Abstract
Colorectal cancer (CRC) is a malignant tumor with high global incidence and mortality rates, often accompanied by an increase in M2 tumor-associated macrophages (TAMs), which may lead to poor prognosis in CRC patients. Zuojin Wan (ZJW), a traditional Chinese medicinal formula, has been demonstrated to have therapeutic effects on CRC. It can inhibit the proliferation of CRC cells and improve the prognosis of CRC by enhancing immunity; however, the precise immune mechanisms remain unclear. A CT26 cell-induced CRC tumor-bearing mouse model in this study was established to investigate the therapeutic effects of ZJW on CRC. In vivo experiments confirmed that ZJW significantly reduced the size of CRC tumors and decreased the levels of M2 TAMs markers, including CD206 and Arg-1, while increasing the levels of iNOS. Furthermore, in a co-culture system with macrophages, ZJW reduced the proportion of M2 macrophages and increased the proportion of M1 macrophages. Simultaneously, ZJW intervention attenuated the proliferation and migration of CT26 and MC38 cells. ZJW may suppress the migration and proliferation of CT26 and MC38 cells by inhibiting M2 TAMs. The results of network pharmacology combined with Western blot indicated that ZJW may suppress M2 TAMs by inhibiting JAK2/STAT3 signaling pathway in TAMs. This study confirmed that Zuojin Wan may suppress the malignant progression of colorectal cancer by inhibiting M2 TAMs, potentially mediated by inhibition of JAK2/STAT3 signaling pathway in TAMs.
Keywords
Introduction
Colorectal cancer (CRC) has been reported as the second largest cause of cancer-related death. In developing countries such as China, the incidence of colorectal cancer is rising rapidly due to lifestyle changes.1,2 At present, the clinical treatment of colorectal cancer mainly adopts traditional western therapy such as surgery, chemotherapy, and molecular targeted drugs; however, patients’ prognosis remains poor. Traditional Chinese medicine plays an important role in the comprehensive treatment of colorectal cancer, which may reduce the toxic side effects of chemotherapy, targeted therapy and immunotherapy, improve immunity, prevent tumor recurrence and metastasis, and improve prognosis. 3 Originating from Danxi Xinfa, Zuojin Wan (ZJW) has been proven to have therapeutic effects on colorectal cancer, inhibiting the proliferation of colorectal cancer cells and improving the prognosis of colorectal cancer by enhancing immunity. 4 However, the immune mechanism of its therapeutic effect is still unclear.
As the most abundant immune cell in tumor immune microenvironment, tumor-associated macrophages (TAMs) are known as the “Big Brother” of tumor immune escape. 5 TAMs are affected by tumor microenvironment (TME). M1 TAMs were induced to be classically activated and M2 TAMs to be alternatively activated. The M1 TAMs usually play an anti-tumor function, while the latter assists the immune escape of tumor cells and promotes the malignant metastasis of tumors. M1 TAMs highly express the co-stimulatory molecule CD80, secrete pro-inflammatory cytokines such as interleukin-12 (IL-12), and produce high levels of inducible nitric oxide synthase (iNOS), which are generally considered as tumor killer cells.6,7 M2 TAMs highly express the mannose receptor CD206 and Arginase-1 (Arg1), and secrete interleukin-10 (IL-10), interleukin-6 (IL-6), and other anti-inflammatory cytokines, which is related to tissue repair. The increased proportion of M2 TAMs may induce the formation of immunosuppressive microenvironment, promote tissue repair and tumor immune escape.8,9 The malignant progression of CRC is often accompanied by increased infiltration of M2 TAMs, thus inducing the formation of an immunosuppressive TME, which leads to poor prognosis in CRC.10,11 Both M1 and M2 TAMs are highly plastic and therefore can transform each other in response to changes in the TME or therapeutic intervention.12 -14 Clinical studies have shown that remodeling the M1 transformation of TAMs can effectively inhibit tumor progression and improve the prognosis of cancer patients, and combining TAMs targeted drugs with current chemotherapy and radiation therapy will improve the specificity of CRC treatment.15,16 Therefore, regulation of TAMs phenotypic transformation is a feasible strategy for the treatment of CRC.
Previous studies have shown that inhibiting M2 TAMs could suppress the malignant progression of CRC. 17 ZJW has been shown to suppress the proliferation of colorectal cancer cells, with its mechanism of action involving the modulation of immune function. 18 Therefore, we hypothesize that ZJW may effectively inhibit the malignant progression of CRC by inhibiting M2 TAMs. The study aims to uncover the pathways involved in the anti-colorectal cancer mechanism of ZJW through network pharmacology.
Methods and Materials
Cell Culture and Reagents
RAW264.7 macrophages (RRID: CVCL_0493, Cat#STCC20020P), CT-26 cells (RRID: CVCL_7254, Cat#STCC20017), and MC38 cells (RRID: CVCL_B288, Cat#PSTCC20018P) were bought from Servicebio (Wuhan, CN).The cells were cultured in RPMI 1640 or DMEM containing 10% of fetal bovine serum (Gibco, NY, USA,Cat#A5256701), 2 mM of glutamine, 100 units/mL of streptomycin and penicillin at 37°C in a humidified 5% of CO2 atmosphere.
Preparation of Zuojin Wan
Zuojin Wan decoction is composed of Coptis chinensis and Evodia rutaecarpa at 6:1 ratio. Coptis chinensis 18 g and Evodia rutaecarpa 3 g were obtained from Nanjing Tongrentang Pharmaceutical Co., Ltd. The medicinal materials were soaked in distilled water for 30 minutes, decocted until boiling, then simmered for 30 to 40 minutes and filtered. Water was added to the residue, the decoction process was repeated, and the 2 filtrates were combined. The mixture was centrifuged at 12 000 rpm for 10 minutes, concentrated to 0.28 g/mL using a rotary evaporator, aliquoted, and stored at −20°C for future use.
