Abstract
Background:
Systematic reviews (SRs) and meta-analyses (MAs) for the use of acupuncture for cancer pain have been increasing, but the evidence has not been systematically and comprehensively assessed. We aimed to perform an overview of the evidence quality of SRs/MAs of acupuncture for improving cancer pain.
Methods:
8 databases were systematically searched to identify SRs/MAs of acupuncture for improving cancer pain. The A Measurement Tool to Assess Systematic Reviews 2 (AMSTAR-2), Risk of Bias in Systematic Reviews (ROBIS), Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), and Grades of Recommendations, Assessment, Development, and Evaluation (GRADE), respectively, were applied by 2 independent reviewers to evaluate the methodological quality, risk of bias, reporting quality, and evidence quality.
Results:
A total of 14 SRs/MAs were included in the present study. By AMSTAR-2, two reviews were rated as having high methodological quality, while 12 were given a critically low rating. All SRs/MAs in Phase 1, Domain 1, and Domain 4, according to ROBIS, were at low risk. Furthermore, 4 reviews in Domain 2, twelve reviews in Domain 3, and ten SRs/MAs in Phase 3, were rated as having low risk of bias. With reporting quality, some reporting flaws were identified in the topic of protocol and registration, additional analyses, and search strategy. According to GRADE, the level of evidence quality was “critically low” to “moderate,” and risk of bias was the most common downgraded factor.
Conclusion:
Acupuncture may be beneficial in improving cancer pain. However, due to the identified limitations and inconsistent findings, we recommend further rigorous, and more standardized SRs/MAs to provide strong evidence for definitive conclusions.
Introduction
It is estimated that more than 70% of cancer patients suffer from pain, yet nearly half do not have their pain effectively managed.1,2 Although the analgesic ladder proposed by the World Health Organization has brought light to cancer pain management, the adverse effects associated with medications and the addictive nature of analgesics pose new challenges to clinical decision making.3,4 In addition, the challenges involved in managing cancer pain have been made worse by the ongoing opioid crisis.5 -8 Therefore, a multidisciplinary and integrated approach to pain management is essential.9,10 Leading organizations in this field, such as the National Comprehensive Cancer Network and the American Society for Clinical Oncology, call for non-pharmacological interventions that should be sought to manage cancer pain. 11
As a traditional non-pharmacological therapy, acupuncture has been used in China as an adjunctive treatment for a variety of painful conditions.12 -16 The role of acupuncture for cancer pain has been continuously debated, but no unanimous conclusion has yet been reached. As the top of the evidence pyramid, systematic reviews (SRs) and meta-analyses (MAs) are used by researchers to explore the effectiveness of acupuncture for cancer pain, and the number of SRs/MAs published on this topic is increasing. However, not all evidence derived from SRs/MAs is reliable, and poor-quality SRs/MAs may even lead to clinical decisions being made in the wrong direction. 17 Hence, when a large number of SRs on the same topic have been published in a relatively short period of time, an overview is needed to comprehensively evaluate the quality and reliability of the evidence. 18 Therefore, this overview was carried out to systematically collect, analyze, and synthesize the existing evidence on the role of acupuncture on cancer pain.
Methods
Protocol Registration
The protocol for this overview has been prospectively registered with PROSPERO (ID: CRD42023422670).
Criteria for Inclusion and Exclusion
Criteria for inclusion were as follows: (a) types of studies: only SRs/MAs that based on randomized controlled trials (RCTs) were included; (b) participants: patients with cancer and suffering from pain, regardless of cancer stage and current treatment; (c) types of interventions: acupuncture was an eligible intervention, while comparative intervention could be analgesics or sham acupuncture; (d) types of outcomes: response rate, reduction of pain intensity, onset time of pain relief, duration of pain relief, reduction of analgesic dose, and side effects. Updated SRs/MAs and experimental animal studies were excluded.
