Abstract
Introduction:
Recent advancements in oncology have significantly improved cancer treatment outcomes and patient survival rates. Nonetheless, the therapeutic effects of cancer treatment often impair the patients’ quality of life (QoL). The most common symptoms include fatigue, pain, sleep disturbances, depression, and anxiety. This study aims to assess the impact of a structured mind-body medicine (MBM) group intervention on these specific symptom domains in patients with cancer.
Methods:
A cohort of patients with cancer engaged in an 11-week MBM program (6 hours weekly, March 2016-August 2017), combining psycho-oncology, lifestyle medicine, and integrative approaches including mindfulness, nutrition counseling, and movement therapies. Participants’ outcomes were evaluated for within-person (pre-post) changes using standardized questionnaires, including the Multidimensional Fatigue Index (MFI), the Perceived Stress Scale (PSS), the Pittsburgh Sleep Quality Index (PSQI), the Functional Assessment of Cancer Therapy (FACT-G), and the Hospital Anxiety and Depression Scale (HADS).
Results:
Assessments from 113 participants (94.7% female, 58.9% with breast cancer) revealed significant improvements (all P < .001) across all outcome measures. Effect sizes were medium to large: fatigue (Cohen’s d = 0.707), stress (d = 0.708), quality of life (d = 0.981), anxiety (d = 0.635), depression (d = 0.526), and sleep quality (d = 0.635). Clinical significance analysis showed that 50% to 70% of participants achieved minimal clinically important differences, with FACT-G showing the highest clinical relevance (70.1% improved ≥5 points). Wild bootstrap regression analysis with multiple testing correction identified baseline sleep quality as the strongest predictor: worse pre-program sleep was associated with greater improvements in depression (β = −.282 [95% CI: −0.467 to −0.099], P = .002) and sleep quality (β = −.432 [95% CI: −0.620 to −0.244], P < .001), surviving Bonferroni correction.).
Conclusion:
MBM programs may be a valuable addition to oncology care and they could improve patients’ quality of life. Incorporating these programs into standard cancer treatment protocols could provide a more holistic approach that addresses both the physical and psychological facets of cancer therapy.
Introduction
Cancer remains a global health challenge, consistently ranking as one of the most prevalent diseases worldwide.1,2 Although recent advancements have significantly improved cancer treatments and patient survival rates, 3 maintaining and improving quality of life (QoL) during and after treatment remains a major challenge for healthcare providers. The complex nature of cancer and its treatment often results in a diminished QoL that affects patients’ physical,4,5 psychological,5,6 social, and spiritual 7 well-being long after the active phase of the disease.
In response to the urgent need for supportive care that addresses these dimensions, complementary medicine has emerged as a significant contributor to improving the QoL of patients with cancer.8-12 The Mindfulness-Based Stress Reduction Program, pioneered by Kabat-Zinn at the University of Massachusetts, 13 is a standardized 8-week group-based intervention that integrates mindfulness meditation, yoga, and body awareness exercises. It was originally developed for patients with chronic pain and stress-related conditions, and has since been widely applied in oncology and other clinical settings. 9 Adapted programs like Mindfulness-Based Cancer Recovery (MBCR) 14 demonstrate positive effects on stress, depression, and anxiety in patients with cancer6,7,9,15 as well as on fatigue, 16 sleep quality, 17 immune response, and inflammation markers.18,19
The proactive inclusion of evidence-based multimodal interventions, focusing on relaxation techniques and mind-body medicine (MBM) strategies is now advocated in both national and international oncology guidelines.19-23
The specialized MBM program implemented at a tertiary care hospital in Germany builds upon these findings, integrating elements such as nutrition, exercise, stress management, disease coping, and order therapy in a structured group setting. Comparable intervention programs have already shown first hints of effectiveness in previous studies.10,24-26 The program is based on the “Essen model” of mind-body medicine, developed by Dobos et al, and evaluated by Cramer et al. 27 It consists of 11 weekly group sessions (6 hours each) of 8 to 12 patients. Each session integrates theoretical input and experiential components, addressing key areas such as mindfulness meditation, yoga, Qigong, nutrition, cognitive behavioral strategies, stress management, and self-care strategies. The group format fosters social support and peer learning. For a detailed overview, see the Supplementary Appendix.
