Abstract
Background/hypotheses. Doxorubicin is a standard adjuvant therapy for early-stage breast cancer, and it significantly improves disease-free and overall survival. However, 3% to 20% of breast cancer patients develop chronic cardiomyopathic changes and congestive heart failure because of doxorubicin therapy. Doxorubicin-induced cardiotoxicity is thought to be due to the increased generation of reactive oxygen species within cardiac myocyte mitochondria. Coenzyme Q10 (CoQ10) is a lipid-soluble antioxidant that may protect against mitochondrial reactive oxygen species and thus prevent doxorubicin-induced cardiotoxicity. Despite the potential benefits of CoQ10 in preventing cardiotoxicity, it is not known if CoQ10 diminishes the antineoplastic effects of doxorubicin therapy. Study design. In vitro cell culture experiments. Methods. Breast cancer cell lines (MDA-MB-468 and BT549) were tested for their ability to uptake exogenous CoQ10 using high-performance liquid chromatography. Breast cancer cell lines were then treated with doxorubicin and a range of CoQ10 concentrations to determine the effect of CoQ10 on doxorubicin’s cytotoxicity. Results. This study demonstrated that intracellular and mitochondrial CoQ10 concentrations increased substantially as higher exogenous concentrations were administered to breast cancer cells. CoQ10 had no effect on the ability of doxorubicin to induce apoptosis or inhibit growth or colony formation in both the cell lines tested when applied over a wide dose range, which encompassed typical basal plasma levels and plasma levels above those typically achieved by supplemented patients. Conclusion. The clinical testing of CoQ10 as a supplement to prevent doxorubicin-induced cardiotoxicity requires confidence that it does not decrease the efficacy of chemotherapy. These results support the hypothesis that CoQ10 does not alter the antineoplastic properties of doxorubicin. Further in vivo studies, as well as combination chemotherapy studies, would be reassuring before a large-scale clinical testing of CoQ10 as a cardioprotective drug.
Introduction
Doxorubicin (adriamycin), an anthracycline antibiotic, plus cyclophosphamide is a standard combination for adjuvant therapy for early-stage breast cancer. This combination significantly improves disease-free and overall survival. Despite these benefits, 3% to 20% of doxorubicin-treated breast cancer patients develop chronic cardiomyopathic changes and congestive heart failure because of doxorubicin therapy. 1 Currently, no treatments are available to prevent this cardiotoxicity that have been proven to be safe and effective in breast cancer patients receiving the current standard dose of doxorubicin. A recent Cochrane review suggests that dexrazoxane may decrease the risk of cardiac toxicity from anthracyclines, but the majority of studies were not conducted in breast cancer patients. 2 Effective agents need to be identified as being both safe and effective for the prevention of doxorubicin-induced cardiac toxicity.
The differential toxicity of doxorubicin to cardiac myocytes as compared with other tissues is hypothesized to be due to an interaction between doxorubicin and a mitochondrial nicotinamide adenine dinucleotide dehydrogenase enzyme specific to cardiac myocytes. 3 In all cells, the mitochondria are the site of adenosine triphosphate generation via the electron transport chain. Coenzyme Q10 (CoQ10, also known as ubiquinone) is located within the mitochondrial inner membrane and plays an integral role in generating intracellular adenosine triphosphate by serving as an electron acceptor within the electron transport chain. CoQ10 also quenches intracellular oxidative stress by accepting electrons.
