Abstract
Cancer survivors experience a range of side effects during and after treatment. There is a need for a rigorous synthesis of the most recent and best available evidence on the role of nutritional supplements for supportive care in cancer, to inform shared decision-making. We searched 5 databases for umbrella reviews, meta-analyses and systematic reviews on nutritional supplements for supportive cancer care, excluding studies on pain, anxiety and depression, which are covered in recent guidelines. We found 52 reviews that reported on 250 RCTs on 18 supplements for 16 indications. Almost all reviews were of low/critically low quality (assessed using A MeaSurement Tool to Assess systematic Reviews version 2). There was moderate-certainty evidence for benefit from the following supplements: amino acids and oral proteolytic enzymes for severity of radiation-induced dermatitis, N-acetyl cysteine for prevention of chemotherapy-induced peripheral neuropathy (CIPN) in individuals with gastrointestinal cancers. There was low to very low certainty evidence that glutamine, zinc, probiotics and melatonin may be effective for oral mucositis; Vitamin E, omega-3 fatty acids, glutamine and other amino acids may be effective for preventing CIPN. Serious adverse events were reported for high-dose Vitamin A, and dose-related adverse events were reported with zinc and Vitamin E. However, the majority of nutritional supplements were associated with only minor adverse events. Due to the low to very low certainty of the majority of evidence, firm clinical recommendations cannot be made. Further research to conclusively evaluate benefit and harm, including potential impact on efficacy of standard treatments, should be conducted.
Introduction
Globally, 1 in 5 people will be diagnosed with cancer in their lifetime. 1 Advances in early detection, novel therapeutics and other aspects of clinical care have led to overall improvements in cancer survival. 2 An estimated 53.5 million people worldwide are cancer survivors with a personal history of a cancer diagnosis within the last 5 years.1,3 Whilst the concept of survivorship is often associated with the period following treatment completion, a cancer survivor is defined as any individual diagnosed with cancer from the day of diagnosis until the end of life. 4
Cancer survivors are a heterogeneous population with diverse and dynamic healthcare needs that vary along the cancer continuum.5,6 Their experiences and healthcare needs are often shaped by treatment side effects – the undesired yet often expected consequences of medical interventions. 7 Acute side effects are most amplified during treatment, impacting quality of life (QoL) and treatment adherence.8,9 Although some side effects resolve post-treatment, many survivors face persistent and late-onset sequelae that can negatively impact well-being and contribute to long-term morbidity.10,11
Treatment-specific toxicities vary widely. 8 Fatigue is the most prevalent, persistent and burdensome symptom affecting over 50% of survivors regardless of treatment modality.8,12,13 Chemotherapy is associated with nausea and vomiting (CINV; 30%-60%),14,15 oral mucositis (OM; 30%-90%) 16 and peripheral neuropathy (CIPN; up to 70%), 8 while radiotherapy-associated dermatitis (RD) affects up to 95% of recipients. 17 Additional acute effects such as xerostomia, weight loss, hot flushes and insomnia vary in incidence and severity according to cancer type, stage, treatment regimen and performance status. 18 While individual symptoms can be distressing and debilitating on their own, cancer survivors often undergo multimodality treatment with diverse regimens, and symptoms rarely occur in isolation.9,18
Advances in early detection, immunotherapy and targeted treatments have significantly improved outcomes and increased survival rates.11,19 However, these gains are accompanied by long-term challenges and latent effects of treatment. Targeted therapies may cause hypertension, skin toxicities and metabolic disturbances.19,20 In addition to acute side effects, long-term complications include lymphoedema, cardiotoxicity, cognitive impairment, sexual dysfunction, genitourinary complications and endocrine disruption encompassing weight gain and cardiometabolic diseases. 11
National and international multidisciplinary organisations, such as the Multinational Association of Supportive Care in Cancer (MASCC), 21 the European Society of Medical Oncology (ESMO) and the American Society of Clinical Oncology (ASCO) 22 have developed evidence-based guidelines for supportive care management across the most common and debilitating side effects. However, the inclusion of nutritional supplement interventions – defined here as vitamins, minerals, phytochemicals, pre/probiotics and foods containing live cultures, fatty acids or amino acids provided in supplement form 23 - remains limited, largely due to the magnitude and heterogeneity of the available literature.
Despite widespread usage among cancer survivors 24 and general perceptions of safety, current guidelines offer limited recommendations on nutritional supplements. The Society of Integrative Oncology (SIO), in collaboration with ASCO, has issued specific guidelines for the integrative management of anxiety, depression, pain management and fatigue, which include some guidance on supplement use. 25 Yet, a comprehensive synthesis of evidence on the efficacy and safety of nutritional supplements across treatment-related side effects is lacking.
To support evidence-informed shared decision-making between clinicians and patients, a rigorous synthesis of the most current evidence is needed. This umbrella review aims to synthesise findings from existing umbrella reviews, meta-analyses and systematic reviews, to provide an evidence base on the efficacy and safety of nutritional supplements in managing cancer treatment–related side effects.
Materials and Methods
An umbrella review, also known as an overview of reviews, is a novel form of evidence synthesis that aims to collate and assess evidence from multiple reviews on a specific topic and provide a synthesis of the current body of evidence. 26
Protocol Registration
This umbrella review was designed and reported in accordance with the Preferred Reporting Items for Overviews of Reviews (PRIOR) reporting guidelines. 27 A protocol was developed a priori and prospectively registered on the Joanna Briggs Institute (JBI) systematic review register 24 April 2024.
Search Strategy
A comprehensive search strategy was developed by CE, SG and SBD. Two authors (SBD, AO) conducted a systematic search of Medline, CINHAL and EMBASE and Cochrane Database of Systematic Reviews and Epistemonikos for relevant publications from January 2019 to May 2024, limited to the last 5 years to capture and synthesise the most recent evidence. All umbrella reviews, meta-analyses and systematic reviews with data from randomised controlled trials (RCTs) available in English were considered. The full search strategy is available in the Supplemental Material (Table S1). Titles and abstracts of all retrieved studies were screened in duplicate independently. All full text articles were screened in duplicate by 2 of the 6 reviewers (SBD, AO, SG, CE, ALM, CP or JL). Disagreements were resolved following discussion.
Selection Criteria
The eligibility criteria for this review were defined according to the Population, Intervention, Control, Outcome, Study Design (PICOS) framework and shown in Table 1. Criteria were developed by our multidisciplinary team with expertise across oncology (medical and radiation), surgery, and supportive care. Given that there have been recent, comprehensive, evidence-based guidelines published on integrative oncology for anxiety, depression and pain management in cancer care, 28 we excluded these outcomes from our review. Although gastrointestinal (GI) side effects are common, they were not included in this review, with the exception of mucositis, nausea/vomiting, postoperative ileus.
Inclusion Criteria-PICOS Framework.

There is little consensus on what is included and excluded in defining nutritional supplements. 23 For purposes of this review, we have defined nutritional supplements as products with ingredients that may include vitamins, minerals, phytochemicals, pre/probiotics, and foods containing live cultures (eg, fermented dairy products), fatty acids or amino acids in supplement form. Probiotics were defined as “live microorganisms which when administered in adequate amounts confer health benefits” with inclusions classified according to the International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement. 29 For phytochemicals, we included isolated or standardised compounds administered in supplement form and excluded whole plant materials and/or unprocessed herbs used in their natural form, including extracts of whole plant materials, as we considered these to be herbal medicines rather than nutritional supplements. 30
Data Collection and Analysis
Hierarchal Evidence Gathering
Due to the broad scope of the research question and resource limitations, it was not practicable to extract and collate data from all eligible reviews retrieved. Another common methodological challenge with umbrella reviews is the problem of overlapping primary studies. Including all systematic reviews without consideration of the likelihood of significant overlap of primary studies introduces the risk of increasing the relative weight of each primary study if it is included more than once in synthesis. This reduces trustworthiness of the final result. There is no consensus on the ideal method of managing overlap. 31 In this review, we use a hierarchical evidence approach to systematically gather data from the top tiers of the evidence. We have previously piloted this method successfully in our other reviews.32 -35 Our methods are similar to those used in other umbrella reviews. 31 Using this approach, 1 author (SBD) reviewed all studies deemed eligible for inclusion following full text review to identify the most recent and highest tier of evidence available. The hierarchical order was structured as follows: (1) Umbrella reviews; (2) Network meta-analyses; (3) Meta-analyses of double-blind, placebo-controlled RCTs; (4) Meta-analyses of RCTs; (5) Systematic reviews of RCTs.
