Abstract
Objective
2-Deoxy-2-[18F]fluoro-D-glucose ([18F]FDG) is widely used for noninvasive imaging of atherosclerosis. However, knowledge about metabolic processes underlying [18F]FDG uptake is mostly derived from in vitro cell culture studies, which cannot recapitulate the complexities of the plaque microenvironment. Here, we sought to address this gap by in situ mapping of the activity of selected major dehydrogenases involved in glucose metabolism in atherosclerotic plaques.
Methods
In situ activity of lactate dehydrogenase (LDH), glucose-6-phosphate dehydrogenase (G6PD), succinate dehydrogenase (SDH), and isocitrate dehydrogenase (IDH) was assessed in plaques from murine aortic root and brachiocephalic arteries and human carotid arteries. High-resolution 2-deoxy-D-[1,2-3H]glucose ([3H]2-deoxyglucose) autoradiography of murine brachiocephalic plaques was performed.
Results
LDH activity was heterogeneous throughout the plaques with the highest activity in medial smooth muscle cells (SMCs). G6PD activity was mostly confined to the medial layer and to a lesser extent to SMCs along the fibrous cap. SDH and IDH activities were minimal in plaques. Plaque regions with increased [3H]2-deoxyglucose uptake were associated with a modestly higher LDH, but not G6PD, activity.
Conclusions
Our study reveals a novel aspect of the metabolic heterogeneity of the atherosclerotic plaques, enhancing our understanding of the complex immunometabolic biology that underlies [18F]FDG uptake in atherosclerosis.
Introduction
Metabolic imaging of atherosclerosis by positron emission tomography (PET) has been extensively investigated, aiming to enhance plaque characterization and predict the risk of vulnerability beyond what can be achieved by conventional imaging methods which primarily focus on detecting the severity of luminal stenosis. 1 In addition to supporting the bioenergetics and biomass demands, metabolism regulates diverse functions of cells residing within the atherosclerotic plaques, such as the leukocytes’ response to inflammatory stimuli.1,2 However, the metabolic states of the cells involved in atherogenesis have been mostly explored using in vitro culture conditions, which cannot recapitulate the complexities of the plaque microenvironment.1,2 While 2-deoxy-2-[18F]fluoro-D-glucose ([18F]FDG) PET allows for in vivo detection of metabolism, its low spatial resolution precludes accurate mapping of the heterogeneities across different plaque components. 1 Moreover, the biological correlates of [18F]FDG uptake in atherosclerosis have remained incompletely understood. 1 For example, while enhanced glycolytic activity of proinflammatory macrophages has been considered as the primary driver of [18F]FDG uptake in atherosclerotic plaques, 1 recent studies have suggested that other cell types, particularly smooth muscle cells (SMC),3,4 or other activation states of macrophages5,6 may significantly contribute to [18F]FDG uptake. Additionally, the association between [18F]FDG uptake and intracellular utilization of glucose through different metabolic pathways has not been studied within the plaque microenvironment.
Most of the literature on the contribution of different cell types to [18F]FDG uptake in atherosclerosis is based on in vitro metabolic studies of SMCs and macrophages cultured in the presence of various chemokines, growth factors, or environmental stimuli (eg, hypoxia, oxidized low-density lipoproteins, and lipopolysaccharide).1,5–10 However, these conditions do not accurately replicate the complex microenvironment of the vessel wall. 1 While alternative approaches such as transcriptomics, proteomics, and metabolomics analysis of tissue lysates have been utilized, these methods do not assess the heterogeneity of the cellular metabolism within tissues. 11 Additionally, the regulation of metabolic enzyme activity is markedly influenced by posttranscriptional and posttranslational mechanisms, which cannot be adequately assessed by transcriptional and proteomics approaches. 12 To address the knowledge gaps in metabolic assessment of cells within the native microenvironment of plaques, we employed in situ enzyme histochemistry technique to provide a high-resolution map of the activity of selected key dehydrogenases involved in glucose metabolism in murine and human atherosclerotic plaques. By applying the metabolic substrates of specific enzymes of interest along with a chromogenic substrate, this technique allows for visualization and quantitative assessment of the enzymatic activities of different cell types within their native tissue microenvironment. 12 This technique has been previously used to reveal the metabolic heterogeneity of neoplastic tissues,11,12 but to our knowledge the current study represents its first report in atherosclerosis. Our results demonstrated a significant heterogeneity in the activity of the selected dehydrogenases in three major glucose utilization pathways, including: (1) lactate dehydrogenase (LDH, a critical enzyme for the continuation of glycolytic activity through regeneration of nicotinamide adenine dinucleotide [NAD+] 13 ); (2) glucose-6-phosphate dehydrogenase (G6PD, a rate-limiting enzyme in pentose phosphate pathway [PPP] 14 ); and (3) succinate dehydrogenase (SDH) and isocitrate dehydrogenase (IDH) (critical enzymes in tricarboxylic acid (TCA) cycle). We also examined the relationship between LDH and G6PD activities with the uptake of 2-deoxy-D-[1,2-3H]glucose ([3H]2-deoxyglucose), as a surrogate for [18F]FDG, in murine brachiocephalic artery plaques.