Components Analysis from Aqueous Extracts of ZJW
The extracts were mixed with acetonitrile and water (ratio: 1:1 v/v) and filtered through 0.45-µm microporous membrane prior to high performance liquid chromatography (HPLC) analysis. The Welch Ultimate C18 column (5 µm, 4.6 mm × 250 mm, Welch Materials, Inc in Shanghai) was applied for chromatographic separations. The mobile phase was composed of A (0.4% phosphoric acid water) and B (acetonitrile) with a linear gradient elution: 0 to 5 minutes, 4% to 12.4% B; 5 to 20 minutes, 12.4% to 13.4% B; 20 to 25 minutes, 13.4% to 14.8% B; 25 to 28 minutes, 14.8% to 17% B; 28 to 43 minutes, 17 to 17.3% B; 43 to 50 minutes, 17.3% to 20% B; 50 to 52 minutes, 20% to 21.5% B; 52 to 55 minutes, 21.5% to 21.6% B; 55 to 70 minutes, 21.6% to 22.1% B; 70 to 71 minutes, 22.1% to 22.5% B; 71 to 76 minutes, 22.5% to 23.5% B; 76 to 86 minutes, 23.5% to 25% B; 86 to 90 minutes, 25% to 29% B; 90 to 95 minutes, 29% B. The eluate was monitored by a diode array detector at the wave length of 335 nm. The injection volume was 5 μL at a flow rate of 0.8 mL/min. The column oven temperature was maintained at 25°C. Peaks were assigned by comparing their retention time with that of each reference compound eluted in this mobile phase and by spiking samples with references dehydroevarine (D22GB172079, purity ≥ 98.0%), pharmacorhizine (Z05D10X104878, purity ≥ 98.0%), palmatine hydrochloride (Z16J10X79792, purity ≥ 98.0%) and berberine hydrochloride (S01A10K94340, purity ≥ 98.0%), which were purchased from Shanghai Yuanye Bio-Technology Co., Ltd. In addition, fingerprints of ZJW from 10 batches were identified.
Animals, Induction of CRC and Treatments
BALB/c mice (male, 20-22 g, 6-8 weeks old) were purchased from Hunan Slack Jingda Experimental Animal Co., Ltd. (Hunan, China). After 1 week of acclimatization under specific pathogen-free conditions (55% ± 2% humidity, 25°C, 12-h light/dark cycle), CT26 cells in logarithmic growth phase were adjusted to 1 × 10⁷ cells/mL. A 200 μL suspension was injected subcutaneously into the right axilla of each mouse. And mice with tumor sizes of 100 to 150 mm3 were selected for follow-up experiments. To investigate the effect of ZJW, all tumor-bearing mice were randomly divided into six groups: control (sterile water), model (sterile water), ZJW (8.75, 4.37, and 2.19 mg/kg), 5-fluorouracil (23 mg/kg) groups. ZJW were orally administered once a day from day 7 to day 21, and 5-fluorouracil was administered once every 3 days (Figure 1). All animal experiments complied with the experimental animal ethics committee of GuangXi University of Chinese Medicine.

Diagram of Animal modeling. CT26 cells was injected subcutaneously into the underarm of the right forelimb of mice. On day 7, mice with tumor sizes of 100 to 150 mm3 were selected for follow-up experiments. All tumor-bearing mice were randomly divided into six groups: Control, Model, ZJW-L (2.19 mg/kg), ZJW-M (4.37 mg/kg), ZJW-H (8.75 mg/kg), and 5-FU (23 mg/kg) groups. From day 7 to day 21, ZJW were orally administered once a day, and 5-fluorouracil was injected administered once every 3 days.
Tumor Size and Spleen Index
On day 21, the mice were sacrificed by cervical dislocation, and tumors were gathered and photographed. Then, the volume and weight of tumor was measured and recorded. At the end of this study, the tumor volume of the mice adhered to the requirements of animal ethics. 19
Analysis of Cytokines in Serum
After sacrificed, blood was collected from mice and left for 2 hours, centrifuged at 3000 rpm for 10 minutes at 4°C, and serum was collected. Serum immune cytokines TGF-β (Enzyme Linked, Cat#YJ057830), IL-1β (Enzyme Linked, Cat#YJ098416), IL-6 (Enzyme Linked, Cat#YJ063159), IL-10 (Enzyme Linked, Cat#YJ037873), TNF-α (Enzyme Linked, Cat#YJ002095), and IL-12 (Enzyme Linked, Cat#037868) were detected by ELISA kits according to manufacturer’s instructions. Results were expressed as pg/mL of serum in each sample.
Immunohistochemistry (IHC) of Tumor Tissues
Paraffin-embedded sections (4 µm) of tumor were analyzed for Ki67 (1:400, Proteintech, Cat#27309-1-AP, RRID: AB_2756525), iNOS (1:400, Proteintech, Cat#22226-1-AP, RRID: AB_2879038), and Arg-1 (1:400, Proteintech, Cat#16001-1-AP, RRID: AB_2289842). After extracting the antigen, the expression was detected by primary antibody at 4°C overnight. Biotin-labeled secondary antibody and streptavidin HRP were incubated at room temperature for 30 minutes. The immune reaction was detected with 3-3-diaminobenzidine and then re-stained with hematoxylin.
Immunofluorescence (IF) of Tumor Tissues
TSA Fluorescence System Kit (Servicebio) was used to detect CD3 and CD8.Briefly, rabbit anti-CD3 monoclonal antibody (1:200; Servicebio, Cat#GB13014, RRID: AB_2920578), rabbit anti-CD8 monoclonal antibody (1:250; Servicebio, Cat#GB15068, RRID: AB_3246431), and goat anti-rabbit HRP (1:200, Bioss) were used as secondary antibodies. The tissue sections were then incubated with an Alexa Fluor-488 secondary antibody (1:1000, Servicebio, Cat#GB25303, RRID: AB_2910224) for 2 hours. Following washed with 5% PBST, and the cell nuclei were counterstained with DAPI. The sections were finally sealed with anti-fluorescence quenching mounting agent, all tissue sections were examined using fluorescence microscopy (NIKON).
