Abstract
Background. Chronic fatigue is one of the most restricting symptoms following primary breast cancer treatment, but clinical studies on symptom management are rare. The objective was to evaluate the impact of a multimodal mind–body program (MMMT), including moderate physical activity as compared with a walking intervention alone, on chronic fatigue symptoms of women with stage I to IIIA breast cancer. Patients and Methods. Sixty-four women (mean age = 56.7 years) suffering from chronic fatigue after active tumor treatment were randomly assigned to either an experimental or a control (n = 32 each) intervention (10 weeks). Fatigue, quality of life (QoL), functional well-being, anxiety, and depression were measured with standard questionnaires at baseline, after 10 weeks, and after 3 months. Results. Compared with baseline, both groups had reduced fatigue scores after treatment without any significant difference between groups (posttreatment, Δ = −0.3, confidence interval = −1.6 to 1.0, P = .678; follow-up, Δ = −0.4, confidence interval = −1.8 to 0.9, P = .510). All patients also improved regarding QoL and general functional well-being. Conclusion. Since both interventions reduced fatigue symptoms and enhanced QoL to a similar extent, we observed no verifiable add-on effect of the MMMT regarding fatigue symptoms. Considering the higher costs with additional expenditure related to MMMT, home-based walking intervention is recommended.
Introduction
Fatigue is one of the most frequent symptoms in cancer patients 1 with accompanying symptoms such as pain, psychological disorders, sleep disturbances, and mental/cognitive impairments. The physical, psychological, and social functions of fatigue patients are disturbed2-4 and moreover patients’ quality of life (QoL) is enormously impaired.5,6 Rather than being an isolated complaint, “cancer-related fatigue” (CRF) is a manifold symptom complex, for which Cella et al 7 proposed a set of diagnostic criteria. 8
Acute CRF accompanies tumor and actual therapy whereas chronic CRF shows no proof of a current tumor. 1 About 33% of breast cancer patients suffer from the latter and its etiology is yet unclear with only a limited number of clinical studies available. 9 Multifactorial processes, including dysregulation of cytokine-dependent homeostasis in hormonal vagal–neuronal immune functions, are discussed as contributing factors.10,11 Especially the loss of physical activity due to disease and its therapy as well as increased mental distress caused in the course of illness and therapy have both been frequently linked to CRF.12,13 With overweight and psychological distress identified as predictors of CRF, 9 2 feasible and beneficial therapeutic strategies are currently advocated for the rehabilitation period: (a) moderate physical activity and (b) psychological/educational interventions,3,4,14-16
Physical training programs, however, show comparably smaller success and less adherence values than psychological programs, especially in patients with high fatigue values. 17 A setting such as multimodal mind–body medicine settings, in which diet, physical activity, and psychological intervention are combined, might enhance compliance and treatment effects. Multimodal mind–body medicine treatments (MMMTs) successfully combine physical and educational exercises with relaxation techniques and are beneficial for a variety of conditions including pain and stress disorders. 18
The aim of the present randomized controlled trial (RCT) was to evaluate if a MMMT program reduces CRF symptoms with accompanying psychological parameters in breast cancer survivors more than a moderate endurance sports intervention alone.
Methods
Patients
Sixty-four female stage I to III breast cancer patients with written informed consent were included in the study, if they had completed their tumor treatment (operation and/or chemotherapy and/or radiotherapy) at least 3 months before, if they scored “unusual fatigue during the past month” >40 mm on an 100 mm visual analogue scale (VAS); and if they were willing and physically/mentally able to participate in a 10-week walking treatment (WT) and a 6-hour MMMT day once a week.
Exclusion criteria included anemia (hemoglobin <10 g/dL), a limitation for endurance sports (peripheral vascular abnormalities, hypo/hypertension, metabolic diseases, neuropathy without connection to the cancer or tumor treatment, limitations in the musculoskeletal system), insufficient knowledge of German language, an established routine practice of physical activity/relaxation (more than 3 × 30 min/wk), concomitant psychotherapy, or a supportive treatment with stimulants such as amphetamines, methylphenidate, or erythropoietin.
All patients were recruited by local newspaper, radio, and television announcements, screened via standardized telephone interviews, and subsequently invited to the clinic for further explanation and evaluation of eligibility.
