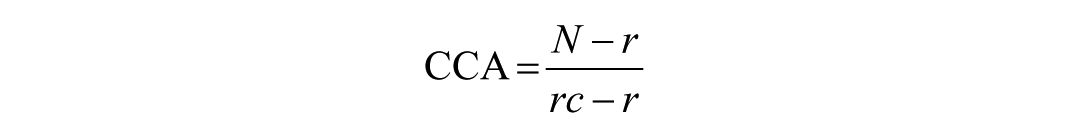Abstract
Objective:
To evaluate the effectiveness and safety of Shenyi Capsule (SC) combined with conventional anticancer therapies through a comprehensive assessment of systematic reviews/meta-analyses (SRs/MAs), identify limitations in current evidence, and provide evidence-based guide for clinical practice and research.
Methods:
Registered in PROSPERO (CRD42024573610), this study searched 8 Databases from inception to November 10, 2024. Two separate reviewers were responsible for selecting studies, collecting data, assessing quality, and evaluating the potential for bias, with this research incorporating only SRs/MAs based on randomized controlled trials and utilizing 4 tools (AMSTAR-2, PRISMA 2020, ROBIS, GRADE) to evaluate methodological quality and risk of bias.
Results:
Thirteen SRs/MAs were included, focusing on 4 cancer types. AMSTAR-2 evaluations revealed no high quality studies. Compliance with PRISMA 2020 guidelines was below 50% for most items, with major deficiencies including lack of protocol registration, incomplete abstracts and search strategies, inappropriate or missing risk-of-bias assessments, insufficient descriptions of data transformation methods, absence of heterogeneity and sensitivity analyses, and failure to report funding sources. ROBIS indicated high bias risk in 11 studies. GRADE assessment of 109 outcomes showed evidence quality as moderate (40.37%), low (34.86%), or very low (24.77%).
Discussion:
SC may enhance efficacy and safety as an adjuvant cancer therapy. However, methodological flaws in existing SRs/MAs limit reliability. Future research should prioritize standardized SR/MA protocols and RCT designs to improve evidence quality, reduce bias, and strengthen clinical applicability.
Introduction
In 2022, approximately 20 million new cancer cases were documented worldwide, and the count of cancer-associated deaths hit 9.7 million. 1 The elevated incidence and fatality rates of cancer place a substantial load on patients and their families while presenting a formidable challenge to global public health systems. Although conventional anticancer treatments such as chemotherapy, radiotherapy, and targeted therapy remain the cornerstone of cancer management, their adverse effects and limited efficacy have driven researchers to explore combination treatment strategies.
In China, the application of traditional Chinese medicine (TCM) for cancer treatment ranks among the most frequently employed therapeutic approaches. Some plant-derived compounds have been validated through pharmacological studies as promising new cancer therapies. For instance, Icaritin, a compound containing icariin, has been recommended for advanced hepatocellular carcinoma and is applicable regardless of liver function classification. 2 Shenyi Capsule (SC) is a preparation containing Ginsenoside Rg3, a monomeric saponin derived from ginseng. As the first Class I TCM monotherapy anticancer drug, its antitumor effects have been preliminarily confirmed. 3 Ginsenoside Rg3 wields its anti-cancer impacts via multiple mechanisms. These encompass curbing the proliferation and dissemination of cancer cells, triggering cancer cell apoptosis, modulating non-coding RNAs, facilitating mitochondrial autophagy, and impeding the growth of tumor-related endothelial cells. Additionally, its diverse immunomodulatory properties suggest that SC may be a promising therapeutic agent.4,5 In clinical practice, SC has demonstrated significant benefits as an adjuvant therapy in various solid tumors, including lung cancer, esophageal cancer, breast cancer, and liver cancer. It enhances chemotherapy sensitivity while alleviating adverse effects.4,6 In 2016, SC was initially included in the suggestions put forward by the National Comprehensive Cancer Network. 7
Although SC has demonstrated therapeutic potential for use as an adjunct in cancer treatment, the numerous SRs/MAs assessing its efficacy and safety display marked disparities in quality. Moreover, a holistic assessment of the overall evidence is still absent. Thus, it is of the utmost urgency to conduct a methodical and scientific assessment of the existing studies to determine the real worth of SC in the context of cancer treatment.
This overview aims to comprehensively evaluate the efficacy and safety of SC in combination with conventional anticancer therapies in cancer patients by assessing existing SRs/MAs using Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020, A MeaSurement Tool to Assess systematic Reviews 2 (AMSTAR-2), the Grades of Recommendation, Assessment, Development and Evaluation (GRADE), and the Risk of Bias in Systematic Reviews (ROBIS) frameworks. By identifying the limitations of current SRs/MAs, this study seeks to provide directions for future high-quality clinical research and offer evidence-based guidance for clinicians and patients regarding SC combination therapy.