In most tissues, the mitochondrial inner membrane serves as a barrier to pro-oxidant doxorubicin metabolites. However, the mitochondrial inner membrane of cardiac myocytes uniquely contains a specific form of mitochondrial nicotinamide adenine dinucleotide dehydrogenase that reduces doxorubicin to its semiquinone form and subsequently generates high concentrations of reactive oxygen species (ROS) within the mitochondrial cytosol. 3 Doxorubicin semiquinones donate electrons to oxygen, which generates superoxide radicals, and are also auto-oxidized and cleaved to form aglycones. The aglycones displace CoQ10 from the mitochondrial inner membrane and serve as electron acceptors from complex I and complex II (which is a function of CoQ10). However, instead of transferring electrons to complex III, as CoQ10 does, the aglycones transfer electrons to molecular oxygen, resulting in the formation of superoxide radicals. The cumulative high levels of ROS cause irreversible damage to mitochondrial DNA, which blocks the regenerative capability of the organelle and leads to cardiac myocyte apoptosis or necrosis. Animal and human studies support this proposed mechanism of doxorubicin-induced cardiotoxicity. 3 During doxorubicin treatment, an acute rise in plasma CoQ10 4 is followed by a marked posttreatment decrease in the CoQ10 content of cardiac and skeletal muscle. 5 Damage to the mitochondria of cardiac myocytes is one of the earliest and most prominent histological findings of doxorubicin-induced cardiomyopathy.
It has been hypothesized that dietary supplementation of CoQ10 prior to and during doxorubicin treatment can prevent doxorubicin-induced cardiotoxicity by preventing or slowing the displacement of CoQ10 by doxorubicin metabolites. 3 CoQ10 is a lipid-soluble antioxidant that is synthesized in the mitochondria and in most intracellular membranes and is ingested via the diet. Intake of CoQ10 via dietary supplements is generally considered to be safe at doses of up to 1200 mg per day. 6 A daily dose of 300 mg per day for 11 days has been shown to raise plasma CoQ10 concentrations by 300% to 400%. 7
In vitro studies have shown that administering CoQ10 either before or during doxorubicin administration can prevent doxorubicin-induced changes including cardiac beat inhibition, increase in lipid peroxidation, and reduced cardiac contractile tension. 3 Animal studies suggest that the impact of doxorubicin on mitochondria is cardioselective and that CoQ10 can prevent doxorubicin-induced mitochondrial damage and progression of cardiomyopathy. 3 Clinical studies of CoQ10 administered before or during doxorubicin treatment have shown that CoQ10 prevented changes in left ventricular ejection fraction, systolic time intervals, fractional shortening, intraventricular septal wall thickening, cardiac output, and electrocardiograms. 3 In addition, CoQ10 allowed for increased doses of doxorubicin to be administered. 3 However, these studies were small and included patients with mixed tumor types. No studies have focused on aggressive early-stage breast cancer, in which doxorubicin-containing chemotherapy regimens are a standard of care.
In addition to the cytotoxic mechanisms of topoisomerase II inhibition, DNA intercalation, and inhibition of DNA-dependent RNA polymerase, doxorubicin treatment also produces DNA-damaging cytotoxic oxygen free radicals. 8 Previous in vitro and animal studies in a lymphoma model suggested that high doses of vitamin C may diminish doxorubicin-induced cytotoxicity, perhaps by preserving mitochondrial membrane potential. 9 Another study found that blockade of CoQ10 synthesis increased the sensitivity of cancer cells to the chemotherapeutic agent camptothecin. 10 Despite the potential benefits of CoQ10 in preventing doxorubicin-induced cardiotoxicity, there is concern that CoQ10 may decrease the desired pro-oxidant treatment effects of doxorubicin. One previous study assessed the effects of CoQ10 on the in vitro efficacy of doxorubicin. CoQ10 increased the cytotoxic activity of doxorubicin in L-1210 leukemia cells and had no effect in Ehrlich ascites carcinoma cells. 3 Similar preclinical studies examining the effects of CoQ10 in breast cancer models are lacking. Here we report on a series of in vitro breast cancer cell culture experiments examining the effects of CoQ10 on doxorubicin cytotoxicity.
