Abstract
Objective
To determine if the pretreatment of hypoxic human oesophageal carcinoma cell lines (EC109, TE1 and KYSE170) with ginsenoside Rg3 (Rg3) increases their radiosensitivity to X-rays.
Methods
The growth inhibitory effect of different Rg3 concentrations was measured using the 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) assay. Radiation sensitivity was measured using a clone formation assay and flow cytometry was used to measure the effects of Rg3 on radiation-induced apoptosis. Western blot analysis was used to measure the effects of Rg3 on the levels of hypoxia inducible factor (HIF)-1α and vascular endothelial growth factor (VEGF).
Results
Rg3 inhibited EC109, TE1 and KYSE170 cell growth in a dose- and time-dependent manner. Pretreatment with 10 µmol/ml Rg3 increased EC109, TE1 and KYSE170 radiosensitivity. Rg3 plus radiation significantly increased the apoptosis rate compared with radiation alone. Rg3 also decreased VEGF and HIF-1α protein levels in EC109 cells in a dose-dependent manner. The combination of Rg3 and radiation increased the fragmentation of double-stranded DNA.
Conclusion
Rg3 enhanced the radiosensitivity of human oesophageal carcinoma cell lines cultured under hypoxic conditions possibly by downregulating VEGF and HIF-1α protein levels.
Keywords
Introduction
Oesophageal carcinoma is the ninth most common cancer worldwide, and is ranked sixth in terms of mortality.1,2 Oesophageal carcinoma is also one of the most common cancers in China and has one of the highest mortality rates, 2 with >50% morbidity worldwide. 1 More than 95% of patients with oesophageal carcinoma are diagnosed in the middle and late stages of the disease; as these patients can then receive only radiotherapy because surgery is not an option, the prognosis is poor. 3 The radiotherapeutic effect is limited by the sensitivity of the tumour cells to radiation, resulting in limited success in controlling the primary oesophageal carcinoma. 4 Currently, the local recurrence rate is 60–80% in patients with oesophageal carcinoma receiving radiotherapy alone.5–7 There is therefore a need for new therapeutic approaches that enhance the radiosensitivity of the tumour cells and decrease local recurrence, which are key to improving survival following oesophageal carcinoma.
Hypoxia inducible factor (HIF)-1α and vascular endothelial growth factor (VEGF) are activated by hypoxia and play key roles in the developing hypoxic environment of tumours. 8 Previous research has demonstrated that HIF-1α is highly expressed in multiple tumours including cervical carcinoma, 9 and breast 10 and gastric cancer. 11 It is negatively correlated with the remission rate following radiochemotherapy, total survival time and tumour-free survival time. 8 Owing to the rapid proliferation of tumour cells during tumour growth, local anoxia is readily generated and quickly induces the expression of HIF-1α. 8 HIF-1α binds to the HIF-1α binding sites of target genes, activating the expression of multiple hypoxia response genes, and increasing protein production by modulating VEGF-dependent signalling pathways and inducing expression of glycolysis-related enzymes. 12 These pathways indicate that HIF-1α plays an important role in the adaption of tumours to anoxia, energy metabolism, neovascularization, metastasis, radiotherapy resistance and chemotherapy tolerance. 13 Blocking HIF-1α expression therefore becomes an important strategy for inhibition of tumour growth and enhancement of the sensitivity of tumour cells to radiation and chemotherapy.
Ginsenoside Rg3 (Rg3) has recently been found to have antitumour potential by inhibiting angiogenesis in experiments using cell lines and animals. 14 There do not appear to be any reports on the use of Rg3 in combination with radiotherapy in oesophageal carcinoma. Therefore, the present study was designed to investigate the in vitro radiosensitizing effect of Rg3 on hypoxic human oesophageal carcinoma cell lines.
Materials and methods
Cell culture
Human oesophageal carcinoma cell lines (EC109, TE1 and KYSE170) were provided by the Central Experimental Laboratory, Nanjing Medical University (Nanjing, China). The cells were cultured in RPMI-1640 medium (KeyGen, Nanjing, China; containing 10% Sijiqing newborn bovine serum) in a thermostatic incubator (Thermo Fisher Scientific, Rockford, IL, USA) in a humidified atmosphere of 5% CO2 at 37℃. Adherent cells were passaged every 3 days and subjected to hypoxic conditions for 24 h in a homemade sealed tank filled with 97% N2 + 3% O2.
