, and plant health and the environment; (2a) the severity of these adverse effects; (2b) the likelihood (ie, the probability of occurrence) that these adverse effect will be realized; and connected to this aspect, the characteristics of the activity with the GMO (eg, complexity of handling or scale of operations). Following the procedure (indicated in Figure 1) in identifying and assessing the adverse effects, a risk class is assigned to the activity. This assignment is a case-by-case professional judgement of scientists, biosafety officers, and regulators. The determination of a risk class enables the assignment of a proportionate set of control measures to activities that pose similar levels of risk but may have adverse effects with slightly different severity and/or likelihood of occurrence. The general principles of a biological risk assessment for a conventional GMO, as outlined in Figure 1, are utilized and expanded for a GDO.
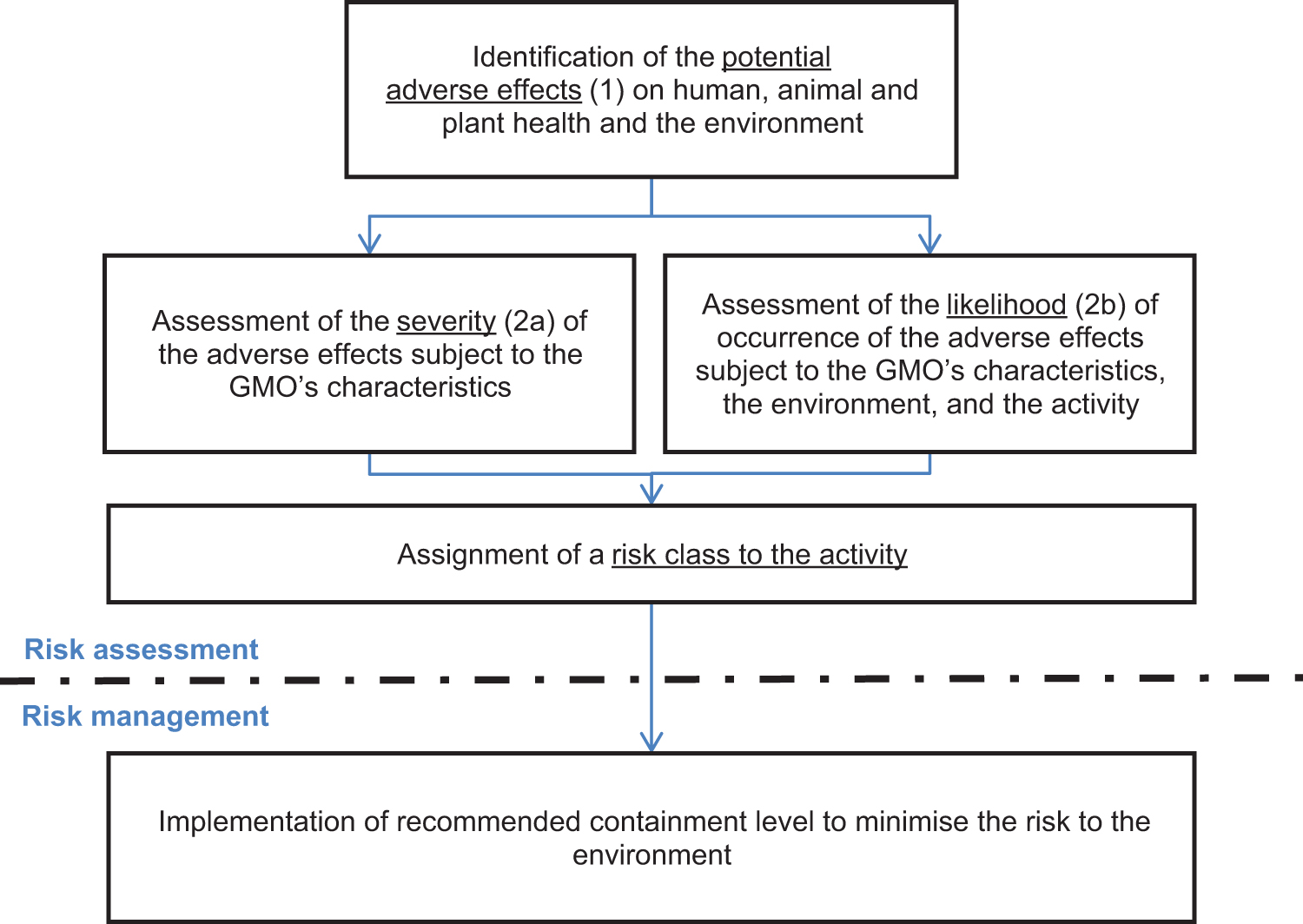
General principles of a biological risk assessment of a contained use activity with a genetically modified organism according to the European Directive 2009/41/EC. 13
Identifying a GDO’s Potential Adverse Effects
The starting point of the risk assessment procedure is the identification of the possible adverse effects on the environment and health due to exposure to the GDO. As already mentioned, it is the environment that will be most at risk if unintentionally exposed given that a GDO typically will not possess any pathogenic characteristics detrimental to human or animal health. The potential adverse effects of a GDO are primarily identified as consequences of (1) establishment or dissemination of the GDO in the environment or (2) the natural transfer of the inserted genetic material to other organisms. In a contained