Abstract
Prangos ferulacea is a widely recognized and valuable medicinal herb known for its diverse biological characteristics. Our study was conducted to evaluate the anticancer and immunomodulatory effects of P. ferulacea. Ethyl acetate, methanol, and aqueous extracts were prepared from P. ferulacea, and the MTT assay was used to measure antiproliferation activity against 5 cancer cell lines (MDA-MB231, T47-D, HeLa, EMT6/P, and Vero). The 3 extracts underwent a qualitative LC/MS spectrometry analysis to determine each extract’s composition. The MTT test was used to measure the proliferation of lymphocytes. Extracts were evaluated for their effects on macrophages phagocytic activity and pinocytosis function using the nitro blue tetrazolium assay and the neutral red method, respectively. The impact of ethyl acetate and methanol was assessed in mice inoculated with EMT6/P using the in vivo model, and the tumor size was measured. Results showed that ethyl acetate and methanol extracts had the highest activity against T47-D with IC50 values of 0.019 mg/ml. The ethyl acetate extract exhibited moderate effectiveness against the following cell lines: MDA-MB-231 and EMT6/P with IC50 values of 0.104 and 0.255 mg/ml, respectively. Meanwhile, methanol extract showed activity against MDA-MB231 and EMT6/P cell lines and had IC50 values of 0.214 and 0.31 mg/ml, respectively. Regarding immunomodulatory assays, the methanol extract, followed by the ethyl acetate extract, significantly increases lymphocyte proliferation and macrophage phagocytosis activity. Aqueous extracts increase the activity of pinocytosis. Both ethyl acetate and methanol extracts reduced tumor size by 61.46% and 34.47%, respectively. They also decreased the average weight of the tumor cells in the in vivo model. The results of P. ferulacea have valuable activity against cancer cells and stimulate the immune system. However, further molecular investigations are required to understand the mechanism of action of P. ferulacea activity.
Introduction
Cancer is increasingly recognized as a major global health challenge, contributing significantly to premature mortality and imposing severe societal and economic burdens. 1 Recent projections estimate that by 2030, the annual incidence of new cancer cases and cancer-related deaths will reach 21.4 million and 13.2 million, respectively, positioning cancer as one of the leading causes of death worldwide in the twenty-first century. 2 Cancer arises from abnormal neoplastic progression, often resulting in the formation of malignant cell masses. 3 Cancer therapy involves multiple strategies. Examples of these strategies include surgery, radiation therapy, chemotherapy, immunotherapy, and hormonal therapy. These approaches are typically employed individually or in combination, depending on the type of cancer, location, and stage 4
Among these, chemotherapy remains a cornerstone in cancer management across various stages and types. However, its clinical use is limited by poor bioavailability, the need for high-dose administration, low therapeutic indices, the risk of recurrence, and the growing threat of multidrug resistance (MDR). 5 Recently, the integration of immunotherapy in the treatment protocol for various solid tumors has marked a remarkable advancement in oncology. 6 In contrast to conventional treatment, this therapy primarily aims to boost the immune system to target and eradicate both local and spread metastasis, develop long-term immune memory, and mediate immune protection against cancer recurrence. 7 These challenges have sparked an increasing interest in discovering novel anticancer agents, particularly from natural sources such as plants, which offer a diverse array of bioactive compounds. 8
Phytochemicals have demonstrated the ability to modulate carcinogenesis through various mechanisms, including halting, delaying, or reversing tumor progression before the onset of invasive malignancy. 9 Immunomodulatory compounds, which can either stimulate or inhibit immune responses, are essential for maintaining immune homeostasis and protecting against diseases such as cancer. 10 Natural compounds with immunomodulatory properties are of growing interest for future applications in cancer therapy. 11 Among Apiaceae, different Prangos species are widely used in traditional medicine. Prangos ferulacea (L.) Lindl., a perennial herb native to western Asia and the eastern Mediterranean, is one of the most commonly utilized species. 12 Previously categorized as Cahrys ferulacea (L.) Calest. under the genus Cahrys, it is now classified under Prangos and is one of the most studied medicinal plants in traditional medicine. 13 Traditionally, its leaves are used as animal food, while its fruits and roots are used for therapeutic purposes.14,15 Previous studies have reported a range of biological activities for P. ferulacea, including cytotoxic, antioxidant, antispasmodic, analgesic, antimicrobial, antidiabetic, sedative, and anti-inflammatory effects, as well as benefits in treating gastrointestinal, liver, and nervous system disorders.16,17
Previous studies have investigated the cytotoxic effects of coumarins isolated from P. ferulacea on cancer cell lines such as PC3 (Prostate cancer cell line), SKNMC (neuroblastoma cell line), and H1299 (non-small cell lung carcinoma cell line), where compounds like isoimperatorin and osthol demonstrated apoptosis-inducing activity through caspase activation and mitochondrial pathways. However, these studies were limited to specific isolated compounds and specific cancer cell lines. 18 In contrast, this study examined the anticancer and immunomodulatory activities of P. ferulacea crude extracts across various cell lines, concentrations, and experimental models. Also, the study tested for the first time P. ferulacea plant samples collected from the north of Iraq.
