Abstract
Objective. The adverse effects of 5-fluorouracil (5-FU) are well recognized. Fuzheng-Yiliu granule (FYG) is capable of enhancing the immune function and suppressing tumor growth. In the present study, the authors evaluated if FYG could synergize with low-dose 5-FU in inhibiting tumor growth. Methods. Hepatoma 22 (H22) tumor-bearing mice were treated with FYG (18 g/kg, ig), 5-FU (10 mg/kg, ip), or 5-FU plus FYG for 5 days. The relative tumor proliferation rates, tumor weight and apoptosis of tumor tissue were measured. White blood cell (WBC) and lymphocyte (LY) were counted. Interleukin-2 (IL-2) and tumor necrosis factor (TNF-a) in the serum were measured. Results. FYG alone had antitumor effect. Combination of 5-FU and FYG produced a more potent antitumor effect and caused more marked apoptosis in tumor tissue (compared with vehicle, P < 0.01; compared with 5-FU or FYG, P < 0.05). Mice treated with 5-FU plus FYG had higher thymus index (P < 0.05) compared with the vehicle group. The numbers of both WBC and LY were decreased by 5-FU (compared with vehicle, P < 0.01), which was significantly reversed after FYG was administered (5-FU + FYG vs 5-FU, P < 0.01 and P < 0.05). Mice receiving FYG alone or FYG plus 5-FU had higher serum levels of TNF-a (P <0.01) compared with the vehicle. Conclusions. Traditional Chinese medical herbs capable of strengthening the body’s vital energy have great potential to be used as an adjuvant therapy for cancer patients who cannot tolerate the adverse effects of chemotherapy.
Introduction
Hepatocellular cancer (HCC) is the fifth most common cancer worldwide and the third most common cause of cancer death. 1 It has a high incidence in China also. Curative treatments for HCC include surgery, local destruction techniques (radiofrequency ablation or percutaneous ethanol injection) and liver transplantation. But unfortunately only about 40% of patients can benefit from curative treatments, and only one third of patients are typically resectable. 2 Hence, for most patients, palliative treatments, which include transarterial chemoembolization, systemic therapy, and radiotherapy, are their only choice. 3 Chemotherapy has been shown to improve the survival of patients with HCC. 4 The most commonly used systemic chemotherapeutic agents are 5-fluorouracil (5-FU) and doxorubicin (Adriamycin). When used as monotherapy to HCC patients, 5-FU showed an improved median survival of 14 weeks. 5 Recent studies suggest that 5-FU given together with gemcitabine, cisplatin, or oxaliplatin can shrink the tumors in some HCC patients.6,7 However, the routine dose of 5-FU is accompanied by serious side effects such as diarrhea, nausea and vomiting, poor appetite, and low blood counts. 8 Many approaches have been pursued to alleviate these avert effects, including low-dose chemotherapy.
Interest in finding anticancer herbs from traditional Chinese medicine (TCM) remains high because they have low toxicity and few side effects. Moreover, these herbs may be combined with chemotherapy drugs that can strengthen the vital energy of the patients to alleviate the adverse effects of chemotherapeutics and improve patients’ quality of life. A number of clinical practices and experimental studies have shown that some Chinese herbal formulas of Fu Zheng Gu Ben are effective in combination with chemotherapy to treat cancer.9-13 Fu Zheng Gu Ben is a major principle for TCM physicians to compose a formula for patients characterized by weakness and fatigue, such as those with tumors. “Fu” is to help, “Zheng” means Zheng Qi (the body’s resistance to disease), and “Gu Ben” means strengthening the body’s basic function. Fu Zheng Gu Ben can enhance vital energy and improve resistance to disease. Huang et al 10 showed that Shenqi Fuzheng injection, composed of Astragalus membranaceus and Codonopsis pilosula, can alleviate bone marrow suppression caused by chemotherapy, improve quality of life, and prolong the survival of patients with advanced breast cancer. Pan et al 11 reported that Fuzheng Yiliu decoction (Radix Hedysari, Angelica sinensis (Oliv.) Diels, Curcuma zedoaria (Christm.) Rosc., Patrinia heterophylla Bunge) can enhance the efficiency of chemotherapy on malignant gastrointestinal tumor and also reduce the gastroenterological reactions and bone marrow suppression. Pharmacological studies demonstrate that herbs of Fu Zheng Gu Ben can improve the efficiency of chemotherapy in fighting cancer by regulating immune function. These herbs can increase the numbers of immune cells (lymphocytes, natural killer [NK] cells, and monocytes) and production of some cytokines (interleukin [IL] family, tumor necrosis factor [TNF] family, and interferon [INF] family), which can bind receptors on cancer cells and activate the death receptor pathway to eliminate tumor cells.12,13
FYG, a Chinese herbal formula according to Fu Zheng Gu Ben principle, is composed of Astragalus membranaceus, Fructus ligustri lucidi, Ganoderma lucidum, and Rhizoma dioscoreae oppositae. FYG has been used in clinical trials to treat HCC, colon cancer, gastric cancer, and lung cancer and has been shown to have an obvious antitumor effect when given as an adjunctive therapy to surgery and chemotherapy. 14 In this study, we investigated the antitumor effect of low-dose 5-FU when given concomitantly with FYG on H22-tumor-bearing mice and the underlying mechanisms.