Methods
Reagents
MDA-MB-468 and BT549 breast cancer cell lines were obtained from American Type Culture Collection (ATCC; Manassas, VA) and cultured in DMEM and RPMI media, respectively, supplemented with 10% fetal bovine serum and penicillin and streptomycin. Doxorubicin hydrochloride was obtained from the medical center’s pharmacy and diluted in water. Powdered fermented CoQ10 was obtained via Thorne Research (Sandpoint, ID; original source: Sinphar Group, I-lan, Taiwan) and dissolved in 1-propanol.
Coenzyme Q10 Quantification
Cells from both cell lines were grown to 80% confluency in 6-well dishes before treatment with CoQ10 and/or doxorubicin for 48 hours. All cells receiving both drugs were pretreated with CoQ10 for 24 hours prior to doxorubicin exposure. On the day of harvest for whole-cell quantification, cells were washed thrice with phosphate-buffered saline, trypsinized, and spun down. Cell pellets were stored at −20°C until CoQ10 level was quantified using a modified form of a previously established high-performance liquid chromotography (HPLC) method.11,12 For mitochondrial fractions, Mitosciences (Eugene, OR) Mitochondria Isolation Kit for Cultured Cells (MS852) was used to separate the mitochondrial portion of the cells before following the same HPLC protocol to quantify the CoQ10 levels. Immunoblotting was used to validate the separation of mitochondria. For all samples, the Bio-Rad DC Protein Quantification Microplate Assay protocol was used to quantify and normalize the amount of protein per sample.
Proliferation and Half Maximal Inhibitory Concentration Assays
Cells (1 × 104) were plated on 48-well dishes. After 16 to 24 hours, the cells were treated with the drug for 72 hours. For CoQ10-plus-doxorubicin studies, cells were incubated with CoQ10 for 24 hours before doxorubicin was added. On the day of harvest, the cells were stained with 0.05% crystal violet in 10% formalin, washed, and incubated with 10% acetic acid before absorbance was measured at 590 nm (BioTek, Winooski, VT). The half maximal inhibitory concentration (IC50) values were determined using the dose–response curve fitting model y = A + ((B − A)/(1 + ((C/x) D ))), designated as model 205 using XLfit4 with parameters A = 0 (min y value), B = 100 (max y value), C = IC50, and D = slope.
Colony Formation Assays
Cells (1 ×105 to 10 × 105) were plated on 6-well dishes and treated with the drug for 48 hours. CoQ10-plus-doxorubicin–treated cells were pre-incubated with CoQ10 for 24 hours. Cells were collected and plated in 500 to 1000 dilutions on fresh 6-well dishes. After 10 to 12 days, media were removed, and cells were fixed with cold methanol for 5 minutes before staining with 0.25% crystal violet in 10% formalin for 30 minutes. Cells were washed, and colonies were counted using ImageJ software (National Institutes of Health, Bethesda, MD).
Immunoblotting
Whole-cell lysates and mitochondrial fractionations were used in immunoblots, as indicated. Cleaved Caspase-3 (Asp175) antibody from Cell Signaling (Danvers, MA) was diluted 1:1000 in 5% wt/vol nonfat dry milk, 1× Tris-buffered saline, and 0.1% Tween-20 and incubated at 4°C overnight before incubation in anti-rabbit secondary diluted 1:5000 for 1 hour. COX IV antibody from Cell Signaling was diluted 1:1000 in 5% wt/vol nonfat dry milk, 1× Tris-buffered saline, and 0.1% Tween-20 and incubated at 4°C overnight before incubation in anti-rabbit secondary diluted 1:5000 for 1 hour.
Statistical Analysis
One-way analysis of variance was used to examine differences in CoQ10 concentration assessed using HPLC. T tests were used to examine differences in the cell proliferation assays. Analyses were conducted using StatPlus:mac LE.2009 (AnalystSoft, Alexandria, VA).