The current study aimed to evaluate the real-world effectiveness of a structured 11-week mind-body medicine (MBM) group program on improving quality of life, stress, sleep, and fatigue in patients with cancer. Further we wanted to investigate the influence of disease-related factors (eg, tumor stage, type, treatment modalities) and patient-related factors (eg, educational level, prior experience with complementary and alternative medicine) on the effectiveness of MBM interventions.
By considering the specific context and real-world conditions, this study contributes to bridging the gap between international research findings and practical application.
While mind-body interventions are increasingly supported by research, their integration into the care structures of oncology centers, particularly in Germany, remains incomplete. This study supports the ongoing efforts to bridge that gap by providing structured outcome data from a comprehensive cancer center.
Materials and Methods
Design
This study is a retrospective, anonymized evaluation of the mind-body medicine (MBM) program as implemented in routine clinical practice and described in Supplementary Appendix. In addition to patient data, quality assurance questionnaires were also collected and evaluated. Our study followed a 2-stage approach: (1) analysis of the primary outcome, which was changes in quality of life after the program, assessed by the Functional Assessment of Cancer Therapy – General (FACT-G), fatigue (MFI), perceived stress (PSS), Pittsburgh Sleep Quality Index (PSQI), and psychological distress (HADS) administered before and after the MBM program; (2) secondary exploratory analysis of other questionnaire items and after supplementing the questionnaire database with clinical data from hospital electronic health records to investigate predictive factors for treatment response. This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies.
Patients
Patients with cancer participated in the MBM program between March 2016 and August 2017 as part of routine care at a tertiary care hospital in Germany. All patients aged 18 years or older with a confirmed oncological diagnosis were eligible to join. Only patients who completed at least 1 baseline and 1 follow-up questionnaire were included in the analysis. Patients who did not complete the MBM program or did not provide outcome data were excluded. Questionnaires were completed before and directly after the MBM program for quality assurance and personal feedback. This data has been retrospectively analyzed, and additional patient information, such as oncological diagnosis, tumor staging (TNM classification), oncological pharmacotherapy, long-term medications, and date of diagnosis, was extracted from the clinical information system (CIS). No a priori sample size calculation was conducted due to the retrospective nature of the study.
Questionnaire
The assessment comprised 5 standardized questionnaires – the Multidimensional Fatigue Index (MFI),28,29 which indicates higher fatigue with higher scores; the Perceived Stress Scale (PSS),30,31 where higher scores reflect greater perceived stress; the Pittsburgh Sleep Quality Index (PSQI), 32 with higher scores indicating more severe sleep disturbances; the Functional Assessment of Cancer Therapy (FACT-G), 33 where higher scores denote better quality of life; and the Hospital Anxiety and Depression Scale (HADS),34,35 where higher scores indicate greater anxiety or depression.
Several of these instruments contain validated subscales that assess distinct domains. For example, the MFI includes subscales for general, physical, and mental fatigue. The FACT-G comprises 4 domains: physical well-being, social/family well-being, emotional well-being, and functional well-being. The HADS includes separate subscales for anxiety (HADS-A) and depression (HADS-D). These subdomains were analyzed to gain a more nuanced understanding of the intervention’s effects.
Additionally, a non-standardized section collected sociodemographic details, and past and current complementary and alternative treatments.