Methods
Mouse Model of Atherosclerosis
Apolipoprotein E knockout (apoE−/−) mice (N = 9, 3 males and 6 females) on C57BL/6J background (Jackson Laboratories) were fed by an atherogenic western diet (21% anhydrous fat, TD88137, Envigo). Mice were euthanized at ∼3 months (early plaques) or ∼7 months (advanced plaques) after western diet feeding. Aortic roots and brachiocephalic arteries were excised for histological and autoradiographic experiments. Experiments were conducted in accordance with a protocol approved by the University of Pittsburgh Institutional Animal Care and Use Committee.
Immunofluorescent Staining of Murine Atherosclerotic Plaques
Unfixed 10-µm thick plaque cryosections were stained for CD68 and α-smooth muscle actin (α-SMA), as surrogates for monocytes/macrophages and SMC, respectively, using commercially available antibodies (Supplemental Table 1). Tissues were temporarily mounted using ProLong™ Gold Antifade containing 4′,6-diamidino-2-phenylindole (DAPI, Invitrogen). Fluorescent images were acquired using an Axiovert 200 inverted microscope (Zeiss) equipped with a digital camera, controlled by ZEN software (Zeiss).
In Situ Enzyme Histochemistry
After the acquisition of immunofluorescent images, coverslips were removed by submerging the slides in PBS for subsequent mapping of the activity of 4 selected enzymes catalyzing key steps in glucose metabolism, ie, LDH, G6PD, SDH, and IDH, according to previously described methods11,12 with minor modifications. Tissues were incubated with enzyme-specific buffers (Supplemental Table 2) at 37 °C to allow for in situ formation and precipitation of blue-colored formazan at sites of the enzymatic activity.11,12 After washing, tissues were dehydrated and cleared in xylene and permanently mounted using (VectaMount mounting medium, Vector labs). Brightfield images were acquired using an Axiovert 200 microscope.
Quantification Technique
For quantification of enzymatic activity in different cell types, the corresponding immunostaining (fluorescent) and enzyme histochemistry (brightfield) images were overlaid and image layers were converted into a grayscale format. Two mask layers were manually drawn to define the total vessel wall and the medial layer (Supplemental Figure 1). A grid was then applied to the overlaid fluorescent and brightfield images to record the intensity of CD68, α-SMA, and the enzyme activity in each 13 × 13 µm square of the grid (Azure software). These multiparametric data were then imported to FlowJo software to analyze enzymatic activities in CD68-positive and α-SMA-positive regions of the medial and intimal layers of the vessel wall. Enzymatic activities were reported after correcting for background (ie, signal from negative control slides incubated with the buffers lacking the metabolic substrates) and normalization to standard curves (prepared by adding known amounts of enzymes to assay buffers) to account for batch-to-batch variations in enzyme assay buffers. Quantifications of enzymatic activities were performed on 2-3 tissue sections per mouse as technical replicates, and the average values were used as biological replicates.