Methods
This research has been registered in PROSPERO (Identifier: CRD42024573610). The review was carried out in accordance with the PRISMA 2020 statement 8 and the Cochrane Handbook for Systematic Reviews of Interventions. 9
Search Strategy
Searches were conducted in 6 English databases PubMed (1996-2024.11.10), Embase (1974-2024.11.10), Cochrane Library (1996-2024.11.10), Epistemonikos (2025.07.25), NIHR (1997-2025.07.25), and Web of Science (1997-2024.11.10) as well as 4 Chinese databases CNKI (1984-2024.11.08), VIP (1989-2024.11.08), Wanfang (1980-2024.11.08), and CBM (1994-2024.11.08). Moreover, manual searches were carried out for conference proceedings and gray literature, along with reference tracing for publications that met the full-text screening criteria. There were no limitations regarding the language of the studies.
For example, the search strategy for Embase was executed through the official website (https://embase.wscience.org/), covering literature from 1974 to the present, with searches conducted on November 10, 2024.
#11 #3 AND #7 AND #10
#10 #8 OR #9
#9 (“analysis, meta”:ti,ab,kw OR metaanalysis:ti,ab,kw OR “meta analysis”:ti,ab,kw OR “system review”:ti,ab,kw OR meta:ti,ab,kw) AND [01-01-1974]/sd NOT [11-11-2024]/sd
#8 (“meta analysis”/exp OR “meta analysis”) AND [01-01-1974]/sd NOT [11-11-2024]/sd
#7 #4 OR #5 OR #6
#6 (“shenyi capsule”/exp OR “shenyi capsule”) AND [01-01-1974]/sd NOT [11-11-2024]/sd
#5 (“shenyi”/exp OR “shenyi”) AND [01-01-1974]/sd NOT [11-11-2024]/sd
#4 (“ginsenoside rg 3”/exp OR “ginsenoside rg 3”) AND [01-01-1974]/sd NOT [11-11-2024]/sd
#3 #1 OR #2
#2 (“acral tumor”:ti,ab,kw OR “acral tumour”:ti,ab,kw OR neoplasia:ti,ab,kw OR neoplasms:ti,ab,kw OR “neoplastic disease”:ti,ab,kw OR “neoplastic entity”:ti,ab,kw OR “neoplastic mass”:ti,ab,kw OR tumor:ti,ab,kw OR “tumoral entity”:ti,ab,kw OR “tumoral mass”:ti,ab,kw OR “tumorous entity”:ti,ab,kw OR “tumorous mass”:ti,ab,kw OR tumors:ti,ab,kw OR tumour:ti,ab,kw OR “tumoural entity”:ti,ab,kw OR “tumoural mass”:ti,ab,kw OR “tumourous entity”:ti,ab,kw OR “tumourous mass”:ti,ab,kw OR tumours:ti,ab,kw OR neoplasm:ti,ab,kw OR cancer:ti,ab,kw OR cancers:ti,ab,kw OR “malignant neoplasm”:ti,ab,kw OR malignancy:ti,ab,kw OR malignancies:ti,ab,kw OR “neoplasm, malignant”:ti,ab,kw OR “malignant neoplasms”:ti,ab,kw) AND [01-01-1974]/sd NOT [11-11-2024]/sd
#1 “neoplasm”/exp AND [01-01-1974]/sd NOT [11-11-2024]/sd
The Supplemental Materials contain the search tactics for the remaining databases.
Inclusion Criteria and Exclusion Criteria
Inclusion Criteria
Study Type: SRs/MAs based on randomized controlled trials (RCTs).
Study Population: Individuals who have received a cancer diagnosis via pathological assessment. There are no limitations regarding age, sex, ethnicity, geographical location, or the disease stage.
Interventions: SC as a single variable. The experimental group receives SC plus conventional antitumor therapy, while the control group receives conventional therapy alone.
Outcomes: Primary outcomes: Short-term efficacy, including objective response rate (ORR) or disease control rate (DCR); long-term efficacy, including overall survival (OS) or overall survival rate. Additional outcome: Safety, assessed by different adverse events (AEs). Language: No language restrictions.
Exclusion Criteria
Network meta-analyses. Qualitative systematic reviews. Articles that do not provide full text or complete extractable data.
