Abstract
Doxorubicin (Dox) is a first-line chemotherapeutic agent applied in cancer treatment. Its long-term anticancer efficacy is restricted mainly due to its subsequent cardiotoxicity for patients. Platycodon grandiflorum (PG), an important traditional Chinese herb, has been reported to eliminate phlegm, relieve cough, and reduce inflammatory diseases. Previous clinical studies found that PG has cardioprotective effects for early breast cancer patients who received Dox-based chemotherapy. However, the cellular and molecular mechanisms underlying PG-mediated cardiotoxic rescue remain elusive. This study aimed to explore the protective role and potential molecular mechanisms of PG on Dox-induced cardiac dysfunction in a mouse model of breast cancer. PG significantly alleviated myocardial damage and prevented cardiomyocyte apoptosis induced by Dox. The expression levels of cytochrome C and cleaved caspase-3 significantly decreased, and the levels of Bcl-XL and B-cell lymphoma-2 (Bcl-2)/Bcl-2-associated X protein increased following PG treatment. Furthermore, PG remarkably enhanced the antimetastatic efficacy (versus the Dox group) by regulating the balance of matrix metalloproteinases/tissue inhibitors of metalloproteinases.
Introduction
Doxorubicin (Dox) is broadly administered in the treatment of various cancers, including breast cancer, lymphoma, and soft tissue sarcomas. 1 However, the long-term usage of Dox is limited by its cardiotoxicity. A clinical trial reported heart failure incidences of 3%, 7%, and 18% in cancer patients who had received a cumulative dose of 400, 550, or 700 mg/m2 Dox, respectively.2,3 Thus, developing a drug that confers cardioprotection for Dox treatment is important.
Several pathogeneses have been proposed to explain the cardiotoxicity induced by Dox, such as oxidative stress, 4 disruption of the normal catalytic cycle of Top 2β, and dysfunction of cardiac mitochondrial bioenergetics. 5 Cell apoptosis through the activation of caspase-dependent signaling and suppression of B-cell lymphoma-2 (Bcl-2)/Bcl-2-associated X protein (Bax) signaling may play vital roles in the pathogenesis of Dox-induced cardiotoxicity.6-8 Thus, the decrease in myocardial apoptosis can be a potential strategy for Dox-induced cardiomyopathy.
Some traditional Chinese herbs display effective anticardiotoxicity and improve cardiac function, such as Platycodon grandiflorum (PG), Salvia miltiorrhiza, and Astragalus membranaceus.9-11 “The Holy Husbandman’s Classic on Roots and Herbs” and “Synopsis of Prescriptions of the Golden Chamber” (Han Dynasty) recorded that PG had been used to relieve cough and reduce sputum. Modern pharmacological studies suggest that PG and its active components suppress the inflammatory response, 12 inhibit cardiomyocyte apoptosis, 13 and protect renal and liver functions. 14 Our previous clinical study on 120 patients indicated that PG treatment (6 g/day) had a significant cardioprotective effect compared with placebo treatment in early breast cancer patients receiving anthracycline-based chemotherapy. 15 However, the specific molecular mechanisms underlying the cardioprotective activity of PG were not fully elucidated.
In this study, we explored the protective role and potential molecular mechanisms of PG on Dox-induced cardiac dysfunction in a mouse orthotopic breast cancer model.
Materials and Methods
Reagents and Antibodies
RPMI-1640 medium was purchased from Hyclone Laboratories, Inc. (Hyclone, Logan, USA). Fetal bovine serum (FBS) and 0.25% trypsin were acquired from Gibco Life Technologies (Gaithersburg, MD, USA). Mouse cardiac troponin-I (CTn-I), creatine kinase-MB (CK-MB), malondialdehyde (MDA), and reactive oxygen species (ROS) ELISA kits were obtained from Hengyuan Biotech Co., Ltd. (Shanghai, China). The Masson assay kits were obtained from Beyotime Co., Ltd. (Shanghai, China). Primary rabbit antibodies, such as cytochrome C (#11940), Bax (#2772), Bcl-2 (#3498), Bcl-XL (#2764), caspase-3 (#9662), cleaved caspase-3 (#9661), GAPDH (#5174), and HRP-conjugated secondary antibody (#7074s) were purchased from Cell Signaling Technology, Inc. (Beverly, MA, USA). Dexrazoxane (Dex) was purchased from Aosaikang Pharmaceutical Co., Ltd. (Nanjin, Jiangsu, China). Dox (#603456) and some primary rabbit antibodies TIMP2 (#221342), TIMP3 (#221339), MMP2 (#261447), MMP9 (#220097), COL3A1 (#220454), and COL1A2 (#220244) were purchased from Sangon Biotech Co., Ltd. (Shanghai, China). All other reagents used were of analytical grade and obtained from commercial sources.
