Abstract
Nigella sativa seeds, which are also known as black cumin or black seed, contain a bioactive substance called thymoquinone (TQ). It has garnered attention due to its potential health benefits. TQ is a natural substance that has the potential to be beneficial due to its anti-platelet and anti-cancer properties. We can attribute its anti-cancer effects to its capacity to prevent platelet activation and aggregation, as well as to promote platelet apoptosis. Despite the fact that the exact processes underlying thymoquinone’s anti-platelet and anti-cancer effects remain unclear, a number of studies have provided insight into the subject. However, no research has investigated the mechanisms causing TQ’s direct impact on platelet-induced cancer spread. The purpose of this study was to examine the effect of TQ-pretreated platelets on platelet-induced MDA-MB-231 metastasis in vitro. The results demonstrate that TQ effectively inhibits platelet-cell adhesion (53.38% ± 5.69%), reducing platelet-induced migration by about 50% and transmigration of MDA-MB231 cells by 36% ± 9.4%. Additionally, 10 µM TQ suppresses MDA-MB231 cell-induced the protein tyrosine phosphorylation in platelets as well as Btk activation by 45% ± 6.7%. 10 µM TQ and LFMA13 have the capacity to suppress ZEB2 expression to 73% ± 2.8%, increase the expression of FBXW7, β-catenin 1 to 147% ± 4.33% and 136% ± 5.3%, respectively, and decrease vimentin (38% ± 3%) and N-cadherin (29% ± 4%) expression, and prevent platelet-induced transmigration. The findings demonstrate that TQ suppresses both the platelet-inducing epithelial-to-mesenchymal transition (EMT) and MDA-MB231 cells transmigration through mediating the FBXW7-ZEB2-β-catenin signaling pathway and downregulating Bruton’s tyrosine kinase (Btk).

Introduction
Breast cancer is the most prevalent type of cancer in women worldwide, and metastasis is the primary cause of mortality. 1 While the exact mechanisms behind breast cancer metastasis are uncertain, current research has identified platelets as important actors in the process. 2 Platelets are small blood cells that originate from the fragmentation of their hematopoietic precursor, the megakaryocyte, which is located in the bone marrow. Platelets are essential components of hemostasis and thrombosis; they are directly involved in primary hemostasis, where they undergo various modifications to halt bleeding. 3 Breast cancer cells are known to interact with platelets which promote migration and invasion. 4 This interaction is facilitated by a variety of processes, such as adhesion, activation, and the release of bioactive compounds. Platelets that have been activated secrete pro-inflammatory and pro-metastatic substances, including TGF-β, PDGF, and VEGF.5,6 These factors increase cancer cell motility, invasion, and angiogenesis.5,6 Platelet TGF-β induces epithelial-to-mesenchymal transition (EMT) and enhances matrix metalloproteinases, thereby promoting cancer cell migration and invasion. 6 Furthermore, platelet-derived PDGF promotes cancer cell motility and invasion by activating the PI3K/Akt signaling pathway. 7 Angiogenesis, a critical phase in the progression of malignancy, is induced by platelets. The proliferation and migration of endothelial cells are stimulated by platelet-derived VEGF, leading to the formation of new blood arteries that facilitate BC cell metastasis. 8
In traditional medicine, plant extracts have been used for centuries. Among them, Nigella sativa L., a species belonging to kingdom Plantae, subkingdom Tracheobionta, division Magnoliophyta, class Magnoliopsida, order Ranunculales, family Ranunculaceae, genus Nigella, sometimes known as black cumin, is a Mediterranean and Asian annual plant. 9 It has been utilized in traditional medicine to treat arthritis, lung illness, hypercholesterolemia, and cancer.10 -17 The therapeutic effects of Nigella sativa are attributed to its complex chemical composition, which includes thymoquinone (TQ), carvacrol, and alpha-hederin, among others. TQ, a bioactive molecule produced from black cumin seed oil, has been proven to be the primary component. 18
TQ shows tremendous promise as a medicinal drug; various research groups have described TQ’s high superoxide and free radical scavenging properties. 19 TQ has the ability to regulate cellular antioxidant defense enzymes, suppress platelet activation and aggregation.20,21 Moreover, it has been reported that TQ alters platelet signaling pathways, including PI3K/Akt and mitogen-activated protein kinase (MAPK) pathways, resulting in inhibition of platelet activation.22 -24 Additionally, TQ inhibits platelet granule release and thromboxane A2 production, thereby exerting antiplatelet effects. 25 Also, it inhibits the production of pro-inflammatory cytokines and chemokines, which both are involved in platelet activation and cancer progression. 26
Cancer is a dynamic disease marked by uncontrollable cell growth and metastasis. TQ has been studied for its anticancer properties against a variety of cancer types, including breast, lung, prostate, and colorectal cancer.27 -30 The anticancer effects of TQ are the result of intricate processes that involve the regulation of numerous molecular pathways. In cancer cells, TQ reduces cell growth, induces cell cycle arrest, and increases apoptosis by modulating critical signaling pathways such as PI3K/Akt, MAPK, nuclear factor-kappa B (NF-B), and Wnt/β-catenin.31 -33 In addition, it can decrease the expression of cancer-promoting genes and proteins such as VEGF and MMPs.34,35 TQ has also demonstrated potential in the prevention of cancer cell invasion, angiogenesis, and metastasis.36,37
Interestingly, studies have revealed that when combined with conventional chemotherapeutic drugs or other natural substances, TQ may have synergistic benefits. TQ-based combination therapy has been proven to improve anticancer drug cytotoxicity and reduce drug resistance.38,39 Furthermore, TQ has been demonstrated to sensitize cancer cells to radiation therapy, increasing its efficiency. 40 These synergistic effects emphasize TQ’s potential as an adjuvant medication for increasing the efficacy of currently available cancer therapies.
