Abstract
Aim:
To determine effects of Coenzyme Q10 (CoQ10) supplementation in breast cancer patients receiving doxorubicin treatment.
Methods:
Phase I randomized, placebo-controlled, cross-over, dose-finding study among women with stage I-III breast cancer receiving 4 cycles of doxorubicin plus cyclophosphamide. The study was designed to test effects on doxorubicin pharmacokinetic parameters when administering up to the maximum tolerated dose of CoQ10 of 1200 mg/day. Eligible patients were randomized to Arm A (CoQ10 after Cycle 3, followed by placebo after Cycle 4) or Arm B (placebo after Cycle 3, followed by CoQ10 after Cycle 4). CoQ10 concentrations and total antioxidant capacity (TAC) were measured before and after chemotherapy cycles. Non-compartmental pharmacokinetic parameters of doxorubicin and its active metabolites were measured with and without CoQ10. Paired t-tests assessed intra-patient differences in pharmacokinetic parameters, serum CoQ10 concentrations, TAC, and adverse events.
Results:
Six patients received 300 mg/day of CoQ10 (Arm A [n = 3], Arm B [n = 3]). One patient received 600 mg/day of CoQ10 but was discontinued due to non-adherence. Serum CoQ10 concentrations were increased in patients receiving 300 mg/day (mean ± SD change: CoQ10, 1.6 ± 0.9 µg/mL; placebo, −0.01 ± 0.3 µg/mL; P = .01). There were no clinically significant pharmacokinetic interactions between 300 mg/day CoQ10 and doxorubicin and no differences in TAC or adverse events during treatment and nontreatment periods. The trial was closed early due to slow accrual.
Conclusion:
300 mg/day of CoQ10 with doxorubicin did not change doxorubicin pharmacokinetics and was not associated with treatment-related adverse events. Future studies should evaluate the long-term effects of CoQ10 at 300 mg/day and safety studies should examine higher doses.
Trial registration:
ClinicalTrials.gov: NCT00976131.
Introduction
In 2023, an estimated 297 790 women will be diagnosed with invasive breast cancer in the US, and there are currently more than 3 million breast cancer survivors.1 -3 Doxorubicin (Adriamycin), an anthracycline, plus cyclophosphamide (AC) is standard neoadjuvant and adjuvant therapy for early-stage breast cancer.4 -6 However, despite the benefits of these therapies in preventing breast cancer recurrence and improving overall survival, 3%-20% of breast cancer patients develop acute and chronic cardiomyopathic changes and congestive heart failure due to doxorubicin therapy, as assessed by changes in left ventricular ejection fraction (LVEF), especially when doxorubicin cumulative doses exceed 450 mg/m2 and/or are co-administered with other cardiotoxic medications such as trastuzumab.7 -10 It is well established that doxorubicin cardiotoxicity is a cumulative dose-related effect. 11 Once a dose threshold is reached, irreversible myocyte damage occurs, likely due to increased myocyte mitochondrial reactive oxygen species (ROS), which is exacerbated by further exposure to doxorubicin and trastuzumab. 12 There has been longstanding interest in identifying possible cardioprotective agents for use during anthracycline treatment. While a recent meta-analysis shows that dexrazoxane is effective in preventing or decreasing cardiotoxicities adult oncology patients treated with anthracyclines, the results were not consistent in pediatric patients, and adverse event data were not of high quality in both populations. 13 The authors concluded that more research is needed before making any definitive conclusions. Moreover, while dexraxozane is may be efficacious for reducing cardiotoxicity in anthracycline-treated patients, it has had low clinical uptake to date given some of these concerns.
Coenzyme Q10 (CoQ10), also known as ubiquinone, is a lipid soluble antioxidant that may protect cardiac myocytes against mitochondrial ROS. CoQ10 is synthesized in the mitochondria and in most intracellular membranes, and is ingested via foods such as meat, poultry, fish, soybean and canola oils, and dietary supplements. CoQ10 plays an integral part in generating intracellular adenosine triphosphate (ATP) within the mitochondrial inner membrane and in quenching intracellular oxidative stress. 12 During doxorubicin treatment, there is an acute rise in plasma CoQ10, 14 and following treatment there is a marked decrease in the CoQ10 content of cardiac and skeletal muscle. 15 The decrease in CoQ10 within muscle tissue is caused when doxorubicin reduces to its semiquinone and the semiquinone displaces CoQ10 in the cardiac myocyte mitochondrial inner membrane. Thus, it has been proposed that CoQ10 supplementation prior to and during doxorubicin treatment may prevent oxidative-stress induced cardiotoxicities. 12
Although CoQ10 is not known to be a strong chelator nor an enzyme inhibitor or inducer, we hypothesized a possible drug-drug interaction effect in doxorubicin treated patients because the antioxidant properties of compounds such as CoQ10 could interfere with the pro-oxidant actions of chemotherapeutic agents.16,17 We previously conducted an in vitro study that supported the hypothesis that CoQ10 does not alter the cytotoxicity of doxorubicin, 18 however that single study is not conclusive. No prospectively conducted, placebo-controlled trials have evaluated the safety of CoQ10 administration during doxorubicin treatment and whether it confers a protective effect against cardiotoxicity in early stage breast cancer patients.
