Abstract
Background:
Neurodegenerative diseases, characterized by progressive neuronal degeneration, are increasingly prevalent due to global aging trends and impose a significant burden on patients. No cure currently exists, with oxidative stress and inflammation serving as key drivers of disease progression. Advances in imaging technologies and artificial intelligence (AI) offer new opportunities for early diagnosis, monitoring, and treatment evaluation. This review aims to summarize the role of advanced neuroimaging modalities and AI integration in improving the diagnosis, monitoring, and management of neurodegenerative diseases, while highlighting current challenges and future directions.
Material and Methods:
A narrative review was conducted based on published literature on neuroimaging techniques in neurodegenerative diseases. Key modalities included structural and functional magnetic resonance imaging (MRI, fMRI), diffusion tensor imaging (DTI), positron emission tomography (PET), and single-photon emission computed tomography (SPECT). The integration of AI in image analysis was evaluated for its impact on diagnostic accuracy and workflow efficiency. Sources were selected from peer-reviewed journals focusing on clinical applications, technical advancements, and multimodal imaging strategies. Results Structural MRI, fMRI, and DTI provide detailed insights into brain atrophy and microstructural integrity, while PET and SPECT enable molecular-level assessment of metabolism and pathology. AI-enhanced analysis reduces interpretation variability and improves diagnostic precision. Despite these advances, high costs, limited accessibility, and inter-expert subjectivity remain major barriers. Emerging multimodal approaches and AI-driven tools show promise in enabling earlier detection and personalized treatment monitoring.
Conclusion:
The integration of advanced imaging and AI holds transformative potential for neurodegenerative disease management. Future efforts should prioritize cost reduction, improved accessibility, and seamless multimodal data fusion to translate these technologies into routine clinical practice.
Plain Language Summary
Neurodegenerative diseases, such as Alzheimer's and Parkinson's, cause gradual loss of brain cells and have no cure. They are becoming more common as people live longer, creating a heavy burden for patients and families. This review explains how modern brain imaging tools-like MRI, PET, and newer methods-help doctors detect these diseases earlier, track their progress, and check if treatments are working. Adding artificial intelligence (AI) to analyze scans makes results more accurate and reduces mistakes. However, these technologies are expensive, not available everywhere, and sometimes hard to interpret consistently. The article highlights the promise of combining different imaging methods with AI to create personalized care plans, while calling for future efforts to lower costs and improve access so more patients can benefit.
Background
Neurodegenerative diseases, also known as neurodegenerative disorders, are characterized by the progressive degeneration of neurons in the brain and spinal cord. These structures are composed of neurons that perform critical functions, including motor control, sensory information processing, and decision-making. 1 Since neurons in the brain and spinal cord generally lack regenerative capacity, significant damage can lead to irreversible and severe consequences.2,3 Neurodegeneration manifests across multiple levels of the brain's neuronal circuitry, ranging from molecular alterations to systemic impacts. 4 Given the absence of established interventions to reverse the progressive neuronal loss, these conditions are deemed incurable. Nonetheless, research has pinpointed oxidative stress and inflammation as the principal contributors to neurodegenerative processes.5,6 While neurodegenerative diseases present with a wide spectrum of clinical features, they can be broadly grouped based on their primary initial manifestations, although considerable symptomatic overlap often develops as the diseases progress. One major group is characterized predominantly by motor dysfunction, including conditions such as cerebellar ataxia, Parkinson's disease (PD), and amyotrophic lateral sclerosis (ALS). 7 Another group primarily affects memory and cognitive functions, with prominent examples being Alzheimer's disease (AD) and frontotemporal dementia (FTD). 8 The incidence of neurodegenerative diseases has risen steadily in conjunction with the global aging trend, placing a significant burden on patients and their families. These diseases typically have a gradual onset and progressive nature, which makes early diagnosis particularly challenging. 9 Current therapeutic drugs often struggle to effectively cross the blood–brain barrier, leading to limited therapeutic efficacy and poor prognosis. Consequently, there is a critical need to develop innovative detection methods and treatment strategies.
In recent years, the diagnosis of neurodegenerative diseases has entered a new era of ultra-early biological detection, moving beyond traditional clinical phenotype analysis. Breakthroughs in biomarker precision such as blood p-tau217 testing, α-synuclein seed amplification technology, liquid biopsy techniques, genetic and epigenetic regulation analysis including multi-gene risk scoring and cfDNA methylation profiles, and artificial intelligence (AI)-driven multimodal data integration have significantly advanced,10–12 collectively expanding the diagnostic window and enabling personalized subtyping. In this context, imaging technology, as a core tool for disease decoding, is undergoing a transformative shift from unimodal approaches, which capture a single type of data (e.g., structural information from magnetic resonance imaging (MRI)), to multimodal integration, which combines data from two or more techniques to provide a more comprehensive pathological and functional assessment. This multidimensional approach is opening new avenues for the early diagnosis and precise management of neurodegenerative diseases. Innovations in imaging have opened new dimensions for the early diagnosis and precise management of neurodegenerative diseases. From traditional structural imaging to cutting-edge multimodal integration, imaging not only reveals the characteristic biomarkers of the disease but also provides unique insights into the pathological mechanisms and optimization of treatment strategies.
In structural imaging, multimodal technologies combine traditional MRI with advanced methods like diffusion tensor imaging (DTI) and arterial spin labeling (ASL) to accurately identify brain pathology. Medial temporal lobe atrophy, especially hippocampal atrophy, is a key marker for differentiating AD from Lewy body dementia. 13 In PD, DTI uncovers neural fiber damage by detecting changes in fractional anisotropy (FA) in the substantia nigra. 14 For instance, new retinal imaging techniques, such as optical coherence tomography (OCT), leverage the retina's connection to the central nervous system to identify retinal changes linked to AD and PD. While these techniques are emerging as powerful non-invasive research tools, their application in routine clinical practice is still limited pending further validation and standardization. Advancements in functional imaging technologies, such as positron emission tomography (PET) and single photon emission computed tomography (SPECT), enable dynamic molecular-level monitoring. PET, using tracers like 18F-FDG, shows metabolic declines in AD patients’ temporal–parietal regions, while SPECT detects abnormal striatal blood flow in PD diagnosis.15,16 Ongoing innovations, including fluorescent probes, enhance imaging sensitivity and early disease detection by visualizing specific biomarkers. 17 The integration of imaging and nanomedicine is revolutionizing diagnostics and treatment. Intelligent carriers like iron oxide nanoparticles (IONPs) enhance MRI contrast and deliver drugs across the blood–brain barrier, which could show promise for diseases like AD, PD, and ALS. 18 The synergy between radiomics and AI is advancing the causal analysis of imaging phenotypes, paving the way for personalized treatments.