These findings indicate that TQ may be able to reduce platelet-related diseases and prevent cancer metastasis caused by platelet activation. Nonetheless, no study has investigated the direct effect of TQ on platelet-induced cancer metastasis and emphasized the processes involved. This study demonstrates that TQ downregulates Bruton’s tyrosine kinase (Btk) to inhibit the platelet-inducing breast cancer cell line, MDA-MB231 transmigration ability, and epithelial-to-mesenchymal transition (EMT).
Materials and Methods
Platelet Preparation and Treatment
Blood samples were collected from 3 healthy volunteers in accordance with the Helsinki Declaration and the approval of King Faisal University Ethics Committee in 2023 under the number ETHICS 1489. The collected blood samples were mixed with one-tenth volume of acid/citrate dextrose (ACD) anticoagulant containing (mM) 85 sodium citrate, 78 citric acid, and 111 D-glucose, pH 5.4. Platelet-rich plasma was then prepared by centrifugation for 5 minutes at 700g, and apyrase (40 µg/mL) was added. Platelets were then collected by centrifugation at 350g for 20 minutes and resuspended in HEPES-buffered saline (HBS) containing 145 mM NaCl, 10 mM HEPES, 10 mM D-glucose, 5 mM KCl, 1 mM MgSO4, pH 7.45, and supplemented with 0.1% w/v bovine serum albumin and 40 µg/mL apyrase. Platelet concentration was adjusted to 3 × 108 cells/mL.
Cell Viability Test
Cell viability was assessed using calcein and trypan blue as previously described. 41 For calcein loading, cells were incubated for 30 minutes with 5-µM calcein-AM at 37 °C, centrifuged, and the pellet was resuspended in fresh HBS or in DMEM culture media. Cells were treated with the different inhibitors, centrifuged, and resuspended in HBS or in DMEM culture media. Fluorescence was recorded from 2 mL aliquots using a fluorescent reader. Samples were excited at 494 nm and the resulting fluorescence was measured at 535 nm. The results obtained with calcein were confirmed using the trypan blue exclusion technique. Ninety-five percent of cells were viable in our platelet suspensions, and no effect was observed on cell viability after treatment with inhibitors.
Cell Culture
Human umbilical vein endothelial cells (HUVEC) were purchased from ATCC (ATCC Cat #CRL-1730, RRID:CVCL_2959) and cultured in RPMI 1640 supplemented with L-glutamine, HEPES, sodium pyruvate, 4.5 g/L glucose, 1.5 g/L NaHCO3, 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin, and 2.5 µg/mL amphotericin B. Cells were maintained at 37 ° in a humidified incubator gassed with 5% CO2. The breast adenocarcinoma cell line MDA-MB-231 (ATCC Cat #HTB-26, RRID:CVCL_0062) was maintained in DMEM containing 4.5 g/L glucose, 584 mg/L L-glutamine, sodium pyruvate, 10% FBS, 100 U/mL penicillin and 100 µg/mL streptomycin, and 2.5 µg/mL amphotericin B at the temperature of 37 ° in humidified 5% CO2 atmosphere.
MDA-MB-231 cells were allowed to reach 70% of confluence in DMEM containing 4.5 g/L glucose, 584 mg/L L-glutamine, sodium pyruvate, 10% FBS, 100 U/mL penicillin and 100 µg/mL streptomycin, and 2.5 µg/mL amphotericin B 24 hours before platelet addition at a concentration of 3 × 108 for direct and indirect contact (cell culture insert).
Transendothelial Migration Assay
Transendothelial migration assay was performed as previously described with modifications. 42 A cell culture insert containing a PET membrane with a pore size of 8 μm was used for cell line migration. The inner side of PET membrane was pre-coated with 100 μL of collagen I (1 mg/mL) overnight. One point five milliliters of culture medium was firstly added into each well of 12-well plates, then the cell culture inserts, and 5 × 104 HUVEC cells were added into each upper chamber of the insert. The cells were cultured for 24 hours at 37 °C and 5% CO2 until reaching total confluence. Following the incubation period, the medium was refreshed, and 1 × 105 MDA-MB-231 breast cancer cells were added to the upper chamber. MDA-MB-231 breast cancer cells were grown with or without platelets that had been pre-treated with TQ, LFMA13, and PP1. Transmigration was detected after 24 hours of incubation at 37 ° and 5% CO2. MDA-MB-231 migrated cells through the HUVEC layer and adhered onto the outer side of the PET membrane, were fixed, and dipped in a mixed solution (5 mL) of acetic acid and methyl alcohol (1:3 in volume ratio) for 30 minutes. After the inserts were washed twice with PBS. The PET membranes were taken out using a scalpel and stained for 10 to 15 minutes with Giemsa solution (0.9%). The redundant Giemsa was washed with Milli-Q water, and then the migrated cells (dyed dark blue) were observed and recorded under an inverted microscope. The migrated cell number was quantified by cell counting from more than 6 microscopic fields for each sample (20× magnification), and 3 PET membranes were used for each experimental group.