Study Selection and Data Extraction
The retrieved studies were imported into Zotero 7.0 and Covidence. After processing duplicates through manual verification and Covidence identification, 2 authors separately screened all titles and abstracts, eliminating studies that were irrelevant, and finally determined the inclusion of literature through full-text review. The full-text screening exclusion criteria focused on 3 aspects: studies employing non-meta-analytic methodologies, uncontrolled single-arm studies or those with incompatible control groups, and studies whose population profiles, intervention strategies, or outcome measures deviated from the PICOS framework. Each exclusion required dual independent adjudication by researchers with archived rationale, culminating in PRISMA diagrammatic representation. Any differences in judgment were settled through consultations with a third researcher to achieve an agreement. Data extraction was independently performed by 2 investigators using a standardized Excel form to collect the following information: first author’s name, publication year, language, cancer type, quality assessment, trial design, sample size, clinical stage, funding source, registration status, intervention details, and outcome measures.
Assessing the Overlap of Primary Research
Systematic reviews may include primary studies that overlap, which could introduce bias due to inflated statistical weight. To address this issue, the corrected covered area (CCA) can be used as a quantitative metric to assess the extent to which original studies have been included more than once. 10
N refers to the total number of publications, including duplicate counts, and represents the number of marked boxes in the citation matrix. R refers to the number of rows in the citation matrix, representing the number of RCTs and c refers to the number of columns, representing the number of systematic reviews. CCA values of 0 to 5 indicate low overlap, 6 to 10 indicate moderate overlap, 11 to 15 indicate high overlap, and values greater than 15 indicate extremely high overlap. 10 This grading system enables researchers to accurately evaluate the degree of overlap in the literature between different systematic reviews. Overlapping systematic reviews were managed by the Cochrane Handbook for Systematic Reviews of Interventions. 9 All Cochrane and non-Cochrane systematic reviews were included. If 2 or more systematic reviews show high overlap (CCA ≥ 11%), the following were prioritized: systematic reviews with the highest methodological quality as assessed by AMSTAR, the largest number of original studies, and the most recent systematic reviews. If 2 or more systematic reviews show mild or moderate overlap (CCA ≤ 10%), all are retained.
Quality Assessment
Two independent researchers conducted the quality assessment. The AMSTAR 2 tool was used to evaluate the methodological quality of the included SRs/MAs. This tool consists of 16 items, with responses categorized as “Yes” (Y), “Partially Yes” (PY), or “No” (N). Studies were classified as follows: High quality: No or only 1 non-critical item rated as “Partially Yes” or “No.” Moderate quality: More than 1 non-critical item rated as “Partially Yes” or “No.” Low quality: One critical item rated as “No,” with or without deficiencies in non-critical items. Critically low quality: More than 1 critical item rated as “No,” regardless of non-critical items. 11
The PRISMA 2020 guideline was utilized to gauge the reporting standard of the SRs/MAs incorporated in this research. This guideline consists of 7 main elements and 27 secondary aspects, which enables an examination of compliance. In particular, for every one of these elements and aspects, a rating of “Yes” or “No” is assigned. This approach permits a comprehensive appraisal of the extent to which the requirements of the guideline are met. 8
The ROBIS instrument was employed to evaluate the bias risk in the incorporated SRs/MAs. ROBIS is composed of 3 distinct domains. The first domain focuses on determining whether the research question of the systematic review corresponds to the intended question. The second domain is made up of 4 components. These components are the eligibility requirements, the process of study identification and selection, data gathering, and the synthesis or findings. The third domain gauges the overall bias risk in the synthesis. For each domain, a rating of “Yes,” “Likely Yes,” “Likely No,” “No,” or “No Information” is assigned. Subsequently, the studies are categorized as having a high, low, or unclear bias risk. 12
Data Synthesis and Analysis
A descriptive assessment was carried out on the results from the integrated studies. Subsequently, a database was created using Excel 2016. As per the methodology outlined in the Cochrane Handbook for Systematic Reviews of Interventions, 9 visual representations were employed to present a summary of the features of the included systematic reviews. Moreover, these graphs also showed the results of the meta-analysis or descriptive analysis for each outcome metric.
Results
Overview of Literature Search
By implementing the search approach, a grand total of 140 research inquiries were discovered. After removing 52 duplicate records, 88 studies remained. Of the screened records, 69 underwent exclusion after title and abstract review, and an additional 6 were excluded after a thorough full-text analysis, leaving 13 studies for inclusion,13 -25 published between 2009 and 2022. Details are presented in Figure 1.