Where multiple reviews with similar PICO criteria were identified, the review with the most recent search date was preferentially selected. In situations where both an umbrella review and systematic review and meta-analysis addressed the same clinical question, the umbrella review was prioritised for inclusion. If the systematic review and meta-analysis was more recent, the umbrella review was updated with the more recent data. Where there was uncertainty regarding the amount of overlap between reviews with a similar clinical question, we assessed primary study overlap by producing a citation matrix and calculating the Corrected Covered Area (CCA) previously described by Pieper et al. 36 Where primary study overlap between reviews was high (>15%), the most comprehensive review (ie, the review that included the most RCTs) with the most recent search date was selected. Hierarchical selection through CCA calculations was performed by 3 authors (SBD, ALM, JL). The hierarchical selection of reviews for inclusion was verified by a second author (CE).
Data Extraction
Seven reviewers (SBD, AO, JL, MI, ALM, JL, CP) independently extracted data using a predefined data extraction tool developed in Microsoft Excel. All extracted data were verified by a second reviewer. The data extraction tool encompassed the PICO of included reviews, number of trials and participants, literature search date, population details, intervention and control details, weighted or standard mean differences and confidence intervals for outcomes (where available), risk of bias assessment and Grading of Recommendations Assessment, Development and Evaluation (GRADE) 37 assessments (where available).
Quality Assessment of Included Reviews
Included reviews were assessed using the AMSTAR Version 2.0. 38 The AMSTAR-2 instrument is a validated and well-accepted measure of the quality of systematic reviews. It consists of 16 items assessing domains such as comprehensiveness of the search, having a clearly defined research question and eligibility criteria and use of appropriate risk of bias assessment. Quality assessment was performed independently in duplicate by SBD, ALM, JL, AO or MI. Disagreements were resolved via discussion and a third reviewer (CE) if required. An overall rating of review quality ranging from “critically low” to “high” was given according to the number of critical and non-critical domains met by the included review. 38
Certainty of Evidence
The GRADE approach 37 was used to assess and report the certainty of the evidence (ie, confidence in effect estimate). Where authors of the included reviews performed a GRADE assessment for outcomes using a validated tool/measure, the assessment was extracted directly from the review. Where possible, all other GRADE assessments were conducted independently by 2 reviewers (ALM, JL). Due to resource limitations, it was not possible to conduct a GRADE assessment for every outcome. Where outcomes were reported by more than 1 outcome measure, GRADE assessments were only conducted for a single outcome measure using the list of pre-defined validated tools or outcome measures, which are ordered in Table 1 in terms of clinical importance as determined by our clinician-researcher team (oncologists, breast surgeons, etc.). GRADE assessments were conducted on the most highly ranked (in terms of clinically important) outcome measure that was reported. Discrepancies were resolved by discussion. Not all reviews reported sufficient information to conduct a GRADE, and GRADEs were not possible for reviews with 1 RCT in the outcome analysis.
Results
Search Results
The search results are presented in Figure 1. After duplicates were removed, the titles and abstracts of 2756 records were screened, and 2623 records that did not meet the inclusion criteria were excluded. The remaining full text of 133 records were assessed for eligibility, and 52 records that did not meet the inclusion criteria were removed following full-text review. A total of 81 studies were considered eligible for inclusion and subject to hierarchical evidence synthesis screening. Of the 81 eligible studies, 29 were subsequently excluded after checking for overlap, leaving 52 reviews included. The excluded studies with reasons for exclusion are listed in Supplemental Table S2.
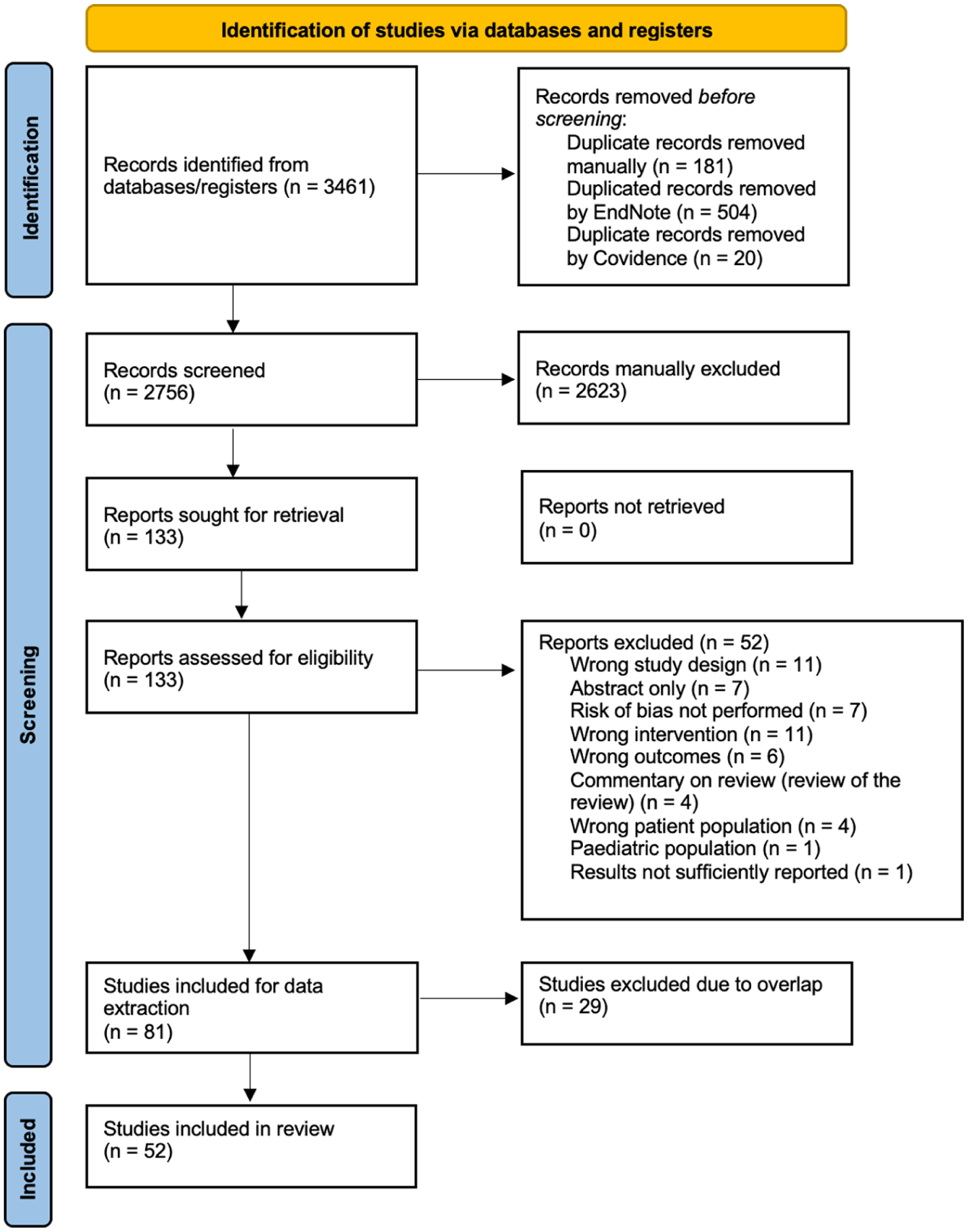
PRISMA flow chart.
Study Characteristics
We report on 250 RCTs extracted from 1 umbrella review, 2 network meta-analyses, 25 meta-analyses and 24 systematic reviews, including 2 clinical practice guidelines.39,40 Of the 52 reviews evaluating the efficacy of nutritional supplements, 17 reviews evaluated QoL,40 -56 11 reviews evaluated OM,41,45,53,56 -63 10 reviews evaluated changes in body weight,43,47,49,51,53,55,64 -67 eight reviews evaluated CIPN40,63,64,68 -72, seven reviews evaluated dermatitis,39,44,73 -77 six reviews evaluated fatigue41,50,53,62,78,79, five reviews evaluated xerostomia45,51,62,63,80, four reviews evaluated CINV48,50,56,62 and three reviews evaluated postoperative ileus.67,81,82 Other symptoms, including hot flushes, 63 lymphoedema,62,65 genitourinary complications,80,83 sleep disorders, 41 cardiotoxicity 84 cognitive changes 47 and appetite 48 were less frequently investigated. No reviews evaluating the efficacy of nutritional supplementation for fear of cancer recurrence were identified.
The characteristics table (Table 2) provides a detailed overview of all outcomes evaluated in the 52 included reviews. Eighteen reviews reported on safety and adverse events related to the nutritional supplement intervention.40,44,46,48,49,53,55,62 -64,70,76,80,85 -89 Most of the reviews included populations with various cancer diagnoses (33 reviews), seven evaluated effects in individuals with breast cancer only, seven in gastrointestinal cancers only, four in reproductive cancers only (prostate, gynaecological) and one in head and neck cancer only.
Characteristics of Included Studies.