To begin to fill this gap, we conducted a phase I randomized, placebo-controlled, cross-over study of intra-patient differences in doxorubicin and its active metabolites, comparing patients with and without administration of CoQ10, and to screen for effects associated with CoQ10 administration. We hypothesized that CoQ10 administration during doxorubicin treatment would be safe and would not affect concentrations of doxorubicin active metabolites and total antioxidant capacity (TAC). Measuring difference in both doxorubicin concentrations and TAC may be able to address some of the concerns of use of antioxidant supplements during chemotherapy.
Methods
Participants
Participants were screened and recruited from the outpatient Breast Medical Oncology Clinic at Columbia University Medical Center (CUMC) between December 2010 and August 2012 with the target of a maximum of 18 evaluable patients. Eligible participants included women aged 21 years or older who spoke English and/or Spanish, were diagnosed with stage I-III breast cancer, and were scheduled to receive at least 4 cycles of dose-dense (every 2 weeks) doxorubicin therapy in the neoadjuvant or adjuvant setting. Patients receiving neoadjuvant dose-dense doxorubicin therapy were permitted to have prior trastuzumab and/or taxane chemotherapy. Patients receiving adjuvant dose dense doxorubicin therapy were required to have single lumen implanted venous access device (i.e., single port) if they had a history of unilateral breast cancer and double lumen implanted venous access device (i.e., double port) if they had a history of bilateral breast cancer. Participants were required to have an ECOG performance status ≤ 2 (Karnofsky > 60%), normal organ and marrow function prior to initial doxorubicin/cyclophosphamide treatment, and normal left ventricular ejection fraction (LVEF) via multigated acquisition scan (MUGA) or echocardiogram. The institutional lower limit for a normal LVEF was ≥50%.
Exclusion criteria included history of CoQ10 supplement use within 30 days of initiating study drug, history of anthracycline use or radiation therapy in the previous 5 years, uncontrolled or significant co-morbid illness, pregnant, breast-feeding, or planning to become pregnant during the study. Patients were excluded if they experienced any significant treatment toxicities related to first or second dose of doxorubicin/cyclophosphamide chemotherapy or biologic therapy that did not resolve to less than a CTCAE 3.0 grade 3 non-hematologic toxicity. Patients were excluded if they were using any other investigational agent, or had severe medical, psychological, or sociological conditions that might interfere with the participant’s ability to follow the protocol or achieve study objectives. Patients were also excluded if they reported use of an over-the-counter nutritional vitamin greater than 5 times the recommended dietary allowance, any form of antioxidant supplements, U.S. Food and Drug Administration (FDA)-approved cardioprotective drugs (i.e., dexrazoxane), or use of warfarin. Due to the use of opaque coloring in a fish-based softgel shell, additional exclusion criteria included titanium dioxide allergy, fish allergy, reporting a tilapia dietary restriction, or following Kosher dietary practices.
A baseline questionnaire collected data on demographics, health history, and fruit and vegetable intake. As part of routine clinical care, prior to doxorubicin/cyclophosphamide treatment all participants received routine blood tests to evaluate organ and marrow function and an echocardiogram to assess LVEF. Prior to enrollment, premenopausal women were screened for pregnancy via urine or serum pregnancy test. Organ and marrow function tests were conducted prior to study initiation. Prior to beginning study drug, blood was collected to measure trough CoQ10 concentrations and total antioxidant capacity. Baseline symptoms were assessed during prior to receiving cycle 2 doxorubicin/cyclophosphamide treatment.
The CUMC Institutional Review Board approved the protocol and informed consent process (initial IRB approval September 2, 2009, CUMC IRB-AAAD8521). The study was administered in English and Spanish. All participants provided written informed consent.
Study Drug
CoQ10 softgel capsules and a matching rice bran oil placebo were manufactured by CanCap Pharmaceutical Ltd. (Richmond, BC, Canada) and distributed by Thorne Research (Sandpoint, ID). Softgel capsule shells were manufactured from fish (tilapia gelatin made from the skins), and softgel shells were rendered opaque using titanium dioxide. CanCap Pharmaceutical holds a Site License and Establishment License from Health Canada. Thorne Research adheres to FDA recommended Current Good Manufacturing Processes (CGMPs) for dietary supplements. CoQ10 was administered in 150 mg capsules with food. The planned dosing at the 3 dose levels was as follows: 300 mg dose level, two 150 mg capsules once per day with food; 600 mg dose level, two 150 mg capsules 2 times per day with food; and 1200 mg dose level, four 150 mg capsules 2 times per day with food. The placebo was administered with the matching number of capsules and timing. CoQ10 and placebo capsules were dispensed by the CUMC Research Pharmacy. The trial was conducted under IND# 105119.