In the future, imaging technologies are anticipated to transition from being solely diagnostic instruments to becoming a pivotal platform for the holistic management of neurodegenerative diseases. This evolution will be driven by ongoing technological advancements and interdisciplinary collaboration, facilitating the discovery and validation of imaging biomarkers, the dynamic analysis of pathological mechanisms, and the monitoring of precision treatments. These developments are expected to generate a synergistic effect, thereby enhancing diagnostic and therapeutic strategies toward earlier, more precise, and personalized approaches. As detailed in Figure 1, multimodal imaging combined with AI enhances diagnostic precision by integrating structural, functional, and molecular data, paving the way for personalized medicine.

A Schematic Overview of the Role of Advanced Imaging and AI in the Management of Neurodegenerative Diseases. The Figure Illustrates the Progression from Early Diagnosis Using Multimodal Imaging Technologies (e.g., MRI, PET, OCT, SPECT, fMRI, and CT) to the Integration and Analysis of This Data by AI Algorithms. This Process Enables the Identification of Disease Subtypes, Prediction of Progression, and Monitoring of Therapeutic Response, Ultimately Facilitating Personalized Treatment Strategies.
Overview of Imaging Diagnostic Technologies
Principles of Key Imaging Technologies
Imaging technologies vary in principles and applications. MRI uses magnetic fields and radio waves to generate detailed structural and functional images without ionizing radiation. PET and SPECT involve radiotracers to visualize metabolic processes, with PET offering higher resolution. Computed tomography (CT) employs X-rays for rapid structural scans, while emerging techniques like near-infrared spectroscopy (NIRS) use near-infrared light for functional monitoring. As research into neurodegenerative diseases progresses, imaging diagnostic technologies have become indispensable for early diagnosis, disease monitoring, and treatment evaluation. These technologies furnish both structural information and insights into functional alterations, thereby enabling clinicians to make more precise decisions in patient care. The advancements in imaging techniques present new opportunities for the early detection and the development of targeted therapeutic strategies for neurodegenerative diseases.
Magnetic Resonance Imaging
MRI is a non-invasive imaging technique that plays a pivotal role in diagnosing neurodegenerative diseases. MRI provides high-resolution structural images of the brain, which are instrumental in identifying pathological changes such as brain atrophy and white matter lesions. For example, in AD research, MRI is used to track critical indicators of disease progression, including amyloid plaque deposition and ventricular enlargement. 19 Moreover, MRI is increasingly utilized to assess brain function, particularly in monitoring cerebral blood flow and metabolism. Techniques such as ASL, like other MRI techniques, enable evaluation of cerebral perfusion without contrast agents, distinguishing it from some invasive alternatives but aligning with MRI's overall non-invasive nature, which is essential for the early detection of neurodegenerative changes. 20
Although MRI offers exceptional spatial resolution and tissue contrast, accurate image interpretation remains challenging in certain cases. For instance, brain tissue characteristics in AD patients may resemble those of healthy elderly individuals, necessitating the integration of clinical symptoms and other diagnostic methods to improve diagnostic accuracy. 21 Recent advances in AI have made the analysis of MRI images more intelligent. Research indicates that AI algorithms can significantly enhance the accuracy and efficiency of imaging diagnoses, particularly in the early detection of AD. 22
Computed Tomography
CT is a traditional imaging modality that, although less widely utilized in neurodegenerative disease diagnosis compared to MRI, still offers significant value in certain situations due to its rapid image acquisition and relatively low cost. CT is particularly critical in diagnosing acute stroke and traumatic brain injury, as it can quickly rule out hemorrhages and space-occupying lesions. 23 In neurodegenerative disease research, CT can help identify patterns of brain tissue atrophy. For instance, in PD, CT scans can reveal changes in the basal ganglia, providing valuable diagnostic insights for clinicians. 24 While CT images generally provide less detailed anatomical information than MRI, their ability to detect early-stage abnormalities may be limited. Preliminary evidence suggests that enhanced CT techniques, such as the use of contrast agents, can improve diagnostic accuracy for neurodegenerative diseases by enhancing the visualization of brain structures and helping to identify potential pathological changes.25,26 In the future, combining CT with other imaging technologies holds promise for further enhancing its diagnostic utility in neurodegenerative disease assessment.
CT provides detailed anatomical images, but lacks functional insights, which SPECT offers by using gamma rays from radioactive tracers to map physiological processes like blood flow and metabolism. This makes SPECT useful for diagnosing conditions such as AD. 27 Combining CT and SPECT in hybrid systems enhances diagnostic accuracy by linking structural and functional data, benefiting fields like neurology and oncology. This integration improves lesion characterization and retains CT speed and cost benefits, addressing neuroimaging limitations.
Positron Emission Tomography
PET is a key molecular imaging technique that allows for the assessment of brain activity and metabolic states at the functional level. PET has demonstrated significant potential in the study of neurodegenerative diseases, particularly in the early diagnosis of AD and PD. By using radiolabeled tracers that are specific to certain biomarkers, PET can detect pathological changes such as the deposition of amyloid plaques and tau proteins, providing preliminary evidence for early diagnosis and treatment efficacy assessment.28,29 In addition, PET can be combined with CT or MRI to form PET/CT or PET/MRI imaging modalities, offering both functional and anatomical information simultaneously. This hybrid imaging approach not only enhances diagnostic accuracy but also facilitates the monitoring of disease progression and therapeutic responses. For example, PET images in AD patients can reveal metabolic abnormalities in various brain regions, which may occur before clinical symptoms appear. 30 With the development of novel radio-labeled tracers, PET will continue to play an essential role in both the research and clinical applications of neurodegenerative diseases.
Comparative Analysis of Imaging Modalities
To synthesize the modalities discussed in Sections Principles of Key Imaging Technologies, Magnetic Resonance Imaging, Computed Tomography, and Positron Emission Tomography, Table 1 provides a comparative overview of major imaging techniques for neurodegenerative diseases. It summarizes their principles, diagnostic performance, practical considerations, and clinical readiness. For example, MRI offers high-resolution structural imaging but lacks the molecular specificity of PET, highlighting the growing trend toward multimodal hybrids (e.g., PET/MRI) that combine complementary strengths but face higher costs and validation needs.
Comparison of Key Imaging Modalities in Neurodegenerative Disease Management.

Overall, PET may offer higher diagnostic accuracy for molecular markers, while MRI remains a foundational tool due to its non-invasive nature and widespread availability. PET/CT might provide better cost-effectiveness and accessibility than PET/MRI, which may offer superior contrast but at a greater expense. The field is shifting toward integrated multimodal strategies to balance accuracy and practicality, with future progress depending on reducing costs and improving access in resource-limited settings.