This study aims to clarify the different phytochemical components of P. ferulacea and provide insights into the anticancer and immunomodulatory characteristics of the aerial parts of plant extracts for the first time, revealing their potential as a treatment and its uses in cancer treatment.
Materials and Methods
Reagents, Instruments, and Commercial Kits
The equipment, reagents, and kits that were used in the current study were CO2 incubator model No 460-1CE (Lab-Line Instrument Incorporation, USA), water bath model No. 1083 (GFL, Germany), hemocytometer (Neubauer, Germany), Lyophilizer (Edwards, UK), rotary evaporator (Heidolph WB 2000, Germany), Dulbecco’s Modified Eagle Medium (DMEM; Sigma, USA), Minimum Essential Medium (MEM; Sigma, USA), and Roswell Park Memorial Institute (RPMI) 1640 Medium (Sigma, USA). Gentamycin, penicillin-streptomycin solution, fetal bovine serum, and L-glutamine (Sigma, USA). Trypsin ethylene diamine tetra acetic acid (trypsin EDTA; Sigma, USA), phosphate buffer saline (PBS), NH₄Cl RBC lysis for humans (bio-world, USA), and trypan blue 0.4% (Sigma, USA). MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay kit (Bioworld, UK) was utilized to evaluate antiproliferative activity. Commercial kits (DiaSys Diagnostic Systems GmbH, Germany) were used to assess liver and kidney function.
Prangos ferulacea (L.) Lindl. Collection and Extract Preparation
The aerial parts of Prangos ferulacea were collected from May to July 2022 on the mountains of Sulaimani/ Kurdistan region of Iraq, and scientifically identified by Prof. Saman A. Ahamad. Fifty grams of the finely ground powdered P. ferulacea were macerated in 500 ml of solvent 1:10 w/v using 3 different solvents with different polarities: aqueous, absolute methanol, and ethyl acetate. With constant stirring, the extraction was carried out for 48 hours. The filter paper and rotary evaporator apparatus are set at 40°C and 60 rpm for methanol and ethyl acetate extracts. A lyophilizer was used to completely dry the aqueous extract after it had been stored for lyophilization. These extracts were then collected and stored for subsequent use at −20°C. The following equation is used to calculate the percentage yield:
Liquid Chromatography-Mass Spectrometry (LC-MS)
The 3 extracts were dissolved in their solvents to create freshly made ethyl acetate, methanol, and aqueous samples. The samples were filtered via a 0.45 μm membrane filter (cellulose acetate membranes) made entirely of cellulose acetate polymer (Sterlitech, Auburn, WA, USA). Each sample was used for analysis immediately as it was prepared. To evaluate the extracts using the Exion LC technique, an X500 QTOF mass spectrometer (SCIEX, Framingham, MA, USA) connected with an ESI was utilized. The separation was performed using an InertSustain C18 column (GL Sciences Inc., Tokyo, Japan, 25 cm × 4.6 mm × 5 m). The mobile phase was a mixture of (A) acetonitrile and (B) 0.1% formic acid in aqueous (1:1000) with a gradient flow rate of 1.0 ml/min. 0.6 μl of volume was set up for injection. LC-MS employed a positive mode with an ion spray voltage of 5000 V, a collision energy of 10 V, and a decluttering potential of 80 V.