Results
Exogenous Coenzyme Q10 Increases Cellular Level of Coenzyme Q10
Triple-negative breast cancer cell lines MDA-MB-468 and BT549 were tested for their ability to uptake exogenous CoQ10 over a dose range of 0.09 to 9 µM (Figure 1). In humans, the standard supplementation dose raises plasma CoQ10 levels from 0.5 to 2 µM; breast cancer patients have a typical plasma level of 0.9 µM. 13 Both cell lines demonstrated that in vitro cellular CoQ10 concentration increases as higher exogenous concentrations are administered to cells. In MDA-MB-468 cells, adding 0.09 µM of CoQ10 increased the amount detected from 30.7 µg CoQ10/µg protein (when no CoQ10 was added) to 53.2 µg CoQ10. Adding 0.9 and 9 µM CoQ10 increased detected levels of CoQ10 even higher to 140.5 and 1265 µg CoQ10/µg protein, respectively. BT549 cells demonstrated a similar dose increase, with approximately 1400 µg CoQ10 detected per microgram of protein—more than a 15-fold increase from the 91 µg CoQ10 detected in the baseline cells.

Exogenous CoQ10 increases the cellular level of CoQ10 in breast cancer cells in vitro. MDA-MB-468 (A) and BT459 (B) triple-negative breast cancer cells were exposed to CoQ10 for 48 hours at concentrations of up to 9 µM CoQ10. HPLC was used to quantify CoQ10 levels in whole-cell lysates. Three separate experiments were conducted in triplicate. Error bars represent 1 standard deviation
Exogenous CoQ10 increases CoQ10 levels in the mitochondria of breast cancer cells in vitro, even in the presence of doxorubicin. To determine if exogenous CoQ10 was able to concentrate in the mitochondria of the breast cancer cells, cellular fractionation was performed to separate out the mitochondria before testing the levels of CoQ10 after exogenous treatment (Figure 2). In both cell lines, the highest treatment of CoQ10, 9 µM, caused a substantial increase in CoQ10 detected over the baseline, up to a third of the amount measured in the whole-cell lysates (Figures 2A and B). The level of CoQ10 was also tested after exposure to doxorubicin, alone and in combination with CoQ10 (Figures 2C and D). In both cell lines, the level of CoQ10 detected after treatment with doxorubicin alone was similar to the baseline CoQ10 levels. However, the 2 cell lines differed in their mitochondrial CoQ10 levels after the combined treatment with CoQ10 and