Imaging Manifestations of Neurodegenerative Diseases
The prevalence, risk factors, and global distribution of diseases like AD, PD, and others underscore the need for advanced imaging to address diagnostic challenges, particularly in early detection and monitoring. By integrating epidemiological data here, we aim to contextualize how imaging technologies are applied to meet the specific burdens of these diseases, including disparities in access to diagnostic tools like MRI or PET, which significantly influence diagnostic rates and clinical outcomes. With technological advancements, imaging techniques provide critical insights into disease progression, thereby assisting clinicians in developing more effective treatment strategies. The hallmark features of neurodegenerative diseases are often closely associated with alterations in brain structure and function, which can be assessed using various imaging modalities, such as MRI, PET, and SPECT. These imaging techniques not only facilitate early diagnosis but also enable ongoing monitoring of disease progression and the assessment of therapeutic outcomes.
Alzheimer's Disease
AD, one of the most prevalent neurodegenerative diseases, plays a critical role in disease prevention and control, with its epidemiological characteristics and advancements in imaging diagnostic technologies being pivotal. Epidemiological data indicate a significant positive correlation between AD prevalence and age: the rate of occurrence in individuals aged 65 and above is 4%–7%, while it rises to 20%–30% in those aged 85 and older.31,32 The global number of AD patients reached 50 million in 2019, and it is projected to increase to 152 million by 2050, with the most pronounced growth expected in low-income countries due to an aging population. 33 This rising burden underscores the importance of imaging for early detection, yet access to advanced modalities like PET remains limited in low-income regions, with only 0.5–1 PET scanners per million people compared to 5–10 in high-income countries, contributing to diagnostic delays. 34 Regarding gender differences, the prevalence and mortality rate of AD are higher in women than in men, potentially due to hormonal factors and genetic susceptibility. Furthermore, AD has become the fifth leading cause of death worldwide, with related economic costs reaching $818 billion in 2015. However, the actual mortality rate may be underreported by 5–6 times. 35 Risk factors for AD include uncontrollable factors such as aging and the APOE ε4 gene mutation, 32 as well as controllable factors such as vascular risks (e.g., hypertension and diabetes) and poor lifestyle habits (e.g., high-salt diets and physical inactivity). In contrast, Mediterranean diets and cognitive training have been shown to have a protective effect. 36 Geographically, high-income countries exhibit higher prevalence rates (e.g., Japan's incidence rate is projected to reach 2.33% by 2024), while northern and rural regions of China experience higher rates due to dietary habits and the prevalence of cardiovascular diseases. In the United States, the southeastern region has the highest prevalence, driven by the concentration of elderly populations. 37 These regional disparities highlight the role of imaging, such as structural MRI, which is more widely available and used to detect hippocampal atrophy, enabling earlier diagnosis in high-prevalence areas.
Advances in imaging diagnostic technologies suggest essential support for the early detection of AD. Structural MRI (sMRI) has become a key biomarker in clinical diagnosis by detecting hippocampal and medial temporal lobe atrophy (with annual volume reductions of 2%–4%), and the extent of this atrophy is closely correlated with the rate of cognitive decline.21,38 Functional MRI (fMRI) and DTI have further enhanced the ability to identify early AD features, including reduced connectivity in the default mode network (DMN) and compromised integrity of white matter fiber bundles, changes that can be observed even before significant brain atrophy occurs. 39 PET imaging techniques provide precise, multimodal imaging to capture the pathological characteristics of AD: 18F-FDG PET reveals decreased glucose metabolism in the temporoparietal regions (diagnostic area under the curve (AUC) of 0.96, sensitivity of 90%, and specificity of 89%) 40 ; β-amyloid (Aβ)-PET and Tau-PET enable visualization of β-amyloid plaque deposition which can occur 10–20 years before symptom onset and tau tangles, helping differentiate AD from other types of dementia, such as Lewy body dementia. 41 The updated Aβ/tau/neurodegeneration (ATN) biomarker framework released in 2024 incorporates Aβ-PET or cerebrospinal fluid abnormalities as diagnostic criteria, advancing the standardization of AD diagnosis.42,43 Additionally, the “Consensus on the Application of Amyloid PET Imaging” published in China may have provided guidelines for technical operations and image interpretation.44,45
Looking ahead, the focus is on integrating multimodal technologies and optimizing clinical translation. Developing new imaging agents targeting inflammation or insulin resistance, and advancing the combined use of PET/MRI and blood biomarkers, are expected to enhance diagnostic precision. A key challenge for achieving global early screening remains the reduction of testing costs and improving access to imaging equipment in developing countries. As the ATN framework becomes more widely adopted and interdisciplinary collaboration deepens, AD imaging diagnostics are evolving from a singular structural assessment to a multidimensional integration of pathology, function, and metabolism, thus providing a scientific foundation for individualized interventions and disease-modifying therapies.
Parkinson's Disease
PD, the second most prevalent neurodegenerative disorder, relies on the combined development of epidemiological insights and imaging diagnostic technologies for early detection and precise intervention. Epidemiological data show a clear positive correlation between PD incidence and age: the prevalence is approximately 1%–2% in individuals aged 65 and older, and it rises dramatically to 1903 per 100,000 in those aged 80 and above. 46 This age-related burden drives the need for imaging modalities like SPECT, which is more accessible than PET in middle-income countries, facilitating early diagnosis in high-risk populations. 47 Gender differences are notable, with men having a 1.5 times higher incidence than women, potentially due to variations in hormonal levels or environmental exposures. 48 Key risk factors include environmental elements, such as long-term exposure to pesticides and heavy metals, as well as genetic mutations (e.g., LRRK2 and SNCA genes). Studies indicate that the Mediterranean diet may reduce the risk of PD onset. 49 The global disease burden continues to increase, with 11.77 million cases in 2021, and projections suggest a rise to 25.2 million by 2050. Middle-income countries, particularly in East and South Asia, are expected to experience the most significant increases in cases due to accelerated population aging.50,51 Limited access to advanced imaging like PET/MRI in these regions hampers early detection, emphasizing the role of cost-effective tools like transcranial ultrasound (TCS) in resource-constrained settings.