use setting, an unintentional release of a GDO into the environment may happen through a violation of the physical containment in place and indirectly through exposure of laboratory personnel or laboratory animals and plants housed within the same containment area, which may lead to an unnoticed contamination and subsequent unintentional release from containment.
Considering a potential release of a GDO, several adverse effects within the exposed ecosystem may arise. However, those effects will be difficult to predict as detailed knowledge of the properties and behavior of the GDO will be typically lacking during the initial research phase. Nevertheless, to enable a well-founded risk assessment in the context of this uncertainty, the establishment and dissemination (survival) of the GDO in the environment and the genetic transfer of the gene drive cassette to wild populations or nontarget species per se serve as suitable proxies (Figure 1, Box 1). Once more data have been gathered and a planned release into the field is considered, these proxies may be replaced by any specific adverse effects the establishment, dissemination, or genetic transfer may cause.
Considering the type of activity (open phases) and the type of organisms (ability to fly, small size, and hiding capabilities,) the likelihood that an adverse effect may occur is high. However, due to the location of the laboratory in the northern temperate climate, this mosquito species is ecologically contained and would not survive or transfer the gene drive construct to offspring if unintentionally released in the local environment. The
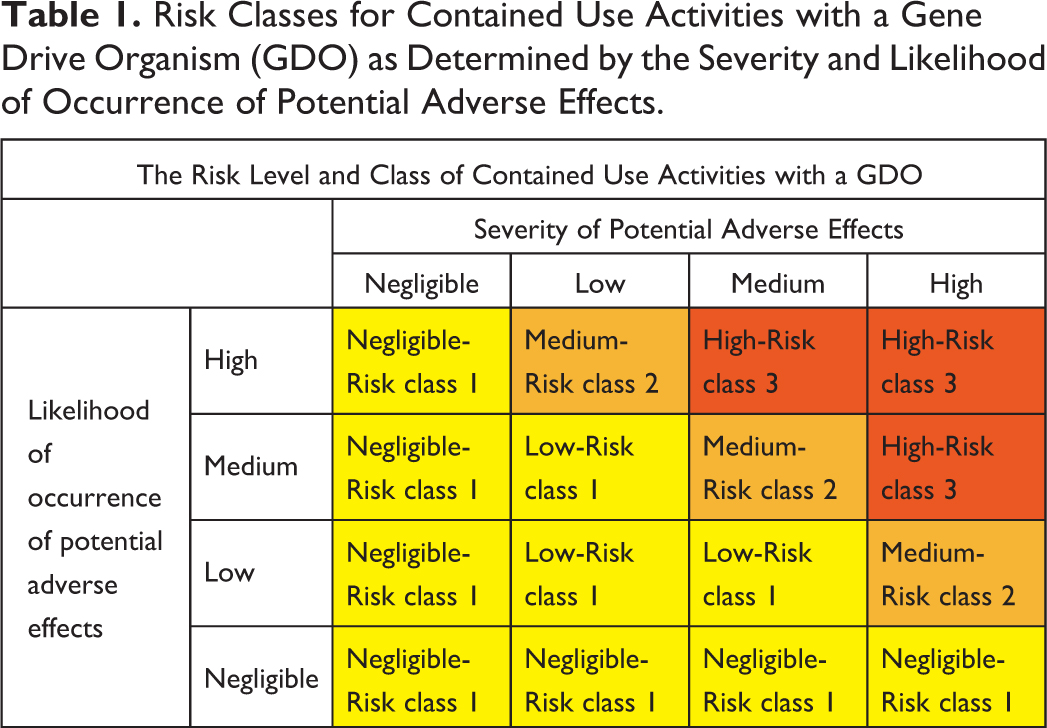
Combining the high severity of potential adverse effects and their negligible likelihood of occurrence in Table 1, risk class 1 is assigned to these activities.