Animals and Drugs
All animal experiments were performed according to protocols approved by the Animal Care and Use Committee of Shanghai University of TCM (Permit Number: PZSHUTCM18122103). Female BALB/c mice (7 weeks, body weight of 20-22 g) were obtained from the Shanghai SLAC Laboratory Animal Technology Co., Ltd. (Shanghai, China). The animals were housed under standardized conditions in animal facilities at 20°C ± 2°C, 45% to 50% relative humidity, and a 12 hours light/dark cycle with dawn/dusk effect. PG was purchased from Shanghai Yanghetang Pharmaceutical Co., Ltd. (No. 171012, Shanghai, China). Morphological, microscopic, and phytochemical identification were performed according to the Pharmacopoeia of the People’s Republic of China (2015 edition). PG (100 g) was immersed in 70% ethanol (1:12, w/v) for 4 hours and refluxed for 1 hour. After filtration, the residue was refluxed again with 70% ethanol (1:10, w/v) for 1 hour and then filtered. Thereafter, the 2 decoctions were combined and concentrated with a vacuum to a final concentration of 1 g/mL. Ethanol in the extract was volatilized completely. The quality of the PG extract was analyzed by high-performance liquid chromatography. PG (1.342 g) and a mixture of 2 reference standards were dissolved in 50 mL of methanol, respectively. After sonication for 30 minutes, the samples were filtered through a 0.22 μm syringe filter, and 10 μL of filtrate was injected into the HPLC-DAD system for analysis. HPLC analysis of PG was performed by Waters 2695 HPLC system (Waters, USA) with an Agilent TC-C18 column (4.6 ×250 mm, 3 μm). The mobile phase was composed of solvent A (acetonitrile) and solvent B (water containing 0.1% phosphoric acid), and the gradient procedure was set as follows: 0 to 14 minutes, 70% A; 14 to 17 minutes, 10% A; and 17 to 20 minutes, 70% A; the UV detection wavelength was set at 260 nm (Supplemental Figure S1).
Cell Culture
The mouse breast cancer cell line 4T1 was purchased from the Cell Bank of the Chinese Academy of Sciences, Shanghai Institute of Cell Biology (Shanghai, China). The cells were cultured with RPMI-1640, supplemented with 10% FBS, and incubated at 37°C in 5% CO2 atmosphere. The cells were used at the logarithmic growth phase in all experiments.
Breast Cancer Xenograft Model and Drug Treatment
In this work, 4T1 cells (2 × 104 cells in 100 μL of PBS per mouse) were injected into the left fat pad of number 2 mammary gland of mice. The tumors formed approximately 14 days after the inoculation. All mice were randomly divided into 6 groups (n = 10): control group (ig saline), Dox group (ip 2.5 mg/kg Dox + ig saline), 3 PG groups (ip 2.5 mg/kg Dox + ig 0.5, 1, and 2 g/kg PG), and Dex group (ip 2.5 mg/kg Dox + ip 25 mg/kg Dex). The mice were administered the PG extract once per day and the solution of Dox or Dex once per 2 days continuously for 2 weeks. These doses were chosen based on the dosage of PG in clinical practice (6 g/day). At the experimental endpoint, all animals were euthanized. Hearts, lungs, and tumors were excised and then fixed in 4% paraformaldehyde overnight or stored at −80°C until further analysis.
Echocardiographic Studies
The mice were anesthetized with 2.5% isoflurane in 95% oxygen and 5% carbon dioxide and then situated in the supine position on a warming platform to maintain the core temperature at 37°C. Cardiac function was evaluated via echocardiography by using a High-Resolution Small Animal Imaging System (Vevo2100, Visual Sonics Inc., Toronto, Canada). Left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS) were measured and calculated using a Vevo Strain Software Work Station (Vevo2100, Visual Sonics Inc., Toronto, Canada). The experimental protocol was performed as described previously. 16
ELISA Analysis
Blood samples (0.5 mL) in different groups were collected and centrifuged at 1500 r/minute for 15 minutes at 4°C. The serum was then obtained. According to the instructions for the ELISA kit, the serum levels of CTn-I, CK-MB, MDA, and ROS were detected.