Western Blotting
Cells were washed with ice-cold TBS buffer (20 mM Tris, pH 7.5, 150 mM NaCl) and lysed by RIPA buffer (Molequle-ON) for 1 hour at 4 ° under constant agitation. Cell lysate supernatant was recovered by centrifugation at 16 000g for 25 minutes in 4 °C. Protein concentration was measured by BCA protein assay kit (Molequle-ON, Cat #BCA-M-001-500), according to manufacturer instructions. Two hundred microliter of the supernatant was mixed with an equal volume of 2 Laemmli’s buffer (4% SDS, 20% Glycerol, 0.004% bromophenol blue, and 125 mM Tris-HCl) with 10% dithioreitol followed by heating for 5 minutes at 95 °. One-dimensional 10% SDS-PAGE was performed, and separated proteins (50 µg) were electrophoretically transferred, to a Nitrocellulose membrane (Molequle-on) for 2 hours at 0.8 mA/cm2 in a semi-dry blotter. Membranes were blocked in TBST Buffer (20 mM Tris, pH 7.5 150 mM NaCl and 0.1% tween 20) supplemented with 5% BSA for 1 hour at room temperature.
Blocked membranes were washed twice with TBST and incubated with primary anti-phospho-tyrosine (Santa Cruz Biotechnology Cat #sc-7020, RRID:AB_628123) anti-Btk antibody (Santa Cruz Biotechnology Cat #sc-1108, RRID:AB_2067819), anti-phospho-Btk antibody (Cell Signaling Technology Cat #5082, RRID:AB_10561017, recognize all isoforms), anti-P-selectin (Santa Cruz Biotechnology Cat #sc-8419, RRID:AB_627151), anti-FBXW7 (Santa Cruz Biotechnology Cat #sc-81829, RRID:AB_2246842), anti-ZEB2 (Santa Cruz Biotechnology Cat #sc-18392, RRID:AB_2217982), β-catenin (Santa Cruz Biotechnology Cat #sc-7963, RRID:AB_626807), N-cadherin (AB-M-027 Molequle-ON), vimentin (AB-M-079 Molequle-ON) antibodies. Anti-β-actin (Santa Cruz Biotechnology Cat #sc-47778, RRID:AB_626632) was used as internal control marker. We used 1:1000 dilutions for all the primary antibodies and 1:10 000 for the secondary antibodies. Target proteins were detected with WBS detection reagent (Molequle-ON, Cat #WSP-M-100). LICOR detector was used for quantification of detected bands.
Cell Adhesion Assay
Cell adhesion to HUVEC was performed as described by Muthusamy S 2024 with modifications. 43 HUVEC were cultured into 24-well plates at ~1 × 105 cells/well and incubated until complete confluence. MDA-MB-231 cells and platelets were pre-labeled with Green CMFDA (GLP BIO, Cat #GC43783) at 10 µM for 30 minutes and seeded at 3 × 105 and 3 × 108/mL, respectively. Cells were allowed to adhere for 24 and 1 hour of incubation at 37 ° and 5% CO2 for MDA-MB-231 or platelets, respectively. Non-adherent cells were removed by gentle washing with PBS. Adherent cells fluorescence intensities were measured with fluorescence plate reader with 485 nm as excitation and 514 nm as emission wavelengths. Adhesion was expressed as the percentage of total cell input by comparing the specific fluorescence in each well with the control.
Human Platelets Membrane Enrichment
To quantify P-selectin expression, we extracted and purified platelet membranes according to DW. Greening et al, 44 then the membrane fraction was electrophoretically separated in SDS-PAGE 10%, transferred, and revealed for membrane P-selectin using anti-human P-selectin antibody (Santa Cruz Biotechnology Cat #sc-8419, RRID:AB_627151).
Statistical Analysis
Analysis of statistical significance was performed using the SPSS 20.0 software package, IBM SPSS Statistics for Windows (RRID:SCR_002865) and GraphPad Prism (RRID:SCR_002798). One and 2-way ANOVA tests were performed for multi-tests comparison. We also performed 2 tailed unpaired Student’s t-test. P < .05 was considered to be significant for a difference.
Results
TQ Mitigates Platelet-Mediated Metastatic Capabilities of MDA-MB-231 In-Vitro
Several factors can significantly affect platelet viability, including culture media. Thus, we tested the effect of DMEM media on platelet viability. As demonstrated in Supplementary Figure 1 platelet viability in DMEM was 90% ± 2.23%, comparable to viability in HBS buffer. A significant viability decrease in DMEM media (70% ± 3.74%) was recorded after 6 hours of incubation compared to HBS buffer (88% ± 3.86%).
Recent research has suggested that TQ suppresses platelet activation and aggregation, 21 potentially lowering the risk of platelet-mediated cancer progression. In order to evaluate this hypothesis, we investigated the impact of TQ on platelet and breast cancer cell interactions.
We have pretreated platelets with 1, 5, and 10 µM of TQ for 10 minutes at 37 °C before quantifying the interaction with MDA-MB-231 cells for 1 hour. As illustrated in Figure 1A pretreating platelets with TQ significantly reduces adhesion to MDA-MB 231 cells in a dose-dependent manner. The maximum effect was observed in 10 µM TQ (53.8% ± 5.69%). We also investigated the effect of TQ on platelet-induced MDA-MB-231 cell migration; we conducted trans-well migration assay using a PET membrane with 8 µm diameter. MDA-MB 231 cells were previously cultured with TQ-treated platelets with no contact in a 0.4 µm PET insert, and platelets are in the upper chamber. Results indicated a significant decrease in the number of migrated cells to the inner side of the insert membrane. At 1 µM of TQ, the number of cells that migrated to the inner side of the insert membrane decreased by nearly 10%. The migration inhibition was dose-dependent, as evidenced by the highest effect at 10 µM, which was ~50% inhibition (Figure 1B).