It is clear that if the ecological containment was not in place, this activity would be assigned to risk class 3 due to the high severity of the potential adverse effects.
It should be noted that if a gene drive is designed for application in a pathogenic organism or if a gene drive cassette may result in pathogenic effects, then adverse effects due to pathogenicity should be considered in addition to the aforementioned adverse effects.
Assessing the Severity of Adverse Effects
Knowing what adverse effects must be addressed, the severity of these effects needs to be estimated (Figure 1, Box 2a) in the second step of the risk assessment procedure. In the context of the adverse effects, severity is defined as the expected persistence, namely, the duration a GDO will remain in the environment, and the spatial dimension of the affected area. It is related to the nature and intrinsic properties of a GDO, and its estimation should consider the characteristics of the inserted gene drive cassette and the recipient organism.
Determination of the Risk Class of a Rodent (Mus musculus ) Gene Drive Organism (GDO).
Manipulations involve feeding and rearing GDOs, including transfer to cages and collecting blood samples.
Mice have a natural habitat outside of the laboratory environment and may reproduce either with wild relatives or with escaped “laboratory mates.”
The
Combining the severity of potential adverse effects and their likelihood of occurrence in Table 1, risk class 2 is assigned to these activities.
As for a GMO, the specific function, location in the genome, and stability of the inserted gene drive cassette should be characterized. In the particular case of a GDO based on CRISPR/Cas9, this cassette contains the endonuclease Cas9 and a gRNA directed to a target DNA sequence, which modifies a particular genetic trait and may influence the fitness of a specific population. It is primarily this modification that will define the severity of the adverse effects because it determines the extent of dissemination and survival and the extent of the development of resistance. Thus, the design of the gene drive cassette could also mitigate the severity of the adverse effects, for example, by employing a split gene drive 15 in which the gRNA and endonuclease are placed at different genomic loci, thus being less effective as a drive. Besides the genetic elements of the gene drive cassette, a cargo gene can be transmitted. The function of this cargo gene in the biology of the recipient organism also needs assessment.
The intrinsic properties of the recipient organism are considered as factors influencing the severity of adverse effects. For instance, the species or behavior of the recipient influences the extent of its spread within and between populations, for example, through limiting environmental conditions, defined movement patterns and ranges, or reproduction cycles and mating behavior. It is therefore essential to consider a recipient’s ecology and its impact on the GDO’s capacity to transmit a gene drive cassette to offspring. Additionally, the possibility of an alteration of the recipient organism’s characteristics after insertion of the gene drive cassette should also be analyzed. Again, to mitigate the severity of adverse effects, laboratory strains may be established that are artificially modified to be susceptible to a certain chemical.
The severity of adverse effects based on the aforementioned characteristics is classified from negligible, low, medium, to high (Table 1).
Risk Classes for Contained Use Activities with a Gene Drive Organism (GDO) as Determined by the Severity and Likelihood of Occurrence of Potential Adverse Effects.
Assessing the Likelihood of Occurrence of Adverse Effects
The next step in the risk assessment procedure is the estimation of the likelihood, namely, the probability of occurrence of any of the identified adverse effects (Figure 1, Box 2b). The likelihood is determined by assessing the characteristics of the intended activity subject to the characteristics of the GDO as described before and the potentially exposed environment.