To detect the co-localization of F4/80 and CD206 in tumor tissues, after antigen repair, tissue sections were incubated with 3% H2O2 for 10 minutes, then washed with PBS, and incubated with the prepared F4/80 antibody (1:500, Servicebio, Cat#GB113373, RRID: AB_2938980) at 4°C overnight. The next day, incubated with HRP-labeled secondary antibody 50 minutes, and incubated for 10 minutes. After antigen repair, incubated with CD206 antibody (1:4000, Servicebio, Cat#GB113497, RRID: AB_3665021) overnight at 4°C. Then incubated with corresponding HRP-labeled secondary antibody for 50 minutes, and with incubated DAPI in the dark for 10 minutes, then incubated with autofluorescence quencher solution B for 5 minutes. The sections were finally sealed with anti-fluorescence quenching mounting agent, all samples were examined using fluorescence microscopy (NIKON) and quantitatively analyzed for the positive proportion.
Cell Viability Assays
To analyze the given experimental setup for determining cell proliferation using the CCK-8 assay, seeding of cells in 96-well plates was performed. Cells were seeded at a density of 1 × 104 cells per well. The CCK-8 assay was performed 48 hours after ZJW treatment. Then treated cells were incubated for 4 hours with culture medium containing the CCK-8 reagent. After incubation, the absorbance of the solution was recorded at 450 nm using a microplate enzyme-linked immunosorbent assay (ELISA) reader (Thermo, USA). The relative survival rate of cell growth was calculated based on the absorbance values.
Transwell Assay and Trypan Blue Assay Detected the Migration and Proliferation of CT26 Cells and MC38 Cells in the Co-culture System
In the transwell assay, the control group (conventional cultured RAW264.7), M2 TAMs group (20 ng/mL IL4/IL13 induced cultured RAW264.7 for 48 hours), M2 TAMs + ZJW group (RAW264.7 co-cultured with 20 ng/mL IL4/IL13 + 32 μg/mL ZJW for 48 hours) were inoculated into the lower chamber of transwell plate with 1 × 105 cells. After the macrophages were fully attached to the wall (6-8 hours), tumor cells were re-suspended using serum-free medium. And 1 × 105 tumor cells were inoculated in the upper chamber of transwell and cultured at 37°C and 5%CO2 for 24 hours. Subsequently, the transwell chamber was removed, the cells in the chamber were gently wiped away, the filter membrane was fixed with methanol, and after staining with crystal violet, 5 fields of view were randomly selected under the microscope, and then photographs were taken, and the mobility was calculated using Image J software.
In the trypan blue viability assay, 1 × 105 tumor cells were seeded in the lower chamber of a Transwell plate. After cell adhesion (6-8 hours), 1 × 105 macrophages from the following groups were added to the upper chamber: control group (RAW264.7 cultured by routine culture), M2 TAMs group (20 ng/mL IL4/IL13 induced RAW264.7 cultured for 48 hours), M2 TAMs + ZJW group (RAW264.7 co-cultured with 20 ng/mL IL4/IL13 + 32 μg/mL ZJW for 48 hours) .Cells were incubated at 37°C with 5% CO₂ for 24 hours. Tumor cell viability in the lower chamber was then assessed using trypan blue exclusion.
Flow Cytometry
The cells in each well were digested with 0.5 mL of 0.25%trypsin-EDTA for 3 minutes at 37°C. The suspension was transferred to centrifuge tubes, centrifuged at 300g for 5 minutes, and washed twice with 4 mL PBS. After discarding the supernatant, the pellet was resuspended in 200 μL PBS containing 2%FBS. Pre-labeled flow cytometry tubes received 100 μL cell suspension per tube. Antibodies were added according to manufacturer specifications: F4/80 (Invitrogen, Cat#11-4801-81, RRID: AB_2735037), CD11b (Invitrogen, Cat#45-0112-80, RRID: AB_953560), CD86 (Invitrogen, Cat#12-0862-81, RRID: AB_465767), and CD206 (Invitrogen, Cat#17-2061-80, RRID: AB_2637419). Negative controls (unstained) and single-stained compensation controls were included. Tubes were vortexed gently, then incubated in the dark for 20 minutes at 4°C. Cells were washed twice with 2% FBS-PBS and analyzed using a Beckman Coulter CytoFLEX flow cytometer. Data were processed with CytExpert software (v2.4).
Isolation of TAMs
Tumor tissue was cut into small fragments, and the tissue fragments were digested using collagenase VIII (Sigma-Aldrich, Cat#9001-12-1) for 1 hours at 37°C. After digestion, the suspension was filtered through a 70 μm mesh to remove larger tissue pieces and clusters and resulted in a single-cell suspension. The cell suspension was centrifuged and washed twice with PBS to remove residual enzyme and other debris. The cells were resuspended in serum-free RPMI 1640 medium and left to adhere for 40 minutes. After the adhesion period, non-adherent cells were washed away, leaving behind primarily macrophages. The remaining adherent cells, which were flat or irregularly shaped and attached to the surface of the petri dish, were characterized as TAMs based on their morphology and positivity for the macrophage-specific marker F4/80. The protocol was designed to efficiently isolate TAMs from tumor tissues, leveraging the inherent properties of macrophages such as adhesion and specific marker expression. The isolated TAMs could then be used for further experiments.
Western Blot Analysis
The cells were collected and lysed using cold RIPA buffer (Beyotime Biotechnology, Cat#P0013C). After the cells were lysed, the protein concentration in the resulting lysate was measured using a BCA protein assay kit (Beyotime Biotechnology, Cat#P0010). A consistent amount of protein, 20 μg, from each sample was loaded onto a Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE) gel. The separated proteins were then transferred onto a 0.22 μm polyvinylidene difluoride (PVDF) membrane. The PVDF membranes were incubated with primary antibodies iNOS (1:1000, Proteintech, Cat#22226-1-AP, RRID: AB_2879038), Arg1 (1:1000, Proteintech, Cat#16001-1-AP, RRID: AB_2289842), JAK2 (1:1000, ABclonal, Cat#A11497, RRID: AB_2758584), p-STAT3 (1:500, Thermo Fisher, Cat#710093, RRID: AB_2532557), STAT3 (1:1000, Proteintech, Cat#60199-1-Ig, RRID: AB_10913811), and GAPDH (1:5000, Proteintech, Cat#60004-1-Ig, RRID: AB_2107436) respectively at 4°C overnight, then the membranes were incubated with secondary antibodies conjugated to horseradish peroxidase (HRP) at room temperature for 2 hours. Finally, the membrane was visualized with chemiluminescence (ECL; Bio-Rad, USA). And western band gray values were analyzed and GAPDH was a reference by using Image J (GE, USA).