Search Strategy
We performed a systematic searched in 4 English databases (Cochrane Library, Embase, Web of Science, and PubMed) and 4 Chinese databases (SinoMed, China Science Journal Database, Chinese National Knowledge Infrastructure, and Wanfang Data) on April 2023. “Acupuncture,” “cancer,” “pain,” “meta-analysis,” and “systematic review” were applied as search terms. In Online Appendix A, the PubMed search methodology is displayed.
Screening and Study Selection Procedure
The titles and abstracts were independently reviewed first by 2 reviewers. Following cross-checking, the full text of eligible publications was further evaluated independently by the 2 reviewers. A third reviewer was involved in any case of disagreements.
Two reviewers extracted data independently by using a standardized form: characteristics of participants (health status, sample size), characteristics of reviews (country, publication year, first author), characteristics of design (quality assessment tool, interventions, comparisons), and findings (outcomes, conclusions).
Review Quality Assessment
To evaluate the methodological quality of the included SRs/MAs, the widely used SR appraisal tool, A Measurement Tool to Assess Systematic Reviews 2 (AMSTAR-2) 19 was applied. Notably, 7 of the 16 items that make up this tool are essential items (2, 4, 7, 9, 11, 13, and 15). The reviewers can make a yes, partial yes, or no decision for each item and ultimately synthesize the results of all items to rate the methodological quality at 4 levels, that is, very low, low, moderate, and high. Online Appendix B contains information on the items for AMSTAR-2.
The risk of bias in the review process, findings, and conclusions of the included SRs/MAs were assessed using the Risk of Bias in Systematic Reviews (ROBIS), 20 a well-known tool that is completed in 3 phases. Achieving evaluation of risk of bias requires focusing on 4 aspects of the review, namely interventions, diagnosis, prognosis and etiology. The results of the risk of bias assessment are classified as “low,” “unclear,” or “high” for each aspect.
To assess the reporting quality of the included SRs/MAs, Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA), 21 was applied. This 27-items tool is focused on providing detailed reporting of the procedures and findings of SRs/MAs. Each item will be rated yes (fully reported), partially yes (partially reported), or no (not reported) based on the completeness of the reported information. Online Appendix B contains information on the items for PRISMA.
To evaluate quality of outcomes of the included SRs/MAs, a popular tool for rating evidence quality, the Grades of Recommendations, Assessment, Development and Evaluation (GRADE) system 22 was applied. Initial evidence is considered the highest grade and is subsequently downgraded based on five dimensions: limitations, publication bias, imprecision, indirectness, and inconsistency.
Results
Results of Publication Selection
A total of 749 publications were retrieved initially, of which 136 were duplicates. After screening titles and abstracts, 592 publications were excluded. Full-text reading of the remaining literature further excluded 7 publications. Finally, 14 publications23 -36 were included. The process of publication selection is shown in Figure 1.

Literature screening flowchart.
Study Characteristics
The basic characteristics of the included studies are given in Table 1. The included SRs/MAs were published between 2010 and 2023. Six of the SRs/MAs were published in Chinese, while 8 in English. The authors were from China, Korea, and USA. The largest sample size was 36 trials with 2213 cases, while the smallest sample size was 5 trials with 285 cases. Acupuncture or acupuncture plus analgesics were administered to the experimental group, while analgesics alone or sham acupuncture were administered to the control group.
Characteristics of the Included Studies.