The intended activity could range from just storing GDOs to manipulating them outside their primary containment, handling mobile organisms versus immobilized organisms, or differences in scale (number of individuals). The likelihood that an adverse effect occurs due to an activity depends on the organism’s characteristics (ability to survive, spreading potential) and the exposed local environment allowing the GDO to establish and spread or not. These characteristics include, in the first instance, the local climate conditions, presence of suitable mating partners, and prevalence of the gene drive target site in the local population. As with the severity of the adverse effects, their likelihood of occurrence may be lowered by introducing artificial properties into the recipient strain. For instance, laboratory strains may be established that for their survival or reproduction depend on specific nutrients not present in the environment, or the gene drive cassette may be directed toward an artificially introduced sequence with no natural homologs. If the environmental conditions for survival, reproduction, and transfer of the gene drive cassette to offspring are nevertheless met, the expected persistence of the gene drive may also directly influence the likelihood of an adverse effect. For example, the gene drive may be permanent or transient (eg, a daisy chain gene drive 16 ) or contain a cargo gene with high fitness cost or rapid resistance development. The level of likelihood of occurrence is, like that of severity, classified from negligible, low, medium, to high (Table 1).
Assignment of a Risk Class to the Activity
The combination of the level of severity and the likelihood of occurrence of the potential adverse effect(s) in case of an unintentional release determine the risk class for the contained use activity with a GDO (Table 1). In assigning this risk class, the following definitions for a negligible-, low-, medium-, and high-risk level are used.
Risk class 1 is a negligible- or low-risk defined by a (1) negligible likelihood of occurrence of adverse effects due to the intended activity, (2) negligible severity of the adverse effects due to the characteristics of the GDO, (3) combination of a low to medium likelihood of occurrence with a likely low severity, or (4) combination of a low likelihood of occurrence with a likely medium severity.
Risk class 2 is a medium-risk defined by a medium likelihood of occurrence of adverse effects with a medium severity of the adverse effects, or a combination of a low likelihood of occurrence with a high severity, or vice versa.
A risk class 2 activity is considered as having nonpermanent (transient) consequences if the GDO is unintentionally released into the environment. The initial situation can be restored naturally or artificially by a reversibility strategy. For example, the impact of the release is limited in space and the number of affected generations of the target population. The (repetitive) release of wild individuals can replace the GDOs, and the natural ecosystem can be reestablished with time.
Risk class 3 is a high-risk defined by a high likelihood of occurrence of adverse effects with a high severity of the adverse effects, or a combination of a high likelihood of occurrence with a likely medium severity, or vice versa.
A risk class 3 activity is considered having permanent (nonreversible) consequences and/or is extensive in distribution if the GDO is unintentionally released in the environment.
The outcome of the risk assessment, the assignment of an activity with a GDO to a risk class, is indicative for an adequate risk management strategy proportionate to the estimated risk.
Risk Management for the 3 Risk Classes
To determine adequate control measures for the 3 risk classes for contained use activities with a GDO, the following considerations should be taken into account. A risk class 1 activity with a GDO has by definition a similar risk as a biosafety level 1 (BSL-1) activity with a GMO in the sense that no additional adverse effects due to the spread of the GDO or its genetic trait into the environment are expected in the case of an unintentional release. The requirements for a risk class 1 contained use activity with a GDO are therefore based on BSL-1 17 and, when applicable, an arthropod containment level 2 (ACL-2), which is the accepted standard for GM insects. 18
For risk class 2 and 3 activities, control measures for preventing unintentional release of a GDO into the environment must be followed according to the BSL-2/3 and/or ACL-3 safety levels. Minimum measures specifically addressing containment of risk class 2 and 3 activities and those for activities with rodents, insects, and fungi, which are most likely to be used for gene drive applications, are presented in Table 2. In general, additional layers of physical containment are essential for risk class 2 and 3 activities with a GDO, and access restrictions are more stringent for a risk class 3 activity to decrease the likelihood of an unintentional release. For organisms like yeast or spore-forming fungi, which may disperse via the air, control measures like working in a Class II biosafety cabinet are recommended if these organisms are used as recipients of gene drive cassettes. Additionally, it is recommended for class 3 activities that laboratory extract air is HEPA filtered. A second group of organisms likely to be used for the construction of GDOs are arthropods. Their size and in part their ability to fly require more stringent control measures regarding prevention of an escape and their effectiveness. Similarly, the control measures for rodents are specifically dedicated to the identification of an escaped animal among wildtype organisms. Furthermore, depending on the specific gene drive to be studied, it may be necessary to implement additional control measures as identified through the local risk assessment process. Measures related to the prevention of infection of the worker and potential transfer of pathogenic microorganisms outside of the laboratory can, however, be omitted for nonpathogenic GDOs.
