Abstract
Backgrounds:
Colorectal cancer (CRC) is one of the common malignant tumors, with a gradually increasing incidence. Due to late detection and poor sensitivity to chemotherapy, it has become a difficult problem in tumor prevention and treatment at present. Exploring or discovering new combinations is a significant strategy for the treatment of CRC. Compound kushen injection (CKI) is a traditional Chinese medicine injection extracted from Sophora flavescens Ait. and Smilax glabra Roxb., which is widely used in the comprehensive treatment of CRC in China. This systematic review is aimed to ascertain the clinical efficacy and safety of CKI combined with chemotherapy in the treatment of advanced CRC based on available data. On this basis, the specific application of CKI in combination with chemotherapy in clinical practice is further discussed.
Methods:
PubMed, Web of Science, the Cochrane Library, EMBASE, China National Knowledge Infrastructure, Chinese Scientific Journals Database, Wanfang Database, Chinese Biomedicine Database Searches, the Chinese Clinical Trial Registry, and ClinicalTrials.gov were searched systematically, from inception to April 20, 2024. We adopted the ROB2 tool to assess quality of the included trials, Stata 16 for data analysis, and evaluated the publication bias with the funnel plot and Egger’s test. The quality of the evidence was justified according to GRADE. We also used trial sequential analysis (TSA) to calculate the final required sample size in this meta-analysis and to verify whether the results present a reliable conclusion. The protocol for this systematic review was registered on PROSPERO (CRD42022380106) and has been published.
Results:
Sixteen trials that examined 1378 patients were included in this study. Meta-analysis revealed that compared with chemotherapy, objective response rate (ORR, RR = 1.30, 95% CI: 1.18-1.44), disease control rate (DCR, RR = 1.08, 95% CI: 1.03-1.13), and KPS score improvement rate were improved (RR = 1.18, 95% CI: 1.07-1.31) by the combination of CKI and chemotherapy in patients with advanced CRC. Additionally, CKI combined with chemotherapy was associated with lower adverse reactions such as leukopenia (RR = 0.74, 95% CI: 0.62-0.87), thrombocytopenia (RR = 0.68, 95% CI: 0.49-0.94), gastrointestinal reactions (RR = 0.72, 95% CI: 0.55-0.94), and liver damage (RR = 0.48, 95% CI: 0.30-0.79), higher CD4+ ratio (MD = 9.70, 95% CI:8.73-10.68) and CD4+/CD8+ ratio (MD = 0.25, 95% CI: 0.22-0.28), and lower CD8+ T cell ratio (MD = −5.25, 95% CI: −5.94 to −4.56). Subgroup analysis demonstrated that ORR and DCR in patients with advanced CRC were improved when CKI combined with FOLFOX and 5Fu + L-OHP. Both 15 and 20 ml/day of CKI combined with FOLFOX provided a significant effect in ORR. Moreover, ORR was improved when the accumulated CKI dose reached 280 ml per course and 420 ml in total. 7 days/course as well as 14 days/course of CKI combined with FOLFOX were effective durations in ORR. As for DCR, 7 days/course of CKI combined with FOLFOX could improve efficacy. Furthermore, CKI + FOLFOX may be useful in ORR and DCR for at least 4 cycles of combination therapies. The TSA showed that firm results in ORR and DCR were established and additional trials were unlikely to change the results.
Conclusion:
CKI combined with chemotherapy provides a statistically significant and clinically important effect in the improvement of ORR, DCR, performance status, ADR reduction, and immune function in patients with CRC. However, more rigorously designed, large-scale, and multi-center RCTs are needed in the future.
Keywords
Introduction
According to GLOBOCAN 2020 data, colorectal cancer (CRC) ranks as the second leading cause of cancer-related deaths and the third most frequently diagnosed cancer globally, with approximately 2 million new cases and about 1 million deaths estimated to occur in 2020. 1 The incidence of CRC has been steadily increasing worldwide, with a particular surge observed in developing countries. This increase is largely attributed to the adoption of unhealthy dietary habits and lifestyles, which are becoming more prevalent in these regions. 2 Due to the subtlety of clinical symptoms in the early stages, CRC is often diagnosed at an advanced stage in many patients. This late diagnosis contributes to high rates of recurrence, metastasis, and mortality associated with the disease.3,4 Current conventional first-line treatment of advanced CRC includes FOLFIRI, FOLFOX, and XELOX. Studies in the past decade have shown similar response and survival rates with an estimated median overall survival (OS) ranging from 15 to 20 months. 5 Emerging treatments including concurrent chemoradiotherapy, targeted therapy, and immunotherapy extend OS to 3 years. 6 Notably, acquired resistance and adverse drug reactions may occur rapidly and lead to disease progression. 7 And treatment options remain limited after recurrence, disease progression or failure to response to initial therapies. Chemotherapy remains the cornerstone of CRC and how to improve its efficacy is an important topic in clinical practice. Consequently, it is urgently needed to identify new effective drugs and improve the therapeutic effect of chemotherapeutic treatment in patients with advanced CRC.