Methods
Reagents
FYG granule was prepared from water extract of a recipe containing Astragalus membranaceus (30 g), Ligustrum lucidum Ait. (30 g), Ganoderma lucidum (15 g), and Rhizoma dioscoreae oppositae (15 g) by Purapharm Co, Ltd (Hong Kong, China). One gram of FYG granule is equivalent to 6 g Astragalus membranaceus, 6 g Ligustrum lucidum Ait., 3 g ganoderma lucidum, and 3 g Rhizoma dioscoreae oppositae. FYG granule was dissolved in normal saline (NS) at a concentration of 0.36 g/mL. 5-FU (25 mg/mL) was from Shanghai Amino Acids Company (Shanghai, China) and diluted to 10 mg/mL with saline solution. PerCP-conjugated hamster anti-mouse CD3, FITC-conjugated rat anti-mouse CD4, PE-conjugated rat anti-mouse CD8, and PE-conjugated rat anti-mouse CD49 were purchased from BD Biosciences (San Jose, CA). Tumor TACS In situ Apoptosis kit was from Roche Bioscience (Palo Alto, CA), and RIA kit was from the Institute of Radioimmunity, General Hospital of People’s Liberation Army of China (Beijing, China).
Mice
Male Institute of Cancer Research mice with body weight ranging from 18 to 22 g were purchased from the School of Basic Medical Science, Peking University, China. The animals were maintained in a pathogen-free facility (23 ± 2°C, 55 ± 5% humidity). Food and water were provided ad libitum. All procedures in treating mice were performed according to the law on Animal Care Guidelines of Chinese, and the Animal Care Committee of Fujian University of Traditional Chinese Medicine approved our protocols.
Tumor Xenograft
Murine H22 cell line was from the School of Basic Medical Science, Peking University (Beijing, China). Cells were cultured in Dulbecco’s modified eagle’s medium (Gibco Laboratories, Grand Island, NY) supplemented with 10% fetal bovine serum (Gibco Laboratories), penicillin, and streptomycin (10 U/L, Gibco Laboratories) in a humidified atmosphere with 5% CO2 at 37°C. The hepatoma model was established by subcutaneous inoculation of H22 cells (1 × 106 cells per mouse) into the right flank of mice. Forty-eight hours after inoculation, mice were randomly divided into 4 groups (n = 8): vehicle-treated group (NS, ig), 5-FU group (10 mg/kg, ip), FYG group (FYG 18 g/kg, ig), and 5-FU + FYG group (5-FU 10 mg/kg, ip, + FYG 18 g/kg, ig). On day 5, the peripheral blood was collected from orbital plexus and the tumor was excised. Tumor volume was calculated according to the following formula: Tumor Volume (TV mm3) = d2 × D/2, where d and D are the shortest and longest diameter, respectively. Relative tumor volume (RTV) = TV5 (tumor volume in the fifth day of every group)/TV0 (tumor volume of baseline in each group). The relative tumor proliferation rate (%) = TRTV/CRTV, where TRTV is the RTV of the treatment group and CRTV is the RTV of the vehicle group.