Cell Lines and Cell Culturing Conditions
Five cell lines were used to evaluate the antiproliferative effects of P. ferulacea: MDA-MB-231, T47-D, HELA, EMT6/P, and VERO. The MDA-MB-231 (ATCC Cat# HTB-26, RRID: CVCL_0062) and T47-D (ATCC Cat# HTB-133, RRID: CVCL_0553), the HeLa (ATCC Cat# CCL-2, RRID: CVCL_0030), and the Vero (ATCC Cat# CCL-81, RRID: CVCL_0059) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). The EMT6/P (ECACC Cat# 96042344, RRID: CVCL_4173) was obtained from the European Collection of Authenticated Cell Cultures (ECACC, Salisbury, UK). MDA-MB-231 is the human breast adenocarcinoma cell line with a triple-negative estrogen receptor, progesterone receptor, and HER2 protein. 19 T47-D is a human epithelial breast cancer cell line. 1 HELA is the human epithelioid cervix carcinoma cell line. 20 EMT6/P is the mouse mammary carcinoma cell line. 21 VERO is the kidney epithelial cells from African green monkeys and represents the normal model in this study. 22
The cells were cultivated in complete media and kept in an incubator with 5% CO2 and 95% humidity at 37ºC. MDA-MB-231 and HELA cell lines were cultured in DMEM medium, while T47D was cultured in complete RPMI 1640 medium. 23 VERO and EMT6/P were cultured in MEM media. 24
Antiproliferative Assay
The antiproliferative activity of the extracts was tested using an MTT (the tetrazolium salt, 3, [4,5-dimethylthiazol-2-yl-yl]-2,5-diphenyltetrazolium bromide), as mentioned in the literature. 25 In the current research, the cells were treated with various concentrations of P. ferulacea extracts (2.5-0.019 mg/ml) and processed as indicated by Mahmod and Talib. 25 In comparison to the negative control (untreated cells), half the maximum inhibitory concentration (IC50) values of the treated cells were determined by SPSS 27. The percentage of cell survival was computed using the following equation adopted from. 24
Animals
All experimental protocols utilized in this study were approved by the Applied Science Private University Research and Ethical Committee’s Faculty of Pharmacy, with approval number 2024-PHA-47, in accordance with accepted standards of ethics. A total of 25 female Balb/C mice, weighing between 23 and 25 g and aged 4 to 6 weeks, were used in this study. The mice were housed in separate cages with bedding made of wood shavings and maintained under standard laboratory conditions, including a consistent temperature of 25°C, relative humidity of 50% to 60%, and continuous air ventilation. They were kept in a pathogen-free environment with a 12-hour light/dark cycle and provided with food and water. At the end of the experiment, the mice were sacrificed by cervical dislocation according to the method described by Davidge et al. 26
This study is an exploratory study, so no pilot study was conducted to estimate sample size. The number of animals was specified based on previous studies27,28
Preparation of Murine Splenocyte
Balb/C mice (N = 3) were sacrificed, and the spleens were extracted aseptically. The cells were maintained in petri dishes with 5 ml of RPMI 0.5 penicillin and 0.5 gentamycin. After being transferred to a centrifuge tube, the cell suspension was centrifuged for 10 minutes at 2000 rpm and 4°C. Red blood cells were removed by centrifugation, discarding the supernatant and re-suspending the cells in 1 mol/l NH₄Cl RBC lysis buffer, and the splenocytes were prepared for seeding and counting in various tests.
Lymphocytes Proliferation Assay
This assay used the MTT test. A 96-well tissue culture plate was seeded with a suspension of splenocytes at a known concentration of 2 × 106 cells/ml. Subsequently, 100 μl of P. ferulacea extracts at varying concentrations (0.3125-2.5 mg/ml) were added, and the experiment was conducted in triplicate. The plate was incubated for 48 hours at 37ºC in a humidified environment with 5% CO2. After that, 10 μl of MTT solution (5 mg/ml) was added to each well, and the wells were incubated for 3 hours. Next, 100 μl of DMSO was added, and an ELISA microplate reader was used to measure the absorbance at 550 nm. The results were presented as a stimulation index (SI) compared with a negative control (untreated). 29
Macrophage Isolation From Peritoneal Fluid
Balb/C mice (N = 3) were first given a peritoneal injection of 5 ml of 3% (w/v) brewer’s thioglycollate medium. The following phase, cell collection, was carried out after waiting 3 to 5 days to initiate the formation of macrophages. After cervical dislocation was used to euthanize the mice, the abdominal cavities were visible enough that 5 ml of ice-cold PBS could be injected. The fluid was cautiously removed and put in a centrifuge tube to obtain the required amount. The resulting cell pellets were resuspended in complete RPM 1640 media after centrifugation at 2000 rpm and 4°C for 10 minutes. 30
In Vitro Phagocytic Assay Nitro Blue Tetrazolium Reduction Test
Generally, a 96-well tissue culture plate was seeded with peritoneal macrophages at a concentration of 5 × 106cells/well. The cells were subsequently treated with varying concentrations of P. ferulacea extracts (0.3125-2.5 mg/ml), and the plate was incubated for 48 hours at 37°C. Next, 20 μl of nitro blue tetrazolium (NBT; 1.5 mg/ml in PBS) and 20 μl of yeast suspension (5 × 107 cells/ml in PBS) were added to each well, except the control wells, which received 20 μl PBS and 20 μl DMSO. The plate was incubated at 37°C for 1 hour. Following that, each well received 140 μl of DMSO and 120 μl of 2 M KOH. Finally, the optical density was measured in the microplate reader at 550 nm. The following equation was used to calculate phagocytic activity. 31