Advances in imaging diagnostic technologies have been pivotal in elucidating the pathological mechanisms of PD and facilitating early diagnosis. Structural MRI, with high-resolution imaging, can accurately quantify atrophy in the substantia nigra compacta of the midbrain, which suggests significant volume loss compared to healthy controls and correlates negatively with motor symptom severity. Furthermore, susceptibility-weighted imaging (SWI) is able to detect the disappearance of the “tail sign” due to iron deposition, with specificity exceeding 90%.52–54 Diffusion DTI reveals microstructural damage to the nigrostriatal pathway, manifested by reduced FA, indicating disruption of white matter integrity. These changes can serve as early indicators of neurodegeneration, often preceding clinical symptoms.55,56 Functional nuclear medicine techniques, such as SPECT and PET, are focused on evaluating dopaminergic system function: ¹²³I-FP-CIT SPECT detects the reduction of dopamine transporter (DAT) density in the striatum, serving as the “gold standard” for early PD diagnosis with a sensitivity of 78% and specificity of 97%.57,58 Meanwhile, ¹⁸F-DOPA PET quantifies dopamine metabolism, with a sensitivity of up to 95%, allowing differentiation between PD and other Parkinsonian syndromes, such as progressive supranuclear palsy.59,60 Moreover, the development of novel tracers targeting α-synuclein or neuroinflammatory markers, combined with AI-driven imaging analysis, is progressively overcoming molecular challenges in PD diagnostic pathology. 61 In future endeavors, the optimization of multimodal imaging strategies, such as the integration of PET-MRI with DTI, will be essential for enhancing the diagnostic precision of atypical Parkinsonian syndromes. Furthermore, the advancement and implementation of cost-effective technologies, including TCS, for screening purposes in resource-constrained regions, are imperative to effectively address the global public health challenges associated with aging populations.
Other Neurodegenerative Diseases
In addition to AD and PD, other neurodegenerative disorders also exhibit distinct epidemiological and imaging diagnostic characteristics. For instance, ALS is a fatal neurodegenerative disease primarily characterized by the degeneration of motor neurons, with an incidence rate of 1.5–2.4 per 100,000, and is more common in men. 62 MRI plays a pivotal role in the diagnosis of ALS, revealing atrophy in the motor cortex and spinal cord, with notable reductions in brain gray matter volume, especially in the motor cortex and brainstem regions. 63 Additionally, fMRI enables the observation of functional changes in the brain during motor tasks, providing valuable insights into the disease's neurophysiological mechanisms. 64 DTI imaging further highlights microstructural damage to the corticospinal tract, reflected in decreased FA, suggesting demyelination and axonal injury in white matter fibers. These imaging features are crucial for early diagnosis and personalized treatment. 65 PET imaging using 18F-FDG tracers detects reduced metabolism in the frontal and parietal lobes, supporting disease diagnosis. OCT and NIRS are primarily experimental, showing promise for non-invasive detection of retinal or cerebral oxygenation changes, but their clinical adoption awaits larger validation studies. 66
FTD is a relatively rare form of dementia, accounting for approximately 5%–10% of all dementia cases, typically manifesting between the ages of 45 and 65. 67 Its imaging characteristics are most notably seen in MRI, which reveals localized atrophy in the frontal and/or temporal lobes, with the most prominent changes observed in the anterior frontal and temporal regions. 68 The extent of this atrophy correlates with the severity and type of cognitive dysfunction. Additionally, 18F-FDG PET scans indicate decreased glucose metabolism in the affected brain regions, aiding in the differentiation of FTD from other dementias, such as AD. 69
Multiple sclerosis (MS) is a common inflammatory demyelinating disease of the central nervous system, with approximately 2.5 million affected individuals worldwide, most commonly diagnosed between the ages of 20 and 40. 70 MRI is the cornerstone of MS diagnosis, revealing multiple demyelinating plaques in the brain's white matter, particularly visible as high-signal lesions on T2-weighted imaging and fluid-attenuated inversion recovery sequences. Monitoring lesion number, size, location, and enhancement patterns with MRI provides critical information for diagnosing, assessing disease severity, and tracking progression. 71
Huntington's disease (HD) is a hereditary neurodegenerative disorder with a global prevalence of approximately 3.92 per 100,000, particularly high in Caucasian populations. 72 MRI scans may indicate atrophy in the caudate nucleus and striatum, while DTI imaging reveals white matter damage in the basal ganglia. PET and SPECT scans detect metabolic reductions in these regions. 73
Spinocerebellar ataxia (SCA) is another neurodegenerative disorder, and the global prevalence of SCAs ranges from 0 to 5.6 cases per 100,000 individuals. 74 SCA presents with cerebellar and brainstem atrophy on MRI, and DTI detects damage to cerebellar white matter tracts, with SCA3 being the most prevalent subtype. 75
As imaging technologies continue to evolve, the complementary advantages of structural and functional imaging become increasingly apparent. MRI and CT scans can accurately localize atrophy in specific brain regions, such as the basal ganglia in HD and the cerebellum in SCA. DTI quantifies white matter fiber damage, while PET and SPECT imaging reveal metabolic and molecular pathological alterations, such as the reduced frontal lobe metabolism in ALS and the widespread metabolic inhibition in prion diseases. Future research will focus on integrating imaging genomics and genetic/biomarker data to further enhance early differentiation and prognostic evaluation.
Clinical Application of Imaging Diagnostic Technologies in Neurodegenerative Diseases
The Role of Imaging Diagnosis in Early Recognition
As the prevalence of neurodegenerative diseases continues to rise, the importance of early recognition has become increasingly evident. Advances in imaging technologies have opened new opportunities for early diagnosis, improving the accuracy of disease identification and laying the foundation for subsequent treatment strategies. Imaging diagnosis, particularly in AD and PD, allows for the detection of potential biomarkers before clinical symptoms appear, providing critical information for clinical intervention.