doxorubicin. MDA-MB-468 cells had a substantially decreased level of mitochondrial CoQ10 in the presence of doxorubicin, decreasing the amount of CoQ10 detected by approximately two thirds (Figure 2E). This decrease in mitochondrial CoQ10 was not observed in BT549 cells, where the amount of CoQ10 detected was the same with or without doxorubicin (Figure 2F).
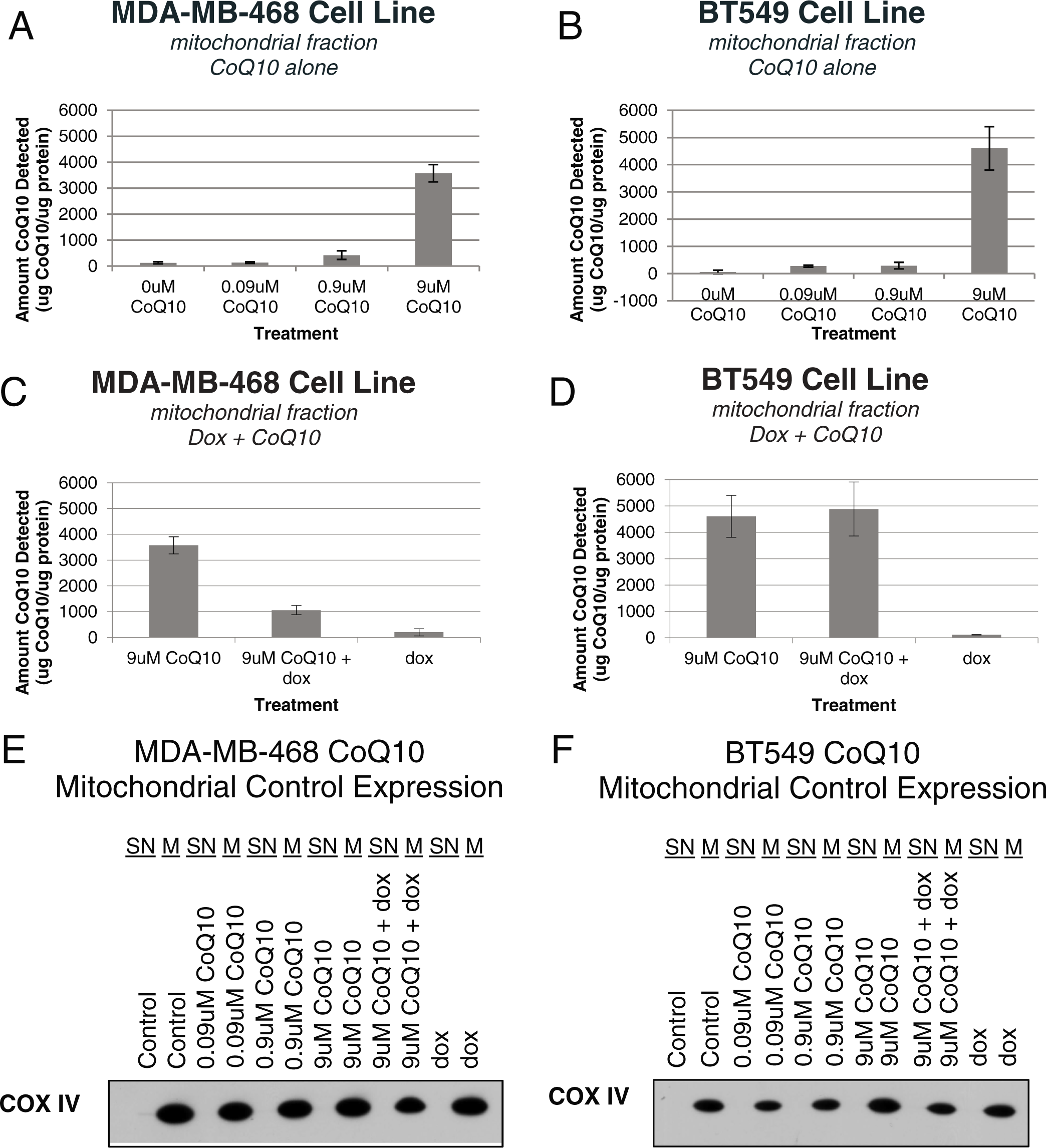
Exogenous CoQ10 increases CoQ10 levels in the mitochondria of breast cancer cells in vitro, even in the presence of doxorubicin. MDA-MB-468 (A, C, E) and BT459 (B, D, F) triple-negative breast cancer cells were exposed to drug before cell lysates were collected and the mitochondrial fraction was extracted. (A, B) Cells were exposed to CoQ10 for 48 hours. (C, D) Cells were pretreated with CoQ10 for 24 hours before treatment with doxorubicin at the cell’s IC50 value (40 nM for MDA-MB-468 and 200 nM for BT549). HPLC was used to quantify CoQ10 levels in the whole-cell lysates. Three separate experiments were conducted in triplicate. (E, F) Extracted cellular fractions were probed for COX IV expression to verify that the mitochondrial fractions were accurate. Error bars represent 1 standard deviation. For all experiments (A-D), P < .05
Coenzyme Q10 Does Not Antagonize the In Vitro Growth Inhibition Effect of Doxorubicin
To determine if CoQ10 affects doxorubicin cytotoxicity, triple-negative breast cancer cell lines MDA-MB-468 and BT549 were treated with doxorubicin at each cell line’s IC50 concentration (40 nM for MDA-MB-468 and 200 nM for BT549—each falling within the peak [5 µM] and steady-state [25-250 nM] plasma concentrations observed in patients after standard bolus infusions 8 ) and a dose range of 0.09 to 9 µM of CoQ10 for 72 hours (Figures 3A and 3B). In both cell lines, CoQ10 did not alter the growth inhibition effect of doxorubicin; nor did CoQ10 alone significantly inhibit cell growth, even at high concentrations (Figure 4). When treated with only doxorubicin, MDA-MB-468 growth inhibition was 53.6%. This inhibition did not change when any concentration of CoQ10 was added. The 53.1% growth inhibition of doxorubicin alone for the BT549 cell line was also not significantly altered by CoQ10 additions (Figures 3A and B). When added alone, up to 180 µM of CoQ10 did not inhibit cellular growth in either cell line (Figure 4).