Immunohistochemistry and Enzyme Histochemistry of Human Carotid Endarterectomy Specimens
Anonymized atherosclerotic plaques were collected from 3 patients who underwent standard-of-care carotid endarterectomy under a “No Human Subject involvement” classification by the University of Pittsburgh Institutional Review Board. Considering the No Human Subject designation, informed consent was not required for this study. Immunohistochemical staining was performed by incubation of 10-µm thick cryosections with anti-CD68 or anti-α-SMA antibodies followed by horseradish peroxidase-conjugated secondary antibodies (Supplemental Table 1) for chromogenic detection (to avoid the high autofluorescence of human plaques). Tissues adjacent to CD68 and α-SMA stained slides were used for in situ enzyme histochemistry, as described above.
High-Resolution [3H]2-Deoxyglucose Autoradiography of Murine Atherosclerotic Plaques
Excised brachiocephalic arteries were incubated in a glucose-free uptake buffer (NaCl: 140 mM, KCl: 5.4 mM, CaCl2: 1.8 mM, MgSO4: 0.8 mM, HEPES: 25 mM), 8 supplemented by 40 µCi/mL (1.48 MBq/mL) of [3H]2-deoxyglucose (American Radiolabeled Chemicals Inc., ART-0103A) at 37 °C for 2 h. Subsequently, arteries were washed with a cold buffer and embedded in optimal cutting temperature (OCT) compound and cryo-sectioned (Leica CM1860) at 10-μm thickness. Tissues were exposed to tritium-sensitive BAS storage phosphor screens (BAS-IP TR 2025e, GE Healthcare) for 3-4 weeks at −20 °C. Phosphor screens were scanned by a Sapphire Biomolecular Imager (Azure Biosystems) at 20-µm resolution. Tissues were then used for immunostaining and enzyme histochemistry, as described above. The average intensity of enzyme activities was calculated in different quartiles of [3H]2-deoxyglucose uptake. Representative autoradiography images are shown after adjustment by the Fire Look-Up Table and smoothing algorithm (Image J).
Statistical Analysis
Statistical analysis was performed using GraphPad Prism software (version 8). Data are presented as mean ± SEM. Mean values between experimental groups were compared using a one-way analysis of variance (ANOVA) followed by Fisher's Exact post hoc test. P < .05 was considered statistically significant.
Results
Metabolic Heterogeneity of Murine Atherosclerotic Plaques
The visual assessment of the murine aortic roots demonstrated heterogenous levels of LDH activity within the intimal and medial layers (Figure 1A). On the other hand, G6PD activity was mostly confined to the media and to a lesser extent along the fibrous caps of plaques (Figure 1B). Notably, the activity of the two TCA cycle dehydrogenases, ie, SDH (Figure 1C) and IDH (Supplemental Figure 2), were minimal within the intimal and medial layers as opposed to the intense activity of the adjacent myocardium.

In situ metabolic mapping of atherosclerotic plaques. Representative low-magnification (top) and high-magnification (bottom) in situ enzyme histochemistry images from aortic root plaques of apoE−/− mice ∼7 months after western diet feeding.
Quantification of LDH Activity in Murine Aortic Root Plaques
We next compared the LDH activity in macrophages and SMCs, as two highly abundant cell types with key functions in atherogenesis. The LDH was selected as a surrogate to assess the glycolytic flux within the vessel wall, considering its critical role in sustaining the glycolysis through the reoxidation of NADH to NAD+ during the conversion of pyruvate to lactate.15,16 To this end, we performed sequential immunofluorescent staining for CD68 and α-SMA followed by in situ enzyme histochemistry of the same tissue sections to allow for their co-registration (Figure 2A). Quantitative analysis demonstrated that LDH activity was ∼50% higher in medial α-SMA+ regions compared to intimal α-SMA+ and CD68+ regions (Figure 2B-E) in both early and advanced plaques (∼3 vs. 7 months of western diet). Moreover, LDH activity of all 3 populations was significantly lower in advanced compared to early plaques.

Quantification of LDH activity of plaque macrophages and smooth muscle cells.
Quantification of G6PD Activity in Murine Aortic Root Plaques
Consistent with the visual assessment (Figure 3A), G6PD activity was markedly higher in the medial α-SMA+ regions compared to the intimal α-SMA+ (∼6-fold) and CD68+ (∼7-fold) regions (Figure 3B-E) both in early and advanced plaques. Most intimal α-SMA+ cells demonstrated no or negligible G6PD activity except for small populations of α-SMA+ intimal cells with moderate G6PD activity along the fibrous cap. Unlike LDH, there was no difference in the G6PD activity of α-SMA+ and CD68+ cells between the early and advanced plaques.