Food Intake and Body Weight
Each cage of mice was provided daily with food. On the following day the remaining food was collected and weighed. The daily food intake was calculated by subtracting this value from the amount of diet provided the previous day. The well-being of the mice was monitored daily and the body weight was determined at baseline and after 5 days of treatment.
Blood Cell Count Assay
A capillary pipette containing anticoagulant (ethylenediaminetetraacetic acid [EDTA] for cell counting, heparin for flow cytometry) was inserted in the lateral canthus and blood collected from the retroorbital sinus. Other blood sample was collected in procoagulant tube and separated for serum. Twenty microliters of whole blood sample was collected and blood cells were counted by an automatic blood cell counter (Abbott Laboratories, Abbott Park, IL).
Thymus Index
Thymus was collected from the mice, washed with phosphate-buffered saline (PBS), and weighed at day 5. Thymus index (TI) was calculated according to the following formula: TI = (thymus weight [mg]/body weight [g]) × 100.
Assay for Percentages of Immune Cells
One hundred microliters of heparin-coated blood was added into a tube and incubated with fluorochrome-conjugated antibodies in the dark for 10 minutes. The 2.5-µL antibodies used varied slightly with individual cases, but most often included a combination of CD3/CD4, CD3/CD8, and CD3/CD49. Erythrocytes were lysed by red blood cell lysis buffer (including 0.155 mol/L ammonium chloride, 0.01 mol/L potassium bicarbonate, 0.1 mmol/L EDTA, and 1% paraformaldehyde in PBS) for 10 minutes. After washing with PBS, the samples were resuspended with 500 µL PBS and analyzed on FACScalibur flow cytometer with CELLQuest software (BD Bioscience, San Jose, CA).
Assay for IL-2 and TNF-α
Serum (100 µL) was measured for IL-2 and TNF-α with radioimmunoassay (RIA) kit according to the manufacturer’s instructions in a gamma radioimmunoassay counter (Kaipu Electromechanical Co, Ltd, Xi’an, China). The levels of 2 cytokines were determined by the competitive binding of the cytokines in the sample with 125I radiolabeled IL-2 and TNF-α standards, respectively. The sensitivity of the assay was 0.3 µg/L.
Assay for Tumor Apoptosis
Sections of tumors from mice treated as above were fixed with 4% paraformaldehyde for 48 hours. The 5-µm-thick sections of tumor samples were analyzed by terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick end labeling (TUNEL) staining using TumorTACS In situ Apoptosis kit (Roche Bioscience, Palo Alto, CA) to detect fragmented DNA according to the manufacturer’s instructions. Microscopic immunohistochemical photos were captured by an Olympus microscope (Olympus, Tokyo, Japan) and a Moticam 5000 C camera of Motic Instruments (Richmond, Canada) and analyzed by Motic Med 6.0 software. The positive cells and total number of cells were counted at 5 arbitrarily selected microscopic fields at 100 magnification (each 7050 µm2 in size). Then the apoptotic index (AI) was calculated according to the following formula: AI = number of positive cells/total number of cells.
Statistical Analyses
Statistical analysis was done with SPSS 16.0 (SPSS Inc, Chicago, IL). Data are shown as mean ± SD, and statistical significance of difference was performed by 2 × 2 analysis of variance. When there was an interaction effect between 5-FU and FYG, we analyzed the multiple comparisons between 5-FU + FYG and 5-FU. If there was no interaction effect between 5-FU and FYG, we would analyze the main effect of 5-FU or FYG. P < .05 was considered significant.
Results
Effects of 5-FU + FYG on Tumor Growth
Compared with the vehicle group, the relative tumor proliferation rates of the 5-FU and FYG group were significant decreased (63.9% and 73.7%, respectively, P < .05 for both comparisons). Importantly, the combination of 5-FU with FYG exhibited additive antitumor activity, as indicated by 55.2% in T/C compared with the vehicle group (Figure 1A, P < .01). Consistently with the above results, 5-FU, FYG, and 5-FU + FYG induced a 32% (P < .05), 30% (P < .05), and 60.7% (P < .01) decrease in tumor weight, respectively, compared with the vehicle group (Figure 1B).