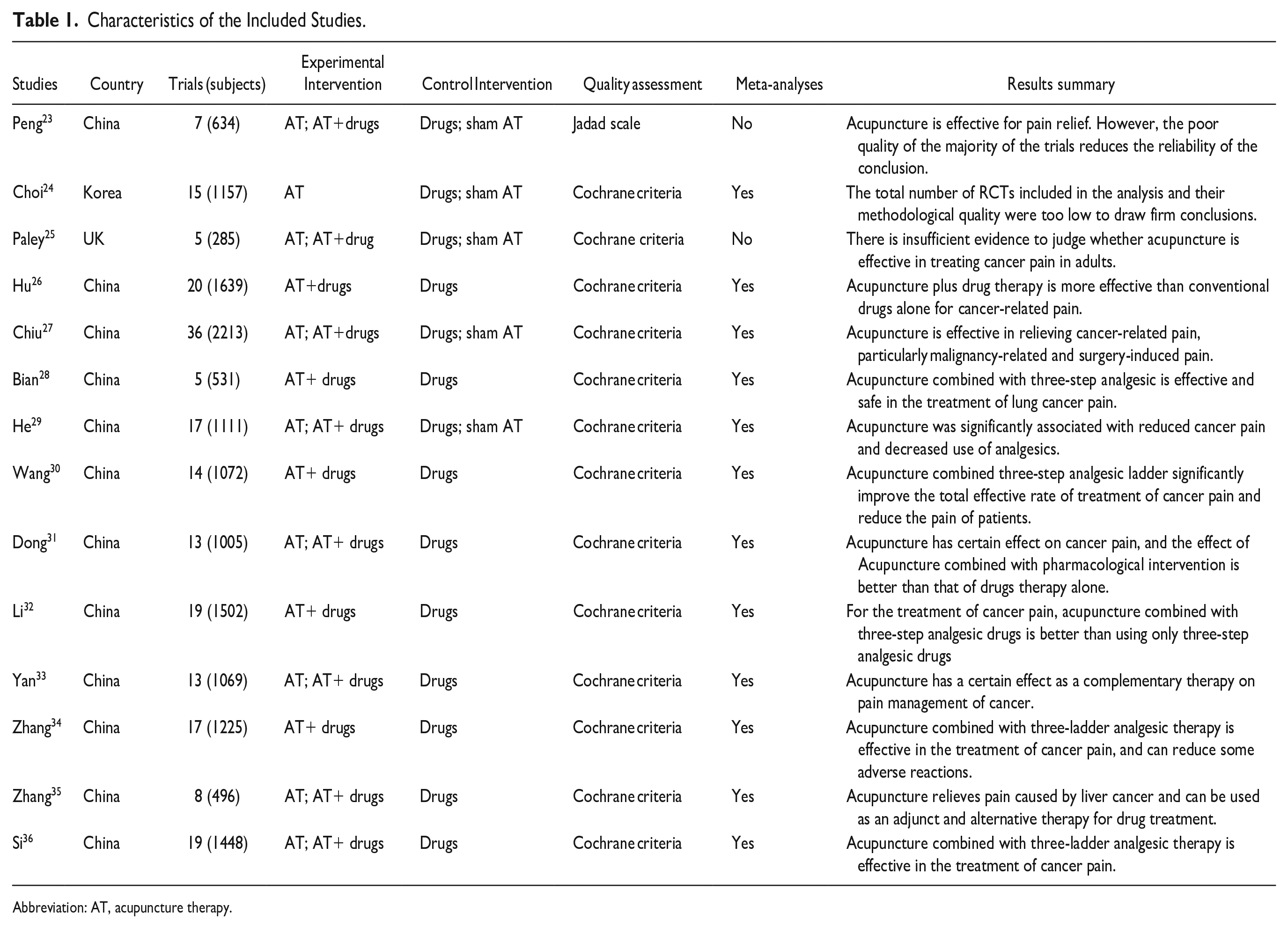
Abbreviation: AT, acupuncture therapy.
Methodological Evaluation
For methodological quality, 2 of the included SRs/MAs were considered to be of high quality, while the rest were considered to be of critically low quality (Table 2). Only 2 reviews reported protocols in advance. A literature screening flow chart was provided for all SRs/MAs, but without explaining the reasons for selection. Furthermore, only 2 reviews provided a list of excluded trials with justification.
Results of the AMSTAR-2 Assessments.