Study Design
Randomization. The study schema is depicted in Figure 1. In this phase I trial, a randomized, cross-over design was used to account for potential differences in cycle effects. At each dose level, 6 participants were planned to be randomized to Arm A (n = 3, Cycle 3 with CoQ10, followed by Cycle 4 with placebo) or Arm B (n = 3, Cycle 3 with placebo, followed by Cycle 4 with CoQ10); however, only a total of 7 patients were accrued. Participants, study staff, and investigators were blinded to randomization assignment; only the research pharmacy knew the study assignment. The randomization list was developed by the trial biostatistician using a random number generator and then provided to the research pharmacy.

Design of randomized, controlled cross-over study design.
Routine doxorubicin/cyclophosphamide treatment. Dose-dense doxorubicin infusions were administered every 2 weeks for 4 cycles. For the 2 doxorubicin cycles administered during this study (Cycles 3 and 4), doxorubicin therapy was administered via infusion at 60 mg/m2 over 30 minutes via an I.V. push, followed by a 45-minutes infusion of cyclophosphamide 600 mg/m2. This dose remained fixed for all cycles unless toxicity necessitated any dose reduction. Anti-emetics were used as needed. Routine hematological assessment was performed 0-3 days prior to Cycles 2, 3 and 4. In order to receive dose-dense doxorubicin/cyclophosphamide neoadjuvant/adjuvant infusion (administered in all cycles) and the subsequent CoQ10/placebo study drug (study drug was administered following Cycle 2 for pharmacokinetic draws during Cycle 3 infusion and study drug was administered following Cycle 3 for pharmacokinetic draws during Cycle 4 infusion), the following parameters had to be met: absolute neutrophil count (ANC) ≥ 1500/µl before starting Cycle 1 and ANC ≥ 1000/µl prior to Cycles 2, 3 and 4; platelets ≥ 100 000/µl; total bilirubin ≤ 1.5 X institutional upper limit of normal (ULN); aspartate transaminase (AST) (or serum glutamic-oxaloacetic transaminase, SGOT)/alanine aminotransferase (ALT) (or serum glutamic-pyruvic transaminase, SGPT) ≤ 2.5 X institutional ULN; and serum creatinine within normal institutional limits. In order to continue participation in the study, up to 3 weeks were allowed between Cycles 2 and 3 and between Cycles 3 and 4, based on the discretion of the medical oncologist.
Coenzyme Q10 administration and adherence. Beginning on the morning of Cycle 2 Day 3 (11-15 days prior to Cycle 3), participants began taking the study drug (either CoQ10 or placebo) and continued taking the study drug through the morning of their Cycle 3 infusion (Day 1 Cycle 3). Participants were then crossed-over to the alternative condition. On the morning of Cycle 3 Day 3, participants began taking the study drug (either CoQ10 or placebo) and continued taking the study drug through the morning of their Cycle 4 infusion (Day 1 Cycle 4). Study drug bottles were collected prior to the infusions and remaining capsules were counted to determine adherence. To ensure that participants understood the dosing schedule and to measure adherence to study drug, participants were provided study drug diaries and signed drug accountability forms when study drug was dispensed and collected. If participants were not ≥80% adherent, they were removed from the study due to non-adherence.
Blood sample collection. Participants remained at CUMC for 8 hours after the infusion and returned to CUMC the following morning for a 24-hours blood draw. A limited sampling strategy was used for pharmacokinetic analysis during Cycles 3 and 4 of doxorubicin/cyclophosphamide therapy and concurrent study drug administration, similar to a previously described limited sampling strategy for doxorubicin metabolism.19,20 Blood was collected in EDTA tubes prior to the start of the infusion (PK1), at the end of the doxorubicin infusion (PK2), and at 30 minutes (PK3), 1 (PK4), 2 (PK5), 4 (PK6), 6 (PK7), and 24 (PK8) hours after the end of the infusion, Blood was collected in heparinized tubes prior to the start of the infusion and at approximately 24 hours (at the same time as PK8) to assess total antioxidant capacity (TAC) and CoQ10 concentrations (CoQ10 was only assessed at PK1 and PK8). The timing of PK2-8 collections were ±30 minutes of the times stated above. Blood samples were immediately placed on ice after collection and immediately centrifuged at 3000 RPMs for 10 minutes at 4°C. Plasma samples were transferred into cryovials. Cryovials were stored at −20°C for 24 hours if needed prior to being transferred to a −80°C freezer.
Side effect and adverse event evaluation. Side effects and adverse events were measured at each clinical visit using NCI Common Terminology Criteria for Adverse Events v3.0 (CTCAE). Patients on statins, hypoglycemic agents, and insulin were monitored for potential interactions with CoQ10, including lowering of cholesterol/triglycerides, blood glucose, and insulin dose requirements. Safety profiles were assessed by recording the nature, severity, frequency, timing, and duration of any adverse events and their relationship to treatment.