Statistical Analysis
Statistical analyses were performed using IBM® SPSS® Statistics software version 29 and RStudio version 4.3.0. Data distribution was assessed through Shapiro-Wilk tests and visual inspection of Q-Q plots. Missing data in questionnaires were handled according to the specific protocols of the respective questionnaires where available. Complete case analysis was applied for each outcome measure, resulting in varying response rates due to partial missing data within individual questionnaires. Due to non-normal distribution of outcome measures and the ordinal nature of most questionnaire data, non-parametric tests were conservatively selected. Pre-post intervention comparisons were analyzed using Wilcoxon signed-rank tests. For independent sample comparisons, the Kruskal-Wallis test was applied across defined subgroups, with Mann-Whitney U tests for individual subgroup analyses. The significance level was set at P < .05. Effect sizes for paired comparisons were calculated using Cohen’s d, with magnitudes interpreted as small (d = 0.2-0.5), medium (d = 0.5-0.8), and large (d > 0.8). Clinical significance was assessed using established minimal clinically important differences (MCIDs): MFI ≥2 points, PSS ≥2.5 points, FACT-G ≥5 points, HADS subscales ≥1.75 points, and PSQI ≥3.5 points. Clinical relevance thresholds were defined as high (≥50% achieving MCID), moderate (30%-49%), and limited (<30%). Multiple linear regression models were constructed to evaluate changes in each predefined quality of life measure (MFI, PSS, FACT-G, HADS), incorporating all explanatory variables simultaneously. To address non-normal and heteroscedastic residuals, wild bootstrap with Rademacher weights (2000 samples) was applied. Bonferroni correction was implemented for multiple testing with α = .0036/outcome (0.05/14 predictors). Ninety-five percent confidence intervals for regression coefficients and Cohen’s d were estimated using bias-corrected and accelerated (BCa) bootstrap methods. The Charlson Comorbidity Index (CCI) was calculated to assess comorbidity impact and estimate 10-year survival probability.36,37 Complete analysis code and results are available upon request.
Results
Patient Characteristics
The study included 113 patients in a retrospective evaluation of a mind-body medicine (MBM) program designed for oncology patients. The participant group was predominantly female (94.7%, n = 107), with a median age of 56 years. The median time from initial diagnosis to program start was 8 months, and the median observation period was 77 days.
Regarding their education background, 37.2% held a university degree, 12.4% had a tertiary school degree, and 50.4% had completed secondary education. Forty-three percent of participants were employed during the study period. The majority (78%) were either married or in a committed partnership at the time of data collection. For details see Table 1.
Demographic Characteristics.

The most common cancer diagnosis was breast cancer (58.9%, n = 66), followed by ovarian cancer (12.5%, n = 14). Other diagnoses included lymphoma, pulmonary cancer, colorectal cancer, and various other types at lower frequencies. The tumor stages were primarily early-stage without metastases, with T1 and T2 stages accounting for 72.1% of cases. Metastases were present in 24.7% of cases.
Treatment modalities prior to or concurrent with the MBM program participation included chemotherapy (78.4%), surgery (73.9%), radiation therapy (40.5%), and hormonal therapy (28.8%). For details see Table 2.
Tumor Type, Staging, and Therapy.

Abbreviation: CLL, chronic lymphocytic leukemia.
Outcome Measures
After completing the mind-body medicine (MBM) program, patients showed a significant improvement (P < .05) in all 5 questionnaires and their subscales (see Table 3).
Outcome Variables at Baseline and Follow-Up.
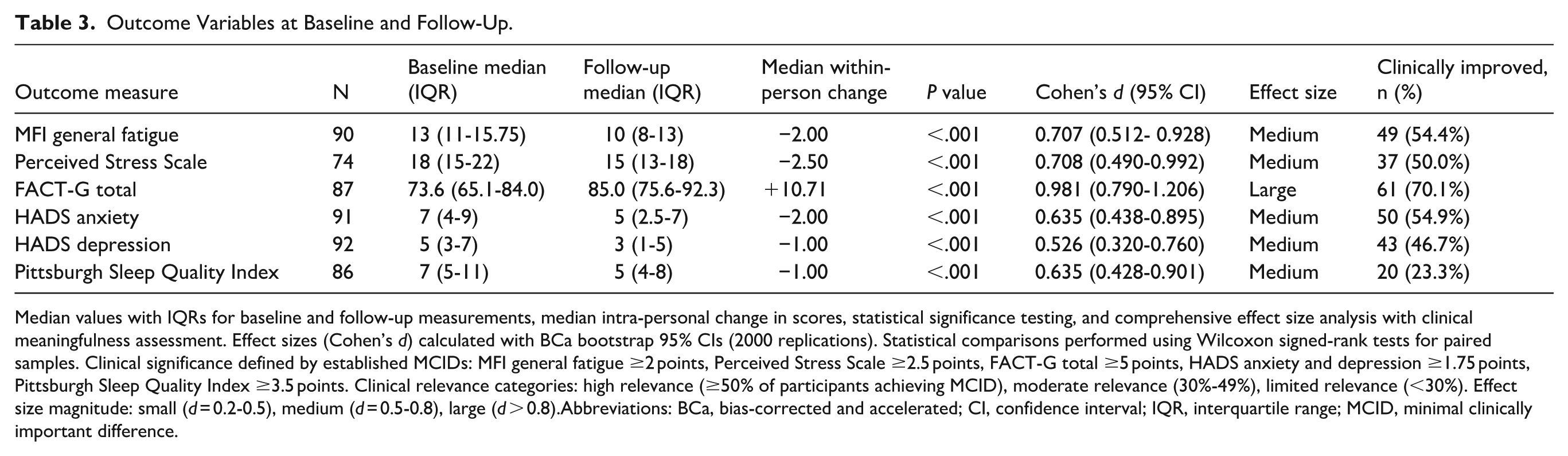