Effect 5-FU + FYG on H22 solid tumor growth
Effects of 5-FU + FYG on Body Weight and Food Intake
The body weights were 19 to 21 g at baseline and were similar among the 4 groups. After 5 days of treatment, body weight per mouse was 25 to 27.5 g. The differences among the 4 groups were statistically insignificant either at baseline or at the end of administration. Average daily diet consumption per mouse was about 5 to 6.5 g and again the differences among the 4 groups were statistically insignificant (data not shown).
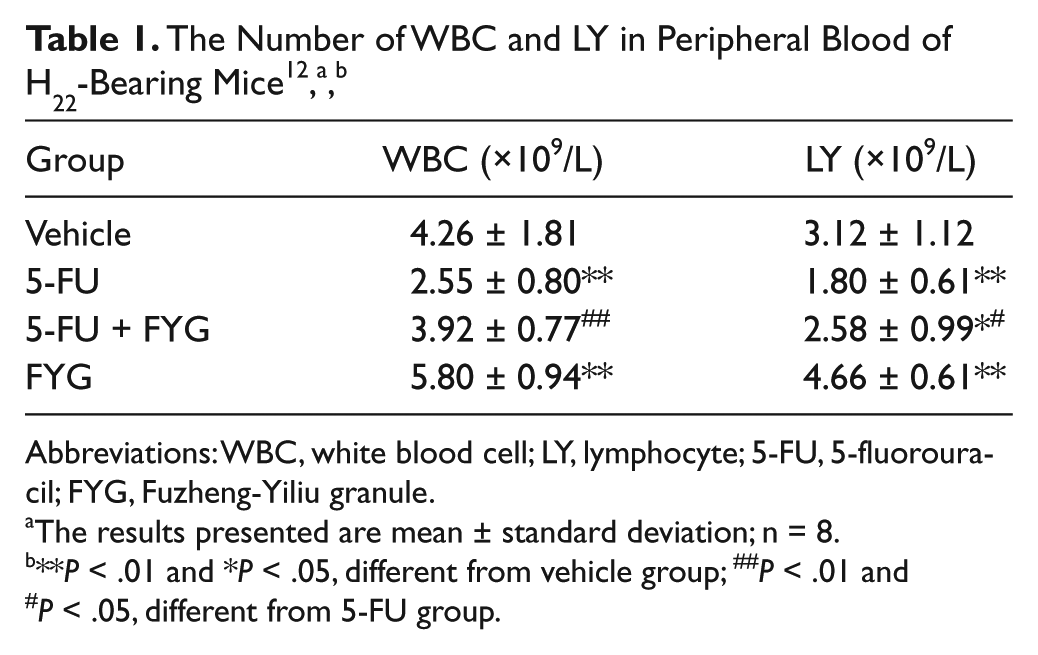
Effects of 5-FU + FYG on Blood Cell Count
As shown in Table 1, the numbers of white blood cell (WBC) and lymphocyte (LY) both decreased significantly in the 5-FU group, which was reversed when FYG was simultaneously administered. The numbers of WBC was higher in the 5-FU + FYG group when compared with the 5-FU group (P < .01) and was highest in the FYG group when compared with the vehicle group (P < .01). Similarly, the number of LY was higher in the 5-FU + FYG group than in the 5-FU group and the vehicle group (P < .05). Mice in the FYG group had the highest LY number (P < .01, compared with vehicle).
Abbreviations: WBC, white blood cell; LY, lymphocyte; 5-FU, 5-fluorouracil; FYG, Fuzheng-Yiliu granule.
The results presented are mean ± standard deviation; n = 8.
**P < .01 and *P < .05, different from vehicle group; ##P < .01 and #P < .05, different from 5-FU group.
Effects of 5-FU + FYG on TI
We also examined the effect of 5-FU + FYG on the thymus index of H22 mice. As shown in Figure 2, the TI of the 5-FU + FYG group was significantly higher than the other 3 groups (P < .05), but there was no significant difference between vehicle and the 5-FU or FYG group.