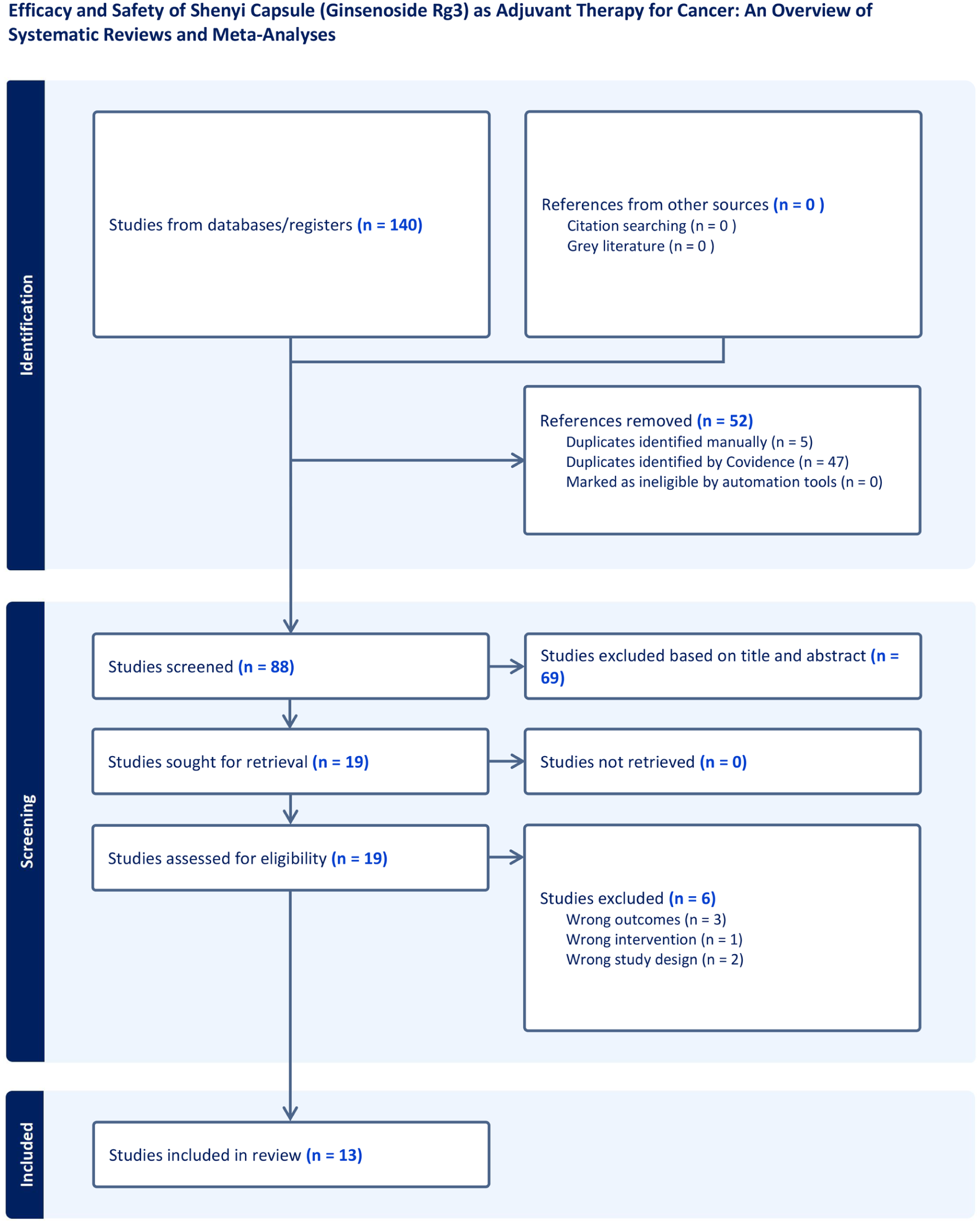
Literature selection process.
Study Characteristics
All authors were from China. A total of 13 SRs/MAs were collected, including 5 English-language articles and 8 Chinese-language articles, published between 2009 and 2022. These investigations encompassed anywhere between 4 and 27 clinical trials. The number of participants in each sample varied from 274 to 2663. A total of 98 primary studies with 7511 patients were included. The analysis incorporated 60 RCTs on lung cancer with 5369 participants (intervention group: n = 2748; comparator group: n = 2621). For breast cancer, 4 RCTs were included with balanced group allocation (137 participants per group). Twenty-five RCTs focused on gastrointestinal tumors (intervention group: n = 549; comparator group: n = 531), while 8 hepatocellular carcinoma RCTs involved 788 patients (experimental group: n = 433; comparator group: n = 355). When it came to evaluating the methodological quality, 10 SRs/MAs adhered to the Cochrane criteria. Two research undertakings utilized the Jadad scale, and 1 incorporated both the Cochrane criteria and the Jadad scale. The clinical characteristics are summarized as follows: Cancer Types: SC, as the first Class I single-component Chinese medicine for cancer treatment, primarily focused on lung cancer (9 SRs/MAs), gastrointestinal tumors (2 SRs/MAs), liver cancer (1 SR/MA), and breast cancer (1 SR/MA). Clinical Stages: SC can be considered for use at various clinical stages. Five SRs/MAs focused on advanced-stage patients, 3 did not limit the cancer stage, and 5 did not provide stage information. Treatment Approaches: SC, as an adjunctive treatment, may be used post-surgery or in combination with chemotherapy, transcatheter arterial chemoembolization (TACE), radiotherapy, and targeted therapies. However, there was heterogeneity in the primary outcome measures across different SRs/MAs. Specific details can be found in Table 1.