Minimum Control Measures for Risk Class 1, 2, or 3 Activities with a Gene Drive Organism (GDO) in Containment.a
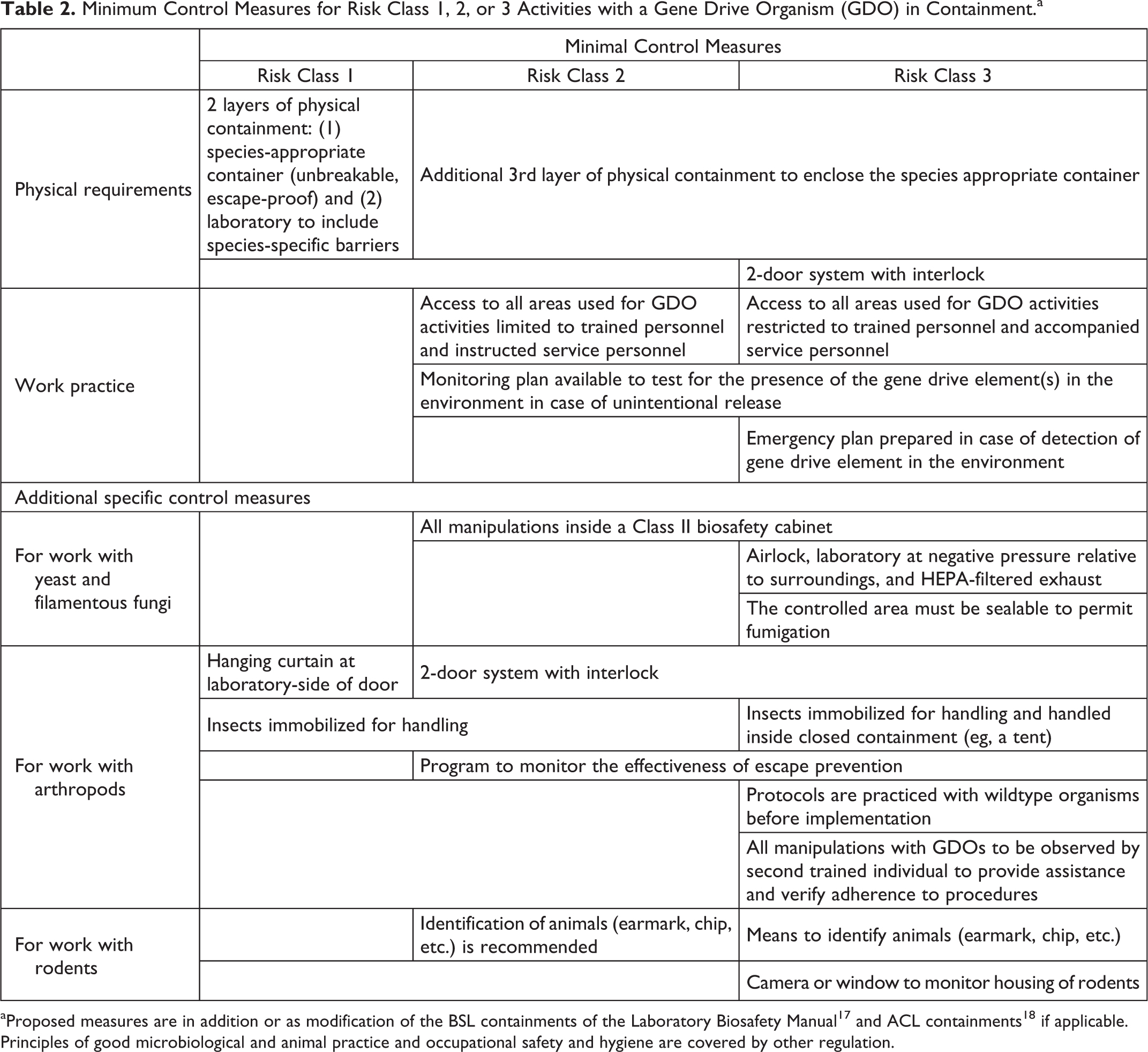
Proposed measures are in addition or as modification of the BSL containments of the Laboratory Biosafety Manual 17 and ACL containments 18 if applicable. Principles of good microbiological and animal practice and occupational safety and hygiene are covered by other regulation.
