Abstract
Emerging technologies in the life sciences call for new models of biosafety risk management. We examine the question of how to address new developments in the life sciences and biosciences in a bottom-up manner—that is, from the concrete level of biosafety practice with a focus on the risk management and risk assessment of emerging technologies in the biology laboratory. We use research on “gene drives” as an example of challenging work with new constructs that have major biosafety implications for the work in the laboratory and beyond. Gene drives are intended for use in ecosystems and require, at an early stage, the consideration of potential future biosafety, biosecurity, and societal impact. We argue for an integrative approach, a truly collaborative model that involves scientists, biosafety officers, institutional leadership, and ethics consultants, with the aim of maximizing safety as well as scientific progress.
Emerging technologies open up avenues to new knowledge, breakthrough discoveries, and the invention of further technologies in a dynamic, cyclical process. 1
Because of their novelty, emerging technologies may challenge existing rules and regulations and established professional practice. 2 In the United States, the higher-level governance of biotechnology, including the oversight structures, is comprised in the so-called coordinated framework, which was established in 1992 and underwent extensive revision in 2015, resulting in an update released on January 4, 2017. 3 New technologies and discoveries in the biosciences can pose a challenge to the professionals—researchers as well as biosafety officers—at the level of the laboratory, where biosafety rules and practices are in use that cannot accommodate for the specific risks and safety requirements of work with the new technologies.
In this article, we examine the question of how to address new developments in the life sciences and biosciences in a bottom-up manner—that is, from the concrete level of biosafety practice with a focus on the risk management and risk assessment of emerging technologies in the biology laboratory. We use research on “gene drives” as an example of challenging work with new constructs that have major biosafety implications, ultimately far beyond the limits of the laboratory.
We propose that the existing integrative approach, combining tried-and-true biosafety methods of risk assessment and risk management, be expanded to cover all biosafety aspects of emerging technologies. Gene drives, the novel technology that we address here, have the unique property of being built to spread through ecosystems in a self-propagating manner and therefore require specific risk management. New technologies in synthetic biology have their own complex risk profiles, and a truly collaborative biosafety risk management model is needed that involves scientists, biosafety officers, institutional leadership, and ethics consultants, with the aim of maximizing safety as well as scientific progress. Preemptively including professional ethicists in the discussion of the biosafety risk management of laboratory-based research in synthetic biology is not yet regular practice. The involvement of ethicists—through institutional review boards or clinical ethics consultation—alongside institutional biosafety committee oversight is known from clinical trials of human gene transfer methods. In that context, however, the specific charge to the ethicists is to ascertain the upholding of highest ethical standards in clinical research, particularly with regard to voluntary, valid informed consent and the rights, interests, and well-being of study participants. The role of ethicists in laboratory-based fundamental synthetic biology research is a different one, as no interests of human study participants are at stake. In addition, the ethical questions get more complex when the research reaches the stage of transition out of the laboratory and into the area of application. In the case of gene drives, this means the transition into the ecosystem in the form of controlled field trials.
While ethical aspects of biological research are often addressed on the basis of their relevance for governance, in terms of higher-level regulation and policies, we want to show the integration of these aspects at the concrete level of biosafety practice in the work of biosafety officers—that is, in the biosafety assessment of research protocols in close collaboration with the researchers. Ethical questions should be addressed from the earliest stages of the research planning, alongside the biosafety assessment of the bench work that involves new technologies.
A model for practical implementation is presented here, but first we examine gene drives as an example of an emerging technology in the broader background of current biosafety practice.
Current Concerns: High-Risk Human Pathogens
Biosafety traditionally focused on the work within the laboratory spaces (and transport between laboratories) and with the containment of select agents, toxins, and pathogens that should never leave the laboratory. Currently, the key criterion for determining the risk of work in biological laboratories is the risk of harm to human health; the most stringent biosafety regulation thus applies to work with “dangerous and exotic agents that pose a high individual risk of aerosol-transmitted laboratory infections and life-threatening disease that is frequently fatal, for which there are no vaccines or treatments, or a related agent with unknown risk of transmission.” 4 Such pathogens have pandemic potential.