Traditional Chinese medicine (TCM), as a form of complementary and alternative medicine, has emerged as a prominent component in the comprehensive treatment of cancer. Numerous clinical studies have found that TCM combined with chemotherapy in the treatment of CRC may improve clinical symptoms, enhance immunity, and alleviate chemotherapy-induced adverse reactions.8 -10 Compound Kushen injection (CKI) is one of the representative and effective Chinese patent medicines, which is extracted from 2 medical herbs: Kushen (Sophora flavescens Ait.) and Baituling (Smilax glabra Roxb.). The main components of CKI, matrine and oxymatrine, exert their anticancer activities primarily by inhibiting cancer cell proliferation, inducing cell cycle arrest and differentiation, accelerating apoptosis, restraining angiogenesis, inhibiting metastasis and invasion, reversing multi-drug resistance, and preventing or reducing chemotherapy and radiotherapy-induced toxicity. 11 One study has suggested that the active components in CKI can exert anti-cancer effects through different signaling pathways, which could have some therapeutic effects on CRC, prostate cancer, bladder cancer, etc. 12 CKI combined with chemotherapy has been shown to improve the 1-year progression free survival rate and immune function in patients with advanced CRC. 13
Now, with the increasing number of published RCTs of CKI on advanced CRC,14 -16 it is necessary to summarize the available evidence, assess the level of evidence, and provide recommendations for future research and practice. The most recent similar meta-analysis was published in 2017 with the search deadline of January 31, 2017. 17 This study demonstrated that CKI combined with chemotherapeutic drugs could improve the efficacy and quality of life and increase safety by reducing the incidence of adverse reactions in patients with advanced CRC. However, several limitations remain unresolved: (1) Which chemotherapy regimen combined with CKI is efficient in improving objective response rates (ORR) and disease control rate (DCR); (2) Whether CKI exerts a positive effect on immune function; (3) Whether current conclusions change with further trials.
To expand upon the existing evidence base and address the limitations identified in prior reviews, we have undertaken a systematic review with a focus on evaluating 2 pivotal questions: (1) Whether the available evidence could adequately demonstrate the effectiveness of CKI combined with chemotherapy; (2) If CKI with chemotherapy improves clinical effects, which combination therapies can maximize the effects? In this study, our purpose was to synthesize the published studies under a unified framework and explore whether current evidence is sufficient to indicate efficacy by using trial sequence analysis (TSA). Furthermore, our meta-analysis aimed to provide decision-making information for the clinical use of CKI combined with chemotherapy in patients with advanced CRC and to further guide future research.
Materials and Methods
The protocol for this systematic review was registered on PROSPERO, the International Prospective Register of Systematic Reviews (CRD42022380106), and has been published. 18 We conducted the study in full accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines, adhering to Supplemental Material 1.
Inclusion Criteria
All of the eligible clinical trials that met the following inclusion criteria were included.
Type of participants (P)
Patients with advanced (stage III or IV) CRC were diagnosed by pathology or cytology.
Type of interventions (I and C)
The treatment group was treated with CKI (intravenous drip) combined with chemotherapy (oral or intravenous drip), and the control group was treated with chemotherapy (oral or intravenous drip).
Type of outcome measures (O)
The primary efficacy outcomes of this study were short-term survival outcomes, including ORR and DCR. ORR and DCR were response evaluations of anticancer treatment, assessed according to the World Health Organization (WHO) criteria or Response Evaluation Criteria in Solid Tumors (RECIST). Tumor disease changes included complete remission (CR), partial remission (PR), stable disease (SD), and progressive disease (PD). ORR was defined as CR + PR and DCR was defined as CR + PR + SD.
The secondary efficacy outcomes included long-term survival outcomes, performance status (KPS improvement rate), immune function indicators (CD4+, CD8+, CD4+/CD8+), and incidence of adverse drug reactions (ADRs) including leukopenia, thrombocytopenia, gastrointestinal reactions, liver damage, and alopecia. ADRs were judged based on the criteria of the WHO for acute and subacute toxicity of antineoplastic agents.
Types of studies (S)
(1) The study design was an RCT, whether the blind method is used or not; (2) All studies described the randomization method precisely, such as the random number table method, and so on; (3) The studies were published in English or Chinese.
Exclusion Criteria
(1) Patients with secondary CRC, or complicated with other malignant tumors; (2) Patients who were receiving neoadjuvant chemotherapy; (3) The inclusion criteria or diagnostic criteria were not clear; and (4) The data report was incomplete and the published literature was repeated.
Search Strategy
Relevant RCTs were searched and identified from PubMed, Web of Science, the Cochrane Library, EMBASE, China National Knowledge Infrastructure (CNKI), Chinese Scientific Journals Database (VIP database), Wanfang database, and Chinese Biomedicine Database (CBM) Searches (from inception to April 20, 2024). We also searched the Chinese Clinical Trial Registry (ChiCTR) and ClinicalTrials.gov to identify ongoing and completed trials.