Identification of Early Biomarkers
The identification of early biomarkers is crucial for the early diagnosis of neurodegenerative diseases. Advances in imaging technologies, such as MRI and PET, have enabled researchers to identify disease-related biomarkers that are detectable before the onset of clinical symptoms. For example, in AD, brain MRI can reveal structural changes associated with cognitive decline, such as hippocampal atrophy and cortical thinning, which can be observed even in the absence of clinical symptoms.76,77 Furthermore, AI techniques applied to imaging data have significantly improved the detection rate and accuracy of these biomarkers. Deep learning algorithms, when applied to MRI scans, have been shown to effectively classify different stages of AD.78,79
In PD, the aggregation of α-synuclein protein is a key early biomarker. Imaging techniques such as SPECT and PET are essential for identifying these aggregates, enabling early recognition of PD.80,81 Additionally, IONPs used as MRI contrast agents may show promise in AD by enhancing image quality and serving as drug carriers, facilitating the delivery of drugs across the blood–brain barrier and potentially offering new avenues for early treatment. 82
The early pathological changes in neurodegenerative diseases primarily involve abnormal protein deposits and changes in brain structure and metabolism. In AD, Aβ plaques and tau protein neurofibrillary tangles begin to accumulate decades before clinical symptoms appear, along with hippocampal and entorhinal cortex atrophy. 83 In PD, the loss of dopaminergic neurons in the substantia nigra and the deposition of Lewy bodies formed by α-synuclein are early signs, often visible as a decline in DAT function in the striatum. 84 Imaging technologies are crucial in capturing these biomarkers or structural changes, allowing for early diagnosis. For example, PET can specifically label Aβ or tau proteins, while MRI can detect gray matter volume reduction and brain network connectivity abnormalities.85,86
Correlation Between Imaging and Clinical Symptoms
The correlation between imaging findings and clinical symptoms is critical for the early diagnosis of neurodegenerative diseases. By integrating imaging data with clinical assessments, physicians can gain a more comprehensive understanding of the patient's condition. For example, in MS, regular MRI monitoring can reveal the relationship between clinical symptoms and visible lesions, helping predict disease progression. 87
In AD, imaging features such as brain atrophy are strongly correlated with cognitive decline. MRI and PET can detect biomarkers related to cognitive impairment, such as the deposition of Aβ and tau proteins, which are characteristic pathological changes of AD.88,89 With imaging, clinicians can identify potential disease progression before symptoms become apparent, enabling early intervention.
In PD, imaging features such as substantia nigra atrophy, iron deposition, and DAT function changes in the striatum are closely related to the onset of clinical symptoms. SPECT and PET scans can detect a decline in DAT function, aiding early diagnosis before motor symptoms appear. Iron deposition in the substantia nigra and neuromelanin imaging (NM-MRI) may provide further pathological insights to assist in evaluating disease progression. 81
Future Directions
The role of imaging diagnosis in early recognition of neurodegenerative diseases cannot be overstated. With the advancement of multimodal imaging technologies, combining structural imaging (such as MRI), functional imaging (such as fMRI and PET), and molecular imaging, it is now possible to more accurately identify early biomarkers, thus enhancing diagnostic accuracy. The integration of AI and imaging genomics, which extracts micro-features from vast imaging data sets to predict individual disease risks, will further facilitate personalized treatment strategies. Additionally, the development of new molecular tracers and contrast agents will continue to unlock the potential of imaging technologies in disease prevention, personalized treatment, and drug development. Multimodal imaging, such as the combined use of PET and MRI, enables the simultaneous assessment of pathological protein deposition and brain structural changes, improving diagnostic accuracy. The fusion of imaging genomics and AI will drive further advancements in the precision of early diagnosis. Looking ahead, with innovations in molecular tracers, the development of imaging genomics, and the growth of non-invasive techniques, imaging technologies are poised to play a larger role in disease prevention, personalized treatment, and new drug discovery.
Imaging Diagnostic Technologies in Disease Monitoring
Imaging diagnostic technologies play an indispensable role in monitoring the progression of neurodegenerative diseases. With the continuous advancement of imaging technologies, particularly MRI and PET, physicians are now better equipped to assess disease progression and treatment efficacy more accurately. These technologies not only assist in early diagnosis but also provide critical information regarding the progression of the disease, thus supporting the development of personalized treatment plans. The application of imaging technologies has significantly enhanced the precision and efficiency of monitoring neurodegenerative diseases, enabling timely adjustments to treatment strategies.
Treatment Efficacy Evaluation
Imaging plays a critical role in the treatment of neurodegenerative diseases, particularly in evaluating therapeutic efficacy. For example, in AD, PET imaging monitors brain glucose metabolism changes, offering initial evidence of treatment effects. Preliminary evidence suggests that as treatment progresses, PET imaging reveals improvements in brain metabolism, especially in cortical and hippocampal areas, suggesting a potential recovery in cognitive function.90,91 PET imaging has proven to be a sensitive biomarker for early treatment responses, allowing physicians to adjust therapy based on these early indications. 92
MRI also contributes significantly to evaluating treatment efficacy. Brain structural changes, such as hippocampal shrinkage and cortical thinning, are key features of AD. As treatment progresses, MRI allows clinicians to observe whether these changes are stabilized or reversed. The reduction in hippocampal volume has a strong correlation with cognitive decline, making MRI an essential tool for both early diagnosis and ongoing monitoring of disease progression.93,94 By combining MRI and PET imaging, clinicians can assess the disease's treatment response from multiple perspectives, helping them decide if treatment adjustments are necessary.
In the management of MS, MRI is widely used for treatment evaluation. MRI not only detects the size and location of lesions but also monitors the surrounding inflammatory activity. 95 Regular MRI monitoring enables doctors to evaluate whether the inflammation is controlled and adjust treatment plans accordingly. Research has shown that persistent inflammation detected on MRI is closely correlated with clinical symptoms, offering valuable insights into treatment efficacy.96,97 Thus, imaging technologies not only support the diagnosis of diseases but also play an essential role in evaluating treatment efficacy.
Disease Progression Monitoring
Imaging technologies are equally crucial for monitoring the progression of neurodegenerative diseases, especially in their early stages. Through regular imaging examinations, physicians can detect subtle changes in the disease, allowing for early intervention to slow its progression. Early imaging changes often act as warning signs for the disease, providing valuable guidance for clinical decisions.
In AD, MRI is able to monitor cortical atrophy, especially in the hippocampus, which is often detectable before clinical symptoms appear. Hippocampal atrophy is one of the key early indicators of AD, and its progression strongly correlates with cognitive decline. Regular monitoring of these structural changes helps physicians identify potential disease progression early and take timely interventions. 98 fMRI and DTI are also increasingly applied in AD monitoring. fMRI evaluates brain network connectivity, particularly the activity of DMN, which often indicates decreased connectivity before significant structural changes, making it a useful early marker. 99 DTI, which quantifies the integrity of white matter fibers, can detect microstructural changes related to cognitive impairment well before clinical symptoms become evident.100,101
For PD, imaging technologies are invaluable for disease monitoring. Research has shown that MRI and PET imaging can detect microstructural changes associated with the disease, such as alterations in the substantia nigra, which indicate a basis for early diagnosis and monitoring.102,103 Techniques like DAT imaging through SPECT and PET are crucial for early monitoring of PD. These technologies enable doctors to track disease progression even before clinical symptoms are evident, helping optimize treatment strategies. MRI techniques like detecting iron deposition in the substantia nigra (e.g., SWI sequence) are also widely used for early assessment of PD, assisting physicians in monitoring disease progression from the early stages.104,105
In conclusion, Imaging diagnostic technologies, including MRI, PET, and AI, are crucial for the early recognition and monitoring of neurodegenerative diseases like Alzheimer's (AD) and Parkinson's (PD). These technologies help identify early biomarkers, such as hippocampal atrophy in AD and α-synuclein aggregation in PD, even before clinical symptoms appear. They also play a vital role in assessing disease progression and treatment efficacy, allowing for timely interventions and personalized treatment strategies. Advancements in multimodal imaging will further enhance diagnostic accuracy and improve patient care in the management of these diseases.