Cell irradiation
Human oesophageal cell lines were irradiated with a 200 cGy/min X-ray beam from a 6 MV medical linear accelerator (Precise Treatment System™; Elekta AB, Stockholm, Sweden). The source–cell distance was 100 cm and the radiation field was 20 cm × 20 cm.
MTT assay
The 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT) assay (Beyotime Biotech, Jiangsu, China) was used to determine the effect of treatment on cell viability. Human oesophageal cell lines at the logarithmic growth phase were inoculated into 96-well plates at a density of approximately 4–6 × 104 cells/ml in a final volume of 100 µl cells/well. Cells were not subjected to hypoxic conditions for this experiment. After adhesion to the cell culture plastic, cells were treated with 10, 20, 50, 100, 200, 400 and 600 µmol/ml Rg3 (six replicates for each group) (Sigma-Aldrich, St Louis, MO, USA) or blank cell culture medium was used as a control. After treatment for 24 h, 48 h and 72 h, the cell culture medium was removed and the cells were washed with 0.01 M phosphate-buffered saline (PBS; pH 7.4) 1–2 times, followed by the addition of 20 µl (5 mg/ml) of MTT. After a further 4-h incubation, the supernatant fraction was removed and 150 µl of dimethyl sulphoxide was added to each well. After vibration at low speed for 10 min to dissolve the purple crystals, the optical density (OD) at 490 nm (with an A630 reference) for six wells in each group was measured using a microplate reader (Bio-Rad, Hercules, CA, USA). The tumour cell survival rate (%) = (OD value in the experimental group/OD value in the control group)×100%. The Rg3 IC20 values were calculated using the SPSS® statistical package, version 19.0 (SPSS Inc., Chicago, IL, USA) for Windows®.
Inhibition of clone formation
Human oesophageal cell lines at the logarithmic growth phase were released from the cell culture plate using a standard trypsinization method and diluted into single cell suspensions in cell culture medium. The cells were inoculated into six-well plates at a density of 200 cells/ml. Adherent cells were randomly divided into a radiation group and an Rg3 plus radiation group. Cells were treated with or without 10 µmol/ml Rg3 for 24 h at the same time that they underwent hypoxia treatment (see above for details). Then the cells were exposed to 0, 1, 3, 6 and 9 Gy X-radiation from a 6 MV beam medical linear accelerator. Cells were washed 1–2 times with Rg3-free cell culture medium and then cultured continuously with the cell culture medium changed every 4 days. When clones were visible to the naked eye, the culture was terminated, the supernatant fraction removed, and the cells rinsed three times with 0.01 M PBS (pH 7.4) before fixing with methanol for 10 min and staining with a commercial Giemsa kit (KeyGen) for 15 min at 37℃. Cells were removed from staining solution, washed under flowing tap water and air dried. The number of clones with >50 cells per field were counted in six randomly selected fields (×10 magnification) using an inverted microscope (Olympus BX40; Olympus Imaging China, Beijing, China). Cloning efficiency was calculated using the following equation: cloning efficiency (100%) = (clone number in the control group/clone number in the experimental group)×100%. The survival fraction was calculated as follows: survival fraction = (cloning efficiency in the experimental group/cloning efficiency in the control group). GraphPad Prism 5.0 (GraphPad Software, La Jolla, CA, USA) was used to fit cell survival curve data according to the single-hit multiple-target model, and the fraction of surviving cells (SF) = 1−(1−e−D/D0)n, where D0 is the dose required when the curve index decreased 63% and D0 = 1/k. Dq is the subthreshold dose required for cell damage and Dq = D0.lnN. The sensitization enhancement ratio (SER) was calculated as follows: SER = D0 in the control group/D0 in the experimental group (performed in triplicate with a deviation <5%).