The search strategy was based on a combination of controlled vocabulary (Medical Subject Headings) and free-text terms. The terms “Colorectal Neoplasms,” “Colonic Neoplasms,” “Rectal Neoplasms,” “Advanced,” “Recurrence,” “Recurrence,” “Metastasis,” “Stage III,” “Stage IV,” “Compound kushen injection,” and “randomized controlled trial” were used to develop the search strategy for PubMed, which is shown in Supplemental Material 2. Modifications to this search strategy were used with other databases. The retrieved studies, reference lists in reviews, clinical studies, and additional Chinese language journals were all considered potential sources of other papers and were separately manually reviewed.
Study Selection
One pair of independent reviewers (JW and XM) screened titles and abstracts of search results in the databases after removing duplicates. Two independent reviewers then reviewed and assessed the full text according to the eligibility criteria. Any disagreement was resolved by discussion and consultation or the interposition of the third review member(XW) until a consensus was reached.
Data Extraction
Microsoft Excel was used in the process of data extraction. The following information was extracted from each trial: (1) identification information (first author and publication year); (2) general information (the randomization method and sample size); (3) participants (clinical stage, average age, and sex); (4) intervention details (dosage and duration of CKI); (5) comparison details (chemotherapy regimen, dose, frequency, and duration of treatment), and (6) outcomes details (efficacy and safety).
Risk of Bias Assessment
We used ROB2 (Version 2 of the Cochrane tool for assessing the risk of bias in randomized trials), provided by the Cochrane Methodology Group to evaluate the quality of the included trials. 19 The ROB2 tool encompasses evaluation across 5 critical domains: randomization process, deviation from intended intervention, missing outcome data, measurement of the outcomes, and selection of the reported result. The investigators JW and XM independently assessed the risk of bias within each field, categorizing it into 3 levels: “low,” “some concerns,” and “high” risk of bias. This categorization was based on the responses to a series of distinct signal questions pertinent to each field. The responses were categorized as: Yes (Y), Probably Yes (PY), Probably No (PN), No (N), and No Information (NI).
Measurement of Treatment Effect
The meta-analysis was performed using the software Stata 16.. We calculated the pooled relative risks (RR) for dichotomous outcomes and mean differences (MD) for continuous outcomes, with the corresponding 95% confidence intervals. Random-effects or fixed-effects models were chosen based on the analysis of heterogeneity. We evaluated heterogeneity using the I 2 statistic. An I 2 value >50% was considered to indicate substantial heterogeneity, and I 2 > 75% was considered indicative of heterogeneity. 20 Quantitative synthesis was conducted for groups with 3 or more randomized controlled trial (RCT) reports. In cases where fewer than 3 RCT reports were available within a group, a narrative synthesis approach was employed. Given the strong correlation between the 2 anti-tumor treatment response evaluation criteria (ORR and DCR), the WHO and RECIST criteria were considered homogeneous. 21 For outcomes informed by more than 10 trials, we visually inspected funnel plots and performed Egger’s test to assess publication bias.
Subgroup Analysis and Sensitivity Analysis
To explore heterogeneity among trials, we performed predefined subgroup analyses to determine if specific baseline factors had an impact on the treatment effects. These subgroups included:(1) different chemotherapy regimens;(2) dosage of CKI; (3) duration of CKI; (4) the number of chemotherapy courses. A sensitivity analysis was undertaken to explore the effect of each study on the overall pooled estimate.
Quality of Evidence
The GRADE method was used to evaluate and grade the quality of evidence for each outcome measure in 5 categories: risk of bias, inconsistency, indirectness, imprecision, and publication bias. 22 We used pro-Guideline Development Tool (GRADE pro GDT) software procedures and guidelines. 23 JW and XM conducted the assessment of GRADE evaluation and use footnotes to downgrade or increase the ratings for justifying decisions.
Trial Sequential Analysis
In conducting a meta-analysis, it is crucial to minimize the risk of drawing a falsely positive conclusion. To achieve this, Trial Sequential Analysis (TSA) employs an alpha-spending function, which establishes thresholds for statistical significance to control the risk of a type I error. 24 It also applied a β-spending function and produced futility boundaries to control type II error. 25 The Z value refers to the statistic provided by log of the pooled intervention effect divided by its standard error. If a Z curve crosses the futility boundaries or sequential monitoring boundary, we can conclude that a sufficient number of patients are included for anticipated intervention effect and further trials are unlikely to change the findings. Therefore, we used TSA Software v.0.9.5.10 Beta (Copenhagen Trial Unit, Copenhagen, Denmark) to determine if the required sample size should reach the threshold of statistical significance. The boundary type was set to be 2-sided, with a 5% Type 1 Error and power of 0.80. We constructed cumulative z-scores and the required information sizes to firmly accept or refute the effect sizes of interest.
Results
Study Selection
We identified 311 citations. Of those, 110 studies underwent full-text review, 94 of which were excluded. A total of 16 RCTs14 -16,26 -38 matched all the eligibility criteria finally. Screening and selection are summarized using a PRISMA flow diagram in Figure 1.

Flow diagram of the literature search.