(A) TQ inhibits platelet-MDA-MB231 interaction. MDA-MB231 were cultured into 24-well plates at ~1 × 105 cells/well and incubated until complete confluence. Platelets were pretreated with 1, 5, and 10 µM of TQ for 10 minutes then, prelabeled with green CMFDA (10 µM) for 30 minutes at 37 °C and added to the wells at ~3 × 108 cells/mL for 1 hour. Values were presented as percentage of the relative fluorescence intensity ± SEM of 4 separate determinations. ANOVA and Tukey’s test were performed for multi-test comparison. *P < .05. ***P < .001 compared to control (non-treated platelets). (B) TQ inhibits platelet-induced MDA-MB231 migration. MDA-MB231 1 × 105 were added to the upper chamber, co-cultured with platelets pretreated or not with 1, 5, and 10 µM of TQ for 10 minutes. Migration was detected after 24 hours of incubation at 37 ° and 5% CO2. Migrated cells onto the outer side of the PET membrane were fixed and stained as described in the Materials and Methods section. Histograms represent the mean of migrated cell number of 3 PET membranes and 6 microscopic fields for each sample ± SEM. ANOVA and Tukey’s test were performed for multi-test comparison. **P < .01. ***P < .001 compared to control (non-treated platelets). (C) TQ inhibits platelet-induced MDA-MB231 transmigration. HUVEC 5 × 104 added into each upper chamber of the insert and cultured for 24 hours at 37 ° and 5% CO2 until total confluence. MDA-MB231 1 × 105 were added to the upper chamber, co-cultured with platelets pretreated or not with 1, 5, and 10 µM of TQ for 10 minutes. Transmigration was detected after 24 hours of incubation at 37 ° and 5% CO2. Migrated cells through the HUVEC layer and adhered onto the outer side of the PET membrane were fixed and stained as described in Materials and Methods section. Histograms represent the mean migrated cell number of 3 PET membranes and 6 microscopic fields for each sample ± SEM. ANOVA and Tukey’s test were performed for multi-test comparison. ***P < .001 compared to control (non-treated platelets).
Cancer cells may interact with platelets in several ways, including platelet-induced angiogenesis, tumor cell-platelet interactions, and platelet-mediated metastasis.4,6 To evaluate the effect of TQ on platelet-induced MDA-MB-231 metastasis, we conducted a transendothelial migration assay with HUVECs, which replicate the vascular endothelial barrier. MDA-MB-231 cells were co-cultured with platelets treated with 1, 5, and 10 µM TQ for 10 minutes at 37 °C. TQ significantly inhibits platelet-induced MDA-MB-231 cells transmigration by 36% ± 9.4% (P < .001) and 53% ± 10% (P < .001) at 5 and 10 µM, respectively. Furthermore, no effect was observed for 1 µM TQ (P > .05; Figure 1C).
TQ Disrupts Tyrosine Kinase Signaling in Platelets Activated by MDA-MB-231 Cells
It has been demonstrated that platelets can be activated by circulating cancer cells. 45 Platelet activation increases protein tyrosine phosphorylation. 46 We investigated MDA-MB-231 cells impact on platelet tyrosine kinase signaling pathways. The MDA-MB-231-educated platelets exhibit around a 138% ± 7.26% increase in protein tyrosine phosphorylation, mainly at 60 and 150 kDa. Platelet pretreatment with 10 µM of TQ for 10 minutes significantly reduces MDA-MB-231 cell-induced tyrosine phosphorylation (Figure 2).

TQ inhibits MDA-MB231-induced platelet tyrosine phosphorylation. Platelets were pretreated with 1, 5, and 10 µM of TQ for 10 minutes then were co-cultured with MDA-MB-231 cells for 24 hours. Proteins were analyzed by SDS/PAGE and subsequent Western blotting with a specific anti-phospho-tyrosine (PY99) antibody as described in the Materials and Methods section. Histograms represent protein tyrosine phosphorylation and are presented as a percentage of control (untreated resting cells) and expressed as mean ± SEM of 5 separate experiments. ***P < .001 compared to control (non-treated platelets).
Thymoquinone Blocks Bruton’s Tyrosine Kinase Activation in Platelets Stimulated by MDA-MB-231 Cells
We have previously described the role of Btk as a platelet tyrosine kinase activated by thrombin or oxidative stress. 46 Our results demonstrated that MDA-MB-231 enhances Btk phosphorylation by almost 20% ± 4.14%. However, the phosphorylation of Btk is substantially reduced by 45% ± 6.7% when platelets are pre-incubated with 10 µM of TQ for 10 minutes at 37 ° (Figure 3). Consequently, we investigated the effect of LFMA13, a specific Btk inhibitor, on MDA-MB-231 cells-induced platelet activation, by quantifying P-selectin exposure. For stimulation, platelets were co-cultured with MDA-MB-231 cells after being treated with 10 µM of LFMA13 for 10 minutes, rinsed, and subsequently co-cultured. Our findings indicate that LFMA13 significantly decreases the relative expression of P-selectin in comparison to the control (50.17% ± 4.65%; P < .001). However, no synergistic effect was observed when cells were treated with both LFMA13 and TQ (41% ± 6.73%; Figure 4).