New constructs, such as viruses with engineered “gain of function” that augments the transmissibility and/or pathogenicity, are of particular concern. They must be categorized within the existing biosafety level system and managed accordingly. As they pose a serious direct threat to human health, work with these organisms may require the highest levels of containment in biosafety laboratory level 3 or even 4 facilities. Research into the airborne transmissibility of the avian influenza strain H5N1 among ferrets and the consequences for transmissibility among humans triggered in 2011 a worldwide debate on gain-of-function research. 5 The research, by a group from the Netherlands and a joint Japan-US team, was conducted in the enhanced animal biosafety laboratory level 3 facility at Erasmus Medical Center Rotterdam, the Netherlands, and at the enhanced biosafety level 3 containment laboratories of the University of Wisconsin–Madison.
A prominent question gaining broad attention was about publication of the studies and the outcomes. Upon initial submission of 2 manuscripts, both
Yet, in spite of the considerable efforts, the fact that these activities were organized by and directed at the scientific community and at actors at the higher levels of governance and science policy elicited fierce criticism. Among the critics, Lipsitch et al called for a broad public dialogue on the policies regarding research into gain of function and pathogens with pandemic potential, pointing to the inclusion of the public in the debates on, for example, the application of genome-editing technologies for intervention in the human germ line or for intervention in ecosystems. 12
New Constructs: Gene Drives
While we may feel familiar with the risks of select agents, toxins, and pathogens, including those with pandemic potential that pose a risk to human health and animal health, we are little prepared for dealing with novel constructs with an entirely different risk profile that can rapidly alter the natural ecosystem, even though these constructs do not threaten human health in a direct way.
“Gene drives” are the prime example of this type of construct. The term “gene drive” stands for novel genetically engineered organisms that contain the combination of a specific genetic alteration—a CRISPR-mediated gene edit—with the biological mechanism of a “driving” gene that ensures biased inheritance surpassing Mendelian rules.13,14 Gene drive systems can rapidly spread a particular trait through sexually reproducing plant or animal populations. Once introduced in a population, gene drives will spread in the environment in a self-propagating manner. In other words, while gene drives are intended for use outside the laboratory, any accidental or otherwise unintended release will have serious consequences. This novel and potentially disruptive technology raises a broad range of questions.
To address the technology and its numerous aspects, the National Research Council of the National Academy of Sciences convened an expert committee to review the state of the science of gene drive research, present case studies, determine the adequacy of existing oversight and risk assessment guidance, and discuss ethical, legal, and social issues. The committee was also asked to examine the role of institutional biosafety committees. A comprehensive report,
Although gene drives do not pose a threat to human health like pathogens with pandemic potential, their propensity to spread beyond control poses a major risk to the environment. Therefore, there should be no accidental or otherwise unintended release of these constructs from the laboratory—even though the ultimate purpose is release and application in ecosystems. The Netherlands is the first country to issue legislation concerning research with gene drives (Box 1).
The Netherlands: Legislation on Gene Drive Research.
On February 8, 2016, the Dutch National Institute for Public Health and the Environment published a policy report on gene drives, including recommendations concerning the requirement of formal authorization of any work with gene drives. 17 On July 1, 2016, the amended regulation on the use of genetically modified organisms came into force. With that, the Netherlands became, to our current knowledge, the first country with gene drive–specific regulation with the force of law. The regulation requires prior authorization of any work with gene drives and places it in the highest restriction category, which means work at biosafety level 4 (BSL-4). The Netherlands, however, do not possess a BSL-4 facility. When a BSL-4 requirement applies, researchers can request an exemption and a special assessment of their planned research to determine if and under what conditions the research can be safely conducted at a lower biosafety level. Thus, the BSL-4 requirement for any gene drives work is intended to enforce/ensure a comprehensive risk assessment on a case-by-case basis.