Emerging Imaging Technologies in Neurodegenerative Disease Applications
Emerging imaging technologies play a pivotal role in the diagnosis and research of neurodegenerative diseases. These technologies not only improve the accuracy of early diagnosis but also facilitate real-time monitoring of disease progression. Over recent years, advancements in fMRI and NIRS have opened new directions for neurodegenerative disease research.
Functional Magnetic Resonance Imaging
fMRI is a technique capable of real-time monitoring of brain activity based on changes in blood flow that reflect neuronal activity. In recent years, fMRI has become a key tool in the study of neurodegenerative diseases, such as AD and PD. Its high resolution and non-invasive nature make it an ideal tool for exploring brain functional and structural changes. Research suggests that fMRI can reveal altered brain activation patterns in AD patients during cognitive tasks, helping identify early pathological changes. 106 For instance, some studies using fMRI have found significant differences in the activation patterns of the prefrontal cortex and hippocampus in AD patients compared to healthy controls, providing important biomarkers for early diagnosis. 107 Furthermore, fMRI is increasingly being used to assess treatment effects. By comparing pre- and post-treatment fMRI data, researchers can evaluate the impact of interventions on brain function, providing valuable insights for ongoing treatment adjustments. However, the use of fMRI also faces challenges, including individual variability, motion artifacts, and the complexity of data analysis. Future research, particularly with the integration of advanced techniques such as deep learning, could enhance the clinical application of fMRI, making it a more robust tool in neurodegenerative disease research. 108
Near-Infrared Spectroscopy
NIRS is an emerging non-invasive brain function measurement technology that monitors brain blood flow and oxygenation by detecting the scattering and absorption of near-infrared light in biological tissues. NIRS offers advantages over traditional neuroimaging techniques like MRI or PET, including ease of operation, portability, and suitability for monitoring elderly or critically ill patients with neurodegenerative diseases. 109 In AD research, NIRS shows promise as a non-invasive tool for assessing cortical brain function, with studies indicating it can detect reduced brain oxygenation in AD patients during cognitive tasks compared to healthy controls, suggesting potential for early screening and monitoring disease progression. 110 However, NIRS remains an experimental modality, limited by low spatial resolution and inability to probe deeper brain structures, requiring integration with techniques like electroencephalography and further validation for clinical adoption. Another key application of NIRS is in evaluating treatment effects. By monitoring changes in brain blood flow before and after different treatments such as medication or cognitive training, researchers can assess the effectiveness of interventions. 111 Additionally, the portability of NIRS makes it flexible for use in clinical settings, enabling real-time monitoring in various environments. Despite its potential, NIRS has limitations, including relatively lower resolution and limited capability for monitoring deeper brain structures. Future research will need to explore the combination of NIRS with other imaging techniques to improve its application in the diagnosis and monitoring of neurodegenerative diseases.
High-Resolution Imaging and Advancements
High-resolution imaging technologies have made significant strides in the diagnosis of neurodegenerative diseases. Among these, ultra-high-field MRI (such as 7 T MRI) is increasingly being used, offering a higher signal-to-noise ratio and providing clearer brain images. This allows for the detection of early subtle morphological changes in neurodegenerative diseases. For example, in AD research, 7T MRI may more clearly reveal the substructure of the hippocampus and is more sensitive in detecting hippocampal atrophy, which aids in early diagnosis. 112 Optical imaging techniques, such as OCT and OCT-Angiography (OCT-A), have also evolved. OCT can perform high-resolution imaging of the retina, which is functionally and anatomically similar to the brain. 113 By measuring the retinal nerve fiber layer thickness and retinal vascular density, OCT can indirectly reflect the impact of neurodegenerative diseases on the visual pathway. Studies have indicated that the retinal nerve fiber layer thickness is reduced in both AD and PD patients, and this reduction correlates with disease severity. 114 OCT-A provides detailed information about retinal blood vessels, which helps identify retinal vascular changes caused by neurodegenerative diseases, offering potential biomarkers for early diagnosis. Additionally, super-resolution imaging technologies, such as stimulated emission depletion microscopy and reversible saturable optical fluorescence transition microscopy, 115 while primarily applied in basic research, have proven valuable tools for understanding the pathological mechanisms of neurodegenerative diseases. These imaging technologies surpass the diffraction limit of conventional microscopes, enabling nanometer-scale visualization of protein aggregation and synaptic changes, and offering novel insights into early disease mechanisms. However, their clinical translation remains limited. Techniques such as OCT and super-resolution microscopy show promise, but their utility is constrained by the lack of standardized protocols, large-scale validation, and ongoing uncertainty regarding the correlation between retinal and brain pathology. At present, they serve primarily as complementary research tools rather than standalone diagnostics.
Multimodal Imaging Integration
The integration of multimodal imaging technologies has led to significant breakthroughs in the diagnosis of neurodegenerative diseases. Combining MRI with PET, the PET/MRI system integrates MRI's high soft tissue resolution with PET's molecular imaging advantages. In AD diagnosis, PET/MRI allows for simultaneous acquisition of structural, functional, and molecular information, such as detecting brain atrophy and white matter fiber changes via MRI and detecting Aβ and tau protein deposits via PET, which improves diagnostic accuracy and specificity. 116 Research indicates that PET/MRI is superior to either MRI or PET alone in detecting AD pathology, offering more comprehensive evaluations for early diagnosis and disease monitoring.117,118 Furthermore, combining optical imaging with other imaging technologies also shows great promise. For example, integrating near-infrared fluorescence imaging (NIRF) with MRI or CT may allow NIRF to provide highly sensitive molecular-specific imaging, while MRI or CT offers precise anatomical localization. In neurodegenerative disease research, this multimodal imaging can be used to track specific neuroinflammatory biomarkers or drug delivery vectors, thereby offering insights that may improve our understanding of disease pathophysiology and treatment responses.119,120 While the integration of PET/MRI offers a powerful, synergistic view of pathophysiology, its clinical adoption is hampered by significant challenges. The high cost, complex workflow, and challenges in attenuation correction for PET data are major barriers. Therefore, while PET/MRI is a superior research tool, its widespread clinical use for neurodegenerative diseases remains limited compared to sequential scanning with standalone PET/CT and MRI systems.