Quantitative Real-Time PCR
Total RNA was extracted by using TRIzol (Thermo Fisher, Cat#15596026CN), the concentration and purity of RNA were calculated, and reverse transcription was performed according to the TaKaRa® RT SuperMix for qPCR (+gDNA wiper) kit (Takara, Cat#RR047A), which was kept at 37°C for 15 minutes and then at 85°C for 5 seconds for PCR reaction. The reaction system was as follows: 2 × SYBR Green PCR Master Mix 10 μL (Takara, Cat#RR820A), 1 μL cDNA, upstream primer 0.4 μL, downstream primer 0.4 μL, RNase Free ddH2O 8.2 μL. The reaction system was as follows: 2 × SYBR Green PCR Master Mix 10 μL, 1 μL cDNA, upstream primer 0.4 μL, downstream primer 0.4 μL, RNase Free ddH2O 8.2 μL. The reaction steps are as follows: predenaturation at 95°C for 30 s, denaturation at 95°C for 5 s, annealing at 60°C for 30 s, 40 cycles. The mRNA levels of iNOS, Arg-1 and GAPDH was calculated by 2−△△Ct. The primer sequences are as follows: iNOS: Forward(TCTAGTGAAGCAAAGCCCAACA), Reverse(TGATGGACCCCAAGCAAGAC); Arg-1:Forward(GTGGAGACCACAGTCTGGCA), Reverse(TTCCCCAGGGTCTACGTCTC); GAPDH: Forward (TGTGTCCGTCGTGGATCTGA), Reverse (CCTGCTTCACCACCTTCTTGA).
Network Pharmacology
The targets of the herbs (Coptis chinensis and Evodia rutaecarpa) in ZJW were obtained from the Traditional Chinese Systems Pharmacology Database and Analysis Platform (TCMSP, https://old.tcmsp-e.com/), and Encyclopedia of Traditional Chinese Medicine 2.0 (ETCM 2.0, http://www.tcmip.cn/ETCM2). The potential targets related to CRC and TAMs were searched from the DISGENET (http://www.disgenet.org/), GeneCards (http://www.genecards.org), and OMIM (https://omim.org/) databases. The network of PPI was established on the STRING (https://www.string-db.org/) and was visually analyzed by Cytoscape software (Version 3.10.2). GO functional enrichment analysis and KEGG pathway enrichment analysis were conducted for the common targets of ZJW, CRC and TAMs using the Database for Annotation, Visualization, and Integrated Discovery (DAVID, https://david.ncifcrf.gov/).
Statistical Analysis
All the statistics shown were presented as mean ± SD with GraphPad Prism 9.0 (GraphPad Software, CA, USA). All data were first tested for normality using the Shapiro-Wilk test, and homogeneity of variances were verified prior to parametric testing. For multiple-group comparisons, one-way ANOVA was conducted. And comparisons between 2 groups utilized unpaired Student’s t-tests. Data were statistically analyzed with SPSS 26.0 (SPSS Inc., Chicago, USA). Statistical significance was established by a P-value <0.05.
Results
Fingerprints of ZJW
To verify the stability of ZJW decoction and ensure its quality, we chose 4 chemical components as standards of ZJW, namely dehydroevarine, berberine hydrochloride, palmatine hydrochloride, and berberine hydrochloride (Figure 2B-E). A rigorous analysis of these components was conducted to reveal the fingerprints of 10 batches of ZJW. As shown in Figure 2A, the similarity between the 10 batches mostly exceeds 0.9. The results clearly reflected the stable quality of ZJW.

The fingerprint of Zuojin Wan decotion (ZJW). (A) The fingerprint of ZJW; (B-E) Extracting ion chromatogram of 4 target standard compounds: (B) Dehydroevodiine; (C) Rhizine hydrochloride; (D) Palmatine hydrochloride; (E) Berberine hydrochloride.
ZJW Inhibited Tumor Growth in CRC Mice
To investigate the therapeutic effects of ZJW on CRC mice, after modeling, mice with tumor sizes ranging from 100 to 150 mm³ were selected for subsequent experiments and were gavaged with ZJW once a day. Figure 3A to C showed that the administration of ZJW significantly inhibited tumor growth compared with model group. After modeling, weight loss was observed (Figure 3F) in mice of model group, and the mice treated with ZJW regained some weight, and no significant differences in body weight were detected between groups. In addition, the results of spleen index (Figure 3D and E) showed spleen enlargement in model group, while ZJW down-regulated spleen index, demonstrating that ZJW may improve immune function in tumor-bearing mice. The immunohistochemical staining (IHC) was used to determine the protein level of the Ki67 in tumors. Compared with the model group, the protein level of Ki67 in tumor of ZJW group was reduced (Figure 3G and H).