Study Characteristics
Overall, 16 eligible trials enrolled a total of 1378 patients, all conducted in China, with 689 cases in the treatment group and control group, respectively. All RCTs described the randomization process, with 14 studies14 -16,26-31,33,35 -38 using the random number table method, one 34 using the stratified random balance method and one 32 adopting the computerized random number generator. In terms of therapy regimen, a total of 11 RCTs14,15,27 -29,31,32,34 -37 employed CKI + FOLFOX, two16,30 used CKI + 5Fu + oxaliplatin and one 33 adopted CKI + FOLFIRI. Of the remaining 2 studies, one 26 used CKI + XELOX and the other adopted CKI + RALOX. Furthermore, chemotherapy courses ranged from 2 to 8, and the dose of CKI ranged from 12 to 15, 20, and 30 ml. Due to the diverse combination therapies of CKI + chemotherapy, a subgroup analysis was considered. Additional details are summarized in Tables 1 and 2.
Basic Information and Treatment Measures.
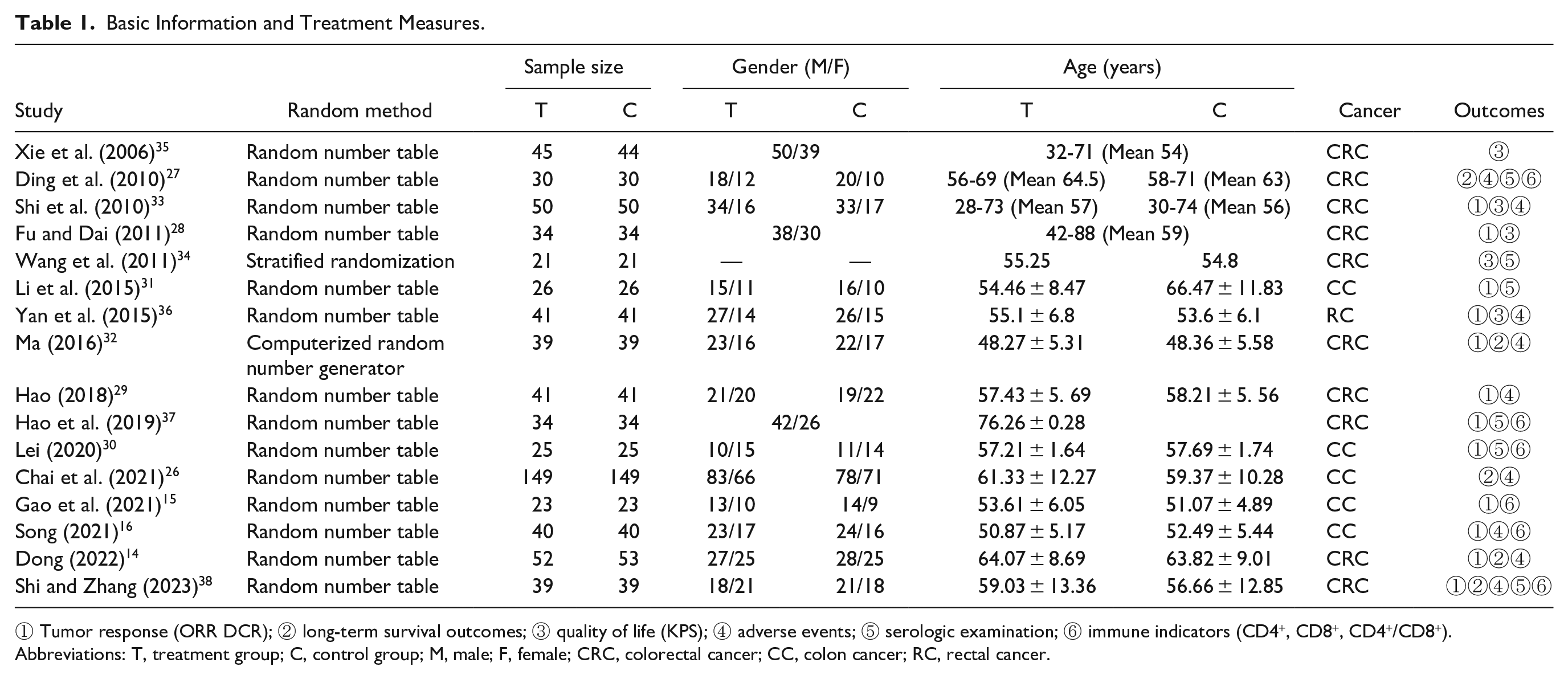
① Tumor response (ORR DCR); ② long-term survival outcomes; ③ quality of life (KPS); ④ adverse events; ⑤ serologic examination; ⑥ immune indicators (CD4+, CD8+, CD4+/CD8+).
Abbreviations: T, treatment group; C, control group; M, male; F, female; CRC, colorectal cancer; CC, colon cancer; RC, rectal cancer.
Intervention Details of Included RCTs.