Apoptosis assessment by flow cytometry
Single-cell suspensions of the human oesophageal carcinoma cell lines at a density of 5 × 106 cells/ml were inoculated into six-well plates and divided into three groups: control, radiation and Rg3 plus radiation. The control cells were subjected to hypoxia for 24 h without any treatment. The cells in the radiation group were treated with 8 Gy X-ray radiation from a 6 MV beam medical linear accelerator after 24 h under hypoxic conditions, followed by incubation for a further 48 h. This dose of X-ray radiation (8 Gy) was selected based on the results of the clone formation study described above. The cells in the Rg3 plus radiation group were cultured with Rg3 (10 µmol/ml) under hypoxic conditions for 24 h and subsequently treated with 8 Gy X-ray radiation from a 6 MV beam medical linear accelerator, and incubated for a further 48 h. The cells were released from the cell culture plate using a standard trypsinization method and suspended in 0.01 M PBS (pH 7.4) to a density of >1 × 106 cells/ml. A sample of each cell suspension (100 µl) was placed in a 5 ml capillary tube and 5 µl of Vybrant® Apoptosis Assay Kit (Invitrogen, Carlsbad, CA, USA) and 10 µl of propidium iodide (20 µg/ml; KeyGen) were added. After mixing, the suspension was incubated at room temperature in the dark for 15 min and the proportion of apoptotic cells was measured using a flow cytometer (BD FACSAria™ III; Becton Dickinson, Franklin Lakes, NJ, USA).
Western blot analysis
For Western blot analysis, EC109 cells were treated with Rg3 (10, 20, 50 µmol/ml) under hypoxic conditions for 24 h. Control EC109 cells were also cultured under increasing durations of hypoxia (12 h, 24 h, 48 h) without Rg3 treatment. Then, 2 × 107 EC109 cells were lysed with 1×sodium dodecyl sulphate buffer and the total cellular protein concentration was quantified using a Bicinchoninic Acid Protein Assay Kit (KeyGen) according to the manufacturer’s instructions. Protein samples were separated using a 10% polyacrylamide gel and 6% spacer gel using electrophoresis at 60 V × 15 min and 120 V × 30 min. Separated proteins were then transferred to a polyvinylidene fluoride membrane (PVDF; Millipore, Billerica, MA, USA) at constant current and 300 mV for 180 min using electroblot equipment (Bio-Rad). The PVDF membrane was then blocked with 5% skimmed milk in Tris-buffered saline Tween-20 buffer (TBST; pH 7.5; 10 mM Tris, 150 mM NaCl and 0.1% Tween 20) for 1 h at room temperature. The PVDF membrane was then incubated with mouse anti-HIF-1α, anti-β-actin (internal control) and anti-VEGF monoclonal antibodies (diluted 1:400; Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at 4℃. The PVDF membrane was washed three times with TBST (pH 7.5) at room temperature. The PVDF membrane was then incubated in horseradish peroxidase-labelled secondary antibody (diluted 1:5000; KeyGen) at room temperature for 2 h. The PVDF membrane was then washed three times with TBST (pH 7.5) at room temperature. The immunostaining of the PVDF membranes were visualized using an enhanced chemiluminescence kit (KeyGen) and exposed to X-radiography film (Kodak, Tokyo, Japan). All Western blot experiments were repeated at least three times.
Measurement of DNA damage by foci formation
EC109 cells were inoculated at a density of 5 × 104 cells/ml into six-well plates. The EC109 cells were then treated with or without Rg3 (10 µmol/ml) whilst being exposed to hypoxic conditions for 24 h and then exposed to 8 Gy radiation. Control cells did not receive either Rg3 or radiation treatment. Cells in each group were treated with 0.01 M PBS (pH 7.4) for 2 h, fixed with 4% paraformaldehyde for 30 min, rinsed with 0.01 M PBS (pH 7.4), and then blocked with 2% bovine serum albumin (KeyGen) and 0.3% Triton™ X-100 (KeyGen) for 1.5 h at –20℃. The cells were then incubated with polyclonal rabbit antihuman phosphorylated H2A histone family, member X (γ-H2AX) antibody (1:250 dilution; Millipore) overnight at 4℃. Following overnight incubation, the cells were rinsed twice with 0.01 M PBS (pH 7.4) and then incubated with goat antirabbit fluorescein isothiocyanate-labelled secondary antibody (1:150 dilution; KeyGen) at room temperature for 1.5 h. Following two rinses with 0.01 M PBS (pH 7.4), the cells were stained with 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (1:50 diluted in 0.01 M PBS, pH 7.4; KeyGen) for 3 min at –20℃ to detect the nuclei. Cells were then mounted with 20% glycerol in a humidified chamber to check for foci formation using a confocal fluorescence microscope (Zeiss LSM, Thornwood, NY, USA).