Study Design
This study was a single-center RCT comparing a MMMT, including WT, with a control group offered a home-based WT alone. The study protocol (No. 04-2575) was approved by the institutional review board of the Medical Institutions of the University of Duisburg-Essen, Germany and complied with the Declaration of Helsinki. 19
The study took place at the Department of Internal and Integrative Medicine, Knappschafts-Krankenhaus, Kliniken Essen-Mitte, academic teaching hospital of the University of Duisburg-Essen, Germany. All patients were included in 3 blocks of equal size for the MMMT intervention.
Treatment Allocation
Patients were randomly assigned to treatment groups by a nonstratified block-randomization with fixed block length of 10. The biometrician drew random numbers from the “ranuni” random number generator of the SAS software (SAS Institute, Cary, NC) and prepared sealed, sequentially numbered opaque envelopes containing the treatment assignments. When a patient fulfilled all enrolment criteria, the study physician opened the envelopes in ascending order to reveal that patient’s assignment. The assignment sequence was kept secret with the biometrician and was not accessible to the study physicians.
Interventions
The MMMT was led in a day clinic setting by a multiprofessional team and included nutrition counseling, relaxation exercises, physical exercises, stress reduction, basics of cognitive restructuring, and hydrotherapy (Figure 1). A special role was assigned to the integration of mindfulness techniques introduced by Kabat-Zinn et al. 20 In each of the 10 weeks, patients attended in steady groups of 10 to 20 patients a 1-day clinic element of 6 hours duration. The program consisted of lectures, group exercises, guided discussions, and medical consultations. Learned methods included different types of meditation (eg, sitting meditation, body scan), whole-food cooking, naturopathic self-help strategies, and mindfulness in everyday situations. Certainly, the cooperation among participants, the contribution of the group to the learning, and the group process are contributing factors for the success of such a program.

Scheme for the multimodal day clinic intervention.
All patients were asked to practice the newly learned methods at home as often as possible. They were additionally given supervised sessions on walking by an experienced sports therapist (weeks 1, 3, and 10) and were instructed to continue walking at home 3 × 30 min/wk. The recommended heart rate during training was 180 − |chronological age| ± 10. A heart rate of 220 − |chronological age| was not to be exceeded.
Patients in the control group received identical supervised sessions on walking and were also instructed to practice 3 × 30 min/wk over a period of 10 weeks.
Outcome Measures
Outcomes were assessed by questionnaires at baseline (week 0), immediately after the interventional period (week 10), and 3 months afterward (week 22).
Primary Parameter
Unusual fatigue of the last month was rated on a 100 mm VAS (0 = not at all, 100 = extremely/completely) of the German Fatigue Assessment Questionnaire 21 (“How strongly did you feel extraordinarily (meaning “extraordinarily” for you) tired in the last month?”). A VAS in general is a psychometric response scale, on which respondents specify their level of agreement to a statement by indicating a position along a continuous line between 2 endpoints. VASs are common and feasible methods to assess fatigue4,22 and were also used in our pilot trial. 23 A symptom reduction of 30% was considered to be clinically significant. 24
Secondary Parameters
Unusual fatigue of the last week (“How strongly did you feel extraordinarily tired in the last week?”) and the intensity of perceived suffering were correspondingly assessed (“If you felt extraordinarily tired in this period of time—last week or month, respectively—, how strongly did you suffer from it?”).
The 30 core items of the European Organization for Research and Treatment of Cancer (EORTC) QoL questionnaire (QLQ-C30) is a cancer specific multidimensional self-report instrument, which is well validated and used worldwide.2,25 The QLQ-C30 incorporates several single-item symptom measures and 9 multi-item scales: 5 functional scales, 3 symptom scales, and 1 global health and QoL scale.
The Multidimensional Fatigue Inventory is a 20-item self-report instrument designed to measure fatigue in cancer patients. 26 It covers the dimensions general, physical, and mental fatigue, reduced motivation, and reduced activity.
The Hospital Anxiety and Depression Scale (HADS) is a reliable scale for detecting states of depression and anxiety in the setting of a hospital, medical outpatient clinic and is also validated for cancer patients. 27 Seven out of 14 questions for assessing depression and anxiety respectively, range from 0 to 21 with higher scores indicating a higher level of symptoms.
The Menopausal Rating Scale (MRS) is a formally validated with 11 items used worldwide for assessing health-related QoL of women in the menopausal transition. 28 The global sum scale ranges from 0 to 44; subscales cover psychological, somatovegetative, and urogenital symptoms.