These developments call for intense scrutiny of biosafety requirements as well as the biosecurity aspects of accidental, unintended—and ultimately intended—release of such novel engineered organisms into the environment. Moreover, the biosafety and biosecurity aspects give rise to ethical questions that, in the case of gene drives, must be placed in the context of ecosystem impact, just distribution of benefits and burdens, and societal acceptance. We use the example of the unfolding gene drive technology to show how an ethics assessment can be integrated in the biosafety risk assessment and risk management model. Our aim is to examine the perspectives that such an integrated risk management model offers for the work of biosafety officers, oversight bodies, and regulators.
Toward Adaptive Risk Assessment
Current risk management in biosafety is agent based, with a focus on the public health implications in the case of accidental release, exposure, or transmission. In the new genomic technologies, risks do not arise from particular agents but from the rapidly evolving work with novel synthetic constructs, requiring adaptive risk assessment to keep pace with developments. With genome engineering, the integration of the construct into the genome is one element of the risk assessment strategy, and the release of the resulting modified organism is another critical aspect. In the case of gene drives, release and effective spread in the environment are the ultimate purpose of the work. The need for a team approach is evident, as multidisciplinary expertise is required to ensure that all aspects of a new technology, including the inference of unknown and unsuspected consequences, are part of the risk assessment and risk management process.
When the core concept of laboratory-acquired exposure is not applicable because a technology poses minimal risk of direct human exposure, the framework of biological risk assessment needs to be adapted to allow for the proper safety evaluation of the new technology. Increasingly, emerging technologies go beyond the framework for known infectious hazards, and agent-based risk assessment is not applicable, so an adaptive approach to risk assessment must be used to develop a risk management plan that fits these technologies.
Adaptive Risk Management in Practice
Risk management design, as the basic element of biosafety practice on which we build here, is a multitiered approach involving scientists and investigators, biosafety professionals, ethicists, and institutional leadership at the very beginning of the design process. This is in line with the best practice of biosafety risk assessment, as currently implemented by biosafety professionals. It ingrains biosafety risk assessment of the technology and provides a manner to evaluate the biosecurity and environmental impact of the proposed research. With emerging technologies, oversight is a moving target, as the risks of new technologies may not yet be known or fully analyzed. For the risk management design, this means that biosecurity risk mitigation must look at known effects as well as potential outcomes that may or may not result from the experiments. This form of adaptive risk management occurs in the absence of specific government regulation, even though components of the experiments might be linked to existing systems that govern biotechnology. The National Academies of Sciences, Engineering, and Medicine, in the report
In practice, the first step is to form the risk management team. This should be a collaboration of scientists, safety professionals, institutional leadership, especially those who will provide review and oversight through institutional biosafety committees, institutional animal care and use committees, and institutional review boards, depending on the nature of the experiment and materials or test subjects involved. At this point, it is also crucial to seek ethical and public affairs consultation to ensure that the proposed experiment can be assessed and receive important nonscience/technical input.
The risk management team will assess the nature of the experiment, including use, release, and exposure, and the consequences for the environment directly and for humans indirectly. The next important concept to discuss is the potential biosecurity risk of the experiment. Does it enhance the lethality or disease transmission? Can it happen accidentally? Is it a type of research that would be considered to have dual-use capabilities or, even possibly, dual-use research of concern, as defined by the Office of Science Policy of the National Institutes of Health? In the case of the gene drive experiments, a significant shift occurred from the prevention of human and animal exposure to a focus on mitigating the risk of accidental environmental release. Even a single organism escaping from a laboratory setting during experimentation can have a significant impact on the wild population of that organism, as confirmed through mathematical modeling. 18
Risk mitigation measures are a key part of the risk management plan and include modification of the design or conduct of the research, the application of specific or enhanced biosecurity or biosafety measures, regular review at the institutional level of emerging research findings, and adaptation of the risk management practice, as needed.