Effects of 5-FU + FYG on TI
Effects of 5-FU + FYG on Percentages of LY Cells
The percentages of CD3+, CD4+, CD8+, and NK subset in peripheral blood of H22-tumor-bearing mice from each group are shown in Table 2. The percentages of CD3+ and CD4+ subpopulations in peripheral blood of the 5-FU + FYG and FYG groups were both higher than the vehicle group (P < .01). The CD3+ percentage in the 5-FU + FYG group was higher than in the 5-FU group (P < .05). The ratio of CD4+/CD8+ was highest in the 5-FU + FYG group (compared with the vehicle group, P < .01; compared with 5-FU group, P < .05). Administration of 5-FU caused a decrease in NK cells, which was reversed by administration of FYG (5-FU + FYG vs vehicle group, P < .05; FYG vs vehicle group, P < .01; Table 2).
Abbreviations: 5-FU, 5-fluorouracil; FYG, Fuzheng-Yiliu granule.
The result presented are mean ± standard deviation; n = 8.
**P < .01 and *P < .05, different from vehicle group; ##P < .01 and #P < .05, different from 5-FU group.
Effects of 5-FU + FYG on IL-2 and TNF-α
To investigate the production of TNF-α and IL-2, we determined their serum levels by RIA. As seen in Figure 3, TNF-α levels in both 5-FU + FYG and FYG groups were significantly higher than that of the vehicle group (P < .01). It was also higher in the 5-FU + FYG group than in the 5-FU group (P < .05, Figure 3A). The production of IL-2 was also significantly increased in the FYG group compared with that in the vehicle group (P < .01, Figure 3B).

Effects of 5-FU + FYG on cytokine production