Simultaneously, multimodal ultrasound imaging is also evolving. The combination of ultrasound imaging and photoacoustic imaging (PAI), such as PA/US imaging, can suggest both structural and functional information of tissues. In neurodegenerative diseases, PA/US imaging can be used to detect morphological and functional changes in brain blood vessels and alterations in the optical absorption characteristics of neural tissues, providing a novel tool for disease diagnosis and monitoring. 121
AI in Imaging Analysis
AI has increasingly played a significant role in the analysis of imaging data, bringing new opportunities to the imaging diagnosis of neurodegenerative diseases. AI-based deep learning algorithms can automatically classify MRI, PET, and other images of neurodegenerative diseases, distinguishing normal individuals from those with diseases and differentiating between various types of neurodegenerative disorders. For instance, convolutional neural networks (CNNs) have been used to analyze MRI images of AD patients, identifying disease-related features with high diagnostic accuracy. 122 Studies found that CNN models could diagnose AD with an accuracy of over 90% based on MRI data of AD patients and healthy controls, thus improving diagnostic efficiency and accuracy.123,124
In disease prediction, AI can analyze imaging data to predict the progression of neurodegenerative diseases. By analyzing longitudinal imaging data, AI algorithms can identify early imaging biomarkers of disease progression and predict the development trend. For instance, machine learning algorithms analyzing DAT-SPECT images in PD patients can predict disease progression speed, offering a basis for formulating personalized treatment plans.125,126 AI can also be used for image segmentation and quantitative analysis, automatically segmenting specific brain regions, such as the hippocampus and substantia nigra, and quantifying their volume and signal intensity. This helps to more accurately assess brain structural changes caused by neurodegenerative diseases, with higher reproducibility and objectivity, reducing human errors.
Despite its immense potential, the application of AI in clinical practice faces significant hurdles. A primary limitation is the “black box” nature of many deep learning models, where the reasoning behind a decision is not easily interpretable, reducing clinical trust. Furthermore, AI models are highly sensitive to variations in data acquisition protocols across different scanners and institutions, which can degrade their performance (the “domain shift” problem). The need for massive, well-annotated data sets for training robust models also presents a major logistical and privacy challenge. Overcoming these issues through the development of explainable AI and data harmonization techniques is a key prospect for future research.
Imaging Diagnostics in Personalized Treatment of Neurodegenerative Diseases
Imaging techniques have become integral to personalized treatment approaches in neurodegenerative diseases, playing a critical role in both early diagnosis and ongoing disease monitoring. These technologies offer a new avenue for obtaining high-resolution, non-invasive insights into brain structure and function, which can assist clinicians in more accurately assessing disease severity and progression. As imaging technologies continue to evolve, they contribute to the development of individualized treatment plans by offering dynamic, real-time data.
Imaging as the Foundation of Precision Medicine
The core concept of precision medicine is to tailor treatment plans based on individual patient characteristics and disease features. Imaging plays a crucial role in this process by providing detailed and accurate assessments of the disease state. High-resolution techniques, such as MRI and PET, enable clinicians to observe structural and functional changes in the brain, helping to identify the most appropriate intervention strategies. For example, in AD, MRI and PET imaging can detect both structural changes, such as brain atrophy, and molecular alterations, such as amyloid and tau protein accumulation. 116 By continuously monitoring brain metabolism and structural changes, these imaging modalities allow for the early detection of disease progression, thus enabling timely therapeutic interventions. Recent studies suggest that combining imaging data with clinical information helps create more accurate predictive models, which can evaluate treatment effectiveness and allow for personalized treatment adjustments based on real-time data. 127 In the case of neurodegenerative diseases, the ongoing evolution of imaging technologies, particularly in functional and molecular imaging, will play an even greater role in shaping personalized treatment approaches. As these technologies improve, they will offer clinicians the tools necessary to monitor disease progression more effectively and adjust treatments to meet individual patient needs.
Imaging in Evaluating Treatment Response
Imaging technologies are also pivotal in evaluating the effectiveness of treatment interventions. By regularly performing imaging exams, clinicians can directly observe changes in the brain and assess whether the current treatment is achieving the desired therapeutic effects. For example, in PD, MRI and PET imaging can be used to monitor changes in brain metabolism and neuronal transmission, providing valuable feedback about treatment efficacy. 128 In AD, imaging plays an equally critical role in evaluating the effectiveness of anti-amyloid and anti-tau therapies. By monitoring changes in amyloid plaques and tau protein accumulation, physicians can determine whether a particular therapeutic approach is successful. This real-time feedback mechanism allows for flexible treatment adjustments based on individual responses, optimizing the therapeutic impact.129,130 Additionally, the rise of AI in imaging data analysis has further enhanced the accuracy and efficiency of these evaluations, enabling clinicians to monitor treatment progress dynamically and adjust treatment plans in real-time.79,131 Ultimately, the application of imaging in assessing treatment response facilitates a more personalized and effective approach to managing neurodegenerative diseases, improving both the quality of care and patient outcomes.
Challenges and Limitations of Imaging in Personalized Treatment
While imaging has significant potential in personalized treatment, several challenges limit its widespread application. One of the main issues is the cost and accessibility of advanced imaging techniques. High-end technologies like PET and SPECT, although valuable in early diagnosis and disease evaluation, come with a high cost that makes them inaccessible to many patients, particularly in resource-limited settings.132,133 The high operational and equipment maintenance costs of these technologies further limit their availability. Moreover, there is a lack of standardization in imaging procedures and interpretation across different healthcare institutions. Variations in equipment, scanning parameters, and expertise among clinicians can lead to inconsistencies in imaging results. For example, studies have indicated that variations in MRI scan parameters can lead to differences in hippocampal volume measurements, affecting the accuracy of early diagnosis of AD. 134 Additionally, subjective interpretation of images remains a challenge, even with advanced imaging techniques, as the interpretation heavily relies on the experience of clinicians. This subjectivity can lead to variability in diagnosis and impact clinical decision-making.135,136 To address these challenges, researchers are exploring the integration of AI algorithms to standardize image analysis and reduce human error. AI has shown great promise in automating image classification and segmentation, improving the accuracy and reproducibility of imaging diagnostics. However, the widespread adoption of AI-based imaging tools requires further research and validation.
Collectively, imaging diagnostics play a crucial role in personalized treatment approaches for neurodegenerative diseases. Through high-resolution, non-invasive technologies like MRI, PET, and advanced functional imaging techniques, clinicians can assess disease progression, evaluate treatment effectiveness, and adjust therapeutic strategies to meet individual patient needs. However, challenges such as cost, accessibility, and subjectivity in image interpretation must be addressed to fully realize the potential of imaging in personalized medicine. Advancements in imaging technologies and their integration with AI could increasingly influence the future of neurodegenerative disease management. These tools might offer a means to provide more timely, accurate, and personalized care.