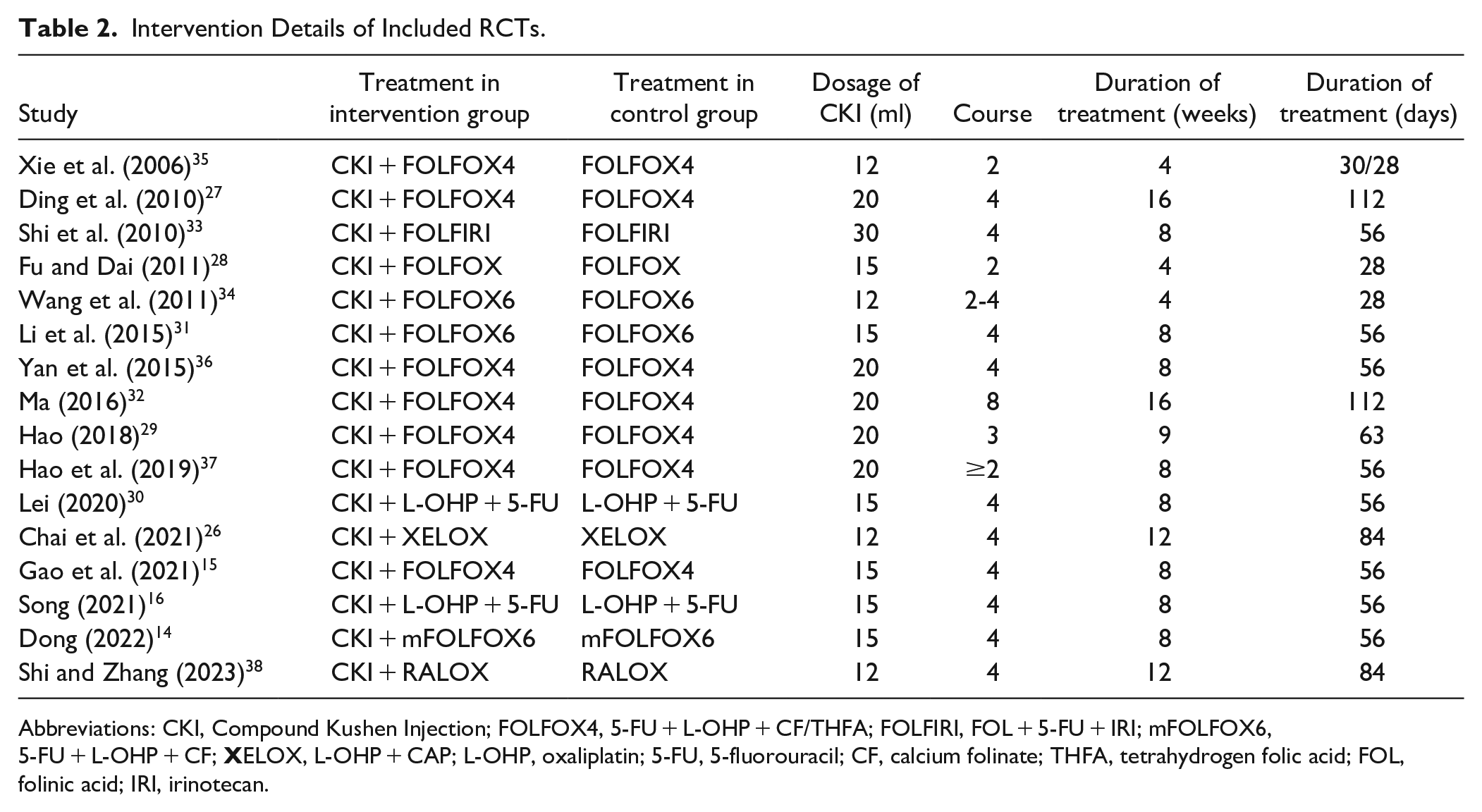
Abbreviations: CKI, Compound Kushen Injection; FOLFOX4, 5-FU + L-OHP + CF/THFA; FOLFIRI, FOL + 5-FU + IRI; mFOLFOX6, 5-FU + L-OHP + CF;
Risk of Bias
In total, 9 trials14 -16,27,30-32,37,38 were deemed to be at low risk of bias. Of the remaining 7 trials,26,28,29,33 -36 assessment of the outcomes might be influenced by knowledge of intervention received and one 35 was rated as high risk for selective reporting (Figure 2).

Risk of bias of included study. (A) risk of bias summary; (B) risk of bias graph.
Outcome Measures
Primary outcomes
ORR
In total, 12 trials14 -16,28-33,36 -38 with 869 patients reported ORR data. To explore the potential effect differences in ORR, we conducted a subgroup analysis according to the different chemotherapy regimens with CKI, namely, CKI + FOLFOX, CKI + FOLFIRI, CKI + 5Fu + L-OHP and CKI + RALOX. As shown in Figure 3, the pooled analysis demonstrated that ORR could be increased by 30% in the CKI + chemotherapy group compared with chemotherapy alone (RR = 1.30, 95% CI: 1.18-1.44). Considering that the different chemotherapy regimens might lead to high clinical heterogeneity, we conducted a subgroup analysis according to chemotherapeutic regimens. The results showed that both CKI + FOLFOX and CKI + 5Fu + L-OHP presented a significantly improved ORR compared with the control group (CKI + FOLFOX: RR = 1.26, 95% CI: 1.12-1.43; CKI + 5Fu + L-OHP: RR = 1.45, 95% CI: 1.17-1.79). The CKI + FOLFIRI and CKI + RALOX subgroup did not show a statistical advantage due to limited sample size.