The Attributes of Relevant SRs/Mas.

TG, treatment arm; CG, control arm; Rg3, Shenyi capsule; TACE, transcatheter arterial chemoembolization; A, overall response rate; B, disease control rate; C, 6-month survival rate; D, 1-year survival rate; E, 2-year survival rate; F, 3-year survival rate; G, median survival rate; H, overall survival; I, Karnofsky Performance Status; J, Decreased VEGF expressions; K, CD4+/CD8+ T Cells level improvement; L, CD3+ T Cells level improvement; M, Leukopenia; N, Grade 3-4 leukopenia; O, thrombocytopenia; P, anemia; Q, Gastrointestinal Reactions; R, liver injury; S, radiological esophageal injury; T, AFP decline rate; U, nausea and vomiting; V, constipation; W, The hepatic and renal dysfunction; X, peripheral nerve toxicity; Y, fatigue; Z, Weight Change; AA, Myelosuppression; AB, Alopecia.
# p > 0.05.
The Degree of Overlap Between Reviews
The citation matrix was generated from references of the 13 included SRs/MAs since no Cochrane reviews were incorporated in this study, with a calculated CCA of 9% indicating moderate overlap, leading to the retention of all SRs/MAs while warranting cautious interpretation of the results. Subgroup analyses demonstrated considerable variation in overlap degrees across cancer types, revealing extremely high overlap for both lung cancer (CCA = 19%) and gastrointestinal malignancies (CCA = 19%), but low overlap for liver cancer (CCA = 0%) and breast cancer (CCA = 0%).
Quality Assessment of the Reviews
Utilizing the AMSTAR-2 framework for evaluation, the 13 SRs/MAs were classified according to quality as follows: 7 were found to be of extremely poor quality, 4 as low quality, and 2 as moderate quality. The main issues identified during the evaluation include: Q2: A significant portion of the SRs and MAs failed to pre-register their research plans in either PROSPERO or the Cochrane Library before commencing the study. Q3: Nearly all authors did not explain why they included only RCTs. Q4: The search for grey literature, clinical trials, or study registration platforms was neglected. Q5: The process of literature screening conducted independently by 2 reviewers was not described. Q6: Hu et al’s study 25 lacked a description of the data extraction process. Q7: The literature screening process was unclear in some studies, with some only providing screening results and not mentioning a screening checklist. Q8: Xu and Zhang’s study 13 did not use tables to describe trial characteristics in detail, and 8 SRs/MAs did not report the dosage information for interventions. Q9: The Jadad scale employed by Xu and Zhang 13 and Hu et al 25 failed to conduct a comprehensive evaluation of the bias risk. Q10: The funding sources were not revealed in 6 SRs/MAs. Q12: The potential influence of the bias risk on the evidence synthesis was not evaluated by 4 SRs/MAs. Q13: During the discussion segment, several studies failed to consider the impact of bias risk on the overall outcome. Q14: 5 studies did not investigate the causes of heterogeneity. Q15: 4 studies did not conduct a systematic inspection of publication bias. Q16: 9 studies did not clearly disclose conflicts of interest or funding sources. More comprehensive details can be found in Figure 2.

The assessment of AMSTAR-2.
Figure 3 presents the detailed reporting quality of the 13 studies, showing significant variation in compliance with the items. For some items, compliance was below 50%. The main defects are as follows: Abstract Section: All SRs/MAs (Q2, 0%) did not fully comply with the PRISMA 2020 Summary List. This non-compliance stems from 2 main problems. First, no summary of the limitations of the evidence included in the review was given. Second, there is no registered name and registration number. Methodology Section: Compliance with Q7 and Q13c was only 23.08%, indicating that most studies did not fully present their database search strategy and results visualization. Q8 compliance was 30.77%, reflecting a severe lack of transparency in the screening process. No study (Q13b, 0%) described how missing statistics and data synthesis were handled. Only 1 study (Q15, 7.69%) used the GRADE tool, highlighting the lack of quantitative evaluation of evidence quality in most studies. Results Section: No SR/MA (Q20a, 0%) disclosed the bias risk in the overall outcomes. Conversely, just 1 study (Q22, 7.69%) evaluated the evidence quality for the meta-analysis results. Some SRs/MAs (Q20d, 46.15%) did not report sensitivity analysis results, although the research methods were mentioned in the methodology section. Other Information: No SR/MA mentioned the registration name and number or stated that the protocol had not been developed or revised (Q24a, Q24b, Q24c, 0%), although 2 studies were registered in PROSPERO. Compliance with Q26 and Q27 was 38.46 and 30.77%, respectively, indicating that more than half of the studies did not adequately disclose potential conflicts of interest and data acquisition methods.