Laboratory Analyses of Blood Sample
The CUMC Core Lab performed laboratory analyses included plasma concentrations of doxorubicin and doxorubicinol measured with high-performance liquid chromatography (HPLC), using methods previously described. 21 Plasma samples were assessed for TAC using a TAC Assay Kit from MBL International Corporation (Woburn, MA). Blood CoQ10 concentrations were measured by high-performance liquid chromatography with electrochemical detection (HPLC-EQ) by use of a reverse-phase column and isocratic mobile phase, as previously described. 22
Pharmacokinetic Analyses
Non-compartmental pharmacokinetic parameters of doxorubicin and its metabolites were determined using WinNonlin software version 5.2 (Pharsight Corporation, Mountain View, CA, USA). Pharmacokinetic parameters were calculated for each patient and each occasion included area under the serum concentration time curve from t = 0 h until 24 hours (AUC0-24h), the maximum concentration reached (Cmax), the time to reach Cmax (tmax) and the terminal half-life (t1/2). These parameters were calculated for the parent compound doxorubicin and for the doxorubicinol metabolites.
Statistical Analyses
We compared the mean age and average daily dietary intakes between study arms using a t-test, and compared the distribution of race, menopausal status, tumor characteristics, chemotherapy regimen and antioxidant use using Fisher’s exact test. Paired t-tests were used to compare the non-compartmental pharmacokinetic parameters of doxorubicin and doxorubicinol during periods with and without CoQ10 administration (treatment effect), and between Cycle 3 and Cycle 4 of doxorubicin infusion (time effect). Treatment effect and time effect were assessed for serum CoQ10 concentration and TAC using paired t-tests. Data analyses, summary statistical tables and concentration versus time curves were generated using R (version 3.3.1, The R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
Between December 2010 and August 2012, a total of 6 women were randomized to receive 300 mg/day CoQ10 (n = 3 in Arm A, n = 3 in Arm B) during 1 cycle of doxorubicin. One patient was randomized to receive 600 mg/day CoQ10 but was discontinued due to non-adherence; of note, the patient indicated that the reason for non-adherence was not due to intolerance of the capsules. The study was closed prior to achieving the target 18 patients due to poor accrual. At enrollment, the average age was 57.7 years old (SD = 14.9 years, range = 30-75 years), and 50% were non-Hispanic white, 33.3% were Hispanic and 16.7% were African American. There was no between arm difference in demographic and clinical, and lifestyle factors (Table 1).
Patient Characteristics (n=6).

Arm A: CoQ10 administered in Cycle 3 followed by placebo in Cycle 4; Arm B: Placebo administered in Cycle 3 followed by CoQ10 in Cycle 4.
Means were tested using t-tests and percentages were tested using Fisher’s exact test.
Abbreviations: SD, standard deviation; ER, estrogen receptor; PR, progesterone receptor; HER2, human epidermal growth factor receptor 2; AC, Adriamycin, and cyclophosphamide; T, Taxol.
Safety
No Grade 3 or Grade 4 adverse events were observed; the most common adverse events were Grade 1-2 alopecia (n = 3, 50%), constipation (n = 3, 50%), diarrhea (n = 3, 50%), abnormal ear, nose and throat examination (n = 3, 50.0%), abnormal hemoglobin (n = 3, 50%) and nausea (n = 3, 50%) (Supplemental Table 1). No adverse effects were “definitely related” to CoQ10 treatment; 5 events were considered “possibly related” to CoQ10 treatment. Treatment with CoQ10 did not result in an increased frequency of doxorubicin-induced adverse events, and no participants discontinued the study because of adverse events.
Doxorubicin Pharmacokinetics
Changes in doxorubicin concentrations and its metabolite doxorubicinol were not appreciably different with and without CoQ10 administration (Figure 2). Table 2 summarizes the mean and median non-compartmental pharmacokinetic parameters for doxorubicin and doxorubicinol in patients treated with CoQ10 and placebo, respectively. Mean AUC (0-24h), Cmax, Tmax and half-life of doxorubicin were generally similar in patients treated with doxorubicin + CoQ10 300 mg/day and patients treated with doxorubicin + placebo. CoQ10 did not change the pharmacokinetic parameters of doxorubicinol or the ratio of doxorubicin AUC (0-24 hours) to doxorubicinol AUC (0-24 hours). Furthermore, there was no statistically significant difference in pharmacokinetic parameters between cycle 3 and 4 of doxorubicin infusion (Supplemental Table 2).