Statistics
In a pilot trial with 20 breast cancer patients, the same primary outcome decreased by 2.3 ± 2.3 cm after the intervention. 23 Assuming a dropout rate of 15% and an improvement in the WT group of 0.5 ± 2.3 cm, we planned to include a minimum of 64 patients. This is enough for a 2-sided t test (α level = .05) to detect the above group difference with a power of (1 − β) = .80.
Group differences at baseline were expressed as arithmetic means ± standard deviations and analyzed by χ2 or Wilcoxon–Mann–Whitney tests.
All safety and effectiveness analyses were based on the intention-to-treat-population, which included each randomized patient who received at least one allocated treatment regardless whether she further adhered to the protocol or not. Missing data in the questionnaires were multiply imputed by Markov chain Monte Carlo methods.29,30 This gave a total of 5 complete data sets, each of which was analyzed separately as described below. Afterward, the results were adequately combined to produce overall effect size estimates, confidence intervals, and P values.
Time courses of each outcome parameter were analyzed with univariate analyses of covariance for repeated measurements, with time (2 levels) as a within-person factor, the respective baseline value as a linear covariate, and the smoking behavior (yes/no) and the actual status of recurrence (yes/no) as binary cofactors. All analyses were conducted with the SAS statistical software (release 8.2, SAS Institute, Cary, NC).
Results
A total of 335 prospects were screened via a standardized telephone interview for study participation, 183 patients were found eligible for further evaluation. Of those, 51 patients stated not to have enough time to participate, 21 patients declined because of the long access route to the clinic, 11 patients could not be recontacted (incorrect telephone numbers), 3 patients denied randomization, and 2 patients suffered from fatigue too severe to participate. The remaining 64 patients were included and randomized, but baseline data of 9 patients could not be assessed: Two MMMT patients withdrew their consent because of time limitations or difficult access routes, respectively. Seven WT patients dropped out before the first intervention, 2 because of dissatisfaction with the result of randomization, 1 because of a new diagnosis of metastatic disease, 2 because of worsening of hip and/or knee pain, 1 for unexpected lack of time, and 1 was not willing to return the questionnaire. Data of 55 patients (MMMT 30, WT 25) were analyzed (Figure 2).