Regarding research with potential adverse biosafety or biosecurity impact, the overall design of the research proposal should be assessed and substitution considered—specifically, replacing high-risk constructs and activities with lower-risk ones that still produce the same experimental results. This type of modification is the best known way to negate a potential high-risk research area; however, it may not be always feasible in the case of entirely new biological constructs.
The next step in the adaptive strategy is the application of specific or enhanced environmental, biosecurity, or biosafety measures. In instances where substitution of high-risk elements is not possible, safety and security measures can be enhanced by leveraging inherent molecular containment mechanisms in a given organism and adapting practices and procedures. Management of the levels of control in the organism can be implemented through molecular, ecologic, and reproductive containment measures. For the laboratory bench areas where work is performed, containment control can provide barriers and other security measures. Finally, risk management and mitigation can be effectuated through procedures that include additional controls, practices, and procedures, beyond biocontainment. One example is the use of specialized caging units, transport containers, and specialized handling procedures that can further control the work with the organisms carrying novel constructs and mitigate the risks of the experiment.
No scientist stands alone with this risk management process. Risk management and risk mitigation are complex steps, and experts must be brought in as necessary, to ensure that communication and actions are appropriate and in accordance with the mission of the institution where the experiments are performed. Every institution must have a system of oversight to review the work. This is a crucial step in ensuring that the “left hand knows what the right hand is doing.”
An emerging technology project should never be treated as a static process, and the review team should aim to review the research before it begins and then periodically assess and evaluate the project concerning changes in the research that may present additional elements of importance for risk management. Information about the progress of the study, changes to the experiments, as well as outcomes and discoveries should be consistently reported to the oversight committees responsible for review.
Adaptive Risk Management in Gene Drive Research
In a landmark article that has set standards for the field, Akbari et al call for safeguards for laboratory experiments with gene drives. 19 The group of leading scientists considers the following requirements a necessary minimum for safe research with gene drive technology: First, a thorough up-front biosafety risk assessment must be in place. Expert advice must be proactively sought whenever needed, and the expert evaluations should be made publicly available. This step perfectly fits with the collaborative team approach in the development of risk management plans for emerging technologies, as outlined here.
Second, Akbari et al require that experiments must employ, whenever possible, at least 2 stringent confinement strategies, to minimize the risk of unintentionally altering wild populations. The authors define molecular, ecologic, reproductive, and barrier types of strategies. Molecular confinement can, for example, be achieved by separating the guide RNA cassette and the Cas9 gene, as that prevents the formation of an autonomous drive system. Ecologic confinement entails limiting the laboratory work to organisms that do not naturally occur in the local environment to reduce the risk that any engineered organism will survive in the wild after unintended release. Reproductive confinement entails the use of laboratory strains that cannot reproduce with wild organisms. In addition, a team approach is recommended for the development of the risk management plan. For new and emerging technologies, enhanced biosafety, biosecurity, and containment control shall be applied, with organismal controls, molecular controls, and enhanced procedures. The provisions described by Akbari et al largely concur with the risk management elements that we propose here.