Pinocytic Activity Assay by a Neutral Red Method
A 96-well tissue culture plate was used to seed peritoneal macrophages at a concentration of 5 × 106 cells/well. Different concentrations of extracts (0.3125-2.5 mg/ml) were then added, and the plate was incubated for 48 hours at 37°C. Next, each well received 100 μl of neutral red solution (7.5 mg/ml in PBS), which was then added, and the medium was incubated for 2 hours. After that, 100 μl of a cell lysis solution, consisting of a 1:1 ratio of ethanol and 0.01% acetic acid, was added to each well to break down the cells. The plate was incubated overnight at room temperature. Ultimately, the plate was set up on a microplate reader. The absolute OD values, which indicate dye uptake, were used to demonstrate pinocytic activity. 31
Acute Toxicity Test of Prangos ferulaea Methanol and Ethyl Acetate Extracts
A limit test was conducted on a small group of mice to determine the dose ranges for the actual measurement of the median lethal dose (LD50). Briefly, PBS containing 5% Tween 20 was used to dissolve the methanol and ethyl acetate extract. Three female mice (6 weeks old, 20-23 g weight) were injected intraperitoneally with a dose of 100 mg/kg of the extract estimated from a previous study. 32 Next, animals were monitored for any deaths for 24 hours. Using fresh animals, the subsequent doses were either lowered by 0.7 if the dose was fatal or elevated by 1.5 if the dose was tolerated. The upper and lower limits that were utilized to create the LD₅₀ doses were the maximum non-lethal and minimum lethal doses. 33
Tumor Cell Inoculation and Antitumor Activity Assay
A suspension of EMT-6/P cells in a MEM medium was prepared for each of the 21 animals. An intraperitoneal injection of 150 000 cells/0.1 ml was given to every mouse. Ten days after injection of the tumor, the mice were randomly assigned to 3 groups, each consisting of 7 mice. Group Ⅰ was treated with the methanol extract at a dose of 100 mg/kg. Group ⅠⅠ was treated with ethyl acetate extract at a dose of 100 mg/kg. Group ⅠⅠⅠ represented the negative control and received PBS. All treatments were continued for 12 days. After 12 days of treatment, a digital caliper was used to measure the length and width of the tumor sizes. The tumor volumes were calculated with the following equation:
Where L = length of a tumor, W = width of the tumor.
Evaluation of Kidney and Liver Function of Treated Mice
Liver and kidney toxicity were assessed after the treatment with P. ferulacea. Aspartate transaminase (AST), alanine transaminase (ALT), creatinine, and uric acid in serum samples were measured to evaluate liver and kidney functions by following the instructions in the relevant kits (DiaSys Diagnostic Systems GmbH, Germany).
Statistical Analysis
The standard error of the mean, or mean ± SEM, was used to present the data. The statistical significance among the groups was determined using SPSS one-way analysis of variance (ANOVA), followed by post-hoc analysis. Differences between groups were considered significant when the P-value was less than .05 (P < .05). IC50 values were calculated using nonlinear regression in SPSS (Statistical Package for the Social Sciences, Chicago, Illinois, version 27).
Results
Percentage Yield of Crude Extract
Different extraction solvents resulted in different percentage yields for the extracts. Water extract showed the highest percentage, followed by methanol extract (Table 1). Ethyl acetate extract exhibited the lowest percentage of yield. The high yield of the water extract is likely due to the good solubility of polar compounds such as phenolics and flavonoids in water, which agrees with results obtained in a previous article. 34
The Percentage Yield Obtained From Extracting 50 g of P. ferulacea Using Maceration.

LC-MS Analysis of Prangos ferulacea Extracts
The phytochemical composition of each extract was determined using LC-MS analysis. The analysis was conducted to detect various bioactive substances, including coumarins, phenolics, flavonoids, alkaloids, iridoid glycosides, and steviol glycosides (Table 2).
Relative Percentage of Phytochemical Compounds Detected By LC-MS of P. ferulacea Ethyl Acetate, Methanol, and Aqueous Extracts.

Abbreviation: m/z, mass to charge ratio.
Antiproliferative Activity of Prangos ferulacea Extracts
When different cell lines were treated with escalating concentrations (0.019-2.5 mg/ml) of P. ferulacea extract, a dose-dependent reduction of cell growth was observed (Figure 1A-C). Both ethyl acetate and methanol were more cytotoxic against T47-D with the same IC50 values of 0.019 mg/ml (Table 3). On the other hand, aqueous extract exhibited lower activity against different cancer cell lines with IC50 values more than 2.5 mg/ml. Both ethyl acetate and methanol extracts were found to be active against the VERO cell line with IC50 values of 0.201 and 0.219 mg/ml, respectively (Tables 3 and 4).