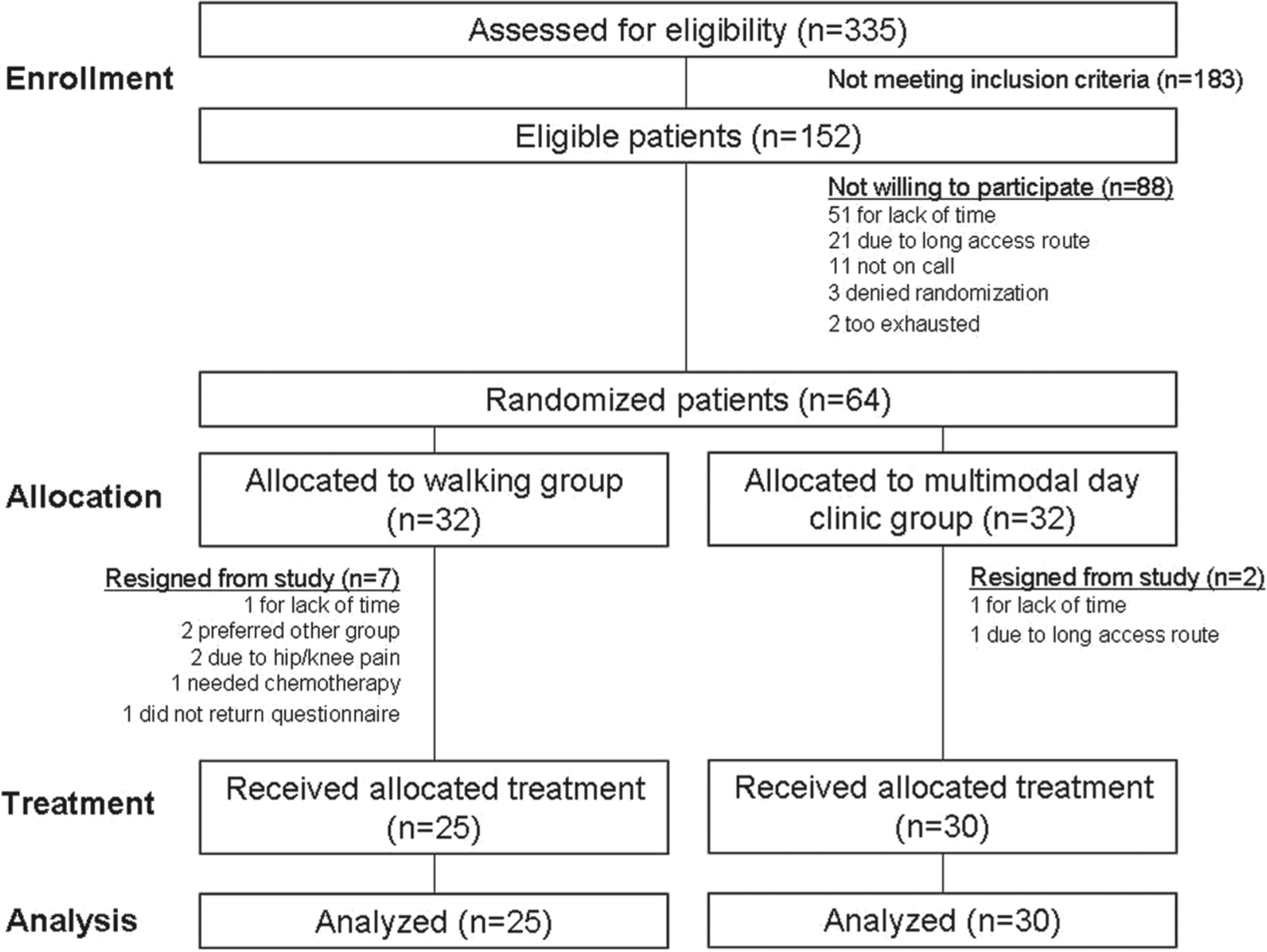
Study flowchart.
Sample Characteristics
Both treatment groups were comparable with respect to sociodemographic and history-taking data (Table 1) with 2 exceptions: smoking behavior and the actual status of recurrence. Both factors were included as adjusting factors in the analyses of covariance.
Sample Characteristics.

Abbreviations: WT, walking treatment; MMMT, multimodal mind–body medicine treatment; VAS, visual analogue scale.
The first diagnosis could not be assessed in 2 patients of each group because of incomplete records.
Statistically significant group.
Difference at baseline: inclusion as adjusting factors in analyses of covariance.
At baseline, the patients were slightly overweight (mean body mass index at 26.9 ± 4.3 kg/m2 for the WT group and 26.6 ± 4.1 kg/m2 for the MMMT group). During the study, body weight stayed constant in both groups changing from 73.7 ± 12.7 to 73.5 ± 12.7 kg and 73.5 ± 12.9 kg in the WT group, and from 73.5 ± 12.9 to 73.4 ± 12.4 kg and 73.7 ± 12.6 kg in the MMMT group.
Safety and Acceptance of the Interventions
No unexpected or adverse effects were observed during the study and the overall acceptance was good to moderate. Ten percent of the MMMT and 16% of the WT group stated to have difficulties with the physical training. The attendance rate in the MMMT group was 92.7%. The number of days per week with physical training was assessed with adherence diaries and was comparable between groups with 2.6 ± 2.0 days in the MMMT and 2.6 ± 1.3 days in the WT group. Seventy-six percent of the MMMT and 80% of the WT patients reported a subjective improvement of physical fitness at the end of the study.
Primary Outcome Parameter
Unusual fatigue of the last month decreased not only for the MMMT group from baseline (6.5 ± 1.6) to posttreatment (4.5 ± 2.3) and to follow-up (4.1±2.2), but also for the WT group (from 6.5 ± 1.6 to 4.6 ± 2.2 and 5.2 ± 2.4, respectively) in a clinically meaningful extent (see Figure 3). But for both time points this resulted in small, nonsignificant group differences of Δ = −0.0 (confidence interval [CI] = −1.2 to 1.2, P = .981) posttreatment and Δ = −1.0 (CI = −2.2 to 0.2, P = .122) at follow-up.

Unusual fatigue in the last month decreases for both groups in a clinically meaningful extent without any significant group differences. Given percentages refer to differences compared with baseline values in each group.