Statistical analyses
The data are expressed as mean ± SD. All experiments were performed at least in triplicate. GraphPad Prism 5.0 (GraphPad Software) was used to fit the survival curve data as described. All statistical analyses were performed using the SPSS® statistical package, version 19.0 (SPSS Inc.) for Windows®. Differences between two independent samples were analysed using a two-sample t-test for independent samples and differences between groups were analysed using single factor analysis of variance. A P-value <0.05 was considered statistically significant.
Results
The results of the MTT assay showed that treatment of EC109, TE1 and KYSE170 cells with Rg3 for 24 h significantly decreased the OD values in a concentration- and time-dependent manner starting at 20 µmol/ml compared with the control group (P < 0.05 for all comparisons; Figure 1). The Rg3 IC20 values for EC109, TE1 and KYSE170 cells were 6.61, 5.44 and 6.32 µmol/ml as calculated by the SPSS® statistical package, respectively (Figure 1). Treatment with 10 µmol/ml Rg3 for 24 h had no significant effect on the OD values of the EC109, TE1 and KYSE170 cells, so 10 µmol/ml Rg3 was chosen for the radiosensitization experiments to avoid Rg3 cytotoxicity.
The effects of different concentrations of ginsenoside Rg3 on the survival rate (%) of human oesophageal carcinoma cell lines cells determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay compared with the control group treated with Rg3-free medium: (a) EC109 cells; (b) TE1 cells; (c) KYSE170 cells. Data presented as mean ± SD.
The EC109, TE1 and KYSE170 cells were exposed to different X-ray doses following pretreatment with 10 µmol/ml Rg3 for 24 h to assess clone formation. The single-hit multiple-target model was used to fit cell survival curves (Figure 2) and to calculate SERs. For EC109 cells, the mean ± SD D0 values were 1.88 ± 0.11 Gy and 1.47 ± 0.08 Gy for the Rg3 plus radiation and radiation groups, respectively, yielding a SER of 1.28. For TE1 cells, the mean ± SD D0 values were 2.44 ± 0.36 Gy and 1.87 ± 0.15 Gy, yielding an SER of 1.30, while for KYSE170 cells, the mean ± SD D0 values were 2.38 ± 0.21 Gy and 1.65 ± 0.18 Gy, respectively, yielding a SER of 1.44. These results suggest that Rg3 has a radiosensitizing effect on all three human oesophageal carcinoma cell lines.
Effect of 10 µmol/ml ginsenoside Rg3 pretreatment on the radiosensitivity of human oesophageal carcinoma cell lines as determined using a clone formation assay: (a) EC109 cells; (b) TE1 cells; (c) KYSE170 cells. Data presented as mean ± SD.
The EC109 cells exhibited an apoptosis rate of 6.46%, 30.68% and 62.33% in the control, radiation, and Rg3 plus radiation groups, respectively (Figure 3). TE1 cells exhibited apoptosis rates of 4.42%, 35.31% and 70.04%, respectively, while KYSE170 cells displayed apoptosis rates of 4.72%, 41.96% and 67.82% , respectively. The apoptosis rates in the Rg3 plus radiation groups for all three cell lines were significantly higher than for the radiation alone groups (P < 0.05 for all comparisons), suggesting that Rg3 plus X-irradiation increases apoptosis of human oesophageal carcinoma cell lines.