CoQ10 does not antagonize in vitro growth inhibition effect of doxorubicin on breast cancer cell lines. MDA-MB-468 (A, C) and BT549 (B, D) cell lines were treated with CoQ10 for 24 hours before treatment with doxorubicin at IC50 value (40 nM for MDA-MB-468 and 200 nM for BT549) for 48 hours. Proliferation assay was used to determine growth inhibition effects of CoQ10, alone and in combination with doxorubicin (A, B). Additionally, colony formation assays were performed (C, D). Error bars represent 1 standard deviation; * indicates P < .05

CoQ10 alone does not affect growth of breast cancer cells. Proliferation assays were performed on MDA-MB-468 (A) and BT549 (B) breast cancer cells after treatment with increasing levels of CoQ10 for 72 hours. Error bars represent 1 standard deviation
To confirm our findings, colony formation assays were also performed on both cell lines after CoQ10 and doxorubicin treatments alone and in combination (Figures 3C and D). Colony formation assays are a more stringent test of cell fitness as they measure the ability of every cell in the population to undergo unlimited growth and are therefore much more sensitive at identifying small differences between treatment groups. 14 In both breast cancer cell lines, doxorubicin severely reduced the number of colonies, and the addition of CoQ10 did not alter doxorubicin’s inhibitory capacity. The addition of CoQ10 alone did not substantially affect the colony formation of the cells.
Coenzyme Q10 Does Not Diminish the Apoptotic Effect of Doxorubicin
Given that doxorubicin kills cancer cells, at least in part, by inducing apoptosis, 14 we directly measured whether or not CoQ10 altered the level of doxorubicin-mediated apoptosis. Cells pretreated with CoQ10 were compared against cells pretreated with vehicle control for induction of cleaved Caspase-3 after doxorubicin treatment for 24 hours (Figure 5). Both cell lines showed substantial cleaved Caspase-3 activity on exposure to doxorubicin, and this effect was not diminished by pretreatment with up to 9 µM of CoQ10. MDA-MB-468 cells were exposed to excessive levels of CoQ10, 180 µM, to determine if CoQ10 might induce apoptosis at high levels, but apoptosis was not detected.