Abbreviations: AAs, amino acids; AEs, adverse events; AIS, Athens Insomnia Scale; ALA, alpha lipoic acid; Arg, arginine; BCAA, Branch chained amino acids; BID, twice daily; BMI, body mass index; CFS, chronic fatigue scale; CFUs, colony-forming units; CI, confidence intervals; CINV, chemotherapy-induced nausea and vomiting; CIPN, chemotherapy-induced peripheral neuropathy; CoQ10, coenzyme Q10; CNS, central nervous system; CRF, cancer-related fatigue; CT, chemotherapy; CTCAE, NCI Common Terminology Criteria for Adverse Events; d, day(s); DHA, docosahexaenoic acid; EORTC, European Organisation for Research and Treatment of Cancer; EPA, eicosapentaenoic acid; FACIT, Functional Assessment of chronic illness; FACT, Functional Assessment of Cancer Therapy; FAs, fatty acids; FLIE, Functional Living Index Emesis; FOS, fructooligosaccharides; g, grams; GERD, gastroesophageal reflux disease; GI, gastrointestinal; GIQLI, Gastrointestinal Quality of Life Index; Gln, glutamine; GOS, galactooligosaccharides; HA, hyaluronic acid; HFS, hand and foot syndrome; HMB, beta-hydroxy b-methylbutyrate; HRQOL, health-related QoL; ISI, Insomnia severity index; IU, international units; KSS, Karolinska Sleepiness Scale; LOS, length of stay; MA, meta-analysis, MD, mean difference, MDASI, MD Anderson Symptom Inventory; MFI-20, Multidimensional Fatigue Inventory; mg, milligrammes; MOS, Medical outcomes study sleep survey; MPQ, McGill Pain Questionnaire; NA, not applicable; NAC, n-acetylcysteine; NCI, National Cancer Institute; nMA, network meta-analysis; NR, not reported, NS, not significant; NTX, neurotoxicity; OM, oral mucositis; OMAS, oral mucositis assessment scale; OIPN, oxaliplatin-induced peripheral neuropathy; OR, odds ratio; PAN, pancreatic cancer; PHGG, partially hydrolysed guar gum; PIPN, paclitaxel-induced peripheral neuropathy; PNP, peripheral neuropathy pain scale; PNQ, Patient Neurotoxicity Questionnaire; PSQI, Pittsburgh Sleep Quality Index; QID, four times daily; QLQ, QoL questionnaire; QoL, quality of life; RCT, randomised controlled trial; RD, radiodermatitis; RDSS, Radiation Dermatitis Severity Score; RINV, radiotherapy-induced nausea and vomiting; RR, risk ratio; RT, radiotherapy; RTOG, Radiation Therapy Oncology Group; SF-36, 36-item Short Form Health Survey; SOMA, subjective objective medical management and analytic evaluation of injury; SMD, standard mean difference; SR, systematic review; TID, three times daily; UR, umbrella review; wk, week(s); VAS, Visual Analogue Scale; WHO, World Health Organization grading system.
Amino acids or amino acid derivatives (eg, N-acetylcysteine [NAC], L-carnitine) were the most commonly investigated nutritional supplement (13 reviews),40,42,47,55,57,66,68,70 -73,76,84 followed by pre/probiotics (11 reviews)48,50,52,54,59,65,67,79,81,82,85, vitamin E (12 Reviews)40,42,44,45,57,58,63,69,76,80,83,84, fatty acids (7 reviews),43,47,51,64,70,72,89 antioxidants (eg, coenzyme Q10 [CoQ10], alpha-lipoic acid [ALA]; 5 reviews)40,42,72,76,78, vitamin A/precursors (4 reviews),45,76,83,87 and zinc (3 reviews).53,60,76 Vitamin D39,80,89, vitamin C,39,45,88 selenium45,61,80, B vitamins40,77 and magnesium 90 were also evaluated though less frequently. Other nutritional supplements included proteolytic enzymes,62,75 hyaluronic acid74,83, melatonin,41,86 curcumin46,49 and various phytochemical supplements (eg, fucoidan, adlay bran).14,42,56,76 Table 2 provides a detailed overview of all nutritional supplements evaluated.
Quality Assessment
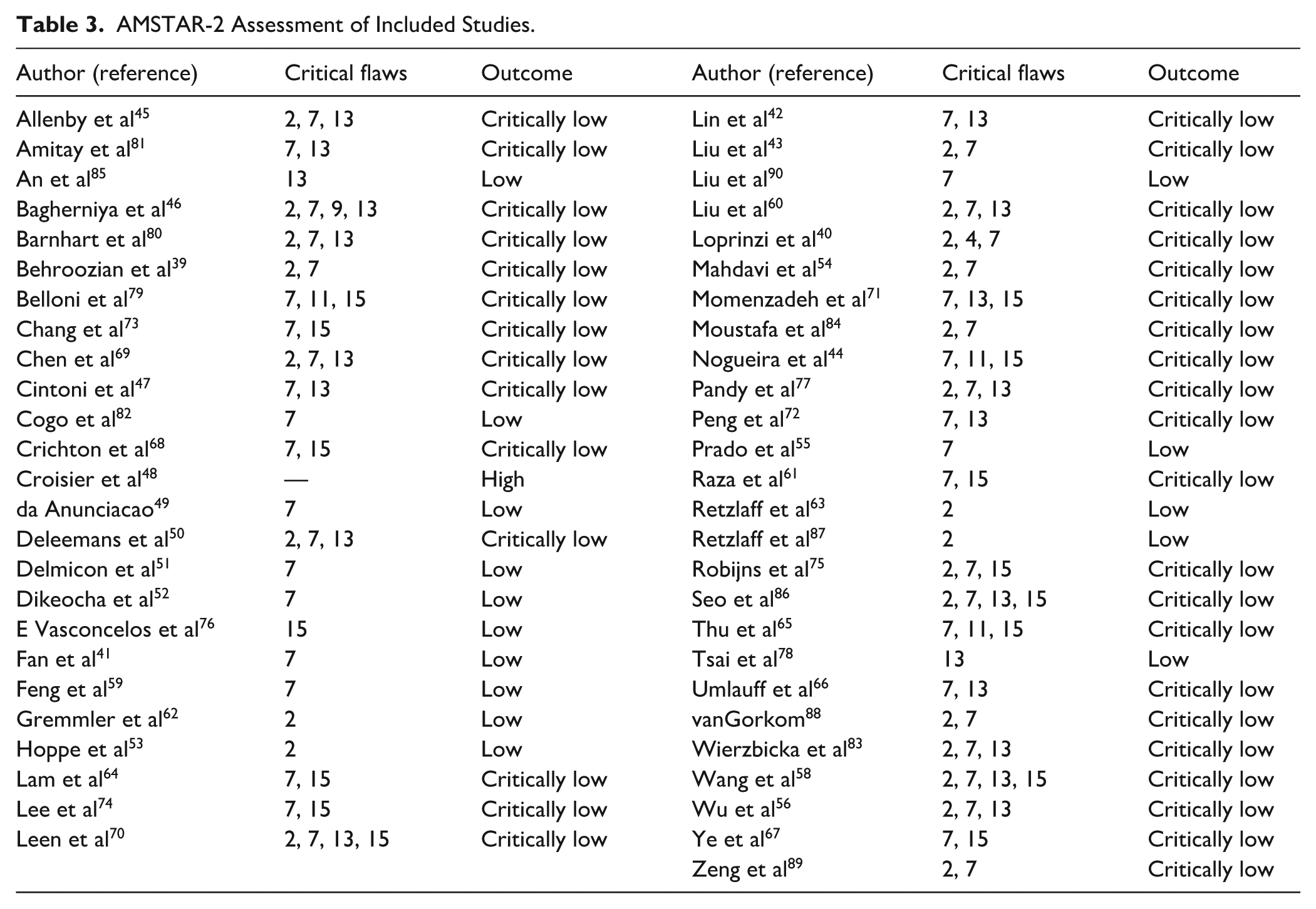
The methodological quality of the 51 systematic reviews and meta-analyses included for review was assessed (the umbrella review Khosroshahi et al 57 was not applicable). The overall quality was generally poor, with 69% (35/51) being assessed as having critically low methodological quality due to the presence of more than one critical flaw. Of the remaining reviews, 29% (15/51) were assessed as low quality, while only one review (2%, 1/51) was assessed as having high methodological quality (Table 3). This review was by Croisier et al 48 on dietary fibre/prebiotic supplementation for gynaecological cancers. 48 Common critical flaws included a lack of justification for excluding individual studies (item 7), a partially inadequate literature search strategy (item 4), absence of prospective protocol registration (item 2) and inadequate discussion of risk of bias with review findings (item 13).
AMSTAR-2 Assessment of Included Studies.