Conclusion
The application of imaging diagnostic technologies in the early detection, monitoring, and treatment evaluation of neurodegenerative diseases is critical. With ongoing technological advancements, imaging plays an increasingly pivotal role in diagnosing diseases, tracking their progression, and assessing treatment efficacy. As outlined in Table 2, different imaging techniques such as MRI, PET, and fMRI each offer unique advantages and limitations in the diagnosis and monitoring of neurodegenerative diseases.
Comparison of Imaging Techniques for Neurological Disease Diagnosis.
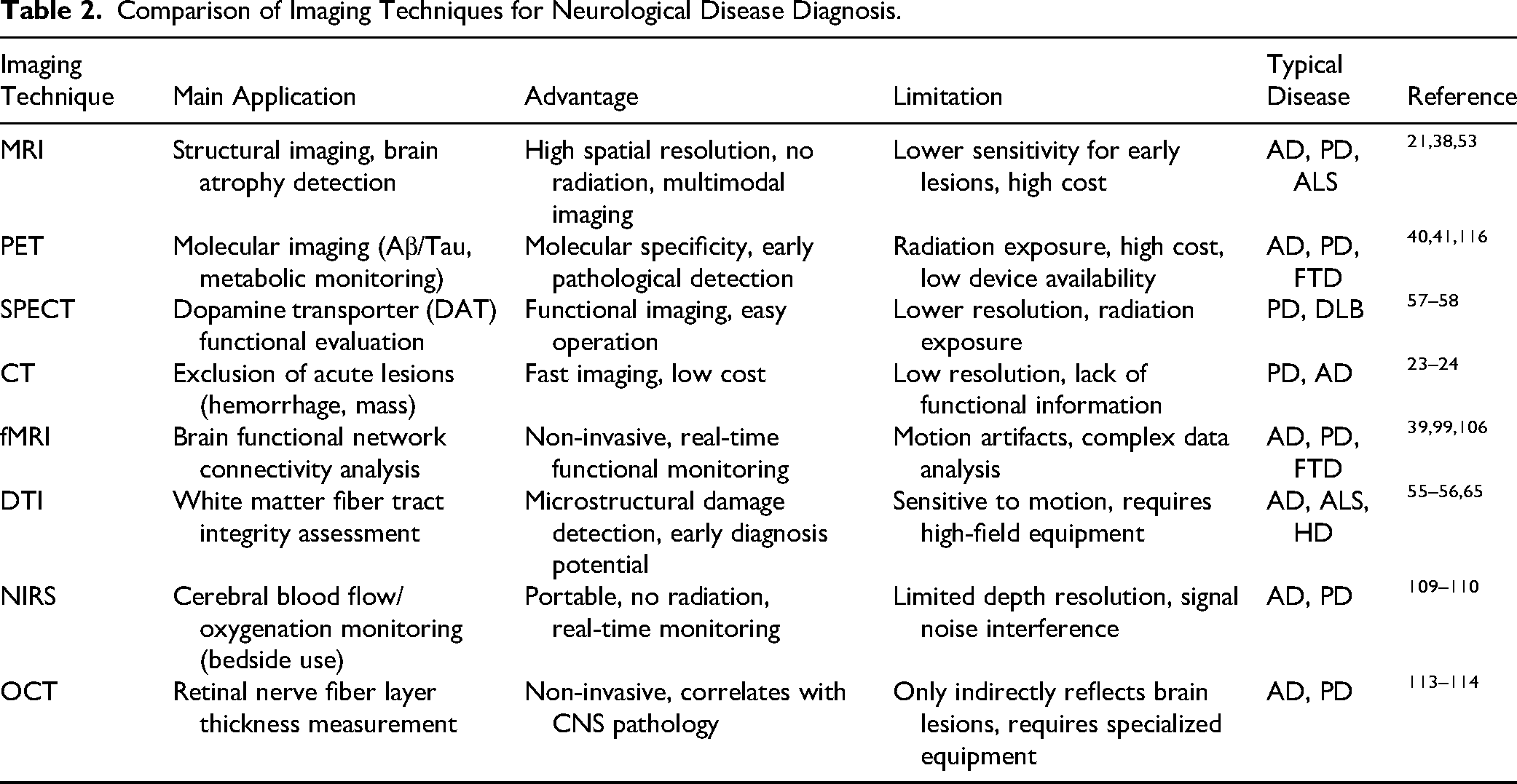
Despite significant progress, several challenges and limitations persist that must be addressed in future research. First, improving the sensitivity and specificity of imaging techniques remains a key focus. Many imaging methods still struggle to detect early-stage neurodegenerative diseases accurately, leading to potential misdiagnosis or missed diagnoses. Future research should prioritize optimizing existing technologies and developing more targeted imaging methods to enhance clinical utility. Additionally, integrating multimodal imaging techniques to gather comprehensive disease data will be an important area of exploration.
Second, personalized treatment strategies are becoming increasingly essential in managing neurodegenerative diseases. Patients exhibit considerable variability in disease progression and presentation, making individualized diagnostic approaches necessary. By combining imaging data with other biomarkers such as genomics and proteomics, a deeper understanding of disease mechanisms can be achieved, leading to more tailored treatment plans. This interdisciplinary approach may not only improve diagnostic accuracy but also offer a scientific foundation for developing personalized therapies.
Ethical and economic considerations also influence the application of imaging diagnostics. Balancing the cost of advanced imaging technologies with their clinical benefits and ensuring patient privacy during data collection and use are critical issues that need to be addressed. Future research should focus on overcoming these challenges while ensuring the accessibility and sustainability of new technologies.
In conclusion, imaging diagnostics hold significant promise for improving the management of neurodegenerative diseases. Although current technologies have limitations, continuous research and innovation offer the potential to overcome these challenges. Integrating emerging imaging technologies with personalized treatment approaches will enhance patient outcomes and quality of life, paving the way for more effective solutions in the management of neurodegenerative diseases.
Footnotes
Abbreviations
Consent for Publication
Not applicable.
Author contribution(s)
Jinshan Xu: Preparation of original draft; Conceptualization; supervision; review. Caiyun Gao, Junhua Zhang, Jialei Lu, Yingyu Xuan, Shiyun Wang, and Chaozhi Bu: Reference collection. All authors contributed to the literature search and information collection, discussion, and revision.
Funding
This work was supported by the Lingying Team project of Obstetrics and Gynecology Hospital of Jiangnan University (Grant number LY2023003).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