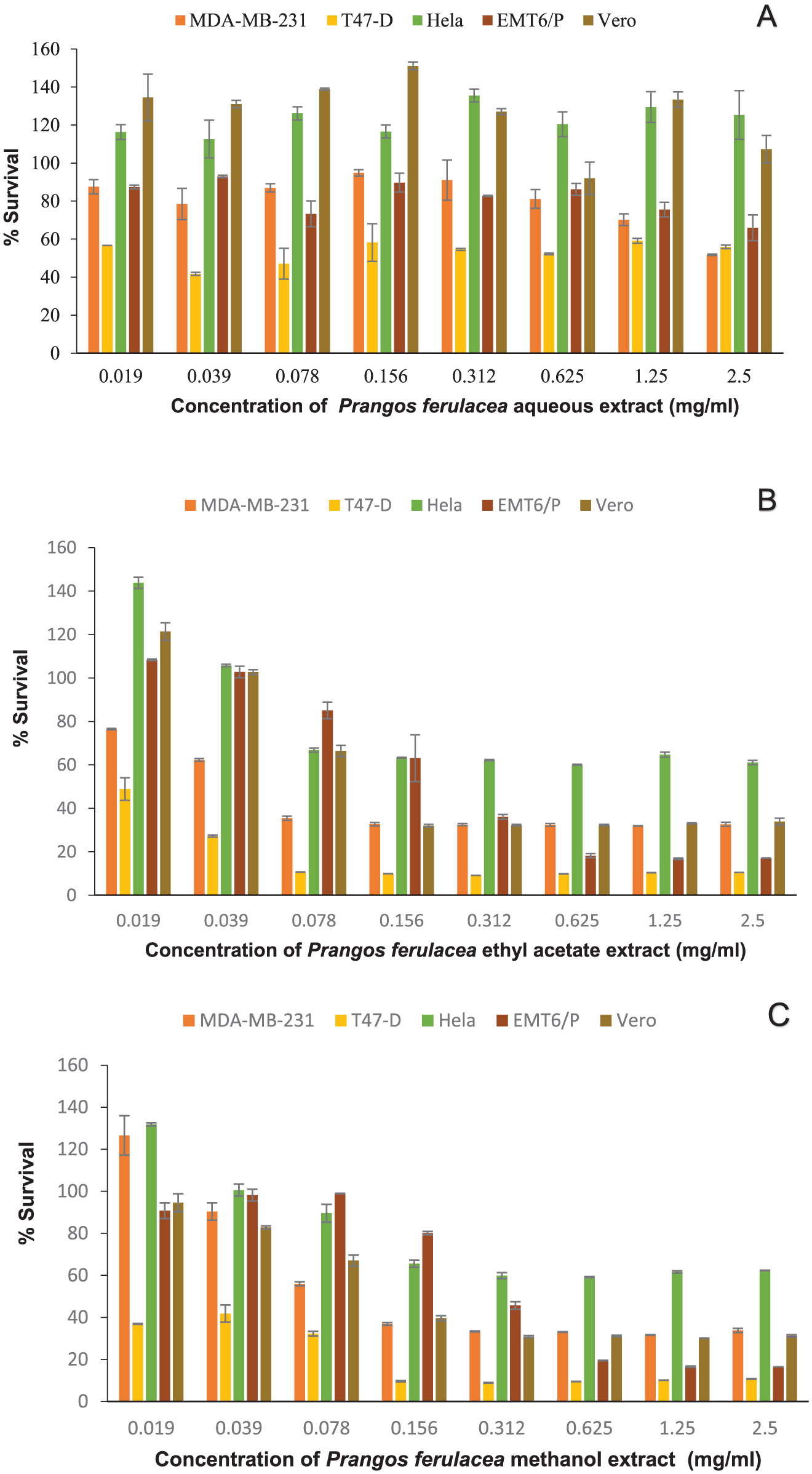
(A) Antiproliferative activity of aqueous extract of P. ferulacea on MDA-MB-231, T47-D, HeLa, EMT6/p, and Vero cell lines. (B) Antiproliferative activity of ethyl acetate extract of P. ferulacea on MDA-MB-231, T47-D, HeLa, EMT6/p, and Vero cell lines. (C) Antiproliferative activity of methanol extract of P. ferulacea on MDA-MB-231, T47-D, HeLa, EMT6/p, and Vero cell lines.
IC50 Values of P. ferulacea Extracts Against Different Cancer Cell Lines.
Results of Ascending Doses in the Limited Study of P. ferulacea Ethyl Acetate and Methanol Crude Extract in Mice.

The Effect of Prangos ferulacea Extracts on the Proliferation of Splenic Lymphocytes
The results showed that methanol and ethyl acetate were the most successful in stimulating lymphocyte proliferation at a concentration of 2.5 mg/ml, with stimulation index values of 3.66 and 3.43, respectively. On the other hand, the aqueous extract showed lower activity with a stimulation index value of 1.88 at the same concentration (Figure 2).

The effect of P. ferulacea extracts at various concentrations (0.3125-2.5 mg/ml) on the proliferation of splenic lymphocytes.