Effects of 10 µmol/ml ginsenoside Rg3 (Rg3) pretreatment combined with X-radiation (8 Gy) on the rate of apoptosis in human oesophageal carcinoma cell lines (EC109, TE1, and KYSE170) as determined using flow cytometry with fluorescein isothiocyanate-conjugate annexin V (FITC-A) and propidium iodide (PI-A) staining. Quadrant 4 (Q4) in each scan contains the apoptotic cells. (a) EC109 control; (b) EC109 treated with radiation; (c) EC109 treated with Rg3+radiation; (d) TE1 control; (e) TE1 treated with radiation; (f) TE1 treated with Rg3+radiation; (g) KYSE170 control; (h) KYSE170 treated with radiation; (i) KYSE170 treated with Rg3+radiation.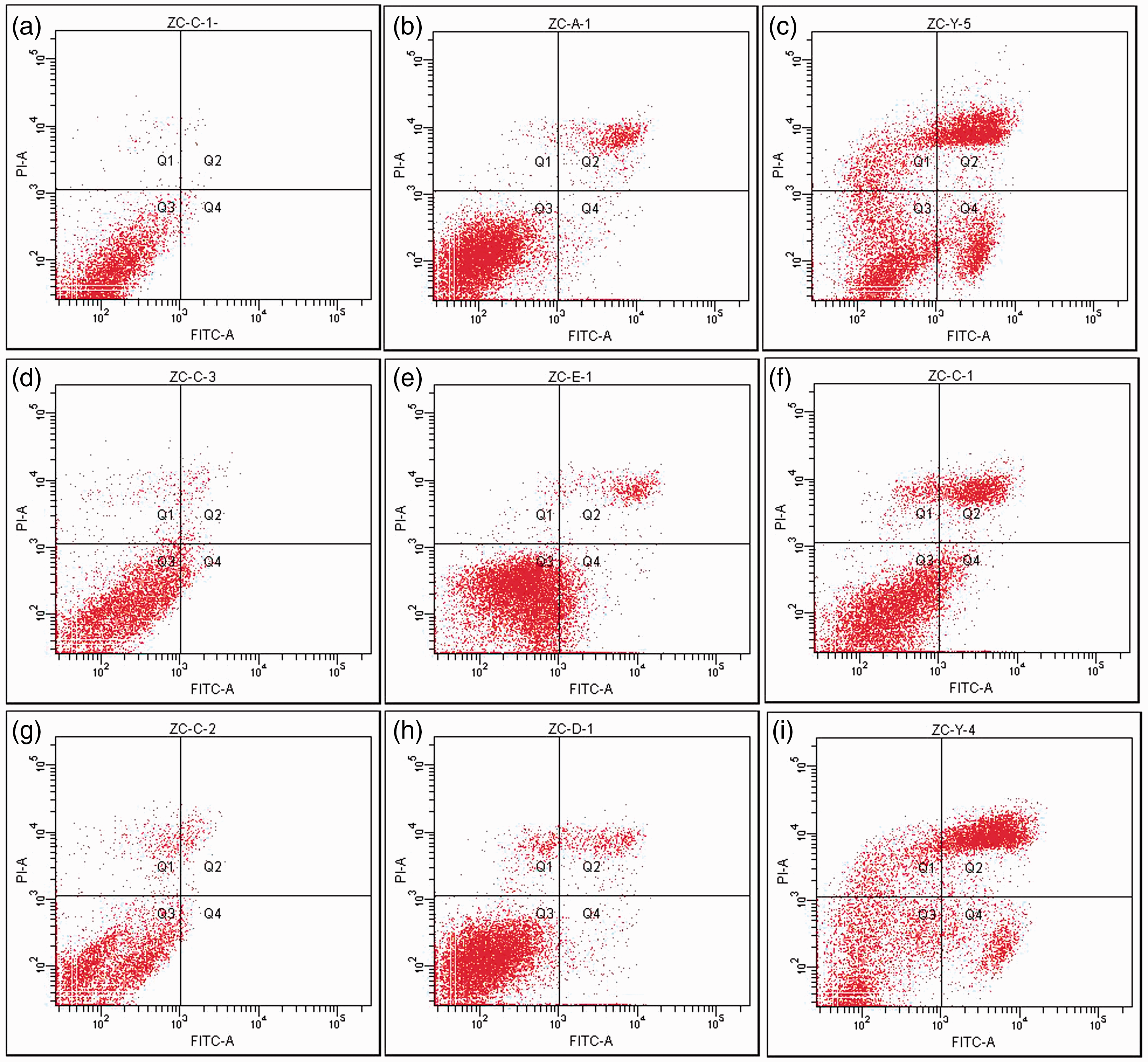
Western blot analysis was used to compare the levels of HIF-1α and VEGF proteins in EC109 cells at different time points under normal conditions (Figure 4). This analysis showed close to normal protein levels after 12 h under hypoxic conditions, which increased to maximum levels for both proteins after 24 h of hypoxia, suggesting a correlation between HIF-1α and VEGF levels and the duration of hypoxia exposure up to 24 h. Treatment of hypoxic EC109 cells with Rg3 (at 10, 20 and 50 µmol/ml) indicated that the levels of HIF-1α and VEGF proteins decreased in a clear dose-dependent manner.