Histopathological and Immunohistochemistry (IHC)
The heart and lung tissues were fixed with 4% paraformaldehyde, embedded in paraffin, and sectioned into 5 mm-thick slices for hematoxylin–eosin (HE) or Masson’s trichrome staining according to each manufacturer’s instructions. 17 For IHC, the sections were washed with PBS for 10 minutes. The antigens were unmasked in 10 mM citrate buffer and then incubated overnight at 4°C with MMP2, MMP9, TIMP2, TIMP3, COL3A1, and COL1A2 primary antibodies. The images were archived using a digital camera (Olympus, BX43, Tokyo, Japan) and analyzed by Image-Pro Plus software 6.0.
Western Blot Analysis
Total proteins were extracted from heart and lung tissues by RIPA lysis buffer with 1% PMSF. The protein concentrations were determined via the bicinchoninic acid method. Proteins (20 μg) were separated on 10% SDS-PAGE gels. The protein was electro-transferred into a PVDF membrane. After incubating in primary antibodies (1:1000 dilution) at 4°C overnight, the membranes were washed 3 times with 1× TBST. The membranes were incubated with the secondary antibody (1:3000 dilution) for 2 hours at room temperature. ECL detection was used to visualize the proteins in the experiment. Band intensity was quantified using Image J software and expressed as the relative intensity compared with the control. GAPDH was used as an internal reference.
Statistical Analysis
The data were analyzed using SPSS 21.0 statistical software (SPSS Inc., Chicago, IL, USA). Continuous variables were expressed as the mean ± standard deviation. Significance between groups was analyzed by one-way ANOVA or Student’s t test using GraphPad Prism 5.0 (GraphPad Software). Statistical significance was set at P < .05.
Results
PG Alleviated the Cardiac Dysfunction Induced by Dox in a Mouse Model of Breast Cancer
After 4T1 breast cancer cell inoculation, a tumor xenograft model was successfully established (Figure 1A). In the tumor-bearing mice treated with Dox (2.5 mg/kg) for 2 weeks at an interval of 1 day per injection, tumor growth and remote metastases were both reduced, but cardiac dysfunction occurred in the mice. Echocardiographic examination showed that LVEF and LVFS significantly decreased in the Dox-treated group compared with that in the control group. Treatment with PG (1 and 2 g/kg) and Dex (25 mg/kg) significantly attenuated cardiac dysfunction in the Dox-administered mice, as indicated by the increased LVEF and LVFS (P < .05; Figure 1B).

PG ameliorated Dox-induced cardiac dysfunction in mice with breast cancer. Echocardiographic assay was used to determine the attenuated left ventricular dysfunction of PG on Dox-induced cardiac dysfunction in mice. PG treatment (1 and 2 g/kg/day) significantly attenuated left ventricular dysfunction in Dox-treated mice as indicated by the increased LVEF and LVFS (A and B). *P < .05 versus the control group; #P < .05 versus the Dox group.
PG Treatment Reduced Dox-induced Myocardial Damage and Oxidative Factors
ELISA results revealed that Dox treatment significantly increased the serum levels of CK-MB and CTn-I (P < .01) in tumor-bearing mice compared with those in the control mice. By contrast, PG treatment reduced the levels of these myocardial damage factors (P < .05; Figure 2A) and decreased oxidative factors ROS and MDA in Dox-treated mice (P < .01; Figure 2B).

PG reduced Dox-induced inflammatory and oxidation factors in mice with breast cancer. ELISA was conducted to determine the inflammatory and oxidation factors in serum. Intuitive quantitative chart analysis showed that PG reduced the levels of inflammatory and oxidation factors (A and B). **P < .01 versus the control group; #P < .05, ##P < .01 versus the Dox group.