The assessment of PRISMA.
In accordance with the guidelines of the ROBIS tool, an evaluation was carried out on the 13 studies. It was determined that these studies presented a low risk in Domain 1 and also in the initial and second phases of Domain 2. In Domain 2, because of incomplete searches, 11 studies were categorized as having a high risk. Regarding Domain 3, 5 studies were recognized as high-risk, and in Domain 4, 8 studies were designated as high-risk. Eventually, during the final assessment of the third stage, 11 studies were placed in the high-risk group. All the comprehensive details are presented in Figure 4.

The assessment of ROBIS.
Quality of Evidence Evaluation
In the course of the GRADE evaluation, it was discovered that the 13 SRs/MAs jointly incorporated a total of 109 outcome measures. Out of these, 44 outcome measures (equivalent to 40.37%) were classified as having moderate-quality evidence, 38 (34.86%) as having low-quality evidence, and 27 (24.77%) as having very low-quality evidence. The primary elements leading to the reduction in the evidence quality were the bias risk (which occurred in 109 instances), inconsistency (29 instances), imprecision (38 instances), and publication bias (26 instances). More comprehensive details are presented in Figure 5.

A systematic evidence landscape of SC as adjuvant therapy for solid tumors.
Outcome Measures Evaluation
Demarcated in Figure 5 (segment A-B), the short-term effect revealed that 11 out of 12 items (5 with moderate quality, 6 with low quality) reported ORR, P < .05. Eight items (5 with moderate quality, 1 with low quality, 2 with very low quality) reported DCR, P < .05. As shown in Figure 5 (segment C-H), the 1-year OS (P < .05) was observed in 7 included studies, comprising 3 of moderate quality and 4 of low quality, based on long-term outcome assessments. The 2-year OS (P < .05) was observed in 4 included studies, comprising 2 of moderate quality and 2 of low quality. One study of low quality documented 3-year OS, P < .05. The remaining 3 studies pertaining to OS were assessed as low-quality evidence: 1 study reported 3-year OS (P < .05), another documented median OS (P < .05), and a third confirmed OS (P < .05).
For quality of life, 11 items (5 with moderate quality, 5 with low quality, 1 with very low quality) reported improvement in the Karnofsky Performance Status (KPS), P < .05. Regarding VEGF expression, 6 items (2 with moderate quality, 4 with very low quality) reported a reduction in VEGF expression, P < .05. For cancer markers, 1 item, rated as moderate quality, reported a decline in AFP rate, P < .05. Regarding body weight parameters, 1 moderate-quality study demonstrated improvement in weight change, P < .05.
For immune response, 2 of 3 items (1 with moderate quality, 1 with low quality) reported improvement in CD4+/CD8+ T Cells levels, P < .05. One item, rated as very low quality, reported improvement in CD3+ T Cells levels, P < .05. For safety evaluation, 11 items reported leukopenia (8 items: 4 with moderate quality, 1 with low quality, and 3 with very low quality), P < .05. Of these, 2 items (1 with moderate quality) reported Grade 3 to 4 leukopenia, P < .05. For thrombocytopenia, 4 of 7 items (3 with moderate quality, 1 with low quality) reported thrombocytopenia, P < .05. Two of 7 items (moderate quality) reported anemia, P < .05. Two items (1 with moderate quality, 1 with low quality) reported myelosuppression, P < .05. Among 12 items, 7 (5 with moderate quality, 1 with low quality, 1 with very low quality) reported gastrointestinal adverse reactions, including nausea and vomiting, radiation esophageal injury, and gastrointestinal reactions, P < .05. Seven items reported liver and kidney damage, but P-values were all greater than 0.05. Three items reported other adverse reactions, including peripheral nerve toxicity, fatigue, and alopecia, but P-values were all greater than .05. These results suggest that compared to traditional treatments alone, SC combined with traditional treatment improves efficacy and reduces related adverse effects caused by traditional treatments. However, the quality of the evidence was, in general, assessed to be moderate.
Figure 5 illustrates that a large quantity of the evidence pertains to non-small cell lung cancer (NSCLC). SC holds the promise of enhancing both short-term and long-term treatment effectiveness. However, there is currently no analysis of the evidence concerning the 5-year survival rate. For breast cancer, SC does not significantly improve short-term efficacy in breast cancer patients, and its evidence lacks statistical significance. Regarding safety, SC shows improvements in reducing adverse reactions caused by traditional treatments, such as blood toxicity, gastrointestinal reactions, and radiation esophageal injury. However, no positive evidence was found regarding its effects on reducing liver and kidney damage, peripheral nerve toxicity, hair loss, and fatigue.