The Effect of Prangos ferulacea Extracts on Macrophage Phagocytosis and Pinocytosis
The results showed that methanol and ethyl acetate were the most successful in stimulating phagocytosis at a concentration of 2.5 mg/ml, with phagocytic index values of 377.272 and 365.277, respectively. On the other hand, the aqueous extract showed lower activity (Figure 3).

The effect of P. ferulacea extracts at various concentrations (0.3125-2.5 mg/ml) on macrophage phagocytosis.
The results of pinocytic assays showed that aqueous extract was the most successful in stimulating pinocytosis at a concentration of 2.5 mg/ml, with an absorbance value of 0.780 nm, followed by methanol with an absorbance value of 0.605. On the other hand, ethyl acetate showed lower activity (Figure 4).

The effect of P. ferulacea extracts at various concentrations (0.3125-2.5 mg/ml) on macrophage pinocytosis.
Evaluation of Prangos ferulacea Ethyl Acetate and Methanol Extracts Toxicity on Mice
A limit test was performed on mice to ascertain the acute toxicity of ethyl acetate and methanol extracts. The maximum solubility of the extract was 281.3 mg/ml for mice weighing 25 mg, which equals 100 mg/kg. This will be considered the highest soluble dose that did not result in death.
Antitumor Effects of Prangos ferulacea Ethyl Acetate and Methanol Extracts on EMT6/P Parent Cells Implanted in Mice
The tumor in the ethyl acetate extract group was inhibited, with a significant reduction in tumor size of (−61.46%), followed by methanol extract (−34.47%), compared to the control group (27.31%). No death was recorded for either group (Figure 5). The percentage of mice with no detectable tumor in ethyl acetate and methanol extracts in the treated group was 57% and 42%, respectively, showing normal activity with no side effects (Figure 6).

An EMT6/P cell line plot showing the mean size of the tumor change (mm3) versus treatment time (days).

Changes in tumor sizes of EMT6/P after dissection at day 12 in all groups at a dose of 100 mg/ml compared to each other, n = 7.
Effect of P. ferulacea Extracts on Serum Levels of ALT, AST, Creatinine, and Uric Acid
Treating mice with ethyl acetate and methanol extracts showed no effect on the serum level of ALT, AST, and creatinine, with non-significant results (P > .05). At the same time, the uric acid values of the methanol and ethyl acetate extract were 4.01 and 2.83 mg/dl, compared to the pre-treatment value of 1.72 mg/dl, with a significant statistic (P < .05) (Tables 5 and 6; Figures 7 and 8).
The Effect of P. ferulacea Methanol and Ethyl Acetate Extracts on Tumor Size and Weight in Mice (n = 7).

Abbreviations: mm3: cubic millimeter; Av, average.
Serum Levels For Several Tests on Pre-Treatment, Negative Control, and Treatment Groups.

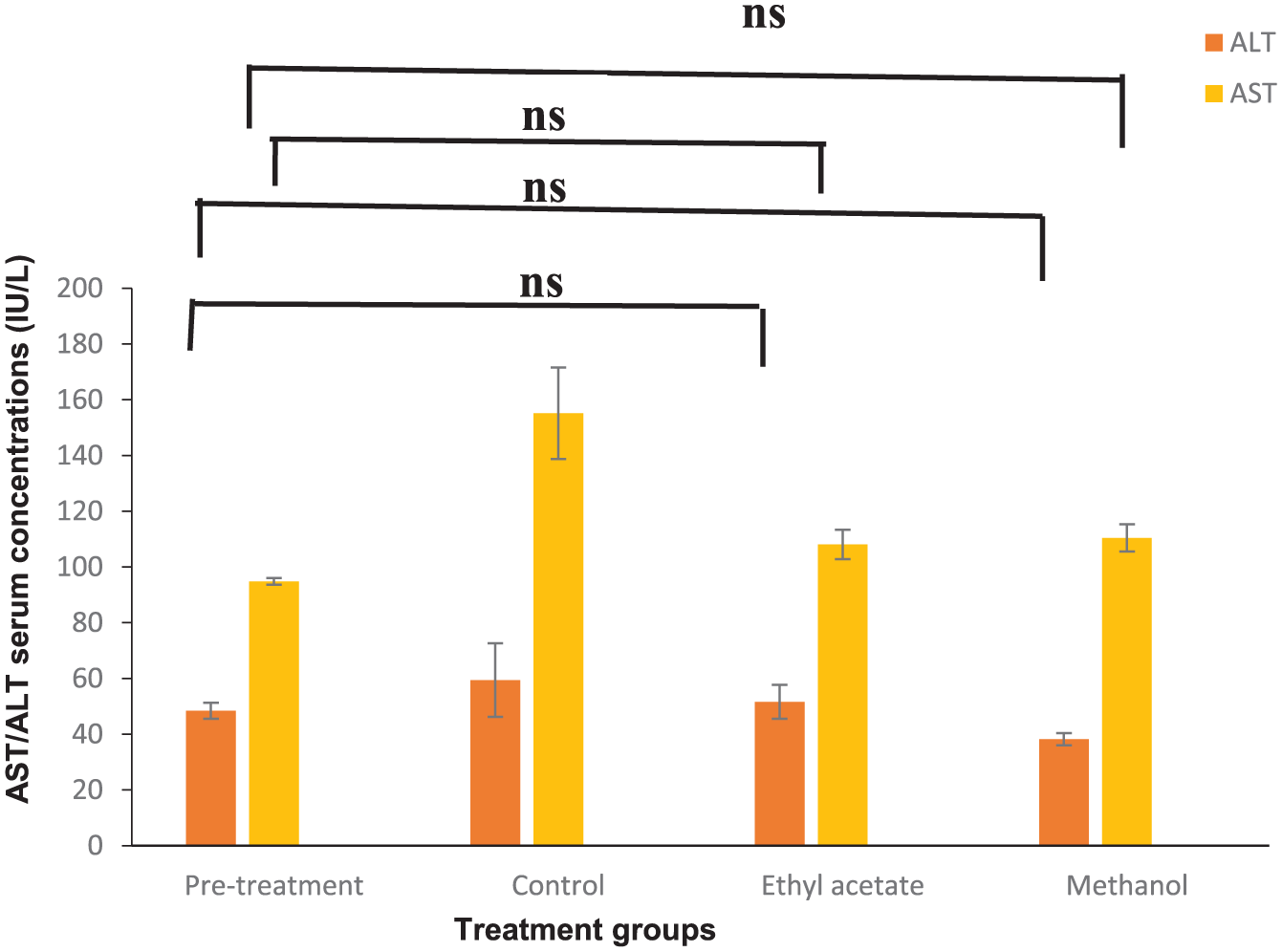
P. ferulacea ethyl acetate and methanol extract’s effect on blood AST and ALT in EMT6/P tumor-inoculated mice (n = 3).

P. ferulacea ethyl acetate and methanol extract’s effect on blood uric acid and creatinine in EMT6/P tumor inoculated mice (n = 3).
Discussion
Cancer is a complex, multifactorial disease that arises from alterations in numerous physiological and biochemical mechanisms. Over recent decades, it has become evident that conventional anticancer drugs can induce multidrug resistance (MDR) in cancer cells, often leading to tumor recurrence. Therefore, the need to develop novel and potent anticancer agents remains constant. Historically and currently, plant-based natural compounds have served as a primary source of anticancer drugs and are expected to continue playing a crucial role. 35 P. ferulacea has been utilized in traditional medicine to treat a variety of diseases due to its demonstrated therapeutic benefits. 15 It has been reported to contain a wide range of bioactive compounds that contribute to its diverse biological and functional properties, including anticancer, analgesic, antioxidant, antidiabetic, and antimicrobial activities. 