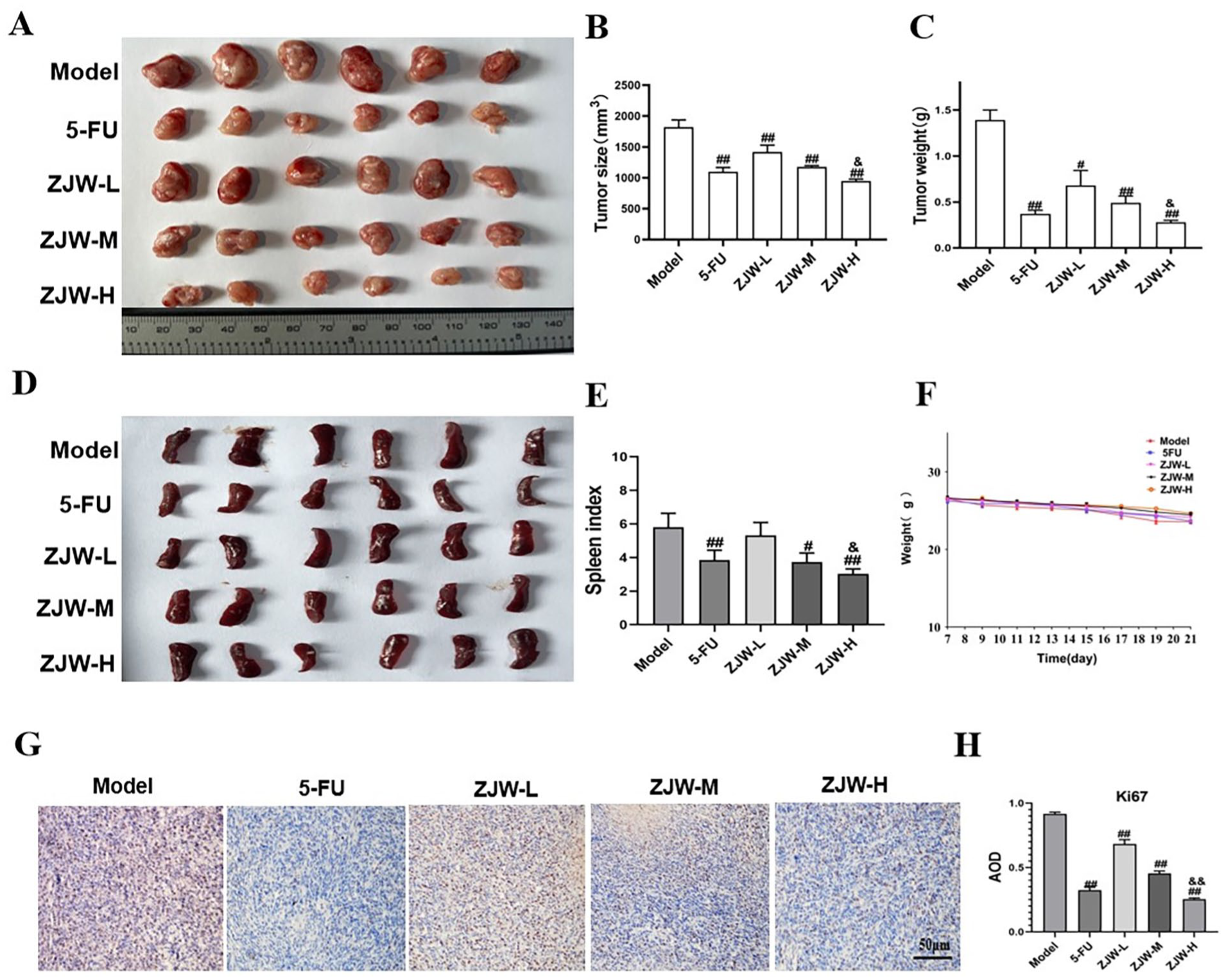
Effect of ZJW on the CRC progression in mice. (A-C) Effects of ZJW on the tumor size and tumor weight in mice. (D and E) Spleen image and spleen index in mice. (F) Weight of mice. (G and H) Quantification of the protein levels of Ki67 by IHC counting the average optical density (IHC, 400×). The bars represent the mean ± SD.
ZJW Enhanced the Anti-Tumor Immune Effect of CRC Mice
The immune cytokines in the serum of mice were determined by using ELISA. The data showed that the levels of immune cytokines IL-6, IL-10, IL-1β, TNF-α, and TGF-β that promote the immunosuppressive microenvironment, were abnormally elevated in the model group compared to the control group, which indicated the intervention of ZJW may significantly down-regulated IL-6, IL-10, IL-1β, TNF-α, and TGF-β. However, IL-12, which have anti-tumor effects, were abnormally reduced in CRC tumor-bearing mice. It was found that the intervention of ZJW may up-regulate IL-12 and promote the anti-tumor immune effect (Figure 4A). These results reveal the role of ZJW in enhancing immunity in CRC tumor-bearing mice. Additionally, the results (Figure 4B and C) demonstrated that ZJW promoted the increase of CD8 and CD3 cells in tumors of CRC tumor-bearing mice, which were related to the enhancement of anti-tumor immune effect, 20 indicating that ZJW could regulate the increase of CD8 and CD3 cells and promote the anti-tumor immune effect and ZJW has an effective protective effect against CT26-induced CRC mice.

Effect of ZJW on the levels of immune cytokines, CD3 and CD8 in mice. (A) Levels of IL-12, TNF-α, IL-6, IL-1β, TGF-β, and IL-10 in mice (n = 6). (B) Images of immunofluorescence of Arg1 and iNOS in tumor tissues (IF, 400×). (C) Quantification of the positive rate of CD3 and CD8 in tumor tissues detected using IF. The bars represent the mean ± SD.
ZJW Inhibits the M2 TAMs In Vivo
To reveal the effect of ZJW on the polarization of TAMs in CRC tumor tissues, IHC and IF were used to determine the protein levels of iNOS, Arg1, and CD206 in tumor tissues. Compared with the model group, the protein level of iNOS was increased, while the levels of Arg1 (Figure 5A-D) and CD206 (Figure 6A and B) in the tumor tissues of ZJW group were reduced. In addition, the mRNA levels of M1 (iNOS) and M2 (Arg1) genes in TAMs of CRC tumor-bearing mice were determined by using RT-qPCR. As shown in Figure 5E, Arg1 was significantly reduced in TAMs in ZJW treated groups, while iNOS were significantly increased. The results indicate that the anti-cancer effect of ZJW seems to be intricately related to the balance of macrophage polarization in CRC tumor-bearing mice.