Efficacy
Body Weight
Ten reviews investigated nutritional supplement effects on body weight, including five meta-analyses43,64 -67 and five systematic reviews.47,49,51,53,55 Table 4 summarises all included reviews that reported on nutritional supplement intervention and body weight. All reviews were assessed as either having low or critically low methodological quality.
Nutritional Supplements for Nutrition-Related Side Effects.

Key: ↓ denotes significant decrease; ↑ denotes significant increase; ↔ non-significant/no difference.
Abbreviations: Arg, arginine; BCAA, Branch chained amino acids; BID, twice daily; BMI, body mass index; CFUs, colony-forming units; CINV, chemotherapy-induced nausea and vomiting; CT, chemotherapy; CTCAE, NCI Common Terminology Criteria for Adverse Events; d, day(s); DHA, docosahexaenoic acid; EORTC, European Organisation for Research and Treatment of Cancer; EPA, eicosapentaenoic acid; FAs, fatty acids; g, grams; GM-CSF, granulocyte-macrophage colony-stimulating factor; HMB, beta-hydroxy b-methylbutyrate; IU, international units; mcg, micrograms; MD, mean difference; mo, month(s); NA, not available; NCI, National Cancer Institute, Common Terminology Criteria; nMA, network meta-analysis; NS, not significant; OM, oral mucositis; OMAS, Oral Mucositis Assessment Scale; QID, four times per day; RCT, randomised controlled trial; RINV, radiotherapy-induced nausea and vomiting; RR, risk ratio; RT, radiotherapy; RTOG, Radiation Therapy Oncology Group; TID, three times per day; UR, umbrella review; WHO, World Health Organization grading system.
Original authors.
Current authors.
Weight Loss Prevention
There is inconsistent evidence for perioperative supplementation with b-hydroxy b-methylbutyrate, arginine and glutamine (HMB/Arg/Gln). 55 In individuals with various solid cancers, oral HMB/Arg/Gln supplementation may be beneficial for increasing body weight compared with isonitrogenous, isocaloric controls (2 RCTs, n = 229). 55 However, oral HMB/Arg/Gln did not have an effect on body weight in individuals with head and neck cancer with the same control (1 RCT, n = 60). 55 Interpretation of these findings of HMB/Arg/Gln is further limited by the absence of the effect estimates and confidence intervals from these RCTs within the systematic review. 55 In a single RCT (n = 72), supplementation with L-carnitine during chemotherapy was effective for preventing weight loss in individuals with pancreatic cancers compared with placebo. 47 This evidence is also limited by sample size and lack of reporting on intervention dosages, effect estimates and confidence intervals.
There was also very low certainty evidence from 2 meta-analyses43,64 and one systematic review 51 that omega 3 fatty acids (O3-FAs) supplementation is not effective for preventing weight loss during treatment in individuals with colorectal cancers (5 RCTs, n = 222), 43 breast cancer (1 RCT, n = 53) and other various cancer types (10 RCTs, n = 317). 64
Oral curcumin supplementation was also reported to demonstrate “significant positive effects for preventing weight loss” in colorectal cancer survivors. 49 However, these results were narratively synthesised, and effect estimates were not provided to support these findings.
Weight Loss Promotion
There was low certainty of evidence that probiotic supplementation is not effective for aiding weight loss in breast cancer survivors, based on pooled data from various Lactobacillus with Bifidobacterium formulations with and without prebiotics (fructooligosaccharides [FOS]; 4 RCTs, n = 298). 65 Following subgroup analysis by probiotic type, Lactobacillus with Bifidobacterium and Enterococcus (2 RCTs, n = 164) or Lactobacillus with Bifidobacterium plus a Mediterranean diet (1 RCT, n = 34; both interventions without FOS) demonstrated efficacy for reducing body weight when compared with placebo or Mediterranean diet only. 65
Supplementation with zinc 53 and protein powders, including both whey protein isolate and soy protein with isoflavones, 66 was not effective for reducing body weight in individuals with colorectal and prostate cancers compared with controls.
Cardiotoxicity
Our review identified 1 systematic review 84 (with critically low methodological quality) evaluating the impact of nutritional supplementation to ameliorate cardiotoxicity outcomes in cancer survivorship. Moustafa et al 84 investigated the prophylactic efficacy of vitamin E and levocarnitine on doxorubicin-induced cardiotoxicity. L-carnitine was reported to be “cardioprotective” compared with silymarin (1 RCT, n = 41) and vitamin E was “cardioprotective” in 3 of the 5 RCTs (n = 102) and “not cardioprotective” in 2 of the RCTs (n = 85) compared with placebo or no intervention. The efficacy of these interventions is significantly limited by the absence of validated cardiotoxic outcome measures and a lack of quantitative data regarding effect estimates and measures of variability. The authors also concluded the RCT data was ambiguous, controversial and of low quality. 84 The available details extracted from this review can be found in Table 5.
Nutritional Supplements for Reproductive and Cardiometabolic Side Effects.

Key: ↓ denotes significant decrease; ↑ denotes significant increase; ↔ non-significant/no difference.
Abbreviations: CFU, colony forming units; CT, chemotherapy; GRADE, Grading of Recommendations, Assessment, Development and Evaluation; HA, Hyaluronic acid; IU, International Units; MA, meta-analysis; NA, not applicable; NR, not reported, NS, not significant; SR, systematic review; TID, three times per day.
Original authors.
n of control group not reported for 2 RCTs. Current authors.
Chemo/Radiotherapy Induced Nausea/Vomiting (CINV/RINV)
Three systematic reviews investigated proteolytic enzymes, 62 fuciodan, 56 and pre/probiotics 50 for CINV with low or critically low methodological quality. One systematic review evaluated the effect of pre/probiotics for RINV 48 with high methodological quality. Among the evaluated interventions, there is limited evidence for the efficacy of nutritional supplementation for CINV/RINV.
The only intervention to demonstrate efficacy was a synbiotic supplement containing L. acidophilus NCFM and Bifidobacterium lactis with inulin. 50 A single RCT (n = 70) reported a significant reduction in the frequency and intensity of CINV in individuals with cervical cancer undergoing chemo/radiotherapy. 50 Pre/probiotic supplementation with lactulose and L. acidophilus-enriched yoghurt and hydrolysed rice bran, however, were not effective for RINV in individuals with gynaecological cancers undergoing radiotherapy (2 RCTs, n = 41). 48 All other interventions showed no benefits over placebo/control (Table 4).
Cognitive Changes
Evidence of the effects of nutritional supplementation on cognition was sparse (Table 6). We identified one review by Cintoni et al 47 reporting on intervention with L-carnitine for cognitive function in individuals with pancreatic cancer undergoing chemotherapy. Evidence from a single RCT (placebo-controlled, double-blinded) with 72 participants showed a significant improvement in cognitive function using validated measures (QLQ-C30, Pancreatic Cancer Module [PAN 26]). 47 The review was assessed as having critically low methodological quality.
Nutritional Supplements for Neurological and Cutaneous Side Effects.

Key: ↓ denotes significant decrease; ↑ denotes significant increase; ↔ non-significant/no difference.
Abbreviations: AAs, amino acids; AIS, Athens Insomnia Scale; Arg, arginine; BID, twice per day; CIPN, chemotherapy-induced peripheral neuropathy; CT, chemotherapy; CTCAE, Common Terminology Criteria for Adverse Events; EORTC, European Organisation for Research and Treatment of Cancer; FACT, Functional Assessment of Cancer Therapy; Gln, glutamine; GRADE, Grading of Recommendations, Assessment, Development and Evaluation; HA, Hyaluronic acid; HFS, hand and foot syndrome; HMB, beta-hydroxy b-methylbutyrate; ISI, Insomnia severity index; KSS, Karolinska Sleepiness Scale; MA, meta-analysis; MOS, Medical outcomes study sleep survey; NA, not applicable; NAC, n-acetylcysteine; NCI, National Cancer Institute; NMA, network meta-analysis; NR, not reported; NS, not significant; O3-FA, Omega-3 fatty acids; OIPC, oxaliplatin-induced peripheral neuropathy; PIPN, paclitaxel-induced peripheral neuropathy; PNP, Peripheral Neuropathic Pain; PNQ, Patient Neurotoxicity Questionnaire; PSQI, Pittsburg Sleep Quality Index; RT, radiotherapy, RTOG, Radiation Therapy Oncology Group; SR, systematic review; TID, three times per day.
Original authors.
Current authors.