PG Treatment Reduced Dox-induced Myocardial Fibrosis and Collagen Deposition
Masson staining and IHC analysis showed that Dox treatment induced collagen deposition and overexpression of collagen type I/III proteins in heart tissues (P < .01). Compared with the Dox treatment group, PG treatment significantly attenuated inflammatory infiltration, myocardial fibrosis, and collagen I/III deposition (P < .01; Figure 3A and B).
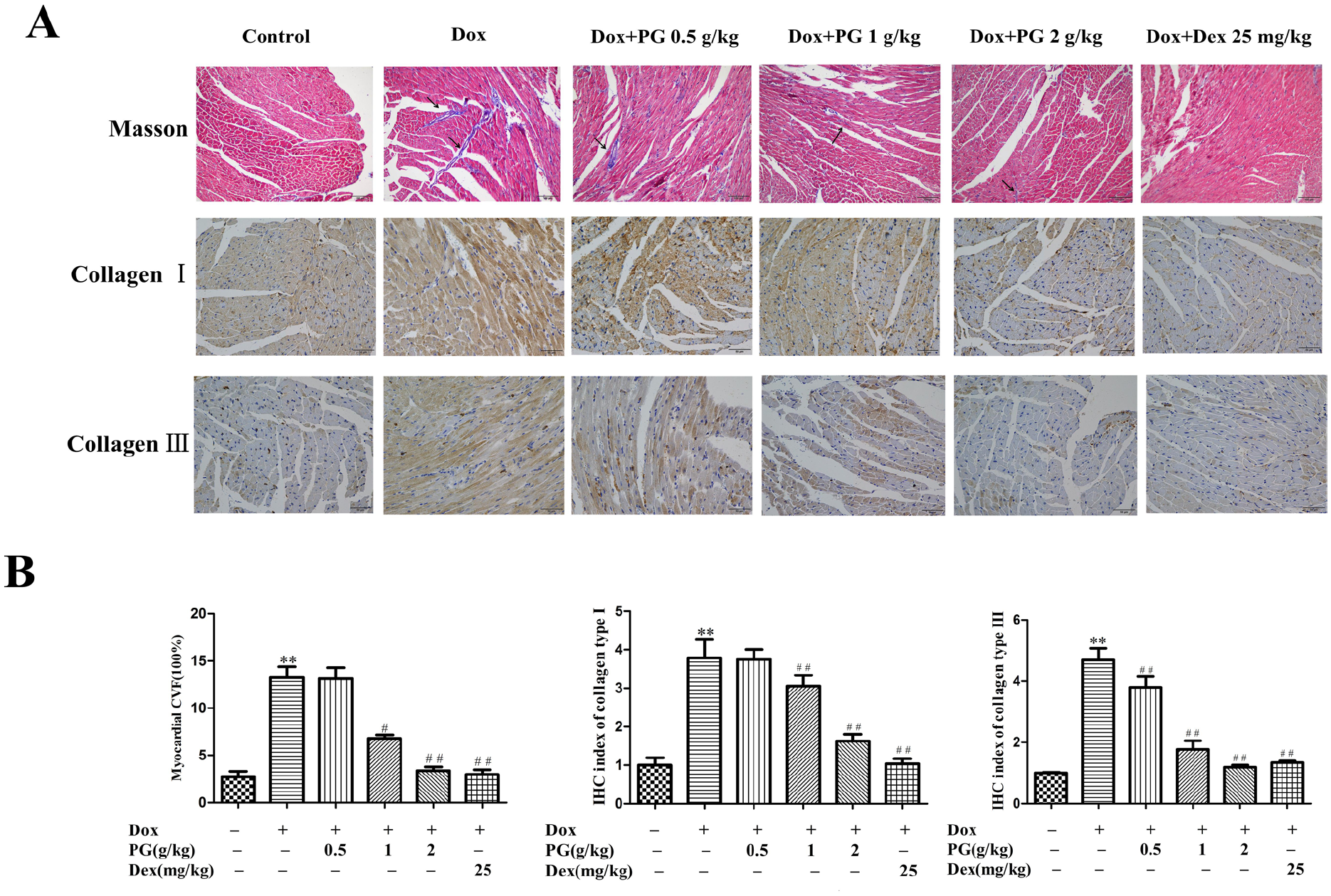
PG reduced Dox-induced myocardial fibrosis and collagen deposition in mice with breast cancer. (A) Masson’s trichrome staining and IHC showed that PG reduced Dox-induced myocardial fibrosis and collagen deposition. (B) Intuitive quantitative chart analysis showed the percentage of the area with collagen type I and III staining. **P < .01 versus the control group; ##P < .01 versus the Dox group. Images were obtained at 400× magnification.