TQ inhibits MDA-MB231-induced Btk activation. Platelets were pretreated with 10 µM of TQ for 10 minutes then were co-cultured with MDA-MB-231 cells for 24 hours. Proteins were analyzed by SDS/PAGE and subsequent Western blotting with a specific anti-Btk antibody (N20 clone) or anti-phospho-Btk antibody (Y-223 clone) antibodies as described in Materials and Methods section. Histograms represent protein expression level and are presented as a percentage of control (untreated resting cells) and expressed as mean ± SEM of 4 or 5 separate experiments. **P < .01. ***P < .001 compared to control (non-treated platelets).
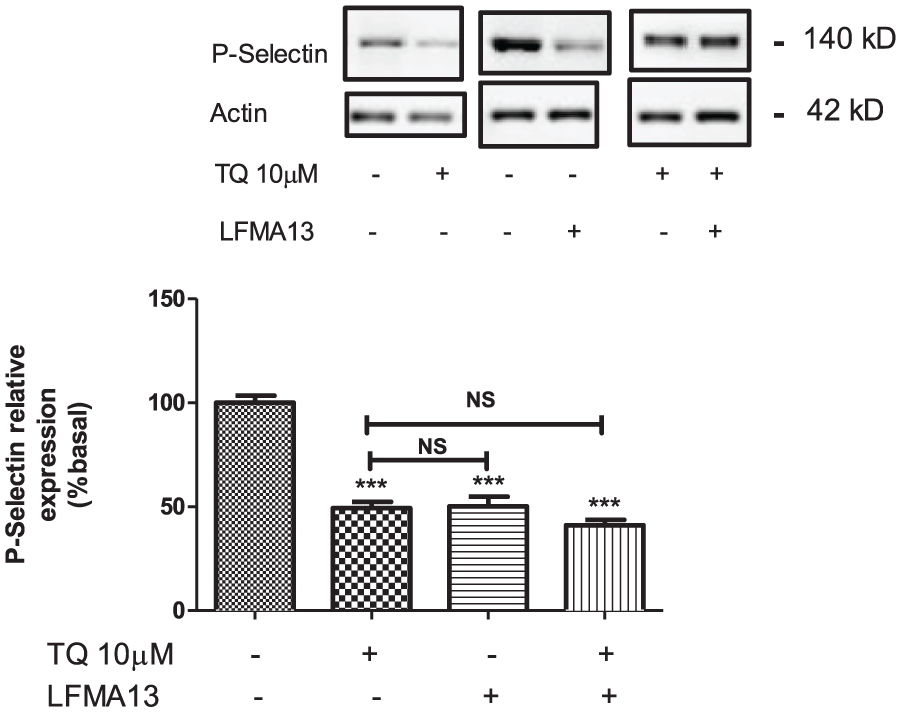
LFM A13 inhibits P-selectin exposure. Platelets were pretreated with 10 µM of TQ and or LFMA13 for 10 minutes then were co-cultured with MDA-MB-231 cells for 24 hours. Proteins were analyzed by SDS/PAGE and subsequent Western blotting with a specific P-selectin antibody (CTB201) as described in Materials and Methods section. Histograms represent protein expression level and are presented as a percentage of control (untreated resting cells) and expressed as mean ± SEM of 6 separate experiments. ***P < .001 compared to control (non-treated platelets).
Targeting Btk and Src Kinases With LFMA13 and PP1 Disrupts Platelet-Driven Adhesion, Migration, and Transmigration of MDA-MB-231 Cells
We have previously reported that PP60src is a substrate of Btk and is the most abundant platelet tyrosine kinase. 46 Given that the foregoing data indicate that TQ inhibitory effect could be driven by Btk, we investigated the TQ impact on PP60src. Pretreating platelets with 10 µM of LFMA13 alone or in combination with 10 µM TQ for 10 minutes significantly reduces platelet adhesion to MDA-MB-231 cells by 50.75% ± 3.9% (P < .001).
1-Tert-butyl-3-(4-methylphenyl)pyrazolo[3,4-d]pyrimidin-4-amine or PP1, is a potent, reversible, ATP-competitive, and selective inhibitor of the Src family of protein tyrosine kinases.
PP1 inhibits the oncogenic protein PP60src, a tyrosine kinase that regulates cell proliferation and transformation. Platelet incubation with 10 µM of PP1 decreases adhesion to MDA-MB-231 by 47.25% ± 4.87%. There were no significant changes in the combination of TQ with LFMA13 or PP1 compared to TQ alone or between LFMA13 and PP1-treated cells (Figure 5A).

(A) LFMA13 inhibits platelet-induced cell adhesion. MDA-MB231 were cultured into 24-well plates at ~1 × 105 cells/well and incubated until complete confluence. Platelets were pretreated with 10 µM of TQ, and/or LFMA13 and PP1 for 10 minutes then, prelabeled with green CMFDA (10 µM) for 30 minutes at 37 °C and added to the wells at ~3 × 108 cells/mL for 1 hour. Values were presented as percentage of relative fluorescence intensity ± SEM of 4 separate determinations. ANOVA and Tukey’s test were performed for multi-test comparison. *P < .05. ***P < .001 compared to control (non-treated platelets). (B) LFMA13 inhibits platelet-induced cell migration. MDA-MB231 1 × 105 were added to the upper chamber, co-cultured with platelets pretreated or not with 10 µM of TQ for 10 minutes and/or LFMA13 and PP1 for 10 minutes. Migration was detected after 24 hours of incubation at 37 °C and 5% CO2. Migrated cells onto the outer side of the PET membrane were fixed and stained as described in the Materials and Methods section. Histograms represent the mean of migrated cell number of 3 PET membranes and 6 microscopic fields for each sample ± SEM. ANOVA and Tukey’s test were performed for multi-test comparison. **P < .01. ***P < .001 compared to control (non-treated platelets). (C) TQ inhibits platelet-induced MDA-MB231 transmigration. HUVEC 5 × 104 were added into each upper chamber of the insert and cultured for 24 hours at 37 ° and 5% CO2 until total confluence. MDA-MB231 1 × 105 were added to the upper chamber, co-cultured with platelets pretreated or not with 10 µM of TQ for 10 minutes and/or LFMA13 and PP1 for 10 minutes. Transmigration was detected after 24 hours of incubation at 37 ° and 5% CO2. Migrated cells through the HUVEC layer and adhered onto the outer side of the PET membrane were fixed and stained as described in the Materials and Methods section. Histograms represent the mean of migrated cell number of 3 PET membranes and 6 microscopic fields for each sample ± SEM. ANOVA and Tukey’s test were performed for multi-test comparison. *P < .05. **P < .01. ***P < .001 compared to control (non-treated platelets).