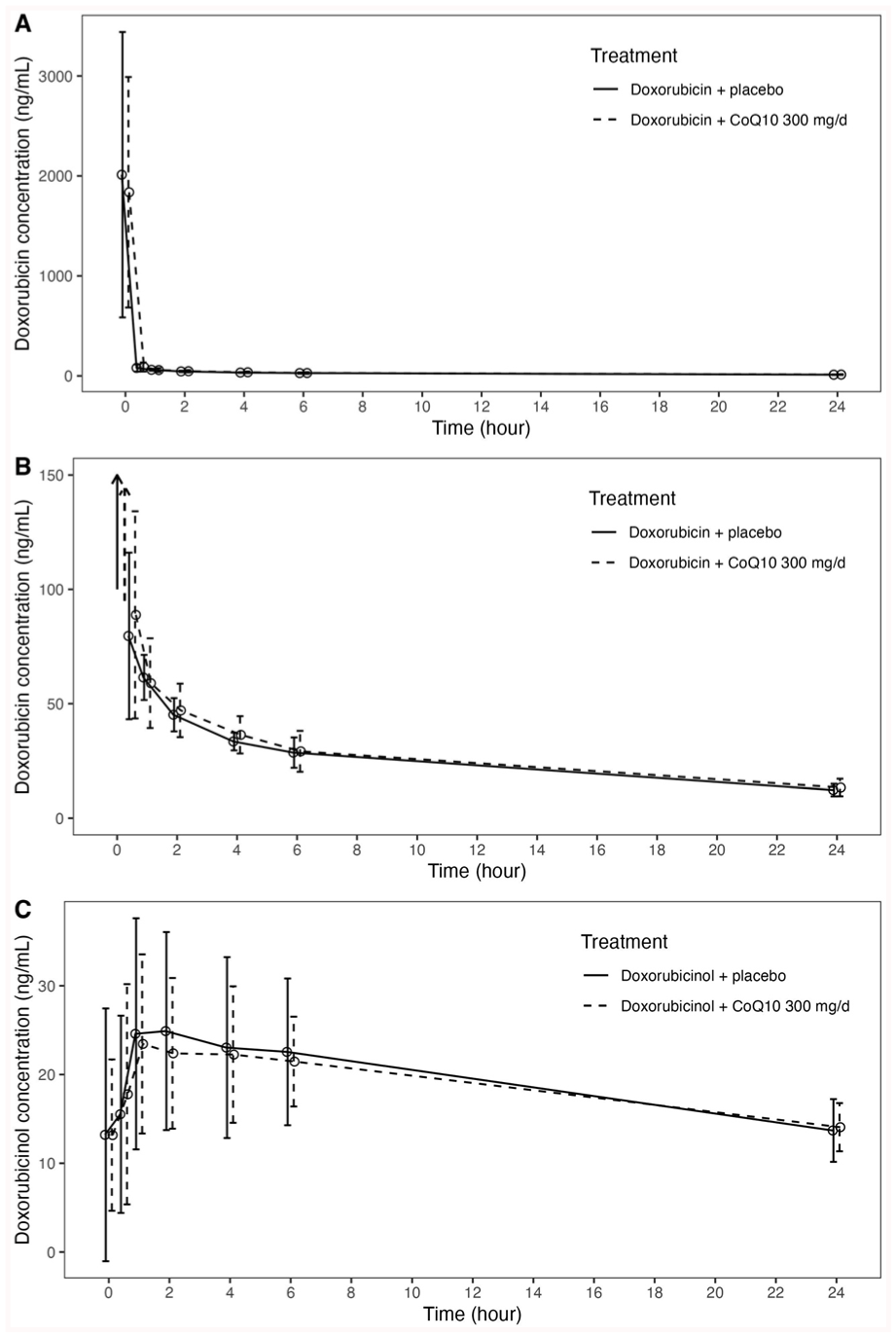
Mean and 95% confidence intervals are shown for doxorubicin (panels A and B; in panel B, the Y-axis is restricted to 150 ng/mL for comparability, with arrows indicating baseline values beyond the Y-axis) and its metabolite, doxorubicinol (panel C), for the 24 hours following doxorubicin infusion (n = 6).
Pharmacokinetic parameters comparing breast cancer patients undergoing 1 cycle of doxorubicin with CoQ10 and placebo (n = 6).