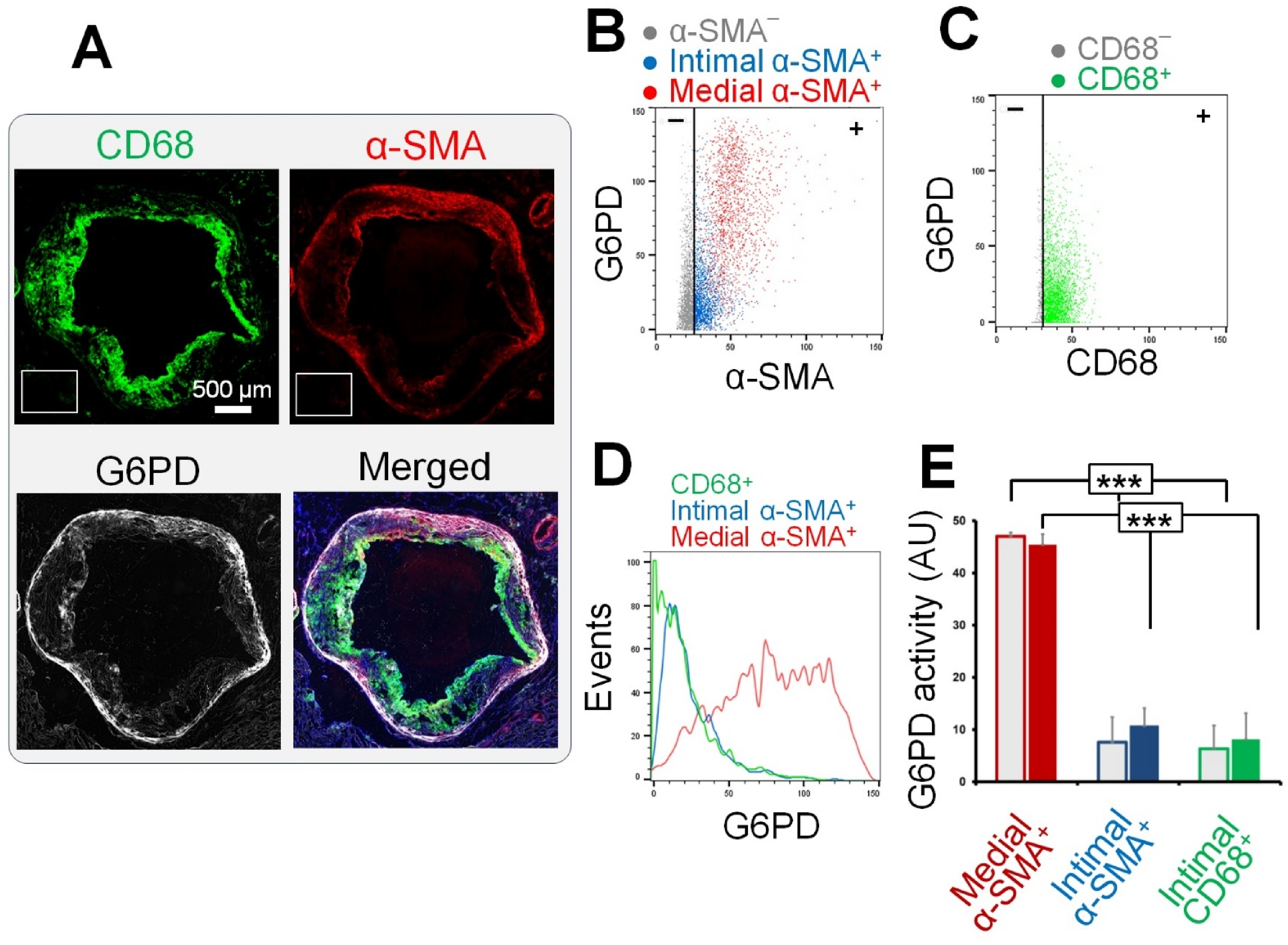
Quantification of G6PD activity of plaque macrophages and smooth muscle cells.