Secondary Outcome Parameters
Unusual fatigue of the last week similarly improved in both groups: MMMT patients rated their fatigue posttreatment (4.1 ± 2.1) as well as at follow-up (4.6 ± 2.5) as lower than at baseline (6.1 ± 2.4), whereas WT patients improved in a similar extent (from 6.8 ± 1.7 to 4.6 ± 2.6 and 5.2 ± 2.9). These ratings resulted in nonsignificant group differences of Δ = −0.3 (CI = −1.6 to 1.0, P = .678) and Δ = −0.4 (CI = −1.8 to 0.9, P = .510). Correspondingly, suffering from fatigue did not show any group difference with Δ = −0.0 (CI = −1.5 to 1.5, P = .999) after treatment and Δ = −1.1 (CI = −2.5 to 0.4, P = .153) at follow-up (MMMT patients improved from 5.7 ± 2.9 to 4.4 ± 2.8 posttreatment and to 4.0 ± 2.7 at follow-up, whereas WT patients improved from 6.3 ± 2.4 to 4.6 ± 2.7 and 5.2 ± 3.0, respectively).
Both groups yielded improvements on all functional scales of the EORTC QLQ 30 C (see Table 2). HADS–depression values of both groups were unremarkable with all assessments and indicated a possible impairment in regard to norm values. Moreover, both groups reduced their anxiety in the course of the study. Although MMMT patients showed more benefit than WT patients after treatment (Δ = −1.6, CI = −3.2 to −0.1, P = .043) this group difference could not be maintained at follow-up (Δ = −0.6, CI = −2.2 to 0.9, P = .422; see Table 3).
Secondary Outcomes Over Time (WT, n = 25; MMMT, n = 30). a

Abbreviations: WT, walking treatment; MMMT, multimodal mind–body medicine treatment; VAS, visual analogue scale; MFI, Multidimensional Fatigue Inventory; EORTC QLQ-30C, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; QoL, quality of life; HADS, Hospital Anxiety and Depression Scale; MRS, Menopausal Rating Scale.
Values are arithmetic means ± standard deviations.
Statistics for Secondary Outcomes (WT, n = 25; MMMT, n = 30).

Abbreviations: WT, walking treatment; MMMT, multimodal mind–body medicine treatment; 95% CI, 95% confidence interval; VAS, visual analogue scale; MFI, Multidimensional Fatigue Inventory; EORTC QLQ-30C, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire; QoL, quality of life; HADS, Hospital Anxiety and Depression Scale; MRS, Menopausal Rating Scale.
CI and P values are based on the intention-to-treat analyses of covariance (see Statistics section). For the MFI 10 and the EORTX QLQ 30C scales, there are neither any significant baseline differences between groups nor any group differences at later points. For HADS and MRS results, see explanation in the text. Being secondary outcomes, significance level for these measures was not α-adjusted.
P < .05.
Psychological menopausal symptoms (respective MRS subscale) decreased in both groups, but these improvements were more pronounced in the MMMT group at end of treatment (Δ = −1.5, CI = −2.9 to −0.2; P = .029) and follow-up (Δ = −1.5, CI = −2.8 to −0.1; P = .035). MRS total scores, however, did not differ significantly between groups at any time point (see Table 3). For the other menopausal symptoms, neither any group differences at any of the assessments nor any relevant improvements over time were obtained, reflecting no relevant changes concerning somatovegetative or urogenital symptoms (see Table 3).
Perceived pain assessed by the subscale of the EORTC QLQ-C30 stayed constant in the WT group, but MMMT patients indicated less perceived pain after the intervention with a significant group difference at follow-up (Δ = −14.6, 95% CI = −27.8 to −1.5; P = .031). All other scales of the EORTC QLQ-C30 showed improvement over time, but no group differences were observed.
Medication
Following the instruction component of the study the majority of patients kept their medication habits as usual. At baseline, 6 patients per group (24% of WT, 20% of the MMMT patients) took pain medication at least once a week, 18 of the WT (72.0%) and 22 of the MMMT (73.3%) group used it once a month or less, and 1 (4.0%) versus 2 (6.7%) provided no details. One MMMT patient stopped her hormone therapy owing to rheumatic side effects in accordance with her gynecologist and 1 WT patient took more beta-blockers. Five (16.7%) of the MMMT patients and 6 (24%) of the WT patients did not complete questionnaires at follow-up.
Discussion
In a group of breast cancer patients with CRF a MMMT and a home-based WT could both reduce fatigue and associated symptoms and improve QoL in a clinically meaningful extent. However, no verifiable add-on effect of MMMT regarding fatigue symptoms was observed.