Effect of ZJW on the levels of Arg1 and iNOS in tumor tissues of mice. (A) Immunohistochemistry images of Arg1 and iNOS in tumor tissues (IHC, 400×). (B) Quantification of the protein levels of Arg1 and iNOS by IHC counting the average optical density. (C) Immunofluorescence images of Arg1 and iNOS in tumor tissues (IF, 400×). (D) Quantification of the positive rate of Arg1 and iNOS in tumor tissues detected using IF. (E) The mRNA levels of Agr1 and iNOS in TAMs of mice. The bars represent the mean ± SD.

ZJW inhibits M2-type TAMs in tumor tissues of mice. (A) Immunofluorescence staining images of CD206 and F480 in tumor tissues from different groups. The images show staining for CD206 (green), DAPI (blue), F480 (red), and a merged view. (B) Immunofluorescence staining analysis of CD206 and F4/80 expression in different groups. The bars represent the mean ± SD (n = 5).
Effects of ZJW on RAW264.7 Macrophages In Vitro
M2 TAMs are typically characterized by promoting tumor growth and associated with high levels of markers such as CD206 and Arg-1. In contrast, M1 TAMs are typically identified by higher levels of CD86 and iNOS. To investigate the cytotoxicity of ZJW on RAW264.7 cells in vitro, dose screening was performed. Based on Figure 7A, doses of 16, 32, and 64 μg/mL were chosen for further study. The Western blot results (Figure 7B and C) demonstrated that ZJW significantly reduced the protein level of Arg1 and increased the protein level of iNOS in a dose-dependent manner, further confirming the role of ZJW in inhibiting M2 macrophages in vitro. The flow cytometry results (Figure 7D and E) showed that ZJW significantly increased the proportion of CD86 (M1 macrophages) in macrophages, while decreasing the proportion of CD206 (M2 macrophages) significantly, and showed a dose-dependent trend. The results of RT-qPCR (Figure 7F) were consistent with the western blot results. The above results indicate that ZJW can inhibit the polarization of M2-type macrophages in vitro.
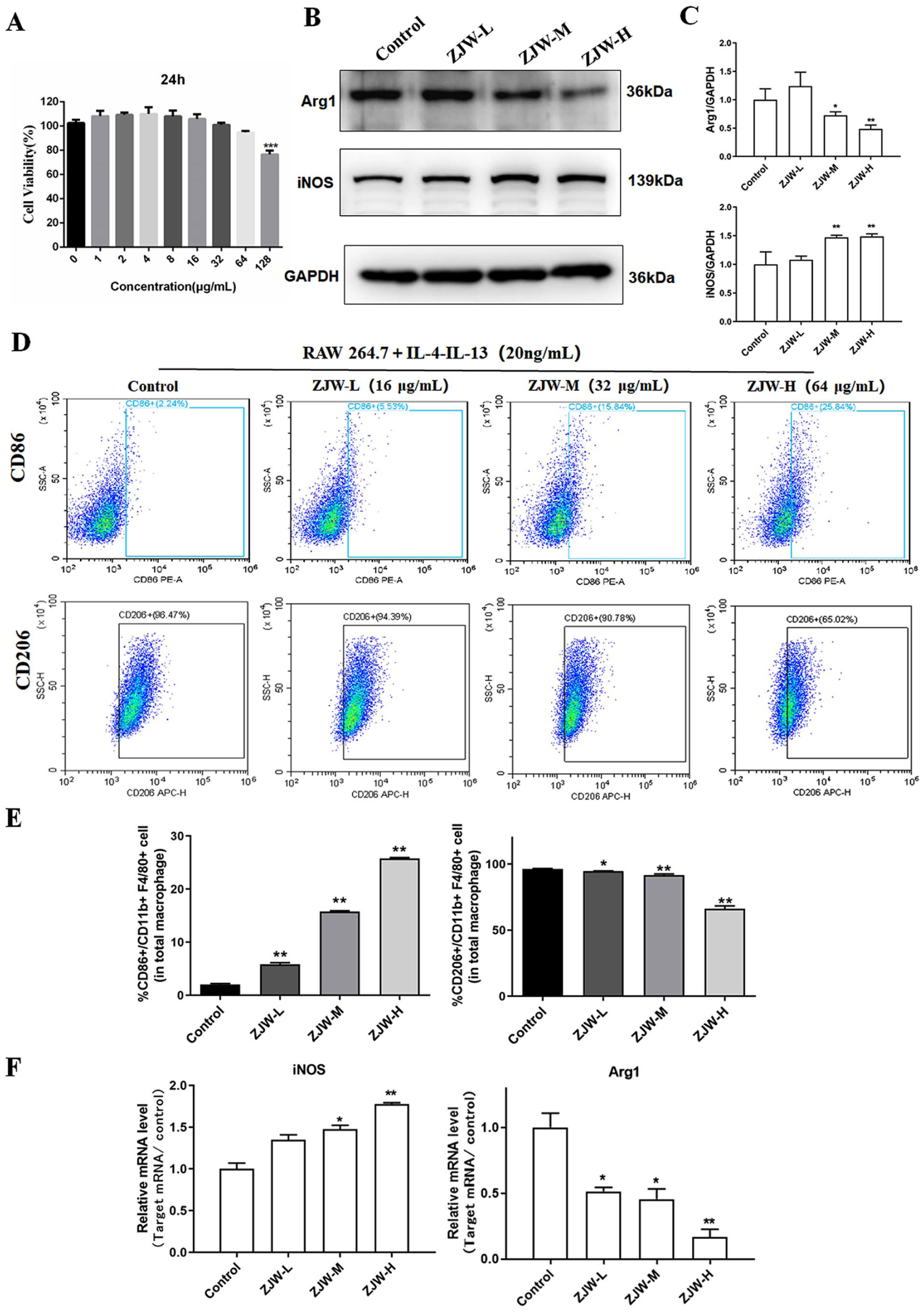
Effect of ZJW on RAW264.7 in vitro. (A) The cytotoxicity of ZJW on RAW264.7 cells. (B and C) Quantification of the protein levels of iNOS and Arg1 by using Western blot. (D and E) The proportion of CD86 and CD206 in CD11b + F4/80 cells were analyzed by using flow cytometry. (F) The mRNA levels of iNOS and Agr1 in macrophages after the intervention of ZJW by using RT-qPCR. The bars represent the mean ± SD.