Metabolic Heterogeneity of Human Carotid Plaques
To address the translational relevance of the observed metabolic heterogeneity of murine atherosclerosis plaques, we qualitatively assessed the in situ activity of the LDH and G6PD in human carotid endarterectomy specimens. Consistent with our findings in murine aortic root plaques, a heterogeneous LDH activity was present within both intima and media (Figure 4A), whereas the G6PD activity was primarily localized to α-SMA+ cells within the media and to a lesser extent along the fibrous caps (Figure 4B).
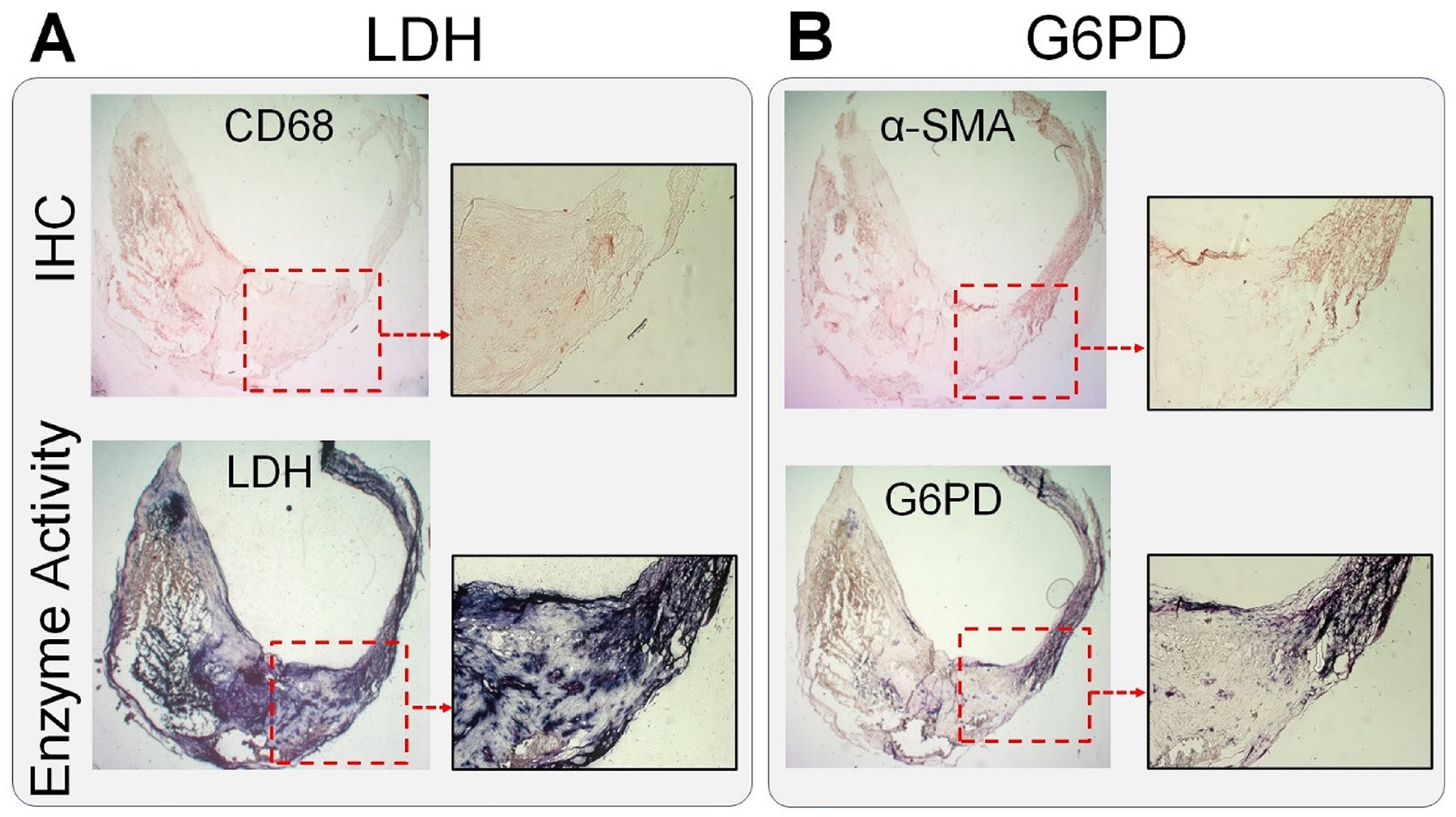
In situ metabolic mapping of atherosclerotic plaques. Consistent with the observed patterns in murine atherosclerotic plaques, human carotid endarterectomy specimens demonstrated a high, but markedly heterogeneous, level of LDH activity
Spatial Correlation of LDH and G6PD Activity with [3H]2-Deoxyglucose Uptake
We next determined if [3H]2-deoxyglucose autoradiography, as a high-resolution surrogate for [18F]FDG PET, distinguishes plaque regions with different levels of LDH and G6PD activity. To avoid scattered radiation from myocardium which interferes with an accurate quantification of glucose uptake in the aortic root, we used brachiocephalic plaques from apoE−/− mice after 7 months of western diet feeding. A modest incremental increase in LDH activity was present across regions of plaques with increased [3H]2-deoxyglucose uptake which reached ∼25% higher activity in the highest quartile compared to the lowest quartile (Figure 5A-B). However, G6PD activity was comparable across different quartiles of [3H]2-deoxyglucose uptake (Figure 5C-D).