Discussion
SC, as the first traditional Chinese medicine type I single-agent anticancer drug, can be combined with chemotherapy or other modern antitumor therapies for various cancer types. 3 Multiple meta-analyses have shown that combined treatments significantly improve patients’ clinical conditions. Nonetheless, basing clinical applications solely on the outcomes of individual clinical trials or systematic reviews comes with substantial drawbacks. This occurs because these investigations are frequently restricted by factors such as sample magnitude, research approaches, or external credibility. To more scientifically and comprehensively assess the efficacy and safety of interventions, constructing a comprehensive evidence map is crucial. Although a previous study conducted an overlapping meta-analysis and systematic review on SC combined with chemotherapy for treating NSCLC, the included studies were limited in number, restricted to only NSCLC, and used the Jadad scale for methodological quality evaluation without conducting bias risk assessment. Only 1 high-quality review, which included more RCTs, was selected for result analysis, so the true extent of SC’s effects has not yet been fully revealed. Therefore, this review included 13 SRs/MAs covering lung cancer, liver cancer, breast cancer, and gastrointestinal tumors. The conclusions of the included systematic reviews almost universally suggest that SC, as an adjunctive therapy for cancer, has significant advantages in both efficacy and safety.
The findings from the AMSTAR-2 assessment indicate that among the 13 articles, not a single one was of high quality. Specifically, 7 studies are extremely low quality, 4 were of low quality, and 2 attained moderate quality ratings. The PRISMA 2020 statement evaluation reveals that the majority of the SRs/MAs analyzed lacked important information, with over half of the items having a completion rate of less than 50%, suggesting poor methodological quality. Consequently, the results of these systematic reviews may not be comprehensive or accurate. According to the ROBIS bias risk evaluation, the comprehensive bias risk level of the 11 SRs/MAs analyzed was deemed high risk, indicating that the included literature might have significant bias, reducing the confidence in the results. Further analysis shows that SC combined with traditional treatment generally outperforms traditional treatment in short-term and long-term efficacy, KPS scores, and immune function. It also better suppresses VEGF and AFP expression and reduces adverse reactions, particularly hematological toxicity and gastrointestinal reactions. Nonetheless, the majority of the evidence indicates a low degree of certainty. Moreover, the quality of the systematic reviews regarding SC as an adjuvant therapy for cancer continues to be substandard. As a result, the credibility of their outcomes is called into question.
The reliability of systematic reviews is closely related to their production processes, and improving methodological quality is crucial for enhancing the translational value of the results. The 13 SRs/MAs showed clear deficiencies in the following areas: (a) They were not pre-registered on relevant platforms. (b) The scope of literature searches did not include grey literature, and search strategies were vague or incomplete, with no clear reasons for excluding certain studies. (c) Literature screening, data extraction, bias assessment, and outcome handling were non-standardized. (d) Sources of heterogeneity were not explored in-depth, and the robustness of the data was not assessed. (e) In certain research endeavors, upon incorporating primary studies that were highly likely to be biased, a full-fledged evaluation of how this bias could affect the overall combined outcome was not carried out. (f) Funding sources were not transparently disclosed.
Given these issues, future researchers should write protocols in advance and upload them to relevant platforms to ensure transparency of the research plan, reduce selective reporting bias, and improve the reproducibility of the results. A comprehensive search strategy should be developed, including multiple databases (covering grey literature and unpublished research), specifying the search dates for each source. The search strategy should follow PRISMA guidelines, using flowcharts to show the selection process and reasons for exclusions to improve reproducibility and comprehensiveness. Furthermore, researchers should use a standardized data extraction form, conduct independent dual-screening of literature, and assess bias with a standard approach to ensure consistency and accuracy. All included original studies should undergo systematic bias risk assessment using the Cochrane tool, with detailed reporting on how bias affects the overall effect. Furthermore, when heterogeneity is apparent, a random effects model ought to be employed in conjunction with subgroup analysis or meta-regression to investigate the sources of the heterogeneity. Alternatively, studies that have a substantial impact can be removed. Although a large number of studies do not carry out sensitivity analysis because of the restricted number of RCTs or low levels of heterogeneity, the absence of sensitivity analysis impedes the interpretation and utilization of the results. Sensitivity analysis methods, such as excluding low-quality studies, changing statistical models, or using the trim-and-fill method, should be considered. If a bias risk assessment is conducted, it should include a detailed report on how bias impacts the combined effect. The protocol number and funding sources should be clearly stated in the abstract or at the end of the article. Implementing these measures will effectively mitigate methodological inconsistencies and avoid reporting bias, advancing evidence-based medical research to higher standards.