Median values with IQRs for baseline and follow-up measurements, median intra-personal change in scores, statistical significance testing, and comprehensive effect size analysis with clinical meaningfulness assessment. Effect sizes (Cohen’s d) calculated with BCa bootstrap 95% CIs (2000 replications). Statistical comparisons performed using Wilcoxon signed-rank tests for paired samples. Clinical significance defined by established MCIDs: MFI general fatigue ≥2 points, Perceived Stress Scale ≥2.5 points, FACT-G total ≥5 points, HADS anxiety and depression ≥1.75 points, Pittsburgh Sleep Quality Index ≥3.5 points. Clinical relevance categories: high relevance (≥50% of participants achieving MCID), moderate relevance (30%-49%), limited relevance (<30%). Effect size magnitude: small (d = 0.2-0.5), medium (d = 0.5-0.8), large (d > 0.8).Abbreviations: BCa, bias-corrected and accelerated; CI, confidence interval; IQR, interquartile range; MCID, minimal clinically important difference.
All outcome measures demonstrated statistically significant improvements with medium to large effect sizes. Quality of life (FACT-G) showed the largest effect (d = 0.981), followed by stress and fatigue measures (both d ≈ 0.71). Clinical significance analysis revealed that 4 outcomes achieved high clinical relevance (≥50% of participants reaching MCID thresholds): FACT-G (70.1%), MFI (54.4%), HADS-A (54.9%), and PSS (50.0%). HADS-D showed moderate clinical relevance (46.7%), while sleep quality improvements, though statistically significant, had limited clinical relevance in the whole cohort (23.3%).
Patients with hematological neoplasia demonstrated substantial improvements across all measures applied, with baseline scores that were generally significantly worse compared to other tumor types (data not shown). For instance, in the PSS, hematological neoplasia patients showed a median improvement of 7.5 points (from 22.0 to 14.5). In the FACTG, the improvement was particularly distinct, with a median increase of 17.7 points (from 66.8 to 84.5). These improvements were more pronounced than those seen in other tumor types, suggesting that patients with hematological neoplasms or those with a particularly affected QoL may have benefited more significantly from the intervention (see also Figure 1A).

Differences in change (before and after the mind-body medicine intervention) in the MFI, HADS-A, and PSQI stratified by clinical subgroups. (A) Changes in MFI general scores stratified by tumor entity, negative values indicate more improvement (lower fatigue). (B) Changes in HADS-A scores stratified by tumor stage, negative values indicate more improvement (lower anxiety). (C) Comparison of change in the PSQI stratified by participants with and without sleep disorders before the intervention. Boxes represent the IQR with the median indicated by the horizontal line. Whiskers extend to 1.5 times the IQR or the most extreme data point if closer. Points beyond the whiskers represent outliers. The red triangle indicates the mean, with error bars showing the 95% confidence interval. The asterisk (*) denotes a statistically significant difference (P < .05) between subgroups with and without sleep disorders (C), respectively.
Regarding tumor stages (TNM classification), early-stage tumors (T1 and T2) showed more significant improvements compared to advanced stages (T3 and T4) or metastases (M1). This was particularly evident in the HADS anxiety dimension, where patients with early-stage disease showed a median reduction of 2 points (from 7.0 to 5.0), compared to a reduction of 2 points (from 6.0 to 4.0) for patients with advanced stages and 0.5 points (from 5.0 to 4.5) for patients with metastasis. Interestingly, it is important to note that despite their disease being less advanced disease, patients with lower T stages (T1 and T2) actually reported slightly higher baseline anxiety scores (median 7.0) compared to those with more advanced T stages (T3 and T4, median 6.0) or metastatic disease (M1, median 5.0; see Figure 1B).