PG Treatment Inhibited Dox-induced Cardiomyocyte Apoptosis
The expression of apoptosis-associated proteins was determined via Western blot analysis. After Dox treatment for 2 weeks, the Bcl-XL and Bcl-2/Bax ratio decreased, whereas cytochrome C and cleaved caspase-3 in heart tissues increased (P < .05 or P < .01). In the PG and Dex treatment groups, the expression levels of these apoptosis-associated proteins were reversed dramatically (P < .01; Figure 4A and B).

PG inhibited Dox-induced apoptosis in the myocardium of mice with breast cancer. (A) Western blot analysis was performed to detect the relative protein expression levels. GAPDH was used as a control. After PG treatment in mice with breast cancer, the expression of apoptosis-related proteins changed; for instance, Bcl-2/Bax and Bcl-XL increased, whereas cytochrome C and cleaved caspase-3 decreased (B). All experiments were performed in triplicate. *P < .05, **P < .01 versus the control group; #P < .05, ##P < .01 versus the Dox group.
PG Enhanced the Anti-metastatic Efficacy of Dox in a Breast Cancer Mouse Model
The synergistic antitumor activity of PG and Dox was investigated in a mouse xenograft model of breast cancer. The tumor volume and weight significantly decreased in the Dox group compared with those in the control group and further decreased in the Dox plus PG group (P < .01 or P < .05; Figure 5A and B).

PG enhanced the antimetastatic efficacy of Dox in a breast cancer mouse model. The weight and size of the primary tumor were measured (A and B). The number of lung metastatic nodules was obviously reduced (C and D). **P < .01 versus the control group; #P < .05 versus the Dox group.
The number of lung metastatic nodules in the Dox plus PG group was obviously reduced compared with that in the Dox group (P < .01 or P < .05; Figure 5C and D). Western blot and IHC analysis further confirmed the antimetastatic activity of PG plus Dox treatment in mice with breast cancer by regulating the expression of metastasis-related proteins MMP2, MMP9, TIMP2, and TIMP3 (P < .05 or P < .01, P < .05 or P < .01; Figures 6 and 7).

Molecular mechanism of PG plus Dox in suppressing tumor metastasis in vivo. PG downregulated the protein expression of MMP2 and MMP9 but upregulated the expression of TIMP2 and TIMP3 in lung metastatic tumor tissues (A and B). GAPDH was used as a control. All experiments were performed in triplicate. *P < .05, **P < .01 versus the control group; #P < .05, ##P < .01 versus the Dox group.

Immunohistochemical staining of MMP2, MMP9, TIMP2, and TIMP3 in lung metastatic tumor tissues. The positive cells in each group were indicated (A and B). **P < .01 versus the control group; ##P < .01 versus the Dox group. Images were obtained at 400× magnification.
Discussion
Anthracycline compounds, especially Dox, are major culprits in chemotherapy-induced cardiotoxicity, which is the chief limiting factor in delivering optimal chemotherapy to cancer patients. 16 Although extensive efforts have been devoted to identify strategies that prevent Dox-induced cardiotoxicity, little consensus has been made regarding the best approach. 18 In the current study, we investigated the cardioprotective effects and underlying mechanisms of PG extract in Dox-treated mice with breast cancer.
Echocardiographic strain imaging has recently emerged as a promising method for the detection of cardiotoxicity prior to left ventricular dysfunction. 19 Cardiotoxicity is defined as an LVEF decline of ≥5% to <55% with heart failure symptoms or an asymptomatic decrease of LVEF ≥10% to <55%. 20 Biomarkers are often used to identify patients with early clinical signs of cardiotoxicity. 21 The peak value of CK-MB and CTn-I is closely correlated with the degree of left ventricular dysfunction. 22 In the present study, 2.5 mg/kg Dox treatment for 2 weeks significantly caused obvious cardiac dysfunction with LVEF decline of 20% and CK-MB and CTn-I elevation of 43.7% and 46.8%, respectively. However, PG significantly increased LVEF and LVFS but decreased CK-MB and CTn-I mediated by Dox in mice with breast cancer.