Effects of ZJW on CRC Cells Through Macrophages In Vitro
To investigate whether ZJW has an effect on the migration and proliferation of colon cancer cells through macrophages, as described in Method 2.10 (Figure 8A), CT26 and MC38 cells were co-cultured with RAW264.7 macrophages in the transwell system, respectively. The trypan blue experiment results (Figure 8B) showed that IL-4-IL13 induced M2 macrophages were co-cultured with cancer cells, and the proliferation of cancer cells significantly increased, while macrophages treated with ZJW could significantly inhibit the growth of CT26 and MC38 cells. In addition, the results (Figure 8C and D) showed that after co-culture of IL-4-IL13 induced M2 macrophages and cancer cells, the migration rate of cancer cells was significantly increased, and macrophages after ZJW intervention could inhibit the migration of CT26 and MC38 cells. These results suggest that ZJW may inhibit the migration and proliferation of colorectal cancer cells through macrophages in vitro.

Effect of ZJW on CRC cells through TAMs in vitro. (A) Schematic diagram of CRC cells co-cultured with conditioned macrophages, trypan blue experiment (left) and Transwell experiment (right). (B) Trypan blue viability assay of CT26 and MC38 cells co-cultured with conditioned macrophages. (C and D) Transwell migration assay of CT26 and MC38 cells co-cultured with conditioned macrophages. The bars represent the mean ± SD (n = 3).
Based on Network Pharmacology Explore the Mechanism of ZJW on CRC
As shown in Figure 9A, 361 ZJW targets, 13209 CRC- and 3338 TAMs-related disease targets were identified, and a total of 103 overlapping targets were obtained, which were considered the potential ZJW targets treating CRC through TAMs. The 103 core targets were then analyzed using the Cytoscape plugin cytoHubba to find the top 10 key proteins, from 1 to 10, in the order of JAK2, STAT3, TNF, MTOR, HSP90AA1, PTGS2, PPARG, MMP9, KDR, and STAT1 (Figure 9B). The results of the GO enrichment analysis showed that ZJW exerted its function by regulating positive regulation of kinase activity, regulation of epithelial cell proliferation, intrinsic apoptotic signaling pathway, and protein serine/threonine/tyrosine kinase activity (Figure 9C). In addition, we performed KEGG pathway enrichment analyses and found that JAK2/STAT3 signaling pathway is the most target-enriched signaling pathway (Figure 9D). Combined with literature study, we hypothesize that ZJW may regulate the M2 polarization of macrophages by affecting JAK2/STAT3 signaling pathway.

Pharmacological analysis of the ZJW-CRC-TAMs network and effects of ZJW on JAK2/STAT3 pathway of TAMs in vitro. (A) Venn diagram for ZJW, CRC and TAMs targets screening. (B) The top 10 targets that Cytoscape plugin CytoHubba has screened. (C) GO enrichment analysis. (D) KEGG pathway enrichment analysis. (E and F) The protein levels of JAK2 and p-STAT3/STAT3 were determined by using Western blot.
Effects of ZJW on JAK2/STAT3 Signaling Pathway in Macrophages In Vitro
In order to verify the results of network pharmacology-related signaling pathway analysis, Western blot was used to determine the protein levels of JAK2, p-STAT3, and STAT3 in macrophages after IL-4-IL-13 induced macrophages were co-incubated with ZJW for 48 h. The results (Figure 9E and F) indicated that protein levels of JAK2 and p-STAT3 decreased significantly after the intervention of ZJW. Combined with the results of network pharmacology, it is speculated that ZJW may inhibit the transformation of TAMs into M2 by inhibiting the JAK2/STAT3 signaling pathway and alleviate the immunosuppressive microenvironment.
Discussion
Colorectal cancer (CRC), an aggressive malignancy of the gastrointestinal tract, ranks as one of the most prevalent and lethal forms of cancer globally. The development and progression of CRC are intricate processes that involve not only genetically aberrant cancerous cells but also numerous elements within the encompassing tumorigenic cellular ecosystem. 20 Due to the complex pathogenesis and poor prognosis of colorectal cancer, it is urgent to find the drugs and methods to treat CRC as soon as possible. Traditional Chinese medicine plays a significant role in the comprehensive treatment of colorectal cancer. 21 It was confirmed that Zuojin Wan (ZJW) has therapeutic effects on CRC, based on existing research. We speculate whether it improve the prognosis of colorectal cancer by enhancing immunity. 22 However, the immune mechanism underlying its therapeutic effect remains unclear. In this study, our findings demonstrated that in comparison with the model group, ZJW significantly suppressed the advancement of implanted CRC tumors and rectified the condition of splenic hypertrophy in CRC mice, meanwhile increasing the infiltration of CD8+ and CD3+ T cells in tumor tissue and improving the tumor immunosuppressive microenvironment This indicated a potential immune enhancement effect by ZJW on CRC tumor -bearing mice.