Forest plot of ORR in CKI combined with chemotherapy versus chemotherapy: subgroup according to chemotherapy regimens.
Owing to the sample size, we continued to focus on the CKI + FOLFOX subgroup to investigate whether the dosage of CKI would influence ORR. Subgroup analysis showed that both 15 and 20 ml/day of CKI combined with FOLFOX were effective in improving ORR compared with the control group (15 ml: RR = 1.26, 95% CI: 1.04-1.53; 20 ml: RR = 1.27, 95% CI: 1.09-1.48; Figure 4A). Moreover, when the cumulative dose of CKI reached 280 ml per course, ORR could be increased by 23% in the experimental group compared with chemotherapy alone (RR = 1.23, 95% CI: 1.05-1.45, Figure 4B). Other cumulative doses of CKI/course did not present statistical superiority, possibly due to the limited sample size. Six trials15,28,29,31,32,36 described the total dose of CKI combined with FOLFOX in detail, as depicted in Figure 4C, we calculated the total dose of CKI during the chemotherapy regimen, and found that 420 ml CKI combined with FOLFOX was an effective total dose to improve ORR (RR = 1.26, 95% CI: 1.04-1.53). Taking into account the limited sample size in other subgroups, the results of 1120, 560, and 840 ml CKI combined with FOLFOX were not evident.

Forest plot of CKI combined with FOLFOX versus FOLFOX in ORR: subgroup according to dosage of CKI. (A) Dosage of CKI/day; (B) dosage of CKI/course; (C) total dosage of CKI.
Furthermore, 8 trials14,15,28,29,31,32,36,37 reported duration of CKI/course combined with FOLFOX in the experimental group and the results showed that CKI of both 7 and 14 days/course could be improved when combined with FOLFOX regimen (7 days: RR = 1.29, 95% CI: 1.07-1.55; 14 days: RR = 1.25, 95% CI: 1.07-1.46; Figure 5).
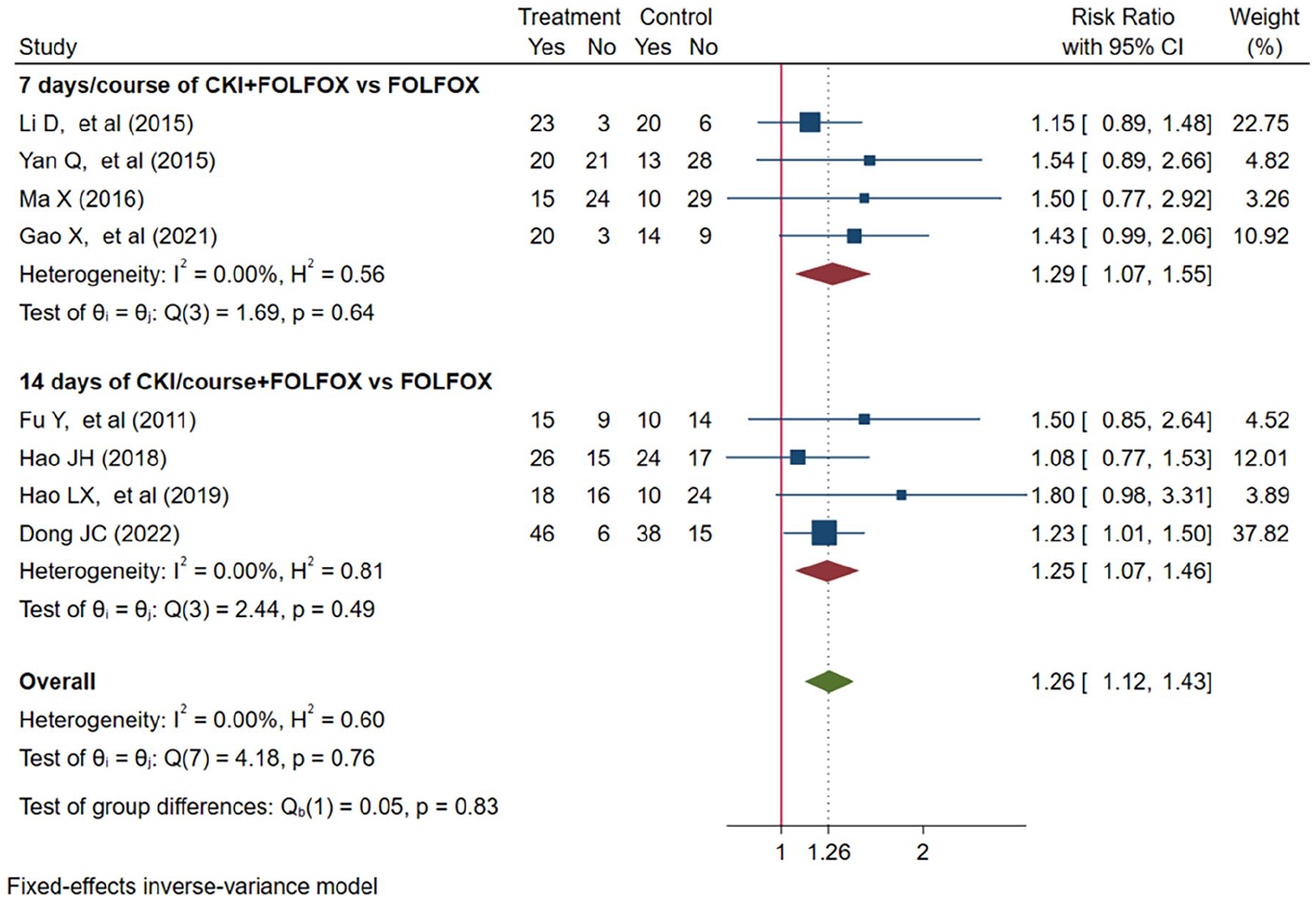
Forest plot of CKI combined with FOLFOX versus FOLFOX in ORR: subgroup according to duration of CKI/course.