Effects of 5-FU + FYG on Tumor Apoptosis
We investigated apoptosis by immunohistochemistry. The apoptosis index in the 5-FU + FYG group was highest among the 4 groups, and the difference between the 5-FU + FYG group and vehicle group, 5-FU group, or FYG group was statistically significant (P < .01, P < .05, and P < .05, respectively; Figure 4A). Representative pictures of immunohistochemistry are shown in Figure 4B to E, where apoptotic cells were stained with brown.
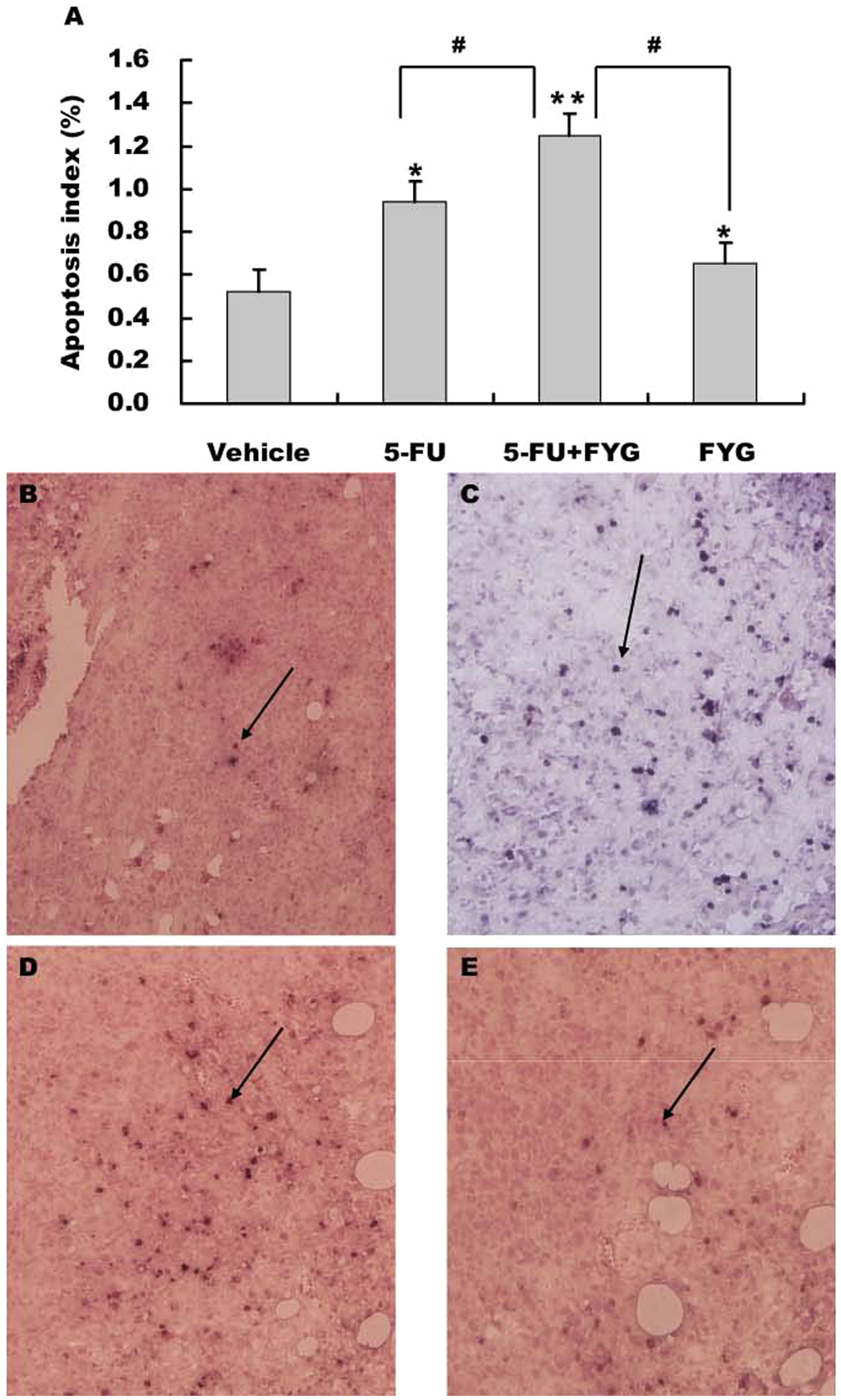
Effects of 5-FU + FYG on apoptosis of tumor
Discussion
5-FU is a widely used agent for patients with unresectable advanced HCC, but at the commonly used dose it can only achieve an objective response rate of about 10%. 15 In addition, some patients have to discontinue the therapy due to serious adverse effects, such as myelosuppression, mucositis, dermatitis, diarrhea, vomiting, nausea, and cardiac toxicity. 8 Low-dose chemotherapy has been used for unresectable cancer as a “tumor dormancy” therapy. 16 For elderly patients with metastatic colorectal cancer, administration of low-dose 5-FU was shown to achieve a favorable survival time with acceptable adverse effects. 17 The use of low-dose 5-FU and low-dose CPT-11/cisplatin for recurrent or metastatic colorectal cancer has also been reported to show favorable survival without toxicity.18,19 Although low-dose chemotherapy is associated with fewer side effects, there is no doubt that low-dose chemotherapy is less effective and it is desirable to promote its efficiency by TCM.