Fatigue, Quality of Life, Psychological, and Functional Aspects
Surprisingly, in our study population the EORTC QLQ-C30 subscale physical function revealed high values of almost 70 (out of 100 possible points) already at baseline. The rather little improvement of about 10% might thus be due to a ceiling effect. General fatigue symptoms as assessed by the VAS and additional questionnaires show indeed relevant reductions with up to 30%. The presented study focused on chronic CRF the intensity of these long-term symptoms is assumed to be more severe and hard to alter. Regarding the long period patients had already been suffering from the symptoms, these are considerable improvements.
Physical activity is already known to be effective in the management of CRF during the rehabilitation period.3,4,14-16 Improvement of physical parameters associated with physical training such as enhanced pulmonary function are positively correlated with a reduction of fatigue symptoms, heightened QoL and better functional values.17,31,32 An advantage of home-based WT is that organization and training are in the hands of the patient and thus self-efficacy is more pronounced, since a main explaining factor for the variability in QoL of breast cancer survivors is the sense of control. 6 Furthermore, with rather minimal costs and small efforts for WTs, this intervention can be logically recommended. However, higher fatigue values are associated with comparably smaller success and less adherence in physical training program. 17
Increased anxiety is found among breast cancer survivors6,33,34 and is presupposed to have a negative impact on various symptoms, while psychological/educational interventions are advocated to be effective in CRF therapy.3,4,14-17 Since mind–body programs incorporate a wide range of different therapeutic aspects, including diet, physical activity, and psychological intervention, we anticipated that such a setting might enhance compliance and treatment effects. Corresponding to our pilot trial, 23 the MMMT was indeed effective in reducing fatigue symptoms as well as in improving QoL and physical, role, emotional, cognitive, and social functions (EORTC subscales). And patients with anxiety and pain had a slightly greater benefit regarding those specific symptoms, if they participated in the MMMT. This might be a worthwhile research topic for future studies. But we were unable to demonstrate a superiority of the MMMT intervention for all the other outcome measures, neither immediately nor 3 months after the invention. Although multimodal therapies are effective in the treatment of symptoms and/or side effects with various diseases, 18 at least for this condition of fatigue there was no substantial add-on effect.
Weighting Strengths and Limitations
With an average of three years between study entry and last active tumor therapy prolonged effects of the last treatment or symptom course are less likely in our sample. The required sample was achieved with an ordinary recruitment rate. Especially in RCTs, the recruitment of cancer patients with both time limitations and physical restrictions is elaborate and unpromising (rates of around 40%).17,35 Although we had based our sample size calculation on a similar pilot trial, the actual sample size in the study with only 55 patients might have been too small to detect any group differences. Also the selection of an adequate control condition is fraught with problems. Patient blinding is impossible and expectancy effects and dropouts due to disappointment of randomization results cannot be excluded. Furthermore, patients had considerable experience with rehabilitation and psychological/educational programs, and the impact of the add-on effect of counseling in the day clinic group may be underestimated. The patients of the MMMT group were asked to practice the learned methods at home as often as possible. However, we only monitored the adherence rate of walking exercise in both groups and did not assess the compliance regarding the other mind–body techniques. It is possible that the patients of the MMMT were unable to cope with the load of both the physical exercise and the other methods. This would, however, also be a conclusion about the feasibility of this program in this condition of fatigue. Future larger studies, including a nontreated control group and measures of actual physical activity behavior will provide a deeper insight in the underlying mechanisms.
After all, there are still only a limited number of RCTs available that include chronic CRF as primary outcome. Fatigue is a major contributor for impaired functions and aggrieved QoL in breast cancer survivors.5,36-38 It is therefore required to further investigate fatigue reducing interventions such as walking or more complex programs. This may be of interest also for health care policy. The immediate rehabilitation period may be an interesting topic in this context. 39 As our study shows, even a basic home-based pulse-controlled WT can relevantly affect fatigue symptoms. Also the questionnaires and subscales assessing QoL and functional aspects underline the benefit of both interventions with an increase of 25% to 30%. Other outcomes including qualitative data may shine a light on possible yet unidentified add-on benefits of MMMT interventions for this condition.
Conclusion/Clinical Practice Points
Both interventions reduced chronic CRF and associated symptoms in breast cancer survivors. There was no considerable add-on effect of the MMMT assessed, and thus home-based WT can be preliminary recommended at first instance in the management of chronic CRF instead. Future research should evaluate other possible benefits of MMMT for cancer patients.
Footnotes
Acknowledgements
We would like to thank all patients, instructors, and therapists for their participation in the study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant of the “Karl and Veronica Carstens Foundation” (Essen, Germany), which is a nonprofit organization for the investigation of naturopathic treatments.