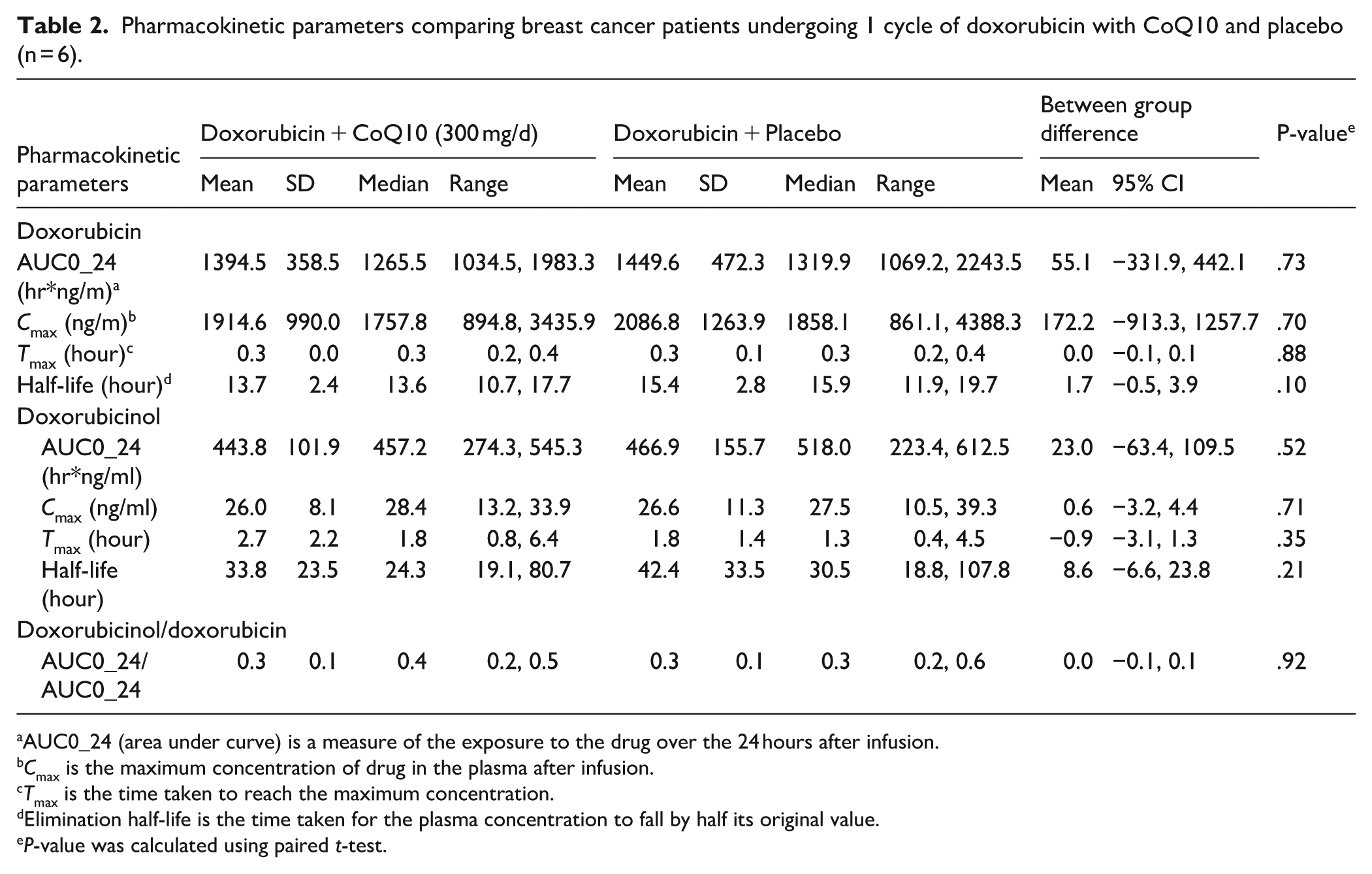
AUC0_24 (area under curve) is a measure of the exposure to the drug over the 24 hours after infusion.
Cmax is the maximum concentration of drug in the plasma after infusion.
Tmax is the time taken to reach the maximum concentration.
Elimination half-life is the time taken for the plasma concentration to fall by half its original value.
P-value was calculated using paired t-test.
Serum Total Antioxidant Capacity (TAC) and CoQ-10 Concentration
Table 3 shows the changes in serum CoQ10 and TAC concentrations after 1 cycle of doxorubicin in patients with and without CoQ10 administration. A statistically significantly greater increase in serum CoQ10 concentration was noted in the patients treated with doxorubicin + CoQ10 compared to patients treated with doxorubicin + placebo (mean ± SD change in serum CoQ10 from baseline: doxorubicin + CoQ10 300 mg/day: 1.6 ± 0.9 µg/ml, doxorubicin + placebo: −0.01 ± 0.3 µg/ml, P = .01). Serum CoQ10 concentrations were not different between Cycle 3 and Cycle 4 (Supplemental Table 3). There was no statistically significant change in TAC between the patients treated with doxorubicin + CoQ10 and doxorubicin + placebo. However, TAC was statistically significantly lower after Cycle 4 of doxorubicin compared to Cycle 3 doxorubicin (mean ± SD TAC: Cycle 3 = 509.7 ± 37.2 nM, Cycle 4 = 482.4 ± 41.4 nM, P = .05; Supplemental Table 3).
Changes in Total Antioxidant Capacity and CoQ10 Concentrations after 1 Cycle of Doxorubicin Infusion (n = 6).

Discussion
In this phase I randomized, placebo-controlled, cross-over study, CoQ10 at 300 mg/day was well tolerated and did not change the pharmacokinetics of doxorubicin. No adverse effects were observed. Patients receiving CoQ10 with doxorubicin experienced a statistically significant approximate doubling of serum CoQ10 concentration, or an average increase of 1.6 µg/mL.