The levels of hypoxia inducible factor (HIF)-1α, vascular endothelial growth factor (VEGF) and β-actin (internal control) proteins in the human oesophageal carcinoma cell line EC109 under increasing durations of control hypoxic conditions and after treatment with 10, 20 and 50 µmol/ml ginsenoside Rg3 (Rg3) as determined by Western blot analysis.
The effect of Rg3 radiosensitization was evaluated by measuring foci formation after γ-H2AX immunofluorescence staining and observing DNA damage (fragmentation of double strand breaks) (Figure 5). Control EC109 cells did not form foci under hypoxic conditions. However, after 8 Gy of X-radiation, there was obvious foci formation, concentrated mainly in the nuclei. Combining Rg3 (10 µmol/ml) with 8 Gy X-radiation increased foci formation and fluorescence intensity, suggesting that Rg3 has a radiosensitization effect. Confocal examination indicated that the nuclear green spots in cells from all groups were most obvious at 30 min and started to disappear after 60 min (images not shown). The green fluorescence observed in the EC109 cells with Rg3 or/and radiation was more obvious than in the control cells at all time-points (data not shown). The mean fluorescence of 50 randomly selected cells was compared, showing that the levels of γ-H2AX in EC109 cells increased relative to controls (data not shown).
The effect of ginsenoside Rg3 (10 µmol/ml) radiosensitization in the human oesophageal carcinoma cell line EC109 as evaluated by observing foci formation (arrows) using immunofluorescence staining of phosphorylated H2A histone family, member X (γ-H2AX) and observing DNA damage (fragmentation of double strand breaks) under a confocal fluorescence microscope. Nuclei are stained blue with 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (DAPI). EC109 cells were treated with either 8 Gy X-ray radiation (8 Gy) or 8 Gy X-ray radiation and 10 µmol/ml Rg3 (8 Gy+10 µmol) and compared with control cells that received neither treatment (0 Gy). The colour version of this figure is available at: http://imr.sagepub.com.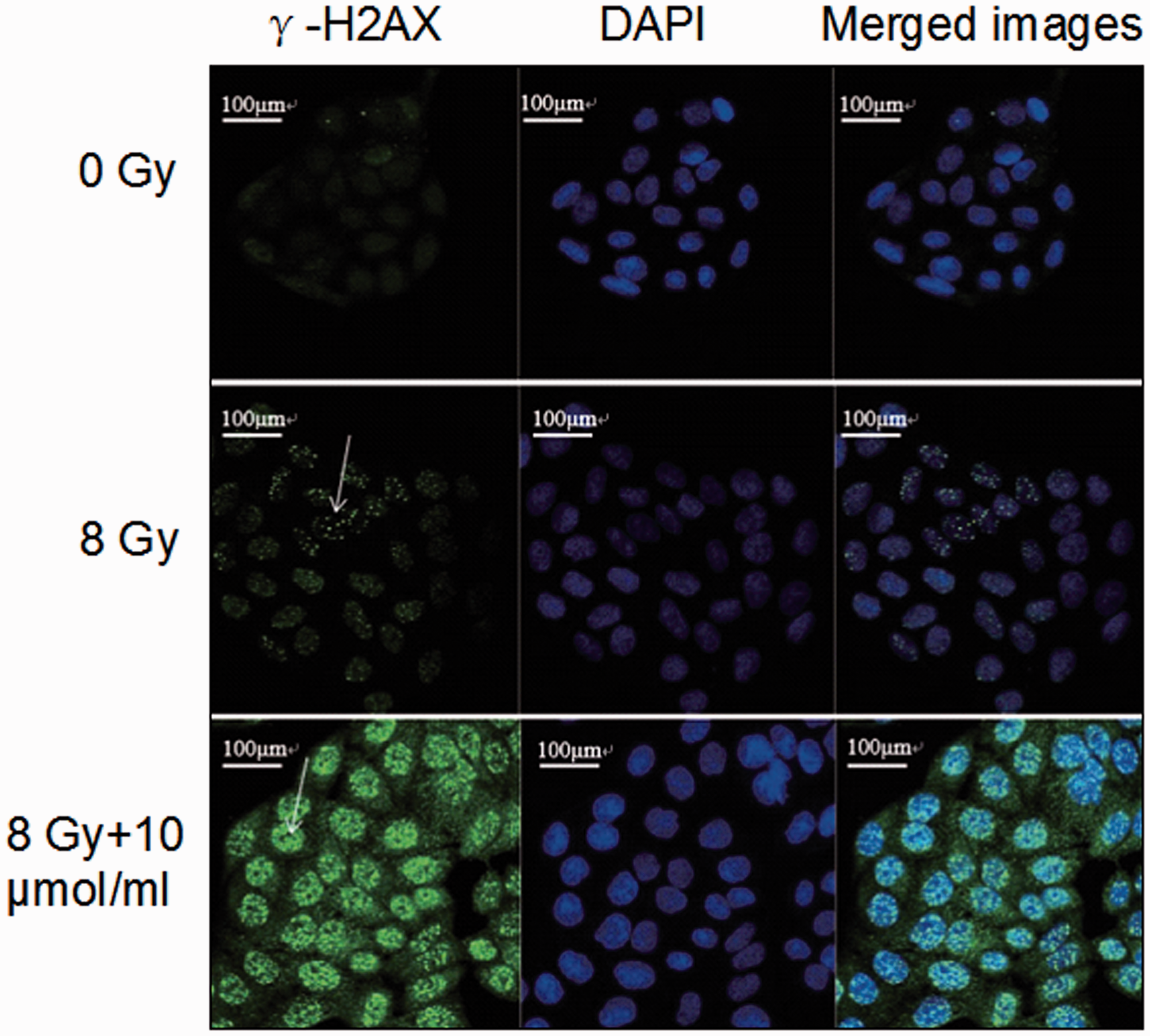
Discussion