Abbreviations: Y, yes; PY, partial Yes; N, no; CL, critically low; L, low; M, moderate; H, high.
ROBIS Evaluation
All SRs/MAs in Phase 1, Domain 1, and Domain 4, according to ROBIS, were at low risk (Table 3). For Domain 2, four reviews were rated low risk, while the remaining were rated high risk. Twelve reviews were rated low risk in Domain 3, while the remaining could not be judged by the available information. Furthermore, 4 SRs/MAs were rated high risk of bias in Phase 3.
Results of Risk of Bias in Systematic Reviews.

☺ = low risk; ☹ = high risk and; ? =unclear risk.
Reporting Quality Appraisal
For reporting quality, the majority of included studies followed the PRISMA checklist for full reporting (Table 4). However, we discovered that the subjects of protocol and registration (85.7%), search strategy (48.1%), additional analyses of Method (46.7%), and additional analyses of Result (46.7%) were not adequately reported. Furthermore, only 2 SRs/MAs provided protocols in advance, and the vast majority of studies lacked an exhaustive search strategy that was applied to at least one database.
Result of the Reporting Quality.

Abbreviations: Y, yes; PY, partial yes; N, no.
Evidence Quality Evaluation
A total of 38 outcome indicators assessing the role of acupuncture in cancer pain were evaluated using the GRADE system (Table 5). Curiously, 26.32% (10/38) of outcomes were deemed of moderate quality, 63.16% (24/38) of low quality, and 10.52% (6/38) of critically low quality. Notably, risk of bias was the primary cause of evidence downgrading. Furthermore, the evidence was downgraded for inconsistency because of significant heterogeneity in effect sizes.
Results of Evidence Quality.

Abbreviations: −1, downgrade; 0, not downgrade; AT, acupuncture therapy; CL, critically low; L, low; M, moderate; RR, relative risk; OR, odds ratio; SMD, standardized mean difference.
Summary of Included Outcomes
Effects of acupuncture on cancer pain
Ten reviews24,26,28,30 -36 reported pain relief rates after the intervention. The results of these reviews24,26,28,30 -36 indicated that the pain relief rate in the intervention group (acupuncture plus drug therapy) was significantly higher than that in the control group (drug therapy). However, acupuncture treatment alone was not superior to drug in improving the pain relief rate.24,26 Ten reviews24,26 -33,36 examined the effects of acupuncture on reduction of pain intensity. The results of the 2 reviews26,29 indicated that acupuncture was superior to sham acupuncture in reducing pain intensity. However, 2 reviews24,27 suggested no difference between these 2 groups. The results of 8 reviews27 -33,36 indicated that the reduction of pain intensity in the intervention group (acupuncture plus drug therapy) was significantly higher than that in the control group (drug therapy). Two reviews26,33 reported the onset time of pain relief after the intervention, and the results indicated that the combined treatment was significantly superior to drug alone. Two reviews26,33 reported the duration of pain relief after the intervention, and the results indicated that the combined treatment was significantly superior to drug alone. However, one review 35 suggested no difference between the acupuncture group and drug group. Furthermore, the results of 1 review 29 indicated that adjunctive therapy with acupuncture helped to reduce the use of analgesic.
Safety of acupuncture on cancer pain
Adverse effects were reported in only 5 reviews,28,30,34 -36 and the pooled results all suggested that the incidence of adverse events was lower in the intervention group (acupuncture plus drug therapy) than in the control group (drug therapy).