As for number of chemotherapy courses, Hao et al. 37 did not depict specific detail in the original article. The remaining 5 trials14,15,31,32,36 found that ORR could be improved in the CKI + FOLFOX group when the combination therapies were used for at least 4 courses (RR = 1.26, 95% CI: 1.10-1.44). The subgroup of fewer than 4 courses did not show a statistical advantage in ORR (RR = 1.18, 95% CI: 0.88-1.59) because the confidence interval exceeded the clinical decision threshold (Figure 6).

Forest plot of CKI combined with FOLFOX versus FOLFOX in ORR: subgroup according to number of chemotherapy cycles.
DCR
The pooled results of 12 trials14 -16,28-33,36 -38 with 869 patients showed that compared with chemotherapy alone, CKI + chemotherapy had a higher DCR (RR = 1.08, 95% CI: 1.03-1.12; Figure 7). Subgroup analysis by chemotherapy regimens suggested that DCR could be improved by 6%, 14%, and 50% in the CKI + FOLFOX group, CKI + 5Fu + L-OHP, and CKI + RALOX group, respectively (CKI + FOLFOX: RR = 1.06, 95% CI: 1.01-1.12; CKI + 5Fu + L-OHP: RR = 1.14, 95% CI: 1.01-1.29; CKI + RALOX: RR = 1.50, 95% CI: 1.01-2.23; Figure 7).

Forest plot of DCR combined with chemotherapy versus chemotherapy: subgroup according to chemotherapy regimens.
Additionally, 8 trials14,15,28,29,31,32,36,37 including 561 patients concentrated on FOLFOX + CKI versus FOLFOX of DCR. Therefore, we made subgroups according to daily dose (Figure 8A), dose per course (Figure 8B), and total dose of CKI (Figure 8C) combined with FOLFOX in DCR, but the results did not show statistical differences. We consider that this may be related to the high DCR in the control group, and there was only a tiny small difference between the experimental group and the control group in DCR.

Forest plot of CKI combined with FOLFOX versus FOLFOX in DCR: subgroup according to dosage of CKI. (A) Dosage of CKI/day; (B) dosage of CKI/course; (C) total dosage of CKI.
Furthermore, the subgroup by CKI duration/course showed that 7 days/course of CKI combined with FOLFOX was associated with higher DCR (RR = 1.09, 95% CI: 1.01-1.17), but the other 14 days/course of the CKI group did not present a significant advantage (RR = 1.05, 95% CI: 0.99-1.12) (Figure 9).