Oesophageal carcinoma is a common malignancy of the digestive tract, and China has a high rate of morbidity due to this cancer. 2 Currently, the major therapeutic strategy for nonresectable oesophageal carcinoma at the middle and late stages of the disease is a radiotherapy-based treatment. 4 Although there have been continuous improvements and developments in the equipment and technology used to deliver radiotherapy, the prognosis of oesophageal carcinoma remains unsatisfactory, with a 5-year survival rate of <30%. 15 The major reason for the failure of radiotherapy is local recurrence owing to solid tumours containing 10–50% hypoxic cells 16 . Oxygen radicals released by tumour cells, which are targeted by radiotherapy, are reduced under hypoxic conditions, resulting in reduced radiotherapy-induced DNA defragmentation. 12 Hypoxic conditions enhance HIF-1α expression, resulting in increased VEGF expression, which induces angiogenesis. This provides an increased energy supply to the tumour, facilitating local recurrence and remote tumour metastasis. 17 Research indicates that many different tumours upregulate VEGF after radiotherapy, 18 which is thought to be a self-protective mechanism. Current studies indicate that the mechanisms of increased resistance of tumours to radiotherapy include the following: (i) hypoxia specifically inhibits the growth cycle of tumour cells; (ii) intratumoural hypoxia–reoxygenation produces massive quantities of free radicals and increases genetic instability, which results in gene mutations and a more aggressive tumour; and (iii) hypoxia increases the synthesis and/or expression of specific proteins including VEGF and P53. 8 Changes in protein synthesis help tumour cells to adapt to hypoxia and increase their resistance to radiotherapy. 8 Research has demonstrated that HIF-1α is independent of P53 and P21, and negatively correlated with sensitivity to radiochemotherapy, with 44.4% of HIF-1α negative cells being sensitive to therapy. 19 A study that transfected gastric tumour cells so that HIF-1α protein levels were reduced observed a concomitant reduction in tumour growth when the tumour cells were grown in vivo. 20 A study of 67 cases of cervical carcinoma found that patients with strong or moderate HIF-1α immunostaining showed less response to radiotherapy and shorter progression-free survival. 21 Therefore, synergistic therapy against HIF-1α and VEGF with radiotherapy during tumour treatment may enhance tumour sensitivity to radiotherapy and serve as an effective strategy to control local tumour growth rates. 22 Therefore, medications and strategies to increase radiosensitization of hypoxic cells is of considerable interest.