Many breast cancer treatments are known to carry a substantial risk of adverse long-term treatment related effects.7 -10 Antioxidant supplements are an unproven yet widely used approach intended to reduce side effects, used by 45%-80% of breast cancer patients after diagnosis.16,23 -25 However, antioxidants may counteract the desired effects of cancer treatment 26 and moreover their effects on cancer recurrence or mortality are not well understood.16,27 Few randomized, clinical trials have examined effects of antioxidant supplement intake during breast cancer treatment, which motivated this study. 16 An observational study (SWOG S0221A-ICSC, PI: Ambrosone) examining effects of antioxidant intake on treatment outcomes nested in a therapeutic trial of adjuvant doxorubicin, cyclophosphamide and paclitaxel for breast cancer, recently reported that use of antioxidants, including CoQ10, before and during treatment was associated with an increased risk of recurrence.28,29 If there is decreased efficacy from doxorubicin when co-administered with CoQ10, based on our findings this does not seem to be related to a change in systemic exposure to doxorubicin or its primary metabolite.
We only tested CoQ10 dosage at 300 mg/d, therefore we cannot report on the pharmacokinetics of higher dose CoQ10 during treatment for breast cancer patients. A daily dose of 300 mg per day for 11 days has been shown to raise plasma CoQ10 concentrations by 300%-400% whereas our study showed less dramatic effects of this dose level on CoQ10 concentrations. 30 An NCI-sponsored multi-center trial tested the effects of 300 mg CoQ10 per day on self-reported fatigue over 24 weeks of treatment among women (n = 236) treated with adjuvant chemotherapy. 31 The study reported significant increase in plasma CoQ10 level in patients receiving CoQ10 supplements but no significant improvement in fatigue. In the current study, we planned to test 3 daily dose levels of CoQ10: 300, 600, and 1200 mg/d. These doses were chosen after reviewing the literature on the use of CoQ10 in oncology, cardiovascular, and neurological patient populations. In oncology clinical studies, daily dosages of CoQ10 for preventing doxorubicin-induced cardiotoxicity ranged from 30 to 120 mg/d.32 -40 Cardiovascular patient populations typically use doses of CoQ10 ranging from 30 to 300 mg/d,41,42 intake via dietary supplements is generally considered to be safe at doses up to 1200 mg per day 30 and high doses of CoQ10 (up to 3000 mg/d) are being investigated to treat neurodegenerative disorders such as Huntington’s Disease and Parkinson’s Disease.43 -53 None of the studies provided rationale for doses used, and the studies showed limited effects, if any.
In our study, all patients in both arms had normal level of CoQ10 at baseline (mean = 1.0 μmol/l), within the adult reference interval of 0.5-1.7 μmol/l.54 -56 The level of CoQ10 was significantly increased by approximately 2-fold after 300 mg/day coenzyme Q10 supplementation. The effect of CoQ10 supplement on serum CoQ10 level was comparable when administered during Cycle 3 and 4 of cancer treatment. The amount of increase was lower to that reported in previous studies of dietary supplementation with 300-900 mg/day CoQ10 and 200 mg/day ubiquinol, in which 3 to 5-fold increases in serum CoQ10 were observed.30,57 -59 The greater effect of CoQ10 reported in previous studies was likely due to the lower baseline CoQ10 levels (0.5-0.8 μmol/l), higher CoQ10 dosage (up to 900 mg/day) and longer duration of CoQ10 supplementation (4-12 weeks).
The leading mechanistic hypothesis for doxorubicin-induced cardiotoxicity is that doxorubicin differentially increases reactive oxygen species (ROS) within cardiac myocyte mitochondria, as compared to other tissues.12,60,61 We did not observe statistically significant differences in total antioxidant capacity, a measure of ROS, between groups who did and did not receive CoQ10. We cannot discern if this lack of difference is due to the small sample size and lack of statistical power, if the 300 mg per day dose was too low to reduce oxidative stress, if there is a discrepancy between system and intracellular total antioxidant capacity, or if in fact there is no true difference. In vitro studies have shown that administering CoQ10 either before or during doxorubicin administration can prevent doxorubicin-induced changes including cardiac beat inhibition, 62 increase in lipid peroxidation as measured by malondialdehyde, 63 and reduced cardiac contractile tension. 64 Animal studies suggest that the impact of doxorubicin on mitochondria is cardioselective, that CoQ10 can prevent doxorubicin-induced mitochondrial damage, and that CoQ10 can prevent the progression of cardiomyopathy induced by doxorubicin.12,65 Clinical studies of CoQ10 administered before or during doxorubicin treatment have shown that CoQ10 prevented changes in LVEF, 32 systolic time intervals (STI), 33 fractional shortening, 66 intraventricular septal wall thickening, 66 cardiac output,34,67 and EKG changes. 35 In addition, CoQ10 allowed for increased doses of doxorubicin to be administered. 32 However, these studies were small and included patients of mixed tumor types. No studies have focused on early stage breast cancer patients, for whom doxorubicin treatment is standard neoadjuvant and adjuvant therapy.