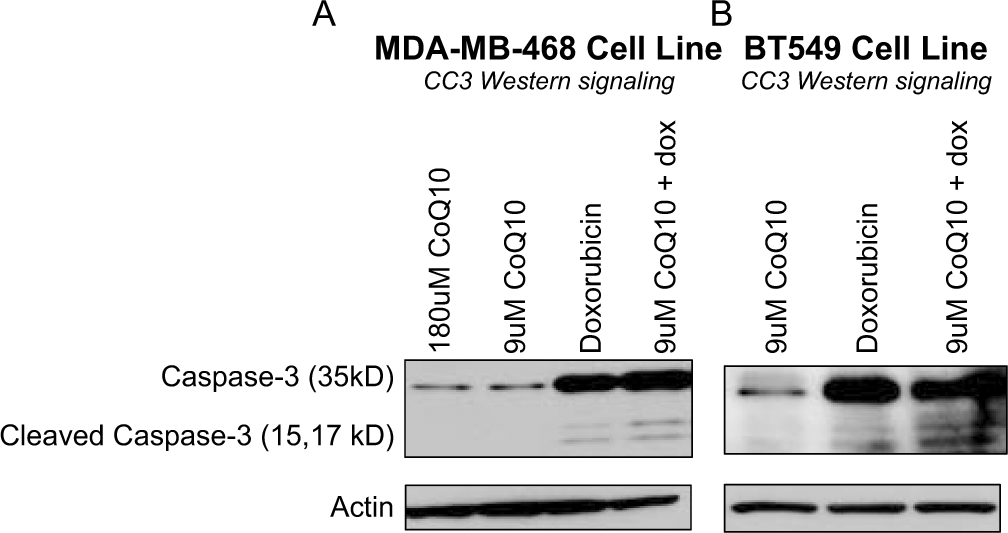
CoQ10 does not diminish the apoptotic effect of doxorubicin. MDA-MB-468 (A) and BT549 (B) cells were treated with CoQ10 alone, doxorubicin alone, or a combination of the two. Cleaved Caspase-3 levels were evaluated using immunoblotting as a measure of apoptotic activity in the cells. MDA-MB-468 cells were treated with 800 nM doxorubicin; BT549 cells were treated with 3.2 µM doxorubicin
Discussion
Doxorubicin-induced cardiotoxicity is a significant concern in the care of breast cancer patients, causing death or disability and limiting the use of potentially more effective concurrent drug combinations (ie, with trastuzumab). Effective treatments are needed to prevent this cardiotoxicity while not decreasing the antitumor effects of doxorubicin. We conducted a series of in vitro cell culture experiments to determine the effects of CoQ10 on doxorubicin cytotoxicity using 2 different triple-negative breast cancer cell lines that are sensitive to doxorubicin treatment. We demonstrated that there was CoQ10 uptake by the breast cancer cells, that the CoQ10 entered the mitochondria, and that CoQ10 did not limit the cytotoxicity of doxorubicin.
The strengths of this work include our expertise and ability to measure and confirm the dose-dependent delivery of CoQ10 to the mitochondria of breast cancer cells. In this setting, we have demonstrated, using 2 separate assays (proliferation and colony formation), that cell death induced by clinically relevant doses of doxorubicin is not altered by CoQ10 administered over a wide dose range and up to doses higher than that achievable through oral supplementation. Our work is consistent with prior studies on non–breast cancer cell lines. 3 The observation that measured CoQ10 levels were decreased in the setting of doxorubicin treatment may be due to a doxorubicin-induced increase in ROS production, which has been observed in other cancer cell lines. 10 Our work sets the stage for much needed in vivo experimentation requiring a large cohort of animals with a noninferiority design and correlative studies showing the activity and effects of both chemotherapy and CoQ10.
There is high use of antioxidant supplements by breast cancer patients after diagnosis, including during the time of treatment.15-17 Many patients use antioxidant supplements in the hope of decreasing treatment-related side effects and preventing recurrence, but the long-term effect of such use has not been rigorously studied for many supplement, disease, and treatment combinations.15,18 Given the high cost of conducting long-term clinical trials, combined with recent findings of harm from antioxidant supplements being tested for the purposes of primary cancer prevention,19,20 we need efficient methods to identify, screen, and test dietary supplements that may be beneficial. Preclinical models, such as those presented here, can be used to efficiently screen agents for potential benefit or harm.
Conclusion
In a series of in vitro cell culture experiments, we found that CoQ10 did not inhibit doxorubicin cytotoxicity in breast cancer cell lines. These results are far from informing clinical practice. Rather, they can inform subsequent preclinical and clinical studies.
Footnotes
Authors’ Note
Heather Greenlee and Jacquelyn Shaw contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Cancer Institute (K23CA141052 to HG) and the Herbert Irving Comprehensive Cancer Center. This publication was also supported by the National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health, through Grant Number KL2 RR024157. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