34 There are limited data associated with the biological activities of P. ferulacea. To the best of our knowledge, the present study is the first to comprehensively evaluate P. ferulacea through immunomodulatory and antiproliferative evaluation in vitro and in vivo using several extracts and several cancer cell lines. Also, it is the first time to study a sample of this plant collected from the north of Iraq. In the present study, the anticancer and immunomodulatory effects of P. ferulacea extracts were evaluated. P. ferulacea exhibited the ability to inhibit the survival of cancer cells in a concentration-dependent manner. According to the results, both ethyl acetate and methanol extracts significantly reduced the viability of all tested cancer cell lines (Figure 1A-C). These extracts demonstrated the highest activity against the T47-D cell line, with IC50 values of 0.019 mg/ml. Additionally, variation in IC50 values of the same extract across different cell lines was observed (Table 3). In contrast, the aqueous extract showed lower activity against the cancer cell lines tested. Kizil evaluated the cytotoxic activity of the methanol extract obtained from the stem of P. ferulacea against the non-small cell lung cancer cell line (H-460) using the WST-8 method. The results showed that the methanol extract activity at concentrations of 125 and 62.5 µg/ml, in contrast to the lung fibroblast cell line’s cytotoxic activity (MRC-5), although this effect was not statistically significant. 36 An additional study was conducted to evaluate the cytotoxic activity of P. ferulacea essential oil on several cancer cell lines, with the strongest activity shown against breast adenocarcinoma (MDA-MB-231 ) at a concentration of IC50 22.41 μg/mL, while the melanoma (A375) and colon carcinoma (HCT116) cell lines demonstrated the lowest sensitivity to the essential oil (IC50 25.08 and 30.35 μg/ml), respectively. 37
For the Vero cell line, ethyl acetate and methanol extracts exhibited some noticeable cytotoxicity, with IC50 values of 0.219 and 0.201 mg/ml, respectively. This toxicity could be attributed to differences in cell-type sensitivity. Given the effective antiproliferative activity of P. ferulacea extracts, further investigation was conducted using a breast cancer mouse model. Treatment led to a higher cure rate and a significant reduction in tumor size. These effects are likely associated with the cytotoxic action of phytoactive compounds that target tumor cells directly. These activities may be explained by the presence of phytochemicals, including alkaloids, iridoids, phenolics, coumarins, and flavonoids. 38 Many alkaloid-rich extracts have demonstrated antiproliferative and anti-metastatic effects against various cancers in both in vitro and in vivo studies. 39 In the present study, the antiproliferative and tumor-reducing effects could, in part, be attributed to the presence of piperine, a compound known for modulating signaling pathways and molecular mechanisms involved in multiple stages of cancer progression. 40 Phenolic acids also exhibit a wide range of biological activities, including anticancer, immunomodulatory, antioxidant, antidiabetic, and antimicrobial effects. 41 One such compound, 4-O-feruloylquinic acid, is an isomer of chlorogenic acid. 42 Chlorogenic acid has been shown to exert antiproliferative effects by inducing apoptosis through upregulation of P53, Bax, and caspase-3, while downregulating Bcl-2 expression. 43 The immune system plays a central role in defending the host against pathogens and includes mechanisms that target altered cells during carcinogenesis. 