Combined high-resolution [3H]2-deoxyglucose autoradiography and enzyme histochemistry of murine brachiocephalic plaques. Combined immunostaining, enzyme histochemistry, and high-resolution autoradiography (Fire Look-Up Table and smoothing algorithm, Image J) of advanced murine brachiocephalic artery plaques was performed to determine if glucose uptake correlates with LDH
Discussion
This study uncovered distinct patterns of the enzymatic activity of selected key enzymes involved in glucose metabolism in plaque macrophages and SMCs. Notably, two key TCA cycle dehydrogenases, SDH and IDH, exhibited minimal activity in the arterial wall and atherosclerotic plaques, whereas a heterogeneous LDH activity was observed in both the medial and intimal layers of atherosclerotic lesions. Interestingly, G6PD activity was mostly confined to the medial layer with a moderate activity found within the SMCs residing along the fibrous cap. Additionally, we observed that the uptake of [3H]2-deoxyglucose had only a modest correlation with the in situ LDH activity, suggesting a weak association between [18F]FDG uptake and the glycolytic state of atherosclerotic plaques. However, [3H]2-deoxyglucose did not significantly correlate with G6PD activity.
Despite extensive investigations of PET in atherosclerosis and the growing recognition of the critical roles of immunometabolic processes in regulating various aspects of cell biology,1,17 most of our knowledge on the biological correlates of [18F]FDG uptake in atherosclerosis has originated from in vitro cell culture studies1,5,6,8,10,18 that do not accurately mimic the complex microenvironment of the vessel wall.
The microenvironment of atherosclerotic plaques is highly complex, involving multiple interconnected mechanisms that contribute to cellular metabolic heterogeneity. 1 Different cells within plaques, such as macrophages and SMCs, exhibit diverse activation states and metabolic profiles. For example, macrophages are highly plastic cells that may exist across a spectrum of activation states as exemplified by the in vitro concept of M1 (pro-inflammatory) and M2 (anti-inflammatory) polarization states. 1 Similarly, SMCs transition across a spectrum from a differentiated contractile phenotype in the medial layer of the arterial wall to a synthetic phenotype in the intima. 19 Local variations in oxygenation, nutrient levels, interactions with neighboring cells, and the composition of noncellular components (eg, extracellular matrix proteins, cytokines, and growth factors) as well as the mechanical forces and shear stress from blood flow create a dynamic microenvironment which influences the metabolic landscape of cells within atherosclerotic plaques.1,20–22 Therefore, the current study attempts to address the limitation of metabolic studies performed in cultured cells in recapitulating the complexity of the plaque environment. We utilized combined in situ mapping of the activity of selected key enzymes involved in glucose utilization along with immunohistology to determine the activity of a few key enzymes involved in glucose utilization in SMCs and macrophages of murine and human atherosclerotic plaques. This approach enables to determine a spatial map of the metabolic heterogeneity of the cells; and therefore complements the growing strategies that delineate the cellular heterogeneity of plaques, such as single-cell RNA sequencing,23,24 spatial transcriptomics, 25 and mass spectrometry imaging. 3 It is noteworthy that metabolism is largely regulated at posttranscriptional and posttranslational levels; hence, the assessment of enzymatic activities provides a more proximate measure of the activity of the metabolic pathways.