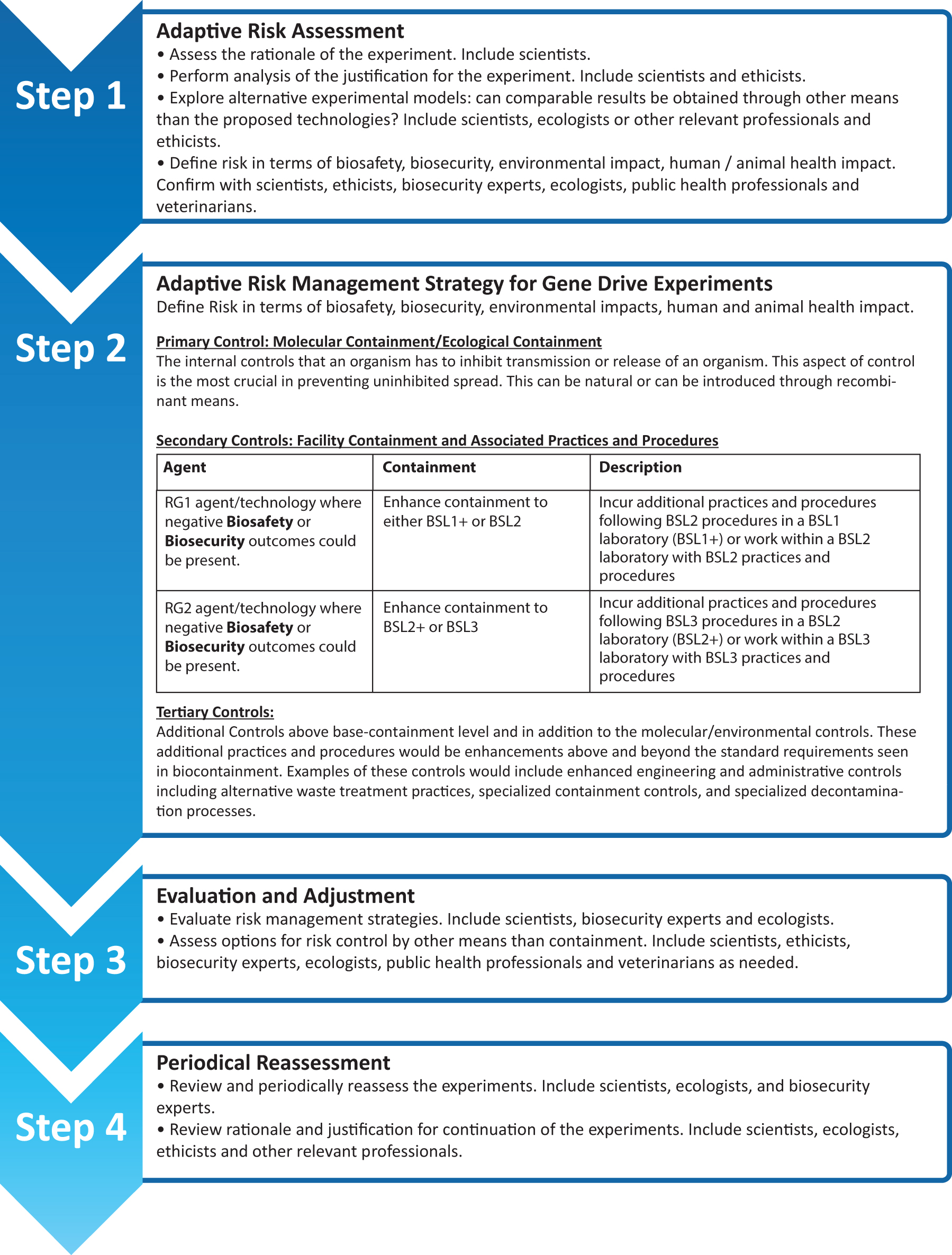
Risk Management Process Flow.
Finally, organisms carrying gene drive systems should not be distributed to other laboratories until formal biosafety procedures have been established. Setting-up such procedures requires collaboration with the institutional oversight bodies to establish a governance structure for the initiation and proper review of the experiments. In the concrete case of gene drive research risk management, the environment should be assessed for the existence of controls that can be used locally, which means that it may not be possible to use the same controls consistently in other locations. This underscores the fact that expertise is needed that expands beyond that of any one single participant, to ensure that the risk management elements are site specific and locally adapted as needed.
Ethical Dilemmas and Emerging Technologies in Synthetic Biology
“Is there anything unique in the ethics of synthetic biology?” The philosopher David Heyd asks this question in the title of an essay and answers that synthetic biology does not create any truly novel ethical dilemmas, nothing that we have not encountered before in the questions related to our actions and interventions in the world around us. 20 Even if this were true in general, we may need to consider new types of dilemmas with some of the most recent emerging technologies, particularly those that are intended for use outside the laboratory in natural ecosystems, forcing us to include complex systems risks in our ethical analysis.