Those with baseline sleep disturbances had significantly worse initial scores in the other QoL questionnaires but also showed significantly greater improvements following the intervention compared to those without sleep disturbances in all QoL measures (see Figure 1C and Supplementary Appendix); these results are reflected in our regression models.
Regression Models
Predictor variables included demographic factors (age, BMI, education), treatment-related variables (tumor stage, metastases, active therapies), program participation (attendance days), clinical characteristics (comorbidity index), and baseline symptom burden (sleep quality, time since diagnosis). Linear regression analysis identified baseline sleep quality (PSQI) as the most consistent predictor of treatment response. After Bonferroni correction for multiple testing (α = .0036), 2 significant associations remained: (1) sleep quality: worse baseline sleep quality predicted greater sleep quality improvement (β = −.432 [95% CI: −0.620 to −0.244], P < .001). (2) Depression: worse baseline sleep quality predicted greater depression reduction (β = −.282 [95% CI: −0.467 to −0.099], P = .002). Additionally, before multiple testing correction, longer time between diagnosis and program start was associated with greater stress reduction (β = −.061, P = .033) and better functional outcomes (β = .088, P = .005), though these did not survive conservative correction. For additional details on these analyses, please refer to Supplementary Appendix.
Discussion
Our investigation into the effects of a structured mind-body medicine (MBM) intervention on quality of life in patients with cancer yielded significant improvements across various domains. We observed statistically significant improvements in quality of life (FACT-G), fatigue (MFI), sleep quality (PSQI), perceived stress (PSS), and psychological distress (HADS-A and HADS-D) following the 11-week MBM program. The clinical meaningfulness of our findings is particularly noteworthy. The median FACT-G improvement of 10.71 points substantially exceeds the established MCID of 5 to 7 points, with 70.1% of participants achieving clinically relevant improvements.
Similarly, fatigue, anxiety, and stress measures showed both statistical significance and clinical relevance, with approximately half of all participants reaching meaningful improvement thresholds. This dual demonstration of statistical and clinical significance strengthens the evidence for MBM program effectiveness in real-world oncology settings. Similar effects have been reported in prior MBM and MBSR studies, including randomized trials, 39 observational cohort studies,24-26 and meta-analyses.9,38 A representative survey by Voiß et al 12 further underscores the high acceptance and perceived value of MBM among breast cancer patients.
After the program, participants reported an average improvement of ~20% on the QoL scales. Our study therefore contributes to the existing body of knowledge by reaffirming the effectiveness of MBM programs in improving quality of life among patients with cancer, mirroring outcomes observed in prior research with this particular 11-week program. 24
Although, similar to other studies, patients with breast cancer in particular made up a large proportion of the cohort in our study, positive results were observed in various tumor entities, suggesting that MBM interventions can be used for more than just certain types of cancer, as has been suggested in other studies.6,9,12,40
However, the composition of our real-life cohort as an unselected sample of participants raises questions about the underutilization of MBM interventions in non-gynecological tumor entities and advanced disease cases. This also appears to be of interest as the greatest effects were observed in patients with early-stage disease. The potential relevance of gender-specific aspects has also not yet been sufficiently well and clearly investigated.9,38,41 One notable aspect of our study was the gender distribution of the participants, with 94.7% being female. This gender imbalance is consistent with previous research on MBM interventions for patients with cancer, which have often reported a higher proportion of female participants or focused on gynecological tumor entities.6,9 This trend may be also attributed to several factors, such as gender differences in health-seeking behaviors, and the perceived acceptability of mind-body practices. 12 Future research should consider stratifying participants based on gender, symptom manifestation, and tumor entity to better understand intervention efficacy and clinical relevance, thereby informing more targeted treatment approaches. 42
Interestingly, we found an association between the time from diagnosis to the start and the effects of the MBM program: For each additional month between diagnosis and the start of the program, participants reported a more pronounced effect on lower stress and better functional assessment. We can only speculate as to why people with a longer waiting time for the program seem to benefit more from it. It may be that the need is particularly great at this time, perhaps because the symptom burden increases with the duration of therapy. Alternatively, it is possible that shortly after diagnosis, patients are still confronted with many different challenges and issues and are not yet able to focus sufficiently on the content of a structured program. However, this observation could alert us to the optimal timing of our interventions in the future, particularly in view of the large resources required of this program for the large number of patients in need.