Strengths and Limitations
This inquiry offers a thorough and methodical assessment of the effectiveness and safety of SC when utilized as an adjunctive treatment for cancer. Moreover, it establishes an evidence-based framework to back its clinical use and direct subsequent clinical inquiries. Nevertheless, it is crucial to recognize that this research has several drawbacks: Limited Scope of Cancer Types: Most of the included studies focused on lung cancer, with fewer studies involving other cancer types. This limits the generalizability of the conclusions. Future SRs/MAs should incorporate more types of cancer and treatment regimens to comprehensively assess SC’s efficacy and safety in various clinical contexts. Lack of 5-Year Survival Data: At present, the incorporated SRs/MAs do not offer an assessment of the 5-year survival rate. This lack of analysis restricts the capacity to gauge the long-term effectiveness of SC therapy. Future studies should include survival rates over longer periods to provide more robust insights into its long-term effects. Data Overlap: There is a potential issue with data overlap, which may lead to an overestimation of effect sizes. This needs to be addressed by properly assessing the uniqueness of included data and avoiding duplicate reporting. Subjectivity in Quality Evaluation: The quality assessment results are subject to the researchers’ subjective judgment, which means there may be variability in the evaluation outcomes between different researchers. This could potentially introduce bias and affect the reliability of the conclusions. These limitations reduce our confidence in the study’s conclusions.
Conclusion
There is evidence suggesting that SC has the potential to be an effective and safe adjuvant treatment for cancer, especially for lung cancer. It may enhance immune responses, reduce VEGF expression, mitigate treatment-related adverse effects, and prolong survival. However, existing systematic reviews and meta-analyses exhibit methodological limitations in protocol registration, bias assessment, and transparency. Therefore, future researchers should focus on conducting more large-scale, scientifically rigorous, and feasible original clinical studies. Additionally, those conducting systematic reviews should aim to produce high-quality SRs/MAs to further enhance the reliability of these conclusions.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251396519 – Supplemental material for Efficacy and Safety of Shenyi Capsule (Ginsenoside Rg3) as Adjuvant Therapy for Cancer: An Overview of Systematic Reviews and Meta-Analyses
Supplemental material, sj-docx-1-ict-10.1177_15347354251396519 for Efficacy and Safety of Shenyi Capsule (Ginsenoside Rg3) as Adjuvant Therapy for Cancer: An Overview of Systematic Reviews and Meta-Analyses by Caixian Wu, Peicong Li, Huiling Yang, Yongchun Zou, Pengrong Hu, Tiantian Yang, Yuetong Zhong, Wanyin Wu, Shunqin Long and Xiaobing Yang in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354251396519 – Supplemental material for Efficacy and Safety of Shenyi Capsule (Ginsenoside Rg3) as Adjuvant Therapy for Cancer: An Overview of Systematic Reviews and Meta-Analyses
Supplemental material, sj-docx-2-ict-10.1177_15347354251396519 for Efficacy and Safety of Shenyi Capsule (Ginsenoside Rg3) as Adjuvant Therapy for Cancer: An Overview of Systematic Reviews and Meta-Analyses by Caixian Wu, Peicong Li, Huiling Yang, Yongchun Zou, Pengrong Hu, Tiantian Yang, Yuetong Zhong, Wanyin Wu, Shunqin Long and Xiaobing Yang in Integrative Cancer Therapies
Footnotes
Ethical Considerations
As this study is an overview of systematic reviews and meta-analyses, it synthesizes data exclusively from previously published literature. No original human or animal experiments were conducted, and no individual patient data were collected, analyzed, or disclosed. Therefore, ethical approval and informed consent were not required for this research.
Author Contributions
Caixian Wu: Conceptualization, Methodology, Investigation, Data curation, Cross-verification, Writing—original draft, Visualization. Peicong Li: Software, Formal analysis, Investigation, Cross-verification, Writing—original draft. Yongchun Zou: Formal analysis, Data Extraction, Cross-verification, Writing—original draft. Huiling Yang: Data Extraction, Investigation, Cross-verification, Visualization. Pengrong Hu: Investigation, Writing—original draft, Visualization. Tiantian Yang: Formal analysis, Writing—original draft, Visualization. Yuetong Zhong: Investigation, Writing—original draft, Visualization. Wanyin Wu: Writing-review & editing, Project administration, Funding acquisition. Shunqin Long: Data curation, Supervision. Xiaobing Yang: Conceptualization, Cross-verification, Final Manuscript Approval, Project administration, Funding acquisition.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by grants from the Key Project of State Key Laboratory of dampness syndrome of Chinese medicine jointly built by the province and Ministry (SZ2021ZZ38). The funders had no role in the study design, data analysis, or decision to publish.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.