Dermatitis
Seven reviews evaluated nutritional management for treatment-induced dermatitis (Table 6). There were 4 meta-analyses44,73 -75, one clinical practice guideline 39 and 1 systematic review on radiation-induced dermatitis (RD) 76 and one meta-analysis on chemotherapy-induced skin reactions, 77 with all reviews being assessed as having critically low or low methodological quality. There is limited evidence for the efficacy of nutritional supplements to improve dermatitis outcomes. Glutamine and proteolytic oral enzymes demonstrated some evidence of effectiveness for RD outcomes in the included reviews. There is limited evidence that zinc and adlay bran are effective for reducing RD severity and inconsistent evidence on the efficacy of topical hyaluronic acid (HA). There were no effective nutritional supplement interventions for chemotherapy-induced skin reactions.
The amino acids HMB/Arg/Gln may be effective for reducing the overall incidence of RD (RTOG/CTCAE) compared with placebo or no treatment (5 RCTs, n = 218) with very low certainty. 73 There was moderate certainty that glutamine or HMB/Arg/Gln reduces the incidence of moderate-severe RD (RTOG/CTCAE; OR 0.49, CI 0.32-0.76, I2 = 52%, 5 RCTs, n = 218). 73 In individuals with head and neck or cervical cancer, there is low certainty that oral enzymes (papain, trypsin, chymotrypsin) may reduce the incidence of moist desquamation and severity of RD compared with no treatment with pooled data from 2 RCTs (n = 219). 75
A 2022 meta-analysis by Lee et al 74 evaluated the efficacy of topical HA compared with active controls in individuals with breast cancer. When compared with phytosterols (2 RCTs, n = 80), vitamin E (1 RCTs, n = 40), and O3-FAs (1 RCT, n = 40), topical HA was more effective in reducing the incidence of RD, however a GRADE assessment was not possible due to the use of outcome measurement scales which were not included in our pre-defined criteria. Topical HA did not demonstrate superior efficacy when compared with simple emollients or grapevine extract. 74
Oral supplementation with zinc and adlay bran extract capsules may also be effective interventions for reducing the severity of RD. There is limited evidence that adlay bran (1 RCT, n = 110) and zinc (1 RCT, n = 100) may be effective for reducing RD severity (RTOG) in breast and head and neck cancer compared with placebo and olive oil capsules. 76
A number of nutritional supplement interventions were not effective for dermatitis. There was very low certainty that pyridoxine is not effective for reducing the incidence of dermatitis compared with placebo, no treatment and active controls (8 RCTs, n = 661). 77 There is low and very low certainty that neither oral nor topical curcumin are effective for reducing moist desquamation incidence or RD severity in individuals with breast and head and neck cancers compared with placebo controls and simple emollients. 75 Oral vitamin E is not effective for preventing fibrosis in individuals with breast cancer when compared with placebo or no treatment (2 RCTs, n = 64), 44 with very low certainty. Intervention with vitamin E combined with vitamin A, 76 anthocyanins, 76 vitamin D 39 and vitamin C 39 also did not demonstrate efficacy.
Fatigue
Six reviews (2 meta-analyses41,78 and 4 systematic reviews50,53,62,79) reported on nutritional supplementation to manage cancer-related fatigue (CRF; Table 7). Nutritional supplements generally did not demonstrate efficacy for CRF. There is limited evidence from 1 systematic review that pre/probiotics may improve fatigue scores. 79 There was no evidence from available meta-analyses demonstrating efficacy against control/placebo.41,78 All reviews were assessed as having low or critically low methodological quality.
Nutritional Supplements for QoL and Fatigue.

Key: ↓ denotes significant decrease; ↑ denotes significant increase; ↔ non-significant/no difference.
Abbreviations: Arg, arginine; BID, twice per day; CFS, chronic fatigue scale; CFUs, colony-forming units; CI, confidence intervals; CoQ10, coenzyme Q10; CT, chemotherapy, d, day(s); CTCAE, NCI Common Terminology Criteria for Adverse Events; DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid; EORTC QLQ-C30, European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire; EORTC QLQ-CR29, QoL questionnaire for colorectal cancer; FAs, fatty acids; FACIT, The Functional Assessment of Chronic Illness Therapy-Fatigue; FACT, Functional Assessment of Cancer Therapy; FOS, fructooligosaccharides; GIQLI, Gastrointestinal Quality of Life Index; Gln, glutamine; HMB, beta-hydroxy b-methylbutyrate; MA, meta-analysis; MD, mean difference; MDASI, MD Anderson Symptom Inventory; MFI-20, Multidimensional Fatigue Inventory; mg, milligrammes; NA, not available; NR, not reported; NS, not significant; O3-FAs, omega-3 fatty acids; PHGG, partially hydrolysed guar gum; QLQ, QoL questionnaire; QoL, quality of life; RCT, randomised controlled trial; RT, radiotherapy; SF-36, 36-item Short Form Health Survey; SMD, standard mean difference; SR, systematic review; TID, three times daily; VAS, Visual Analogue Scale.
Original authors.
Current authors.
Pre/probiotics may be effective for reducing CRF scores in breast and colorectal cancers compared with placebo 79 as found in 3 RCTs (n = 280) evaluating diverse pre/probiotic formulations. One RCT demonstrating efficacy evaluated a combined intervention with probiotics and O3-FAs, however, the baseline fatigue scores of the intervention group were significantly higher than the control group, limiting the interpretation of this evidence (n = 140). 79
All other evaluated interventions did not demonstrate efficacy against controls, including a meta-analysis on melatonin. 41 Melatonin is not an effective intervention for CRF (5 RCTs, n = 855) 41 with very low certainty. Zinc, 53 proteolytic enzymes 62 and coenzyme Q10 (CoQ10) 78 were also not effective for improving CRF.
Genitourinary Complications
Two systematic reviews,80,83 with critically low methodological quality, evaluated the efficacy of nutrients for genitourinary complications in individuals with breast and cervical cancers (Table 5). Vaginal suppository intervention with a combination of HA, vitamin A and vitamin E resulted in statistically significant reductions in vaginal dryness, dyspareunia, vulvar fibrosis and mucosal inflammation among individuals with cervical cancer compared with control. 83 However, the nature of the controls was not reported, and the evidence is limited to 2 RCTs (n = 222). 83 Intervention with vitamin D or vitamin E resulted in a significant reduction in self-reported genitourinary symptoms in individuals with breast cancer compared with placebo. 80 This evidence is also limited to a single RCT (n = 96), and the details of the intervention (dose, duration) were not reported. 80
Hot Flushes
One network meta-analysis 90 and 1 systematic review 63 evaluated the incidence and severity of hot flashes in individuals with breast cancer (Table 5). The reviews were assessed as having critically low and low methodological quality. None of the evaluated interventions demonstrated efficacy. Supplementation with magnesium oxide (1 RCT, n = 358) 90 and vitamin E (1 RCT, n = 125) 63 showed no significant difference compared with placebo.
Insomnia/Sleep Disorders
We identified 1 meta-analysis, with low methodological quality, evaluating melatonin for sleep quality in individuals with various cancer diagnoses. 41 Melatonin does not improve sleep quality compared with placebo and active controls with data pooled from 9 RCTs (n = 1128) 41 with very low certainty. The effects remained nonsignificant following subgroup analyses according to intervention duration, dosage and treatment type 41 (Table 6).
Lymphoedema
We identified 1 meta-analysis on probiotics 65 and 1 systematic review on proteolytic enzymes 62 that evaluated effects on lymphoedema in individuals with breast cancer (Table 5). The reviews were assessed as having low and critically low methodological quality. There were no interventions that demonstrated efficacy for improving lymphoedema outcomes.
Supplementation with probiotics Lactobacillus, Bifidobacterium and Streptococcus (species not reported) alone or combined with FOS were not effective for reducing oedema volume compared with placebo (2 RCTs, n = 223). 65 Intervention with proteolytic enzymes also did not show any significant differences in arm volume, skin fold thickness or skin tension compared with placebo (1 RCT, n = 88). 62
Mucositis
Eleven reviews reported on OM outcomes following nutritional supplement interventions (1 umbrella review, 57 1 network meta-analysis, 58 4 meta-analyses,41,59 -61 and 5 systematic reviews45,53,56,62,63 (Table 4). Several interventions demonstrated promise for reducing the incidence and/or severity of OM during chemo/radiotherapy, including glutamine, 57 melatonin, 41 probioitics 59 and zinc. 60 There is limited evidence for supplementation with proteolytic enzymes 62 and inconsistent evidence for vitamin E.58,63 The meta-analyses and systematic reviews were assessed as having low or critically low methodological quality.