Discussion
A number of SRs/MAs have been conducted to evaluate the effects of acupuncture on cancer pain.23 -36 However, not all SR/MAs were reported according to PRISMA standards or had the assessment of evidence quality by GRADE.37,38 In such cases, an overview of SRs/MAs is preferable to the large number of SRs/MAs of lower quality and with less convincing conclusions.39,40 In addition, an overview can provide limitations that are worth noting and need improvement to drive future high-quality SRs/MAs.41,42
No Definitive Conclusion can be Drawn
Based on the published results, there are no definitive conclusions to be drawn, and acupuncture should be recommended with caution as an adjunctive therapy for cancer pain. All SRs/MAs evaluated the role of acupuncture for cancer pain, but their conclusions were inconsistent. In these SRs/MAs, although most of them came to positive results, their methodological quality was not satisfactory. According to AMSTAR-2, methodological quality was generally rated as being very low, especially in items 2, 4 and 7. In Phase 2, which involved the evaluation of 4 domains to cover the critical review process, some SRs/MAs were classified by ROBIS as high risk. A transparent process of SRs/MAs is essential to produce high-quality evidence, and conversely bias may occur in SRs/MAs. 43 According to GRADE, evidence quality or outcomes regarding to acupuncture on cancer pain was rated as low owing to the risk of bias of RCTs and high heterogeneity. Moreover, authors of SRs/MAs were hesitant to make definite statements about the effectiveness of acupuncture for cancer pain due to the small sample size and poor quality of the original RCTs. Therefore, caution is needed when recommending acupuncture to improver cancer pain.
Research Gaps to be Addressed
Our findings indicate that during the SRs/MAs process, there is a significant need to address issues in methodology, risk of bias, reporting quality, and evidence quality. Methodological flaws can limit the reliability of SRs/MAs, insufficient SRs/MAs reporting can exaggerate the success of interventions or report masked adverse events, and bias risk in SRs/MAs can affect the veracity of the evidence. 44 These shortcomings can ultimately affect the use of evidence and even mislead clinical decision-making. Lack of pre-registered protocols or interpretation of study design choices, comprehensive searches, and lists of excluded studies were common deficiencies among the SRs/MAs we reviewed. Furthermore, a small number of SRs/MAs reached low risk, particularly Phase 3 (risk of bias in the review), and in Phase 2 (identification and selection of studies). In addition, evidence quality varies from moderate to very low given the deficiencies in randomization, blinding, allocation concealment, and small sample sizes or RCTs. Therefore, there is still tremendous room for improvement in RCTs that are rigorously designed and implemented and SRs/MAs that are strictly implemented in accordance with AMSTAR-2 and PRISMA.
Implications for Research and Practice
Aspects of SRs/MAs included were evaluated by AMSTAR-2, ROBIS, and PRISMA to identify areas for common improvement. To ensure transparency of the SR/MA process and reduce any potential risk of bias, it is strongly advised that reviewers register or publish study protocols beforehand. 43 The pre-publication of protocols prior to SR/MA makes it easier to identify any potential bias risk and is essential to guaranteeing the efficacy of the SR/MA methodology. To ensure the study can be replicated, it is advantageous to provide a detailed search strategy. To reduce the risk of publication bias, it is also important to recognize the use of grey literature sources and to provide a list of trials that were excluded and why. 45 When performing data analysis, subgroup analysis and sensitivity analysis should be considered as additional analytical strategies to explore sources of heterogeneity or even to reject performing a pooled analysis if there is significant heterogeneity between studies. Notably, having higher quality SRs/MAs is challenging before having high-quality RCTs. Therefore, it is highly desirable to advocate the development of high-quality RCTs. When performing RCTs, authors should adhere to the fundamental standards for declaring clinical trials, such as the CONSORT statement and STRICTA recommendations, in order to address methodological concerns through meticulous trial design, sound assessment, and thorough analysis.