Ginsenoside Rg3 is a microscale tetracyclic triterpenoid saponin found in ginseng. 23 It has selective inhibitory effects on tumour growth, invasion and metastasis. 24 Rg3 antagonizes tumour angiogenesis, inhibits the expression of matrix metalloproteinase, interferes with endothelial and extracellular matrix interactions, prevents the formation of the tumour vascular network, and inhibits VEGF synthesis in tumours. 25 Research on Rg3 has focused on its antitumour and antiangiogenesis activities, particularly in the treatment of late-stage nonsmall-cell lung cancer (NSCLC) in combination with chemotherapy, which results in better therapeutic outcomes than chemotherapy alone. 26 A multicentre, double-blind, phase III clinical study of patients with NSCLC treated with Rg3 + vinorelbine + cisplatin reported that in 115 patients with confirmed stage III–IV disease, 106 completed treatment. 27 The response rate (complete or partial remission) and median survival time were 33.3% (17/51) and 10.0 months, and 14.5% (8/55) and 8.0 months in the treatment and control groups, respectively. 27 These results demonstrated that Rg3 when combined with standard chemotherapy resulted in improvements in the response rate and survival time compared with standard chemotherapy alone and that this agent warranted further investigation in clinical trials. 27 However, the ability of Rg3 to increase radiosensitivity when combined with chemotherapy has remained unclear. The present study investigated the in vitro effect and mechanism of action of Rg3 on the radiosensitivity of hypoxic human oesophageal carcinoma cell lines.
Based on the findings of the present study, Rg3 inhibited the growth of oesophageal carcinoma cells in a dose-dependent manner as determined by the MTT assay. These present findings were consistent with those of a study that reported that Rg3 inhibited the growth of the lung carcinoma cell line A549. 28 Cell survival data from the present study demonstrated that the survival rate of oesophageal carcinoma cells decreased following an increase in the dose of X-radiation. The dose–survival curves fitted the single-hit multiple-target model, and the cells demonstrated resistance to radiation in the ‘shoulder portion’ of the cold spot area, suggesting that oesophageal carcinoma cells were able to self-repair following sublethal damage. At a low dose without obvious cytotoxicity as demonstrated by the MMT assay, Rg3 (10 µmol/ml) combined with X-radiation increased the lethality of the X-ray dose. The ‘shoulder portion’ of the survival curve was shortened and cell resistance to radiation was reduced. The SER was 1.28 for the EC109 cells, 1.30 for the TE1 cells and 1.44 for the KYSE170 cells. These present findings suggest that pretreatment with Rg3 radiosensitizes oesophageal carcinoma cells cultured in vitro. The findings of the apoptosis experiment in the present study also suggest that Rg3 may have a synergistic effect that increases the rate of radiation-induced apoptosis of hypoxic human oesophageal carcinoma cells.
Previous research has demonstrated a negative correlation between the levels of VEGF in tumour tissues and radiosensitivity. 29 In the present study, treatment with increasing doses of Rg3 decreased the levels of HIF-1α and VEGF proteins in tumour cells compared with cells treated with hypoxic conditions alone. These present findings suggest that Rg3 radiosensitization may be related to the downregulation of HIF-1α and VEGF proteins. A limitation of our study is that the cells were from a Chinese population, so the results may not be applicable to other populations.
In conclusion, pretreatment with 10 µmol/ml Rg3 increased the radiosensitization of hypoxic human oesophageal carcinoma cell lines in vitro. The mechanism of action by which Rg3 increases the radiosensitization of hypoxic human oesophageal carcinoma cell lines may be related to the downregulation of HIF-1α and VEGF proteins. However, this requires further investigation. These present findings provide preliminary scientific evidence that Rg3 might be a useful chemotherapeutic agent to consider for the future treatment of oesophageal carcinoma.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by the Natural Science Foundation of China (grant no. 81272504), the Innovation Team (grant no. LJ201123 [EH11]), Jiangsu Provincial Science and Technology Projects (grant no. BK2011854 [DA11]), and “333” Project of Jiangsu Province (grant no. BRA2012210 [RS12]).