There was low certainty that glutamine supplementation is effective for reducing the severity OM during radiotherapy compared with placebo or no treatment with pooled data from 16 RCTs (n = 1199). 57
There was very low certainty evidence that melatonin supplementation is not effective for reducing the severity of stomatitis (3 RCTs, n = 154), nor was it effective for reducing the overall incidence of stomatitis (7 RCTs, n = 832). 41 However, melatonin was found to be effective for reducing incidence of stomatitis when individuals with head and neck cancer were excluded in a subgroup analysis (4 RTCs, n = 674). 41
There is very low certainty evidence that zinc supplementation, encompassing various dosage forms, may be effective in preventing OM compared with placebo (12 RCTs, n = 783). 60 Following subgroup analysis by cancer location, zinc was not effective for reducing the incidence of OM in individuals with head and neck cancer (6 RCTs, n = 339). 60
Three reviews evaluated the efficacy of vitamin E.57,58,63 In a network meta-analysis by Wang et al 58 vitamin E mouthwash was superior to placebo (1 RCT, n = 59) and showed comparable efficacy to the other active treatments (eg, sodium bicarbonate, natural drugs; Table 4) for reducing the severity of OM in individuals with head and neck cancer (13 RCTs, n = 570). Topical application of vitamin E paste was also shown to be superior to both oral vitamin E supplementation and placebo paste for reducing the severity of OM after cycles 3 and 4 of chemo/radiotherapy in haematological cancer (1 RCT, n = 67). 63 Oral vitamin E supplementation is effective for reducing OM incidence 57 (2 RCTs, n = 72) with low certainty. A single RCT (n = 535) reports that beta-carotene “reduces mucositis” in head and neck cancer, 45 however, the dosage and data to support this conclusion was not provided.
Among the other evaluated supplements, probiotic supplementation covering a diverse range of strains and dosage forms (eg, lozenges, Yakult) was effective for reducing the overall incidence of OM during chemotherapy compared with placebo or usual care 59 (4 RCTs, n = 412). The authors, however, did not find any significant effects on OM severity (7 RCTs, n = 647). 59 Oral proteolytic enzymes demonstrated efficacy for reducing the overall severity of OM in individuals with head and neck cancers compared with placebo in 1 review (3 RCTs, n = 208). 62
Fucoidan 56 and selenium 61 did not demonstrate efficacy for OM outcomes.
Neuropathy
We identified 8 reviews (6 meta-analyses64,68 -72 and two systematic reviews40,63) reporting on nutritional supplements to manage CIPN (Table 6). Vitamin E, glutamine, O3-FAs and NAC may reduce the incidence of CIPN however, the evidence was very uncertain. There is limited evidence that supplementation with various amino acids/amino acid derivatives, vitamin E and a B vitamin complex may reduce severity of CIPN. All reviews were assessed as having critically low to low methodological quality.
Vitamin E supplementation may be effective for reducing the incidence, but not severity, of CIPN (8 RCTs, n = 307) 69 with very low certainty. The type of control was also not reported. 69 In a systematic review by Retzlaff et al, 63 compared with no treatment, vitamin E was significantly associated with a reduction in both the incidence and severity of CIPN and showed significant differences in sensory nerve function (2 RCTs, n = 62).
O3-FA supplementation may reduce the likelihood of developing CIPN by 80% compared with placebo across various cancer diagnoses (3 RCTs, n = 158, OR: 0.20, 95% CI: 0.10-0.40, P < .00001) 64 with very low certainty.
There is very low certainty evidence that glutamine may reduce the incidence of CIPN (1 RCT, n = 86) 68 and low certainty evidence that it may reduce severity (grade 3-4; 1 RCT, n = 86), 72 when compared with standard care and non-active controls. Among the other amino acid/derivative interventions, there was moderate certainty that supplementation with NAC (2 RCTs, n = 46) and low certainty that L-carnosine (2 RCTs, 121) may be effective for reducing the severity of CIPN in individuals with gastrointestinal cancers compared with placebo or non-active controls. 72 However, in another single RCT, supplementation with NAC did not prevent CIPN in individuals with breast cancer (1 RCT, n = 65). 70 Cysteine and theanine supplementation reduced incidence of CIPN in gastrointestinal cancer (1 RCT, n = 28) 72 with very low certainty.
Finally, supplementation with a B vitamin complex was associated with significant improvements in patient perceived sensory CIPN after 36 weeks of treatment (1 RCT, 71), however there were no differences in the overall incidence of CIPN in various cancer types. 40
Other interventions with acetyl-L-carnitine,40,71 glutamate, 70 and alpha-lipoic acid 40 did not reduce incidence or severity of CIPN.
Postoperative Ileus
We identified 2 meta-analyses67,81 and one systematic review 82 evaluating perioperative probiotic supplementation for the prevention of postoperative ileus in individuals with gastrointestinal cancers (Table 4). All the reviews were assessed as having critically low or low methodological quality.
There is very low certainty that probiotics may reduce time to flatus (7 RCTs, n = 693) 67 and time to first stool (4 RCTs, n = 398). 81 The systematic review by Cogo et al 82 included 7 RCTs evaluating post-operative ileus with inconsistent findings across the interventions. The inconsistency is related to the diversity of the probiotic interventions; this heterogeneity precluded the authors from performing a meta-analysis. 82 The details of the probiotic interventions are shown in Table 4.
Quality of Life
Seventeen reviews reported on the efficacy of nutrient supplementation for improvements in QoL including 4 meta-analyses41 -44 and 13 systematic reviews40,45 -56 (Table 7). Overall, perioperative pre/probiotic supplementation may improve QoL in individuals with colorectal cancer. There was less evidence for amino acids/amino acid derivatives, combined nutritional and phytochemical supplementation and O3-FAs in various cancer types. All included reviews on QoL were assessed as having critically low or low methodologic quality, apart from Croisier et al 48 evaluating inulin and FOS in gynaecological cancer which was assessed as high quality.
There is limited and inconsistent evidence on pre/probiotics for improving QoL. Two systematic reviews found significant improvements in QoL in individuals with colorectal cancer using pre/probiotic interventions, irrespective of strain and formulation, when compared with placebo or no treatment (5 RCTs, n = 410).50,52 Prebiotic FOS-enriched jelly (1 RCT, n = 32) was also found to be effective for maintaining QoL (Gastrointestinal Quality of Life Index [GIQLI]) in individuals with head and neck cancer compared with placebo. 50 However, supplementation with synbiotics, a combination of pre and probiotics, 54 and Lactobacillus plantarum 50 were not effective for improving QoL in individuals with colorectal cancer. Lactobacillus acidophilus combined with Bifidobacterium longum, 50 prebiotic fibres including partially hydrolysed guar gum (PHGG), 50 inulin and FOS 48 did not demonstrate efficacy against control across individuals with various cancer diagnoses and treatment schedules.
A single RCT evaluating the amino acid derivative, L-carnitine (n = 72) demonstrated efficacy for improving QoL compared with placebo during chemotherapy in individuals with pancreatic cancer. 47 A combined nutritional supplement (b-carotene, vitamin C, vitamin E, and selenium) may significantly improve QoL in individuals with cervical cancer undergoing chemoradiotherapy (1 RCT, n = 103), 45 while CoQ10 and melatonin, 41 vitamin E 44 and zinc 53 did not demonstrate efficacy.
O3-FA supplementation was reported to significantly improve QoL in individuals with breast cancer (1 RCT, n = 44), 51 however was not effective in colorectal cancer (3 RCTs, n = 332) 43 or mixed cancer types (none in breast cancer; 6 RCTs, n = 33).
A range of other evaluated nutritional supplement interventions did not demonstrate efficacy for QoL. Pooled data from trials evaluating L-carnitine, chlorella, coenzyme Q10 with vitamin E, creatine monohydrate and kefir showed no significant differences in QoL. 42 Fucoidan, 56 melatonin 41 vitamin E 44 zinc 53 and prebiotics, inulin and FOS 48 did not demonstrate efficacy against control.
Xerostomia
Five systematic reviews45,51,62,63,80 reported on xerostomia outcomes following nutritional supplementation (Table 4). Interventions included proteolytic enzymes 62 (1RCT), O3-FAs 51 (1 RCT), selenium 80 (1 RCT) and vitamin E alone63,80 (2 RCTs) or in combination with vitamin C 45 (1 RCT). All of the evaluated nutritional supplements demonstrated efficacy for improving xerostomia,45,51 dysphagia 62 or salivary gland function outcomes.63,80 This evidence, however, is limited to data reported from single RCTs with small sample sizes and deficient reporting on point estimates and measures of variability. All the reviews were assessed as having low or critically low methodological quality.