Akbari et al 9 proposed multiple confinement strategies for gene drive research, which included molecular, ecological, and reproductive barriers in addition to physical barriers. The former 3 barriers are factored into the risk assessment procedure described here. Moreover, if the outcome of the assessment is a risk class 3 activity, the addition of at least one of these confinement strategies should be considered to reduce the risk class.
If a risk class 3 activity with a GDO could result in the spread into the environment due to an unintentional release, a protocol for incident management (ie, an emergency plan) to limit and mitigate the adverse effects must be available. Several strategies are being developed to deal with the effect of an unintentional spread of a GDO. These may include the release of the unaltered recipient organism or a second GDO to restore the natural phenotype (immunity or reverse gene drives 12 ). Research into these strategies is at an early stage, and the level of success is impossible to predict, and some ecological effects may occur before the mitigation strategy takes effect. Moreover, the utilization of immunity or reverse drives as mitigation strategy will require an authorization for the deliberate release of GMOs according to national or supranational regulations, for example, Directive 2001/18/EC 19 in the European Union. As this type of approval will require an environmental risk assessment and potentially multinational agreements due to the possibility of a spread into neighboring countries, this approach is likely not a viable option during the research phase of a GDO.
Safety measures for transport outside the facility are covered by other legislation and not dealt with here. However, the transport of a GDO being assessed as a risk class 3 activity must be avoided if possible. Instead, it would be preferable to send the gene construct and recipient organism separately. Additionally, biosecurity considerations (eg, prevention of theft) are not part of the measures prescribed here but should be considered in line with local/national requirements.
Conclusion
The fundamental purpose of any GDO is its establishment within a wild population, which it concomitantly is supposed to alter, suppress, or even eliminate. However, during the initial stages of the development of such organisms, this gene drive property also poses unique challenges to users and regulators 9 trying to anticipate a sufficient level of safety and security to prevent the unintentional release of GDOs from the laboratory. These conditions to study a GDO may initially call for a precautionary approach to risk assessment, considering the worst case scenario when assessing the risks for the environment.
The risk assessment of a GDO in contained use therefore has a strong focus on the prevention of adverse effects to the environment by preventing the release of any GDO into the environment. This diverges from the typical approach of an environmental risk assessment 19 during the approval of a planned release of a GMO, which assesses the overall acceptability of the environmental introduction of the GMO. Logically, such an assessment requires a substantial amount of prior knowledge about the GMO and the receiving environment. Accordingly, consequences of the gene drive technology like altering complex ecological networks in which the modified population plays a role through predator-prey, competitive, symbiotic, or other relationships are not factored into a risk assessment for contained use. In the same way, the economic impact of an unintentional release of a GDO could have serious consequences. Nevertheless, an economic impact is generally outside the scope of a biological risk assessment of a contained use activity.
The principles of the risk assessment presented here are based on extensive experience with risk assessment of GMOs based on the applicable European Directive (2009/41/EC). 13 Potential adverse effects specific to a GDO are different from those of potentially pathogenic GMMs, but the general principles to address these adverse effects are in essence laid down in the directive. The risk assessment as proposed here describes risk classes for contained use activities with a GDO for which control measures according to existing BSLs need to be adapted. The proposed control measures presented can be used as a starting point to assist with the selection of necessary controls. However, the risk of each activity should be assessed on a case-by-case basis and the selection of control measures adapted to each activity.
The risk assessment framework presented here describes the first structured methodology specifically aimed at assisting in the assessment of GDOs in contained use. It thus addresses the demand from several countries to have a consistent approach to the assessment of the biological risks of gene drive technology as any unintentional release may have international consequences. Likewise, a consistent approach of a GDO risk assessment as well as guidance material is necessary for both users and risk assessors to facilitate the development of the technology while ensuring that risks are appropriately managed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Disclaimer
The views presented in this publication are those of the authors and do not necessarily reflect the positions of the associated institutions.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