44 Our preliminary findings revealed the immunomodulatory potential of P. ferulacea, primarily by evaluating its effects on murine lymphocyte and macrophage activity. Increased splenic lymphocyte counts, as well as enhanced pinocytosis and phagocytosis following treatment, suggest that the extracts exert immunostimulatory effects on both the innate and adaptive immune systems. These findings are consistent with a previous study, which reported that the methanol extract of P. ferulacea at concentrations of 500 to 2500 μg/ml significantly enhanced human lymphocyte proliferation. 45 The observed immunomodulatory activity of P. ferulacea could be linked to the compounds detected by LC-MS. Flavonoids, in particular, are known to possess immunomodulatory activities that are crucial for the management and treatment of various malignancies. 10 Isoquercetin was previously reported to boost the release of inflammatory factors, such as tumor necrosis factor-α (TNF-α), interleukin (IL)-1β, and IL-6, by inhibiting the NF-κB and mitogen-activated protein kinase (MAPK) pathways. 46 Similarly, diosmin has been shown to selectively modulate macrophage activity by promoting anti-tumor macrophages and suppressing pro-tumor ones, while also influencing lymphocyte function. 47 Furthermore, diosmin enhances the expression of molecules involved in the recruitment and activation of natural killer (NK) cells, potentially boosting their anti-tumor efficacy. 48 To assess safety, liver and kidney function markers, including ALT, AST, creatinine, and uric acid, were evaluated, as they are key indicators of drug metabolism and toxicity. 49 The levels of ALT, AST, and creatinine in the treated group remained within the normal range, indicating an acceptable safety profile for all treatments. These results can be attributed to careful dose selection based on LD₅₀ estimation, which showed no harmful effects. Although uric acid levels in the treated group were higher than in the control group, they remained within the normal physiological range for mice and are therefore considered acceptable. Notably, no deaths were recorded throughout the 12-day treatment period.
Further research is needed to determine if there is an association between a specific metabolite or compound of P. ferulacea and a particular form of cancer, as well as to uncover the underlying mechanisms of action of the medicinal compound.
Conclusion
P. ferulacea is rich in phytochemicals with diverse biological activities, including compounds with well-documented anticancer and immunomodulatory effects. The present study demonstrated that P. ferulacea significantly enhances splenic lymphocyte proliferation, phagocytosis, and pinocytosis, reflecting potent immunomodulatory activity. Furthermore, the ethyl acetate extract exhibited pronounced anticancer effects in both in vitro and in vivo models, providing novel evidence of its therapeutic potential. Further investigations are needed to isolate and characterize the active constituents and elucidate their underlying mechanisms of action. Moreover, these findings provide valuable insights into the pharmacological potential of P. ferulacea and its potential for future practical applications.
Footnotes
Acknowledgements
The authors would like to acknowledge the Applied Science Private University (Amman, Jordan) for providing full financial support for this research.
Ethical Considerations
This study was approved by the Ethics Committee of Applied Science Private University (Approval Number: 2025-PHA-24).
Author Contributions
Conceptualization: W.H.T. and S.A.A, formal analysis: Sh.M.Al, investigation: Sh.M.Al, methodology: Sh.M.Al, software: Sh.M.Al, validation: W.H.T, resources: W.H.T, data curation: Sh.M.Al, writing—original draft preparation: Sh.M.Al, writing—review and editing: M.M.Al, S.A.A H.K.Al, A.M.A.O, and W.H.T, visualization: W.H.T.; supervision: W.H.T, project administration: W.H.T.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Applied Science Private University, Amman, Jordan.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.