Supplementation with O3-FAs in breast cancer (1 RCT, n = 52) 51 and vitamin C with vitamin E in head and neck cancer (1 RCT, n = 52) 45 during chemotherapy were associated with significant improvements in xerostomia compared with placebo controls. In individuals with head and neck cancer, proteolytic enzymes also showed a significant reduction in dysphagia during radiotherapy compared with placebo (1 RCT, n = 100). 62 In individuals with thyroid cancer, vitamin E supplementation during chemo/radiotherapy (2 RCTs, n = 118)63,80 and selenium during treatment with radioactive iodine were associated with significant improvements in salivary gland function compared with placebo (1 RCT, n = 16). 80
Safety and Adverse Events
Eighteen of the included reviews reported on nutritional supplement-related adverse events AEs, including 5 meta-analyses44,64,70,85,86 and 13 systematic reviews40,46,48,49,53,55,62,63,76,80,87 -89 (Table 8). Among the reviews that reported AEs, serious side effects (grade 3 or 4) were consistently reported for interventions with high-dose vitamin A, while AEs of zinc and vitamin E supplementation were varied. In a 2020 systematic review by Retzlaff et al 87 oral vitamin A doses of 50 000, 100 000 and 300 000 IU/day were associated with a higher incidence of mucocutaneous side effects including skin rashes and desquamation, hepatic side effects including raised liver enzymes and increased triglycerides, and grade 3 or 4 gastrointestinal side effects, and bone pain compared with placebo controls (4 RCTs, n = 3335). Yellowing of the skin, bone pain, depression, diarrhoea and mucositis were associated with 50 mg and 75 mg beta-carotene intervention doses compared with placebo (n = 2 RCTs, 378). 87 Yellowing of the skin was also reported following a placebo-controlled intervention with 300 mg beta-carotene and 400 IU vitamin E (1 RCT, n = 79). 76 In a 2021 systematic review by Hoppe et al, 53 135 mg of zinc/day was associated with an increased incidence of moderate to severe dysphagia (1 RCT, n = 159), increased mild gastrointestinal side effects were reported in 3 additional RCTs (n = NR) while 7 other RCTs and 1 meta-analysis within the review found there were no side effects or differences between groups following zinc supplementation. 53
Nutritional Supplement Safety and Adverse Events.

Abbreviations: AE, adverse events; Arg, arginine; BID, twice per day; CLL, Chronic lymphocytic leukaemia; CT, chemotherapy; d, day; DHA, docosahexaenoic acid; EPA, eicosapentaenoic acid; Gln, glutamine; HMB, beta-hydroxy b-methylbutyrate; PUFA, polyunsaturated fatty acid; TID; three times a day; MA, meta-analysis; n, number; NAC, N-acetylcysteine; NMA, network meta-analysis; NR, not reported; SR, systematic review; PTX, pentoxifylline; RCT, randomised controlled trial; RT, radiotherapy; TID, three times per day.
Three reviews reported on AEs to vitamin E.40,44,63 Two reviews (5 RCTs, n = NR) reported there were no differences between groups with intervention doses between 300 and 600 mg/day.40,63 Conversely, a systematic review and meta-analysis by Nogueira et al 44 identified a higher incidence of mild gastrointestinal, haematological, and neurological side effects with doses between 300 and 1000 mg vitamin E/day compared with placebo or no intervention (3 RCTs, n = 160).
Minimal mild side effects, primarily gastrointestinal origin, were reported for supplementation with adlay bran, 76 selenium, 80 curcumin 46 , HMB/Arg/Gln, 55 NAC, 70 prebiotics, 48 vitamin C, 88 and proteolytic enzymes. 62 Melatonin supplementation was associated with headaches, fatigue and bad dreams in 2 of 4 included RCTs that reported on AEs in a systematic review by Seo et al. 86
No differences between groups for AEs were reported for supplementation with anthocyanins, 76 fucoidan, 56 O3-FAs, 64 vitamin B3, 89 vitamin D 89 or pro/synbiotics. 85 There is low certainty evidence that perioperative pro/synbiotics supplementation in individuals with colorectal cancer is associated with a 5% minimal clinically important difference in AEs compared with placebo or standard care in perioperative colorectal cancer (7 RCTs, n = 692). 85 All data extracted on safety and AEs to the nutritional supplement interventions are shown in Table 8.
Discussion
To our knowledge, this is the first umbrella review to comprehensively collate and evaluate the evidence on the use and safety of nutritional supplements in supportive cancer care. We synthesised the findings from 52 reviews covering a wide range of cancer and treatment-related effects, including any reported adverse events associated with the supplement intervention. Overall, most included reviews were of low or critically low methodological quality. While some interventions suggested benefit, evidence was largely uncertain or very uncertain, and a number of interventions did not suggest benefit, including for QoL.
We report that amino acids 73 and oral proteolytic enzymes 75 may reduce the incidence and severity of RD. Certainty of evidence varied from very low certainty for HMB/Arg/Gln for incidence to moderate certainty for severity of RD. 73 Supplementation with exogenous enzymes may accelerate skin barrier repair in conditions such as epidermal hyperplasia 91 and their use as a cosmetic application for skin renewal have gained attention. 92 Our review updates evidence from the MASCC clinical practice guidelines for RD which did not recommend glutamine due to insufficient evidence of benefit. 39 Although our updated evidence is based on a greater number of RCTs (5 RCTs vs 2 RCTs in the MASCC guidelines) we note that evidence remains limited and inconsistent, with different studies reporting both harm and benefit for glutamine supplementation, 75 and therefore that strong clinical recommendations cannot be made at the present time.
Our review updates the older evidence on O3-FAs for prevention of weight loss, cited in the European Society for Clinical Nutrition and Metabolism (ESPEN) clinical practice guidelines for nutrition in cancer. 93 The ESPEN guidelines cited 2 systematic reviews published in 2007 and 2015 and made a weak recommendation for O3-FAs to stabilise or improve appetite, food intake, lean body mass and body weight in individuals with advanced cancer undergoing chemotherapy and at risk of weight loss or malnutrition. Our review considers updated evidence from 2 more recent meta-analyses43,64 and 1 systematic review 51 that report that O3-FAs are not effective for preventing weight loss in individuals with colorectal cancer, breast cancer and other cancer types, and includes new evidence from 7 additional RCTs. However, given this evidence was rated as very low certainty due to imprecision, risk of bias and inconsistency, we are uncertain as to the true effect of O3-FAs on prevention of weight loss. We note, as do the authors of the ESPEN guidelines, that there are no significant safety concerns with O3-FA administration and the balance of benefit versus harm may be in favour of O3-FAs.
Oral mucositis remains a debilitating and dose-limiting side effect of treatment. Glutamine, 57 zinc, 60 probiotics 59 and melatonin 41 may be effective for reducing the incidence of OM while glutamine may also reduce severity, 57 however the certainty of evidence was low to very low. Melatonin, a hormone with potent free radical scavenger with antioxidant and anti-inflammatory properties, 94 may protect against chemo/radiotherapy-induced mucosal damage and support mucosal regeneration. 94 However, the evidence suggests its efficacy is limited to non-head and neck cancers. 41 Glutamine is the most abundant amino acid well-recognised for its indispensable role in rapidly proliferating cells, 95 while zinc is a trace element abundant in the skin and necessary to maintain the physiological functions. 96 Our review updates the recommendations in the 2020 MASCC/ISOO Clinical Practice Guidelines for OM, 97 which was based on older evidence (search date June 2016). Our findings concur with and strengthen the evidence that supports the use of oral glutamine (doses 10-30 mg/day) for individuals with head and neck cancer. 97 In the same guidelines, zinc was no longer recommended for prevention due to inconsistent and insufficient evidence, 98 and our review similarly concurs with these recommendations with regard to head and neck cancer, finding that there is no evidence for benefit in subgroup analysis. 60 However, in our review, a pooled meta-analysis of all cancer types suggested very low certainty evidence of potential benefit for zinc, with particular promise for leukaemia and pharyngeal cancers in subgroup analyses. 60 The use of zinc for OM in individuals with these cancers should be further evaluated.
Chemotherapy-induced peripheral neuropathy is a dose-limiting toxicity with limited treatment options. 99 We found very low certainty evidence that vitamin E, O3-FAs and amino acids may provide potential protective effects for CIPN,40,44,63,64,69 with no serious side effects reported for either intervention. Our findings concur with the ASCO 2020 guidelines that were unable to make recommendations as to nutrient supplements for prevention or management of CIPN. Although we present updated evidence, including moderate certainty evidence for potential benefit of NAC in gastrointestinal cancer, 72 this evidence remains limited and sparse. Further research is required to determine the neuroprotective effect of these supplements, especially given the dearth of options available to individuals experiencing CIPN.