Our analysis revealed sleep quality as the strongest predictor of treatment response – a novel finding in MBM research. Participants with worse baseline sleep disturbances experienced greater improvements in both sleep quality and depression measures, with these associations surviving conservative multiple testing correction. This suggests that sleep disturbances may serve as a treatment-responsive phenotype, identifying patients who are particularly likely to benefit from mind-body interventions. Given the established links between sleep quality and cancer survival, this finding has important clinical implications for patient selection and treatment timing. Therefore, recent other studies emphasize the critical importance of sleep quality as well, not only for psychological well-being but also for survival in patients with cancer.43-45 These findings highlight the need for tailored interventions that address sleep disturbances as a key factor in improving both quality of life and clinical outcomes.7,17,46,47
Despite its contributions, our study is not without limitations. Due to the retrospective design of the study, causal attribution of the results is only possible to a limited extent. The predominantly female sample and the predominance of breast cancer and gynecological tumors limit the generalizability of our findings to male patients, so that further studies are necessary to explore gender differences. Additionally, data constraints hindered a comprehensive assessment of socioeconomic status on treatment influences. The absence of a control group and retrospective nature do not allow for hypothesis about causality.
Future research should focus on several key areas to build upon our findings. Investigating the optimal timing of MBM interventions in the cancer care trajectory is crucial, possibly exploring a staged approach offering different support types at various post-diagnosis time points. Studies on MBM-programs should also include measures on sleep quality and how this mediates overall quality of life. Longitudinal research is needed to assess long-term effects on sleep quality, quality of life, and, potentially, survival rates.
In conclusion, our study provides robust evidence for the clinical effectiveness of MBM interventions in cancer care, demonstrating not only statistical significance but meaningful clinical benefits for the majority of participants. The identification of baseline sleep quality as a key predictor opens new avenues for personalized intervention approaches. The integration of MBM into standard cancer care protocols, particularly for patients with sleep disturbances, could substantially improve patient-reported outcomes and quality of life in oncology populations.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251378056 – Supplemental material for Effects of a Mind-Body Medicine Group Program for Cancer Patients: A Retrospective Cohort Study
Supplemental material, sj-docx-1-ict-10.1177_15347354251378056 for Effects of a Mind-Body Medicine Group Program for Cancer Patients: A Retrospective Cohort Study by Severin Schricker, Felix Breßmer, Mark Dominik Alscher, Claudia Löffler, Holger Cramer and Marcela Winkler in Integrative Cancer Therapies
Supplemental Material
sj-html-2-ict-10.1177_15347354251378056 – Supplemental material for Effects of a Mind-Body Medicine Group Program for Cancer Patients: A Retrospective Cohort Study
Supplemental material, sj-html-2-ict-10.1177_15347354251378056 for Effects of a Mind-Body Medicine Group Program for Cancer Patients: A Retrospective Cohort Study by Severin Schricker, Felix Breßmer, Mark Dominik Alscher, Claudia Löffler, Holger Cramer and Marcela Winkler in Integrative Cancer Therapies
Footnotes
Acknowledgements
We sincerely thank Elizabeth Morris for language editing and proofreading to again significantly improve our manuscript.
Ethical Considerations
The Ethics Committee of the University of Tübingen approved this study (project number: 098/2018BO2).
Author Contributions
S.S., F.B., M.D.A., and M.W. contributed to the study concept and to the methodology. S.S., M.W., C.L., and H.C. drafted the manuscript. M.W., S.S., C.L., M.D.A., and H.C. critically reviewed and edited the article. The investigation has been carried out by F.B. and S.S. contributed to the data curation and visualization of the data. F.B. and S.S. were responsible for the formal analysis. Supervision and funding were contributed by M.D.A. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.L.: lecture fees: Celgene GmbH, Roche GmbH, Novartis Pharma GmbH, BMS GmbH and Co. KGaA, Mundipharma GmbH Co. KG, Merck KGaA. The other authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Data Availability Statement
The questionnaire and datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