We also investigated the role of Btk and PP60src on TQ inhibiting platelet-induced MDA-MB-231 migration. Platelets were incubated with 10 µM of TQ alone and in combination with 10 µM LFMA13 or 10 µM PP1 for 10 minutes prior to indirect co-culture with MDA-MB-231 cells. As illustrated in Figure 5B, targeting Btk or PP60src significantly reduces the number of migrating cells by 51% and 49%, respectively, at the same level as TQ alone (50%). The pretreatment of platelets with LFMA13 and PP1 alone or in combination with TQ (48% ± 7.19% and 48.5% ± 8.52%) results in no differences (Figure 5B).
In the foregoing data, we demonstrated that Btk and PP60src pharmacological inhibition reduce platelet-induced MDA-MB-231 cells migration, which most likely affects the platelet induced MDA-MB-231 cells transendothelial migration. Thus, we pretreated platelets with 10 µM of TQ, LFMA13, and PP1 for 10 minutes at 37 °C prior to incubation with MDA-MB-231 cells. Platelet-educated MDA-MB-231 cells were the subject of transendothelial migration assay. As presented in Figure 5C, Btk and PP60src pharmacological inhibition significantly decreases (P < .001) the number of transmigrated cells, 156 ± 7 and 151 ± 8, respectively. In addition, platelet co-incubation with TQ in combination with LFMA13 or PP1 does not exhibit any significant differences compared to platelets incubated with TQ alone.
TQ and LFMA13 Attenuate Platelet-Induced Epithelial-Mesenchymal Transition in MDA-MB-231 Breast Cancer Cells
Platelets interact with cancer cells, promoting migration and invasion via the epithelial-to-mesenchymal transition (EMT). 6 On the basis of our earlier findings, we investigated the involvement of platelet Btk in platelet-induced EMT of MDA-MB-231 cells. Platelets were pre-treated with 10 µM LFMA13 and 10 µM TQ for 10 minutes at 37 °C, then allowed to interact with MDA-MB-231 cells for 1 hour. Vimentin and N-cadherin protein expressions, as EMT markers, were quantified by immunoblotting, as stated in the Materials and Methods section. The research revealed that TQ significantly decreased vimentin and N-cadherin protein levels by 38% ± 3% and 29% ± 4%, respectively. The combination of TQ and LFMA13 demonstrated nearly identical inhibition levels (Figure 6).

TQ and LFMA13 inhibit vimentin and N-cadherin expression. Platelets were pretreated with 10 µM of TQ and/or LFMA13 for 10 minutes then were co-cultured with MDA-MB-231 cells for 24 hours. MDA-MB-231 proteins were analyzed by SDS/PAGE and subsequent Western blotting with specific anti-N-cadherin and anti-vimentin antibodies as described in the Materials and Methods section. Histograms represent protein expression level and are presented as percentage of control (untreated resting cells) and expressed as mean ± SEM of 7 and 4 separate experiments, respectively. ***P < .001 compared to control (non-treated platelets).
In order to investigate the signaling pathway involved in TQ attenuating platelet-inducing EMT, we focused on the expression of FBXW7, an E3 ubiquitin ligase that mediates the proteasomal degradation of proteins. Shen et al and Kang et al described the FBXW7 as a transcription factor modulator that regulates the expression of EMT markers, including ZEB2 and β-catenin.47,48
As presented in the Figure 7, TQ alone can increase the expression of FBXW7 in platelet-educated MDA-MB-231 to 147% ± 4.3% and to 157.8% ± 5.6% in combination with LFMA13. In addition, TQ alone or in combination with LFMA13 significantly increases the protein level of β-catenin (136% ± 5.3% and 134% ± 3.6%, respectively). Conversely, the expression of ZEB2 is reduced by 73% ± 2.8% in comparison to non-treated platelets by the same platelet treatment.

Effect of TQ and LFMA13 on FBXW7, ZEB2, and β-catenin 1 expression. Platelets were pretreated with 10 µM of TQ and/or LFMA13 for 10 minutes then were co-cultured with MDA-MB-231 cells for 24 hours. MDA-MB-231 proteins were analyzed by SDS/PAGE and subsequent Western blotting with specific anti-FBXW7 (A-4 clone), anti-ZEB2 (E-11 clone), and β-catenin (E-5clone), antibodies as described in the Materials and Methods section. Histograms represent protein expression level and are presented as a percentage of control (untreated resting cells) and expressed as mean ± SEM of 6 or 4 separate experiments. *P < .05. **P < .01. ***P < .001 compared to control (non-treated platelets).