FYG, a formula of Fu Zheng Gu Ben, includes 4 herbs, that is, Astragalus membranaceus, Fructus ligustri lucidi, Ganoderma lucidum, and Rhizoma dioscoreae oppositae. In TCM, Astragalus is classified as a herb that can tonify Qi (the vital substance constituting human body and maintaining life activities) and is indicated for symptoms of spleen Qi deficiency such as diarrhea, fatigue, and lack of appetite. It also raises Qi of the spleen and stomach and is widely used to treat prolapses of organs such as uterus, stomach, and anus. 20 Fructus ligustri lucidi can tonify the liver and kidney, nourish Yin (dark, passive, downward, cold, contracting, and weak), and improve vision. 21 Ganoderma lucidum can promote life energy, or Qi of the heart, and benefits those with a knotted and tight chest. 22 Rhizoma dioscoreae oppositae is capable of strengthening the spleen, tonifying the lung, and reinforcing the kidney to benefit renal essence. 21 These herbs have been demonstrated to possess immunomodulatory and antitumor activities in immunodeficiency syndromes and cancer. 21 Astragalus membranaceus and Ganoderma lucidum can enhance immune function by regulating CD3, CD4, CD8, and NK cell activity and production of IL-2, IL-3, IL-6, TNF-α, and INF-γ, which may be responsible for their antitumor properties.23,24 Astragalus membranaceus also can stimulate B cell and antibody production and enhance macrophage activity. 25 Ligustrum lucidum fruit has been used as an immune-modulating agent. 26 It is an important herb for immune-system restoration after chemotherapy (as part of Fu Zheng therapy) and was shown to be capable of increasing WBC count. 27
Cytokines play important roles in controlling immune responses and inflammatory reactions. In the present study, we found that FYG can promote the production of IL-2 and TNF-α, which is consistent with the previous report that Ganoderma lucidum can stimulate the production of TNF-α, IL-6, IL-2, and IFN-γ from human macrophages and PBMC.28,29 Many studies have reported that compounds isolated from the herbs in our recipe have marked immunomodulatory properties, although the underlying mechanisms are still not clear. For example, nuclear factor-κB (NF-κB), a transcription factor involved in regulating the transcription of many of the immunomodulatory mediators, can be activated by β-glucans, leading to enhanced expression of the IL family, TNF family, and IFN family.30-32 Astragalus membranaceus, Fructus ligustri lucidi, Ganoderma lucidum, and Rhizoma dioscoreae oppositae all contain β-glucans. Indeed, it was reported that Ganoderma lucidum can promote production of cytokines through activation of NF-κB. 33
According to the TCM theory, FYG has the functions of invigorating and supplementing Qi, tonifying the liver and kidney, nourishing Yin, and strengthening the spleen. FYG has been used as an adjunctive therapy for surgery and chemotherapy on HCC in clinical trials. In this study, we investigated the effect of low-dose 5-FU plus FYG on H22-tumor-bearing mice and the mechanism mediating its activity. Our results indicated that low-dose 5-FU plus FYG had a potent antitumor effect on H22 tumor mice and was safe as indicated by stable food intake and body weight gain. 5-FU-induced decrease of the number of WBC and LY in peripheral blood of H22-bearing mice, which could be reversed by FYG supplementation. These results support the proposal that Fu Zheng Gu Ben can be used to alleviate the side effects of chemotherapy.
Recently, clinical trials have shown that host immunity of metastatic colorectal cancer was not affected or even improved by low-dose leucovorin/5-FU treatment. 34 Consistent with these observations, our study showed that low-dose 5-FU could increase the percentage of lymphocytes such as CD3+, CD4+, and NK cells in the peripheral blood of H22-tumor-bearing mice. We found that FYG also increased the percentages of T and NK cells and the levels of IL-2 and TNF-α in serum. There was an additive effect when low-dose 5-FU and FYG were concomitantly administrated, which may be responsible for their more potent antitumor effect. Increased percentages of T and NK cells may be responsible for the increased level of IL-2 and TNF-α, which in turn may stimulate and activate T cells and NK cells. Activated T cells and NK cells can interact with death receptors (CD95, TNF-related apoptosis-inducing ligand receptor) on the surface of tumor cells and induce tumor cell apoptosis by forming a death-inducing signaling complex to trigger the cascade of caspase activation.35-37 Indeed, using TUNEL assay we found that 5-FU plus FYG caused marked tumor cell apoptosis.
In conclusion, the current study showed that the combination of low-dose 5-FU and FYG has a more potent inhibitory effect on H22 tumor growth by regulating immune function and inducing apoptosis. Furthermore, almost no side effects were observed. The signaling pathways, including NF-κB pathway, that account for the enhanced immunity function and apoptosis by low-dose 5-FU and FYG will be further investigated.
Footnotes
Authors’ Note
Most of the experiments were carried out in the Cell Biology Lab of Fu Jian University of Traditional Chinese Medicine.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by CHEN Ke-ji Integrative Medicine Development Fund (CKJ2010020), International Science Joint Project of the Ministry of Science and Technology of the People’s of China (2008DFA32200), National Natural Science Foundation of China (81102582), and Fujian Province Natural Science Foundation (2009J01170).