Currently, 2 sham acupuncture techniques are widely used in trials of acupuncture: non-puncture using a sham acupuncture device (sham device control), and minimal insertion of acupuncture needles (shallow needling control). 46 Two conditions for the establishment of sham acupuncture are that it cannot produce the physiological effects of real acupuncture, and that it must also be morphologically similar to real acupuncture for purpose of blinding. 47 However, sham acupuncture has been criticized since it cannot be biologically inactive, whether it pierces the skin or not. 47 In addition, there are also physiological effects regardless of whether the stimulation is at acupoints or non-acupoints. 48 By stimulating the skin, all sham acupuncture procedures activate afferent nerves, which causes a variety of responses in the body, including limbic tactile responses in the brain. 49 As a result, criticism of sham acupuncture controls in acupuncture research persists. 47 Owing to the limitations of sham acupuncture, the effects of acupuncture may be underestimated, leading to the conclusion that acupuncture is “ineffective” in “sham” controlled trials. 50 Therefore, in exploring the real effects of acupuncture, the search for more scientific and rational sham acupuncture protocols should be regarded as one of the focuses of the research work.
Limitations
Limitations to our overview should be acknowledged. First, a small percentage of literature published in other languages might have been overlooked because the included publications were primarily reported in Chinese and English. Second, the majority of the reviews that were included were carried out in China, so whether the conclusions of these studies are applicable to other populations needs to be verified by further studies. Finally, though our overview was assessed by 2 independent researchers, evaluating quality is a subjective process in which different researchers may have their own opinions on each factor.
Conclusion
Acupuncture may be beneficial in improving cancer pain. However, due to the identified limitations and inconsistent findings, we recommend further rigorous, and more standardized SRs/MAs to provide strong evidence for definitive conclusions.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231210288 – Supplemental material for An Overview of Systematic Reviews and Meta-Analyses of Clinical Studies of Acupuncture for Cancer Pain
Supplemental material, sj-docx-1-ict-10.1177_15347354231210288 for An Overview of Systematic Reviews and Meta-Analyses of Clinical Studies of Acupuncture for Cancer Pain by Di Zhang, Yue Ji, Liu Lv, Qiongyang Zhou, Zhijiang Liu, Chenlin Zhang and Shanshan Chen in Integrative Cancer Therapies
Supplemental Material
sj-pdf-2-ict-10.1177_15347354231210288 – Supplemental material for An Overview of Systematic Reviews and Meta-Analyses of Clinical Studies of Acupuncture for Cancer Pain
Supplemental material, sj-pdf-2-ict-10.1177_15347354231210288 for An Overview of Systematic Reviews and Meta-Analyses of Clinical Studies of Acupuncture for Cancer Pain by Di Zhang, Yue Ji, Liu Lv, Qiongyang Zhou, Zhijiang Liu, Chenlin Zhang and Shanshan Chen in Integrative Cancer Therapies
Footnotes
Acknowledgements
Not Applicable.
Authors’ Contributions
Di Zhang and Yue Ji contributed equally to this work and they are the co-first authors. Conceptualization: Di Zhang, Yue Ji; Methodology: Liu Lv, Zhijiang Liu; Supervision, Qiongyang Zhou: Chenlin Zhang, Yue Ji; Writing—original draft: Qiongyang Zhou; Writing—review & editing: Shanshan Chen. All authors read, critically reviewed and approved the final manuscript as submitted.
Availability of Data and Materials
Online Appendix A: the detailed search strategy for PubMed. ![]() : information on the items for AMSTAR-2 and PRISMA. All analyses were based on previously published studies; thus, no other data is required.
: information on the items for AMSTAR-2 and PRISMA. All analyses were based on previously published studies; thus, no other data is required.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Science and Technology Program of Lanzhou, China (2022-ZD-73); Administration of Traditional Chinese Medicine of Gansu Province, China (GZKP-2022-17); the Project of Director of Gansu Famous Doctors Studio, China (No. [2022]50); The Key Talent Project of Gansu Province (No. [2023]20): Development and transformation of clinical talent cultivation based on the needle knife medical platform; National Natural Science Foundation of China (No. 82274481); Implementation plan of high level key discipline construction project of traditional Chinese medicine of the State Administration of traditional Chinese Medicine,China(No.[2022]226).
Ethics Approval and Consent to Participate
This work does not include any studies performed on humans or animals. Thus, no ethics approval and consent to participate are need.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