Discussion
Platelets are small, irregularly shaped blood cells that play an important role in blood coagulation. However, their aberrant activity has been linked to numerous types of diseases, including breast cancer. Platelets and cancer cells can interact in a multitude of ways. Breast cancer cell-platelet interactions can also facilitate the dissemination of cancer by increasing the survival, proliferation, and migration of cancer cells. TQ is an antiplatelet and anticancer natural molecule, that is, highly promising. Its capacity to suppress platelet activation, aggregation, and adhesion, as well as promote death in platelets, may contribute to its anticancer properties.
The present study examined the impact of thymoquinone (TQ) on the platelet-induced metastasis of MDA-MB-231 breast cancer cells in vitro. In a dose-dependent manner, platelet treatment with 1, 5, and 10 µM of TQ reduces adhesion to MDA-MB-231 cells and platelet-induced MDA-MB-231 cell migration. In addition, we demonstrated that TQ significantly suppresses platelet-induced MDA-MB-231 transmigration. TQ has been reported to inhibit platelet activation and granule secretion.21,22,49 This prevents the exposure of a number of adhesion molecules, including integrins, P-selectin, glycoprotein Ib-IX-V, and the immunoglobulin, which are essential for platelet cancer cell interaction and cancer extravasation.50 -52 This suggests that TQ has the potential to inhibit platelet-induced metastatic behavior of breast cancer cells in vitro.
Previous studies have reported that platelet activation leads to an increase in protein tyrosine phosphorylation.46,53,54 Consequently, we investigated the effect of TQ on the signaling pathways of platelet tyrosine kinase. We demonstrated that TQ reduces MDA-MB-231-induced tyrosine phosphorylation. The result is consistent with previous studies that have demonstrated the ability of natural products, such as, flavonoids, catechins, and echistatin, to inhibit platelet tyrosine kinase and tyrosine phosphorylation.55,56
The cytosolic tyrosine kinase protein Btk, is found in large amounts in platelets, and is part of signaling complexes that are linked to the cytoskeleton. 57 The phosphorylation of Btk is reduced by platelet pretreatment with TQ. Similar results were reported using the selective inhibitor of Btk, Ibrutinib. 58 The inhibition of Btk phosphorylation by TQ suggests that TQ may be targeting the Btk signaling pathway to inhibit platelet-induced migration of MDA-MB-231 cells.
Moreover, the results show that the Btk inhibitor LFMA13 also reduces platelet P-selectin expression, 59 and there was no additive effect when TQ was combined with LFMA13. This implies that TQ and LFMA13 may be inhibiting platelets through comparable mechanisms. We investigated the effect of TQ on PP60src since the previous study suggests that Btk may be the driving force behind the TQ inhibitory effect. 41 Platelet adhesion to MDA-MB-231 is significantly diminished by the pretreatment of platelets with TQ alone or in combination with the PP60src inhibitor, PP1. There were no significant differences observed when TQ was administered alone in comparison to LFMA13 or PP1 treatment, or when LFMA13 and PP1-treated cells were compared. Cell migration is significantly reduced when Btk or PP60src are targeted at the same level as TQ. Whether platelets were pre-treated with LFMA13 and PP1 independently or in combination with TQ, no differences were observed. Based on our findings, we proposed that TQ’s inhibitory effect may be mediated through the Btk-Src signaling pathway. Consistent with the previous hypothesis, numerous studies reported that TQ could inhibit the Src family members in various cell types.60-64
Based on the previously mentioned results, we propose that pharmacological inhibition of Btk and PP60src may lessen platelet-induced MDA-MB-231 trans-endothelial migration. The data indicated a significant decrease in the number of transmigrated cells. Furthermore, compared to platelets incubated with TQ alone, co-incubation of TQ with LFMA13 or PP1 does not significantly altered the results. That inhibition of Btk or PP60src in platelets reduces their adhesion and induction of migration in MDA-MB-231 cells, similar to the effects of TQ alone. This suggests that TQ may be targeting multiple signaling pathways, including Btk and Src, to inhibit platelet-induced metastatic behavior of breast cancer cells. Our results are in agreement with earlier findings that have elucidated the role of platelets in cancer dissemination.65,66 We propose that TQ acts through blocking platelet surface markers, granule, and platelet-derived vesicles secretion, all of which promote cancer spread.58,59,67
Platelets can enhance the EMT via different signaling pathways. 68 Based on our earlier findings; we investigated the involvement of TQ in platelet-induced EMT of MDA-MB-231 cells. Consistent with prior studies reporting that platelet-educated cancer cells upregulate the expression of vimentin and N-cadherin, 69 we found that pretreating platelets with TQ substantially decreased vimentin and N-cadherin protein levels in MDA-MB-231 cells. The combination of TQ and LFMA13 exhibited nearly the same level of inhibition. EMT is a critical process in cancer metastasis, where cancer cells acquire a more invasive and migratory phenotype. The results demonstrated that TQ and the Btk inhibitor LFMA13 decrease the expression of mesenchymal markers in MDA-MB-231 cells co-cultured with platelets, suggesting that TQ prevents breast cancer cells from acquiring a more invasive, migratory mesenchymal shape.