A major highlight of this study is the minimal activity of two key TCA cycle dehydrogenases (SDH and IDH) along with a high glycolytic activity in atherosclerotic plaques. This resembles the previously reported immunometabolic response of macrophages to lipopolysaccharide, characterized by breaks in the TCA cycle activity along with augmented glycolysis which promotes a pro-inflammatory response (eg, high interleukin-1β production) through the accumulation of succinate and stabilization of hypoxia-inducible factor-α.26,27 Oxidative metabolism has also been linked to the inflammation-resolving state of macrophages in cell culture studies.5,6,28 Therefore, it may be speculated that sustained glycolysis along with a low level of TCA cycle activity in plaques may promote proinflammatory activation of macrophages and/or their inefficient polarization into inflammation-resolving states; hence, contributing to the chronic nonresolving inflammation as a hallmark of atherogenesis. 29 The implications of metabolic heterogeneity of plaques for understanding the pathogenesis of atherosclerosis and the development of diagnostic/therapeutic approaches require further investigation.
Another important finding of this study was that the activity of G6PD, the rate-limiting enzyme of the oxidative PPP, was highest within the medial SMCs followed by a modest activity by fibrous cap SMCs. PPP as the main source of NADPH plays a major role in regulating the redox state of the cells by contributing to the regeneration of reduced glutathione and as a substrate for NADPH oxidases. 30 Increased G6PD activity promotes the viability of the vascular SMCs and reduces their apoptosis by improving glutathione homeostasis. 31 PPP also plays a crucial role in supporting cell proliferation by supplying ribose-5-phosphate, a key precursor for nucleotide synthesis. 32 Increased G6PD activity and glucose metabolism through PPP have been observed in proliferating vascular SMCs. 33 While it may be speculated that PPP activity may contribute to the phenotypic changes in vascular SMCs, particularly upon the activation to the synthetic phenotype, or to the integrity of the fibrous cap, the mechanistical significance of PPP in atherosclerosis and plaque vulnerability requires further investigation.
PET is a robust technique for in vivo imaging of vessel wall metabolism. 1 Among multiple substrates, such as 11C-acetate,7,34 (2S,4R)-4-[18F]fluoroglutamine, 35 [18F]fluoromethylcholine, 36 and [11C]choline, 37 which have been successfully utilized for PET imaging of atherosclerosis, [18F]FDG is the most extensively studied radiotracer due to its widespread availability. 1 However, the biological basis of [18F]FDG PET in atherosclerosis and its clinical implications has remained incompletely understood. This reflects the limited specificity of [18F]FDG as a surrogate for glucose, a ubiquitous metabolite involved in numerous metabolic pathways, as well as the scarcity of studies examining the histological correlates of [18F]FDG uptake within the vessel wall. For example, while increased [18F]FDG uptake has been broadly considered to be mostly driven by the increased glycolytic activity of the proinflammatory M1 polarized macrophages, 1 recent studies have shown that SMCs 4 and inflammation-resolving M2 polarized macrophages5,6 may have a comparably high contribution to [18F]FDG uptake in atherosclerotic plaques. Furthermore, the results of the current study showed only a modest association between the 2-deoxyglucose uptake and LDH activity, suggesting that the contribution of nonglycolytic utilization pathways of glucose metabolism should be considered in the interpretation of the [18F]FDG PET in atherosclerosis. Notably, a comprehensive radiometabolite study has recently shown that metabolic pathways downstream of [18F]FDG-6-phosphate, such as oxidative PPP, can significantly contribute to the tissue accumulation of the radioactivity reaching up to 28% at 1 h and 39% at 2 h. 38
Metabolism has been a major focus of research over the past decade for its role in the pathogenesis of a wide range of diseases, including cancer and cardiovascular diseases.1,17,32,39 Beyond its significance for imaging, metabolism is increasingly being recognized as a key target for therapeutic interventions. 40 A growing body of evidence supports that several drugs currently used to reduce the risk of atherosclerosis and cardiovascular diseases, such as statins, achieve their effects by reducing inflammation through modulation of immunometabolism. 40 However, the impact of these therapeutic interventions on the heterogeneous metabolism of different cellular components of plaques has not been adequately characterized. The in situ assessment of cell metabolism may provide novel insights into the mechanisms by which these drugs modulate the risk of atherosclerosis and may ultimately pave the way for new therapeutic interventions that target immunometabolism.