How do risk management and ethics fit together? Consider risk assessment using probabilistic terms: outcomes are uncertain, and the risk of harm is never zero. Decisions in risk management, research, and prospective applications of emerging technologies in synthetic biology lead back to ethical questions: What trade-off between the chance of benefit and the risk of harm is justifiable and acceptable and for whom? It is the trade-off, the inevitable price that has to be paid, that turns choices into dilemmas. Such dilemmas place ethical considerations at the core of biosafety risk assessment.
What is the right course of action? In genomic science and engineering, we can find ourselves in a situation where we must pause and decide if and how to move forward or whether to pursue a certain line of research at all. Ethical considerations related to synthetic biology, very roughly, fall into 2 categories.
First is the question of whether it is a morally right action to “remake nature” through biological engineering, to alter living organisms and the environment. This first concern considers the moral quality of such interventions: Are those acts permissible, independent of their consequences? In this context, one can ask whether changes made through the means of synthetic biology should be judged differently from changes that occur spontaneously, by “natural” causes. How we appreciate the “natural” is highly dependent on our value system, including our cultural and religious beliefs. The “natural” is not per se morally superior to the “artificial.” 21 The concrete options for the alteration of the genome of any organism have acquired an entirely new dimension with the developments of high-precision genome editing with CRISPR systems. This changes the empirical context in which ethical questions are asked, even if there may be no unique “genome editing ethics.”
The other traditional approach in ethics looks at the consequences of our actions, which, in the context of genome editing, raises questions such as the following: What are the potential beneficial and harmful effects for human well-being, the human environment (including wild and domesticated plants and animals), and the larger-scale ecosystems? A consequentialist approach in ethics builds on real-world consequences, not dependent on beliefs but observable, measurable, and, as we hope, calculable and predictable outcomes. With gene drives—that are intended for ecosystem management—the assessment of the very large-scale and long-term potential effects, both beneficial and harmful, is critical for any ethical analysis. The sheer magnitude of the possible impact may warrant caution in taking the next steps toward implementation of the technology, but how much precaution is necessary and effective?
Effective Precaution Through Adaptive Risk Management and Shared Responsibility
The uncertainty about outcomes and the proportionality of estimated benefits and harms of emerging technologies is, at the level of public policy and risk governance, often addressed by a call for precaution. 22 While exercising precaution seems wise, invoking the “precautionary principle” as the basis for policies does not provide clear guidance for risk management. In-depth theoretical analysis shows that precaution is a valuable concept but that it cannot be applied in a broad and generic manner to synthetic biology and other emerging technologies. The precautionary principle in decision making about risks can be effective only if applied to a concrete technology or research project: evaluation on a case-by-case basis is an absolute requirement. 23
This strongly reinforces the model that we are recommending: adaptive risk assessment and risk management through an integrative approach, involving scientists, biosafety officers, institutional leadership, and ethics consultants, with the aim of maximizing safety as well as scientific progress.
The professions involved all have their own guidelines, best practice standards, and frameworks outlining professional responsibilities—together a rich and complementary resource in collaborations.24 -26
The biosafety profession must continue to engage the research community in the joint effort of providing effective review of new and emerging technologies. Now, more than ever, the relationship between biosafety professionals and the scientific community should be one of shared responsibility—in cases where regulation is in place and in the yet unregulated cases, such as the “gene drives.” A strong partnership is necessary to uphold awareness of the impact of the experiments associated with new and emerging technologies. Such a partnership must be built on the foundation of risk assessment and the subsequent risk management principles.
Footnotes
Acknowledgment
Jeantine Lunshof receives funding through the Greenwall Foundation, “Making a Difference” program.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