To investigate the signaling mechanism by which TQ inhibits platelet-induced EMT, we assess the FBXW7 expression. 70 TQ on its own can elevate FBXW7 expression in platelet-educated MDA-MB-231 cells. Furthermore, TQ by itself or in conjunction with LFMA13 considerably increases the protein level of β-catenin; conversely, ZEB2 expression is downregulated by the same platelet treatment. The increase of β-catenin implies a possible alteration in the Wnt signaling pathways, which are crucial in regulating EMT and the progression of cancer. 71
Simultaneously, the elevated expression of β-catenin 1, ZEB2, and FBXW7 suggests that important transcriptional regulators of EMT are being modulated. By targeting and degrading EMT-inducing transcription factors like Snail, FBXW7 can inhibit the EMT process. The transcription factor ZEB2 is responsible for driving the transition to a mesenchymal phenotype by activating EMT. 72 Furthermore, β-catenin 1 can co-regulate the expression of EMT genes via interacting with ZEB proteins. It appears that TQ via Btk can change the transcriptional landscape in MDA-MB-231 cells away from a pro-EMT state. This is possible because they upregulate some EMT regulatory proteins. Stabilizing FBXW7 to target EMT inducers for degradation and/or interfering with ZEB2/β-Catenin 1 complexes that regulate mesenchymal gene expression programs may be necessary to achieve this.
Overall, the data suggest that TQ might be able to stop the mesenchymal transition and invasive behavior of MDA-MB-231 breast cancer cells by blocking platelet-derived pro-EMT signals. This could happen by decreasing mesenchymal markers and increasing EMT-regulatory proteins. This emphasizes their potential as cutting-edge treatment options that target platelet-driven breast cancer metastases.
The combined effects of TQ on platelet signaling, adhesion, and EMT highlight its therapeutic potential. TQ has the potential to be a dual-action agent in the treatment of breast cancer because it targets the intrinsic signaling pathways within cancer cells as well as the influence of platelets on cancer cells. TQ and Btk inhibitors do not exhibit additive effects, indicating that they may have a similar mechanism of action. This finding supports the notion that TQ might be developed in conjunction with current treatments to increase their efficacy.
To summarize, TQ inhibits platelet-induced MDA-MB-231 metastasis in vitro via targeting platelet-cancer cell contacts and downstream signaling cascades. The principal mechanism includes inhibiting platelet activation and subsequent signaling processes that are essential for metastasis. TQ’s capacity to diminish platelet adhesion, migration, and transendothelial migration of cancer cells is due to its interference with tyrosine kinase signaling within platelets, specifically the suppression of Btk. By inhibiting Btk activity, TQ significantly reduces platelet-mediated cancer cell spread. TQ’s impact on FBXW7, β-catenin, and ZEB2 expression disrupts the EMT process, preventing cancer cells from migrating and becoming more aggressive. Platelet activation, which is controlled by TQ, has a direct impact on EMT, a vital stage in metastasis, demonstrating the interconnectivity of these pathways. As a result, TQ appears to modulate linked signaling pathways, inhibiting metastatic progression by targeting both platelet-mediated support for cancer cell dissemination and the intrinsic EMT program within cancer cells themselves. The convergence of these effects emphasizes TQ’s potential as a therapeutic drug capable of interrupting various aspects of the metastatic cascade.
This study should consider numerous restrictions and other confounding variables. First, the results are limited in their generalizability since they rely on a single breast cancer cell line (MDA-MB-231) and mostly use in vitro models, which might not adequately represent the intricate interactions in vivo. The incubation time of 10 minutes and the particular TQ doses (1, 5, and 10 µM) studied might not be typical of clinical situations. Furthermore, the study lacks deep mechanistic understanding of the ways in which TQ influences signaling pathways; more research is required, especially with regard to the specific downstream targets of FBXW7 and Btk in connection with TQ therapy. Moreover, variations in cell culture conditions may influence cellular responses, and the effects of co-morbidities and drugs on platelet function and cancer biology were not examined. To improve robustness and practical application, future studies should try to validate these results in vivo and across a wider variety of cancer types and circumstances.
In conclusion, TQ shows encouraging anti-transendothelial migration qualities in vitro by blocking EMT and platelet-induced signaling in MDA-MB-231 breast cancer cells. By modifying platelet connections and cancer cell signaling pathways, this work paves the way for further research on the role of TQ in cancer therapy and may present a novel approach to the management of breast cancer metastasis.
Supplemental Material
sj-pdf-1-ict-10.1177_15347354251378060 – Supplemental material for Thymoquinone’s Role in Suppressing Platelet-Mediated Migration and Epithelial to Mesenchymal Transition in MDA-MB231 Cells: Mechanistic Involvement of Btk Signaling
Supplemental material, sj-pdf-1-ict-10.1177_15347354251378060 for Thymoquinone’s Role in Suppressing Platelet-Mediated Migration and Epithelial to Mesenchymal Transition in MDA-MB231 Cells: Mechanistic Involvement of Btk Signaling by Nidhal Ben Amor, Sana Belkahla, Fahad Abdullah Al-Hizab and Juan Antonio Rosado in Integrative Cancer Therapies
Footnotes
Acknowledgements
Authors extend their gratitude to the Molecular Genetics and Genome laboratory, where some experiments were performed.
Ethical Considerations
The King Faisal University Ethics Committee approved this study in 2023 under the number ETHICS 1489.
Author Contributions
Nidhal Ben Amor: design, investigation, data analysis and interpretation, fund acquisition, writing manuscript draft. Sana Belkahla: investigation, data analysis and interpretation, and writing manuscript draft. Fahad Abdullah Al-Hizab: validation, supervision, and manuscript editing. Juan Antonio Rosado: validation, supervision, and manuscript editing. All authors reviewed and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from King Abdul-Aziz City for Science and Technology (KACST) number 11-BIO1813-06 (وع-37-726).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