Strengths and Limitations
Strengths of the study include prospective data collection in a randomized, controlled, cross-over design with a well-defined single pharmacologic agent. However, there are important limitations to consider. The major study limitation is the small sample size, which led to relative imprecision of estimates, and which also made it impossible to exclude safety concerns. The small sample size was due to the difficulty of enrolling participants as the protocol was burdensome to patients due to time, travel and inconvenience. In the adjuvant setting it is challenging to recruit patients to a trial of a dietary supplement as it can be burdensome to follow the study protocol, and dietary supplements are readily available over the counter. Moreover, this trial required patients to enroll prior to their first cycle of chemotherapy, which is often a period of high stress and anxiety for patients. Future trials of this nature should actively employ methods to expand eligibility criteria and reduce participant burden. It is possible that CoQ10 effects may have been difficult to detect in light of natural between-patient and intrapersonal variations of measured parameters. We lacked data on cardiac biomarkers and circulating CoQ10 metabolites, which is another weakness that limited our ability to examine the effects of CoQ10 on level of cardiotoxicity. There are limited data to guide CoQ10 dosing and future studies should assess differences in CoQ10 AUC and pharmacokinetics in order to better guide CoQ10 dosing. Finally, the goal of this trial was to assess changes in doxorubicin pharmacokinetics following CoQ10 administration, but did not assess the pharmacodynamics between CoQ10 and doxorubicin. Pharmacodynamics are of significance because the concern is that CoQ10 may counter the desired therapeutic effect of doxorubicin. Future trials should include pharmacodynamic analyses.
Conclusion
In conclusion, these findings showed that CoQ10 at 300 mg/day was well-tolerated and did not change pharmacokinetics of 1 cycle of doxorubicin in breast cancer patients. However, our results do not warrant clinical recommendation for CoQ10, but indicate that this is a possible area of study. Future studies will need to balance the potential beneficial protective effect against doxorubicin cardiotoxicity in early stage breast cancer patients with the potential risk of use of antioxidants during treatment.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251388014 – Supplemental material for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment
Supplemental material, sj-docx-1-ict-10.1177_15347354251388014 for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment by Heather Greenlee, Katherine D. Crew, Matthew Maurer, Kevin Kalinsky, Serge Cremers, Ali Naini, Wei Yann Tsai, Zaixing Shi, Frances Brogan and Dawn L. Hershman in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354251388014 – Supplemental material for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment
Supplemental material, sj-docx-2-ict-10.1177_15347354251388014 for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment by Heather Greenlee, Katherine D. Crew, Matthew Maurer, Kevin Kalinsky, Serge Cremers, Ali Naini, Wei Yann Tsai, Zaixing Shi, Frances Brogan and Dawn L. Hershman in Integrative Cancer Therapies
Supplemental Material
sj-docx-3-ict-10.1177_15347354251388014 – Supplemental material for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment
Supplemental material, sj-docx-3-ict-10.1177_15347354251388014 for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment by Heather Greenlee, Katherine D. Crew, Matthew Maurer, Kevin Kalinsky, Serge Cremers, Ali Naini, Wei Yann Tsai, Zaixing Shi, Frances Brogan and Dawn L. Hershman in Integrative Cancer Therapies
Supplemental Material
sj-docx-4-ict-10.1177_15347354251388014 – Supplemental material for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment
Supplemental material, sj-docx-4-ict-10.1177_15347354251388014 for Phase I Randomized, Placebo-Controlled, Cross-Over Dose-Finding Study of Coenzyme Q10 on Doxorubicin Pharmacokinetics during Breast Cancer Treatment by Heather Greenlee, Katherine D. Crew, Matthew Maurer, Kevin Kalinsky, Serge Cremers, Ali Naini, Wei Yann Tsai, Zaixing Shi, Frances Brogan and Dawn L. Hershman in Integrative Cancer Therapies
Footnotes
Acknowledgements
We thank Kirsten Abildskov for her technical work on the Doxorubicin assay and Franco Barsanti for his assistance preparing the IND application. We acknowledge Alan Butcher at Thorne Research (Sandpoint, ID) for coordinating provision of the CoQ10 supplement.
Author Note
The authors confirm that the Principal Investigator for this paper is Heather Greenlee, ND, PhD, MPH, and that as Director of the Breast Medical Oncology Program at Columbia University Irving Medical Center, Dawn Hershman, MD, MS had direct clinical responsibility for patients.
Ethical Considerations
The CUMC Institutional Review Board approved the protocol and informed consent process (initial IRB approval September 2, 2009, CUMC IRB-AAAD8521).
Consent to Participate
All participants provided signed written consent to participate in this research.
Author Contributions
All authors reviewed and approved the final manuscript. Specific contributions are: Greenlee: Designed study, Conducted analyses, Obtained funding, Overall oversight of research. Crew: Designed study, Enrolled participants. Maurer: Designed study, Enrolled participants. Kalinsky: Enrolled participants. Cremers: Designed study, Conducted analyses. Naini: Designed study, Conducted analyses. Tsai: Designed study, Conducted analyses. Shi: Conducted analyses. Brogan: Nil. Hershman: Designed study, Enrolled participants, Obtained funding, Overall oversight of research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute (K23CA141052 to Dr. Greenlee) and the National Center for Advancing Translational Sciences through grant number UL1TR001873.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data and analysis code will be made available under reasonable request, and in accordance with limitations of informed consent language, through correspondence with the authors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