The production of ROS and MDA in cardiomyocytes plays an important role in Dox-induced cardiotoxicity. 23 Myocardial fibrosis characterized by collagen accumulation is the predominant pathological change in myocardial remodeling, which results in both diastolic and systolic dysfunction. 24 It can disrupt mechanical and electrical functions, induce arrhythmia, upregulate myocardial stiffness, and reduce ventricular compliance.25,26 Herein, we found that PG treatment reduced the levels of oxidative stress and inhibited the myocardial fibrosis induced by Dox in heart tissues of mice with breast cancer.
Dox-induced myocardial damage is accompanied with excessive initiation of apoptosis. Cardiomyocyte apoptosis is an essential contributor, causing myocardial dysfunction and heart failure.27,28 When apoptosis occurs, cytochrome C is released from the mitochondria into the cytoplasm and acts as a key regulatory step in apoptosis. 29 Caspase-dependent apoptosis is an essential molecular mechanism that regulates various diseases, including tumor progression. Bcl-XL is a typical anti-apoptotic molecule, whereas Bcl-2 and Bax are pro-apoptotic molecules and key proteins that lead to the release of inner membrane space proteins, including cytochrome C.30,31 In our study, PG deactivated cytochrome C, enhanced Bcl-2/Bax and Bcl-XL, and suppressed cleaved caspase-3 to prevent Dox-induced cardiomyocyte apoptosis in mice with breast cancer.
Taken together, our data indicated that PG ameliorated Dox-induced cardiac injury and dysfunction. Its underlying molecular mechanisms were partly related to the suppression of Dox-triggered myocardial fibrosis, inflammatory response, and cardiomyocyte apoptosis.
Matrix metalloproteinases (MMPs) are a family of proteins composed of 23 different members that are overexpressed in many tumor tissues, including breast cancer. 32 MMP protein degrades the extracellular matrix and plays a key role in the invasion and metastasis of tumor cells. The activity of MMPs is regulated by their endogenous inhibitors, namely, tissue inhibitors of metalloproteinases (TIMPs). Therefore, the MMP/TIMP balance is critical for tumor invasion and metastasis.33,34 Our data suggested that PG improved the antitumor and antimetastatic efficacy of Dox by regulating the balance of MMPs/TIMPs.
Despite the strengths of this study, a number of experimental limitations existed. First, our study was an in vivo-based study lacking the Dox-induced neonatal rat left ventricle myocyte cardiotoxicity in vitro model. Second, the active compounds of PG extract for preventing Dox-induced cardiotoxicity need further investigation in our laboratory.
In summary, PG treatment alleviated Dox-induced cardiac dysfunction in a xenograft mouse model of breast cancer by reducing myocardial fibrosis and cardiomyocyte apoptosis. The combination of PG and Dox significantly inhibited tumor growth and metastasis by regulating the balance of MMPs/TIMPs. Thus, PG might be applied as an adjuvant therapeutic agent for the treatment of Dox in breast cancer.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231164621 – Supplemental material for Extract of Platycodon grandiflorum Prevents Doxorubicin-induced Cardiotoxicity in Breast Cancer
Supplemental material, sj-docx-1-ict-10.1177_15347354231164621 for Extract of Platycodon grandiflorum Prevents Doxorubicin-induced Cardiotoxicity in Breast Cancer by Youyang Shi, Yuanyuan Wu, Man Shen, Jianfeng Yang, Yuenong Qin, Sheng Liu and Chenping Sun in Integrative Cancer Therapies
Footnotes
Author Contributions
Youyang Shi conceived and devised the research, conducted the experiments, analyzed the data, and wrote the manuscript. Yuanyuan Wu, Chenping Sun, Man Shen, and Jianfeng Yang performed the experiments and analyzed the data. Yuenong Qin supplied technical support. Sheng Liu provided all of the reagents and collected data. All the authors edited and commented on the manuscript. All authors have read and agreed on the final manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from National Natural Science Foundation of China (No. 81603629, 81573973, and 81774308) and the Shanghai three-year action plan (ZY2018-2012-ZYBZ-01).
Ethics Approval and Consent to Participate
The protocol was approved by the Animal Research Ethics Committee of Shanghai University of Traditional Chinese Medicine (Permit Number: PZSHUTCM18122103).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