At the time of planning our umbrella review, guidelines on management of CRF had not yet been published, and therefore, this symptom was included in our synthesis. Our findings complement those reported in the recently published ASCO-SIO guideline update on the management of fatigue in adult cancer survivors. 25 Unlike our review, the ASCO-SIO guideline is based on a synthesis of individual RCTs, rather than of systematic reviews or meta-analyses. We also report on some nutritional supplements not included in the ASCO-SIO guideline, including pre/probiotics50,79 and zinc. 53 Taken together, our findings and those in the ASCO-SIO guideline do not suggest a benefit from nutritional supplements (including co-enzyme Q10) for managing CRF. 100 Given the demonstrated effectiveness of other interventions, such as exercise, cognitive-behavioural therapy and mind-body therapies for fatigue during and after treatment, these approaches should be recommended instead of nutritional supplements for management of this debilitating condition. 100
The safety of nutritional supplements during and after cancer treatment is a key concern of clinicians and their patients. Adverse events were inconsistently reported across the included reviews. However, where reported, they were generally minor and severe AEs were rare. Additionally, AEs tended to be dose-related. High-dose vitamin A was associated with significant toxicity (50 000, 100 000 and 300 000 IU/d), 87 and high doses of zinc (≥135 mg/d) led to severe dysphagia, increased nausea and vomiting. 53 These side effects are consistent with other pharmacological doses of zinc, which exceed the recommended tolerable Upper Intake Level of 40 mg/d, across studies in other patient populations where nausea, vomiting, epigastric pain, lethargy and fatigue were reported with doses ranging from 100 to 300 mg/d. 101 Importantly, however, and not captured by this review, are any disturbances in mineral absorption with high doses of zinc. Zinc interacts with mineral absorption, competitively inhibiting calcium, manganese, copper, selenium and iron in high doses for extended durations, with iron and copper most concerning.101,102 Copper deficiency has been associated with zinc doses of >50 mg/d for >10 months. 101 This highlights the need to evaluate the risk-benefit profile while also considering treatment duration and ongoing mineral status with pharmacological doses of zinc.
A key challenge of using nutritional supplementation in supportive cancer care is the balance between benefit and harm, in particular, the potential for interference with the intended effect of curative treatment. 103 For example, NAC increases cellular levels of cysteine and glutathione, restoring the antioxidant potential of cells by replenishing glutathione. 104 During radiotherapy, this is counter-productive to the intended effect of treatment with radiotherapy and may have the potential to reduce the efficacy of radiotherapy. These key clinical concerns should be addressed with improved reporting of AEs and longer-term follow-up of participants to determine any potential impact on efficacy of curative treatments.
Implications for practice: Strong clinical recommendations are not possible due to the overall uncertainty of the evidence. At the present time, we suggest that clinicians discuss the direction of evidence and the uncertainties that preclude strong recommendations and assist their patients to make decisions informed by this evidence.
Implications for research: There is a significant gap in research on debilitating conditions such as cognitive impairment, that should be addressed. There are promising findings for a number of interventions such as glutamine and other amino acids, zinc and vitamin E. However, findings are consistently downgraded for concerns such as risk of bias in included RCTs, imprecision due to small sample sizes and small numbers of trials, and inconsistency due to significant heterogeneity. There is a need for a collaborative, systematic and standardised approach to evaluating the potential efficacy of nutrient supplements in supportive care in cancer. Standardisation of dose and delivery type should be achieved so that findings are comparable across studies, and heterogeneity is minimised. Additionally, evaluation of the impact of different doses should be incorporated in order to determine the minimum effective dose. Comprehensive and longer-term evaluation of adverse events is essential. Researchers should also aim to use core outcome sets where available. Lastly, factors affecting implementation of the use of nutrient supplements in clinical practice should also be evaluated.
This umbrella review has several strengths, among which is the methodology encompassing inclusion of the most up-to-date literature, selection of only the highest levels of evidence available and the broad scope of outcomes examined. A rigorous process was followed to eliminate or minimise overlap. Only reviews reporting a relevant quality assessment were included for review, although the quality assessment for each review was not presented. We used validated appraisal tools (AMSTAR-2, GRADE) and prioritised outcomes with validated measures. However, the strength of the umbrella review hinges on the quality and rigour of the included studies, most of which were of low methodological rigour. Whilst the AMSTAR-2 quality appraisal tool considers causes of heterogeneity, the use of sensitivity analysis in meta-analyses and the impact of publication bias in all included reviews, these assessments were not presented individually. Our search was restricted to articles available in English, and due to resource limitations, grey literature and non-randomised trials were excluded, which may have resulted in a less comprehensive review. Due to the breadth of the outcomes, GRADE assessments were not feasible for all reported outcomes. Finally, heterogeneity in dosages, formulations, probiotics strains and numbers, treatment duration and control limit the synthesis of findings.
Conclusion
Cancer survivors, depending on the patient population, stage of disease and cultural background, are highly motivated to use nutritional supplements to manage side effects, improve QoL and provide a sense of autonomy and self-advocacy. 105 This evidence synthesis serves to fill an important gap in supportive cancer care by equipping healthcare providers with the best available evidence to clarify misconceptions, guide evidence-based shared decision-making and overcome the historic and persistent lack of disclosure to healthcare providers regarding supplement use. 105 Despite the generally low risk of adverse effects with nutritional supplements, the low to very low certainty of the majority of evidence limits the ability to make firm clinical recommendations. Moving forward, the provision of high-quality, evidence-based, patient-centred care requires larger, high-quality RCTs that include patient-reported outcomes as well as clinician-reported outcomes. These studies should assess the potential impact, positive or negative, of nutritional supplements on cancer treatment.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251405267 – Supplemental material for The Efficacy and Safety of Nutritional Supplements for Cancer Supportive Care: An Umbrella Review and Hierarchical Evidence Synthesis
Supplemental material, sj-docx-1-ict-10.1177_15347354251405267 for The Efficacy and Safety of Nutritional Supplements for Cancer Supportive Care: An Umbrella Review and Hierarchical Evidence Synthesis by Sarah Benna-Doyle, Suzanne Grant, Alison Maunder, Jing Liu, Melik Ibrahim, Adele Cave, Chhiti Pandey, Monica Tang, Eng-Siew Koh, Geoff Delaney, Deep Jyoti Bhuyan, Victoria Choi, Ki Kwon, Maria Gonzalez, Susannah Graham, Ashanya Malalasekera and Carolyn Ee in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354251405267 – Supplemental material for The Efficacy and Safety of Nutritional Supplements for Cancer Supportive Care: An Umbrella Review and Hierarchical Evidence Synthesis
Supplemental material, sj-docx-2-ict-10.1177_15347354251405267 for The Efficacy and Safety of Nutritional Supplements for Cancer Supportive Care: An Umbrella Review and Hierarchical Evidence Synthesis by Sarah Benna-Doyle, Suzanne Grant, Alison Maunder, Jing Liu, Melik Ibrahim, Adele Cave, Chhiti Pandey, Monica Tang, Eng-Siew Koh, Geoff Delaney, Deep Jyoti Bhuyan, Victoria Choi, Ki Kwon, Maria Gonzalez, Susannah Graham, Ashanya Malalasekera and Carolyn Ee in Integrative Cancer Therapies
Footnotes
Acknowledgements
We gratefully acknowledge the assistance of Allana O’Fee and Atekah Albrehee with project management.
Author’s Note
Monica Tang is now affiliated to Prince of Wales Hospital, Sydney, Australia; The George Institute for Global Health, Sydney, Australia; School of Clinical Medicine, University of New South Wales, Sydney, Australia
Ethical considerations
Not applicable.
Author contributions
Conceptualisation, CE, SG(Suzanne Grant), SBD, MT, ESK, GD; Data curation, CE, SBD, ALM (Alison Maunder), JL, MI, AC, CP; Writing-original draft, SBD, CE; Writing- reviewing & editing, SBD, CE, SG(Suzanne Grant), ALM, JL, MI, AC, CP, MT, ESK, GD, DP, VC, KK, MG, SG (Susannah Graham), AM (Ashyana Malasekera). All authors read and agreed on the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a philanthropic donation by an anonymous donor to Western Sydney University. The donor had no input into the design of the study, interpretation of the findings or preparation of the manuscript for publication. Open access publication fees were provided by Western Sydney University.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: CE declares that she is the Jacka Foundation Principal Research Fellow, Chair of the RACGP Integrative Medicine Specific Interest Network (voluntary role), Programme Lead of an academic integrative healthcare centre (no financial interest), past GP Advisory Board member for Blackmores Research Institute, has received industry funding from nutraceutical and acupuncture device companies to conduct clinical trials, and has received honoraria and had travel expenses covered for presenting at complementary medicine events. As a medical research institute, NICM Health Research Institute receives research grants and donations from foundations, universities, government agencies, and industry. Sponsors and donors provide untied and tied funding for work to advance the vision and mission of the Institute. ALM declares that she is a naturopathic practitioner at a clinic in Sydney, Australia. She is the recipient of a scholarship from the Jacka Foundation of Natural Therapies for her PhD. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data availability statement
Data are available from the authors upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