Over the past 2 decades, mounting empirical evidence has demonstrated that the tumor microenvironment (TME) assumes significance equal to genetic and epigenetic mutations in tumor genesis, progression, and metastasis.23 -26 Paget 27 proposed the hypothesis of tumor “seeds and soil,” that is, tumor cells act as “seeds,” and their occurrence and metastasis depend on the tumor microenvironment as “soil.” Through scrutinizing the “soil” shaped by cancer cells and host immune cells, researchers discovered that immune cells typically adhere to cancer cells and acquire distinct biological traits through reciprocal interactions. 28 Tumor-associated macrophages (TAMs), integral components of the TME’s immune landscape, critically regulate tumor progression through cytokine/chemokine secretion. Their inflammatory mechanisms facilitate invasion, metastasis, immunosuppression, angiogenesis, and drug resistance.29 -31 TAM subpopulations exhibit functional plasticity, dynamically adapting to stimuli from malignant cells or microenvironmental factors. While TAMs generally portend poor prognosis in solid tumors, their role in CRC is notably complex: reprograming TAMs polarization may enable novel immunotherapies. 32 Macrophages polarize into 2 functionally distinct phenotypes: classically activated (M1) and alternatively activated (M2). 33 M1 macrophages promote Th1 responses while phagocytosing and eliminating tumor cells. 34 Within the TME, M1 TAMs secrete IL-12 and other pro-inflammatory cytokines that activate inflammatory cascades and exert antitumor immunity. 35 Conversely, upon IL-4/IL-13 stimulation, M2 macrophages release anti-inflammatory cytokines including IL-10, thereby promoting angiogenesis, tissue remodeling, wound healing, and tumor progression.36,37
Animal experiments demonstrated that Zuojin Wan (ZJW) reduced serum levels of TNF-α, IL-1β, TGF-β, IL-6, and IL-10 while increasing IL-12 in CRC-bearing mice. In CRC models, elevated TNF-α triggers pro-inflammatory cytokines (eg, IL-1β and IL-6), initiating an inflammatory cascade. These cytokines collectively activate STAT3 signaling in tumor cells, sustaining proliferation. IL-1β further promotes M2 macrophage polarization, inducing immunosuppression. TGF-β activates cancer-associated fibroblasts (CAFs) that secrete collagen and fibronectin, forming a dense stromal barrier to create an immunosuppressive microenvironment facilitating CRC progression.38,39 The immunosuppressive factor IL-10 inhibits infiltration, differentiation, and maturation of tumor-infiltrating dendritic cells (DCs) and T cells while suppressing their cytotoxic functions.35,40 Reduced serum IL-12 levels in the model group indicate impaired anti-tumor immunity in colorectal cancer. ZJW counteracts this immune suppression by upregulating IL-12 to activate the JAK/STAT pathway, thereby inducing M1 macrophage polarization and enhancing anti-tumor responses to inhibit malignant progression. Collectively, these findings demonstrate that ZJW suppresses CRC progression through coordinated modulation of serum cytokines: downregulating TNF-α, IL-1β, TGF-β, IL-6, and IL-10 while elevating IL-12, which potentiates anti-tumor immunity.
In vivo experiments demonstrated that Zuojin Wan (ZJW) downregulated CD206 and Arg1 expression while upregulating iNOS in CRC tumors, indicating inhibition of M2 TAMs polarization. Flow cytometry in vitro confirmed IL-4/IL-13-induced M2-like polarization in RAW264.7 macrophages, whereas ZJW suppressed M2 polarization and increased M1 macrophage proportion. Furthermore, Transwell assays revealed that ZJW-pretreated macrophages (stimulated with IL-4/IL-13 for 48 hours) significantly inhibited migration of co-cultured CT26 and MC38 cells. The trypan blue exclusion assay further showed ZJW enhanced macrophage-mediated suppression of CT26 and MC38 proliferation. Collectively, ZJW inhibits CRC cell migration and proliferation by blocking M2 TAMs polarization.
Integrating these findings, we investigated the pathway through which ZJW inhibits M2 polarization of TAMs. The network pharmacology analysis revealed that ZJW could modulate TAMs polarization and exert anti-CRC effects by regulating kinase activity, epithelial cell proliferation, intrinsic apoptotic signaling pathways, and protein serine/threonine/tyrosine kinase activity. Additionally, our KEGG pathway enrichment analysis identified the JAK2/STAT3 signaling pathway as the most enriched among the targeted pathways. Research indicates that the JAK2/STAT3 signaling pathway serves as a central regulatory hub in the tumor microenvironment, driving the polarization of tumor-associated macrophages (TAMs) toward the pro-tumor M2 TAMs. 41 Multiple tumor-derived factors persistently activate this pathway, upregulating M2 TAMs-associated proteins (eg, Arg1, IL-10) while suppressing M1 TAMs-associated proteins (eg, IL-12, iNOS),42,43 and the process enables TAMs to acquire and maintain pro-tumor functions such as immunosuppression, pro-angiogenesis, and pro-metastasis. 44 Therefore, targeting the JAK2/STAT3 pathway represents an effective therapeutic strategy to reverse TAMs polarization, reshape the immune microenvironment, and enhance anti-tumor immunity. Based on these findings and literature evidence, supported by Western blot analysis, we propose that ZJW suppresses M2 TAMs polarization by inhibiting the JAK2/STAT3 pathway in macrophages.
Conclusion
In conclusion, integrated in vivo and in vitro experiments combined with network pharmacology demonstrate that Zuojin Wan (ZJW) suppresses the malignant progression of CRC by inhibiting M2 polarization of tumor-associated macrophages (TAMs). This inhibition is mediated through suppression of the JAK2/STAT3 signaling pathway. A limitation of this study is the need for CRC in situ models and pathway agonists to further validate this mechanism.
Footnotes
Acknowledgements
We are very grateful to all the staff of the First Affiliated Hospital of Guangxi University of Chinese Medicine for their help in the study.
Ethical Considerations
All animal experiments were approved by the Animal Care and Use Committee of Guangxi University of Chinese Medicine (License No. DW20240507-106). Clinical trial number: not applicable.
Author Contribution
Feifei Nong and Shuye Deng designed the experiment, Shuye Deng and Wende Cheng conducted the experiment, and Feifei Nong wrote the paper, Shangping Xing revised the paper. All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Natural Science Foundation of Guangxi Zhuang Autonomous Region (No. 2025GXNSFBA069323, 2023GXNSFBA026237, and 2022JJB140189); the Middle-aged and Young Teachers’ Basic Ability Promotion Project of Guangxi (No.2023KY0308 and 2022KY0280); the PhD start-up fund of Guangxi University of Chinese Medicine (No. 2022BS033); the National Natural Science Foundation of China (No. 82460718); the Guangxi Young Talents Program (No.0502405914 and DC2400003575); the Guangxi Medical University Student Innovation and Entrepreneurship Training Program (S202310598115).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This article contains all the data graphs and tables that support the conclusion.