A limitation of the current study is that in situ assessment of the enzymatic activities does not fully recapitulate the in vivo metabolism, which is regulated by various microenvironmental factors, such as the tissue oxygenation level, substrate availability, and hormonal factors. Furthermore, mapping of the activity of selected enzymes cannot provide a complete picture of the metabolic flux through a pathway. Nevertheless, in situ enzyme histochemistry provides a new complementary tool to the existing armamentarium to study plaque metabolism, particularly the metabolic heterogeneity of the different cellular components across different regions of the plaques. It is important to note that this study focused on delineating the metabolic heterogeneity of cells within atherosclerotic plaques, providing the biological basis for [18F]FDG uptake and underscoring that [18F]FDG uptake cannot simply be attributed to the pro-inflammatory activation of plaque macrophages. However, several technical limitations of PET hinder its applicability for noninvasive high-resolution mapping of plaque metabolism. First, the spatial resolution of PET is generally in the 3-5 mm range, 41 precluding cellular-level resolution. Second, high blood pool activity following [18F]FDG administration impacts the accuracy of quantifying tracer uptake by the arterial wall, though advances in PET technology with the new-generation total-body scanners and dynamic imaging have improved the resolution and distinction between blood pool and vessel wall activity.41,42 Third, arterial wall motion during the cardiac cycle deteriorates image quality and affects the accuracy of [18F]FDG uptake quantification by plaques. 43 Lastly, [18F]FDG uptake by myocardium is a significant challenge for detecting and quantifying its uptake by atherosclerotic plaques in the coronary arteries. 44 Consequently, extensive efforts over the past decade have focused on detecting various aspects of atherogenesis using other radiotracers. For example, fluorine-18-labeled sodium fluoride ([18F]NaF) is a highly promising and readily available radiotracer for detecting calcification, a key pathogenic process in atherogenesis, in coronary arteries and other arterial beds. 45
In summary, this study underscores the remarkably diverse metabolic activities present within atherosclerotic plaques which may not be accurately captured by in vitro investigation of the individual cell components. A comprehensive multidimensional approach, extending from well-regulated in vitro conditions (such as stable isotope metabolic tracing of cultured cells) to in vivo metabolic studies, is crucial to gain a deeper insight into the metabolic intricacies of various cell components within the vessel wall. Unraveling the metabolic complexities of the vessel wall may ultimately provide novel opportunities for the diagnosis and treatment of atherosclerosis.
Supplemental Material
sj-docx-1-mix-10.1177_15353508241280573 - Supplemental material for In Situ Mapping of the Glucose Metabolism Heterogeneity in Atherosclerosis: Correlation With 2-Deoxyglucose Uptake
Supplemental material, sj-docx-1-mix-10.1177_15353508241280573 for In Situ Mapping of the Glucose Metabolism Heterogeneity in Atherosclerosis: Correlation With 2-Deoxyglucose Uptake by Joseph Haddad, Selim Demirdelen and Clayton E. Barnes, Steven A. Leers, Sina Tavakoli in Molecular Imaging
Footnotes
Abbreviations
Data and Materials Availability
The data needed to evaluate the conclusions of the paper are present in the paper and/or the Supplemental Materials.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Animal experiments were conducted in accordance with a protocol approved by the University of Pittsburgh Institutional Animal Care and Use Committee. Anonymized atherosclerotic plaques were collected from patients who underwent standard-of-care carotid endarterectomy under a “No Human Subject involvement” classification by the University of Pittsburgh Institutional Review Board. Considering the No Human Subject designation, informed consent was not required for this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Institute of Health (NHLBI, K08 HL144911) and Radiological Society of North America (RSD-1820) as well as a Seed Fund from University of Pittsburgh/UPMC Departments of Radiology and Medicine to S.T.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