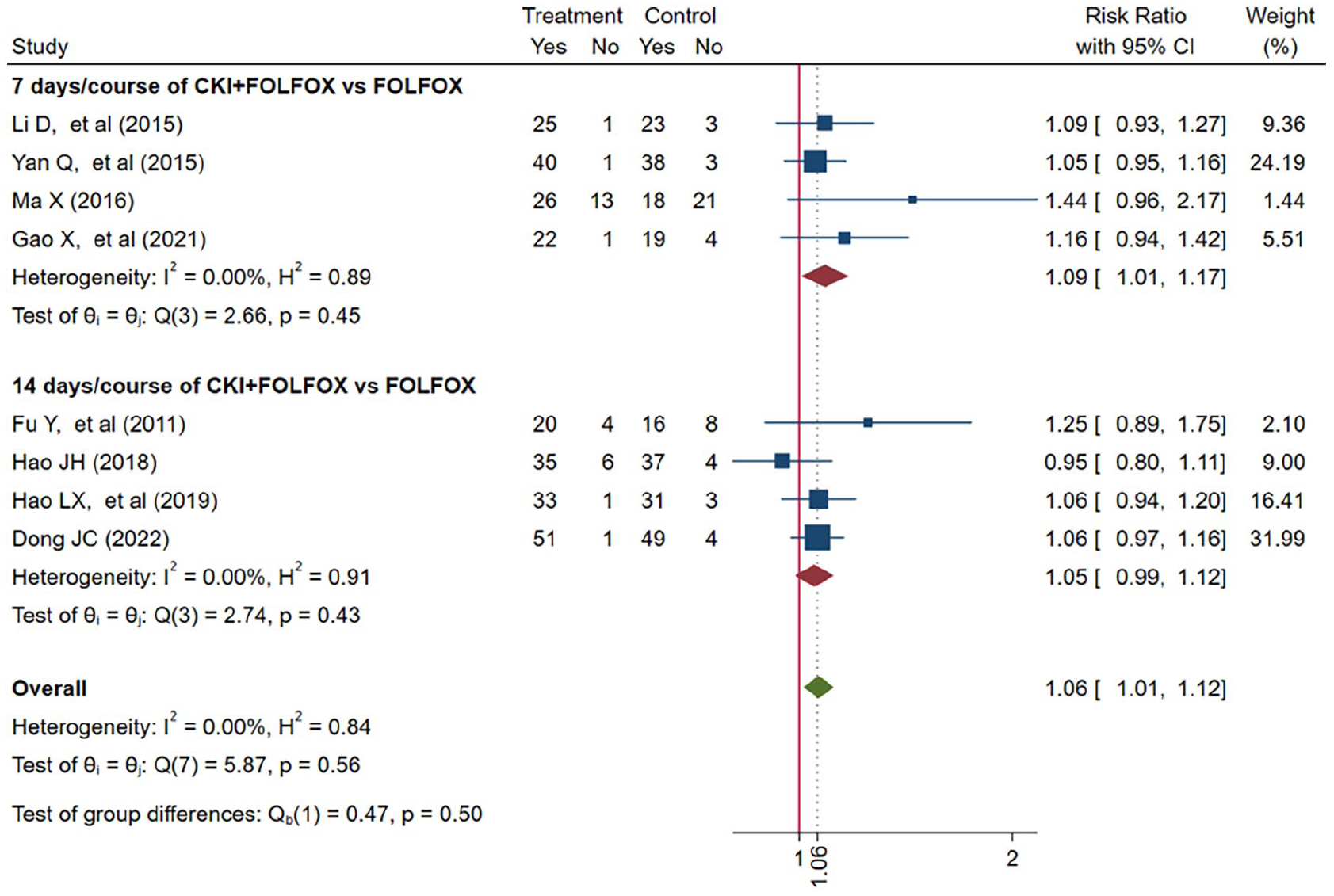
Forest plot of CKI combined with FOLFOX versus FOLFOX in DCR: subgroup according to duration of CKI/course.
Regarding the number of chemotherapy cycles, DCR could be improved when combination therapies (CKI + FOLFOX) were used for at least 4 courses (RR = 1.08, 95% CI: 1.02-1.14) compared with FOLFOX, but the effect on fewer than 4 courses did not show a statistical advantage (RR = 1.00, 95% CI: 0.86-1.15; Figure 10).

Forest plot of CKI combined with FOLFOX versus FOLFOX in DCR: subgroup according to number of chemotherapy cycles.
Secondary outcomes
Performance status
As presented in Figure 11, 5 trials28,33 -36 including 354 patients reported KPS improvement rate. Compared with chemotherapy alone, CKI + chemotherapy exhibited a better KPS improvement rate (RR = 1.18, 95% CI: 1.07-1.31), suggesting a superior performance status in patients with CRC.

Forest plot of KPS improvement rate.
ADRs
10 trials14,16,26,27,29,32 -34,36,38 referred to ADRs in Figure 12. Among them, the main outcomes were leukopenia (8 trials16,26,27,29,33,34,36,38), thrombocytopenia (4 trials26,33,36,38), gastrointestinal reactions (4 trials14,16,32,36), liver damage (5 trials16,32,33,36,38), and alopecia (4 trials26,32,33,36). CKI + chemotherapy group decreased the risk of leukopenia (RR = 0.67, 95% CI: 0.54-0.82), thrombocytopenia (RR = 0.68, 95% CI: 0.49-0.94), gastrointestinal reactions (RR = 0.72, 95% CI: 0.55-0.94), and liver damage (RR = 0.48, 95% CI: 0.27-0.83). However, no statistically significant difference was detected in the occurrence of alopecia.

Forest plot of ADRs.
Immune indicators
Among immune indicators, we mainly focused on CD4+ T cell ratio (4 trials,15,16,27,30 Figure 13), CD8+ T cell ratio (4 trials,15,16,27,30 Figure 14), and CD4+/CD8+ ratio (6 trials,15,16,27,30,37,38 Figure 15). CKI + chemotherapy increased CD4+ ratio (MD = 9.70, 95% CI: 8.73-10.68) and CD4+/CD8+ ratio (MD = 0.25, 95% CI: 0.21-0.28), and reduced CD8+ ratio (MD = −5.25, 95% CI: −5.94 to −4.56).

Forest plot of CD4+.

Forest plot of CD8+.
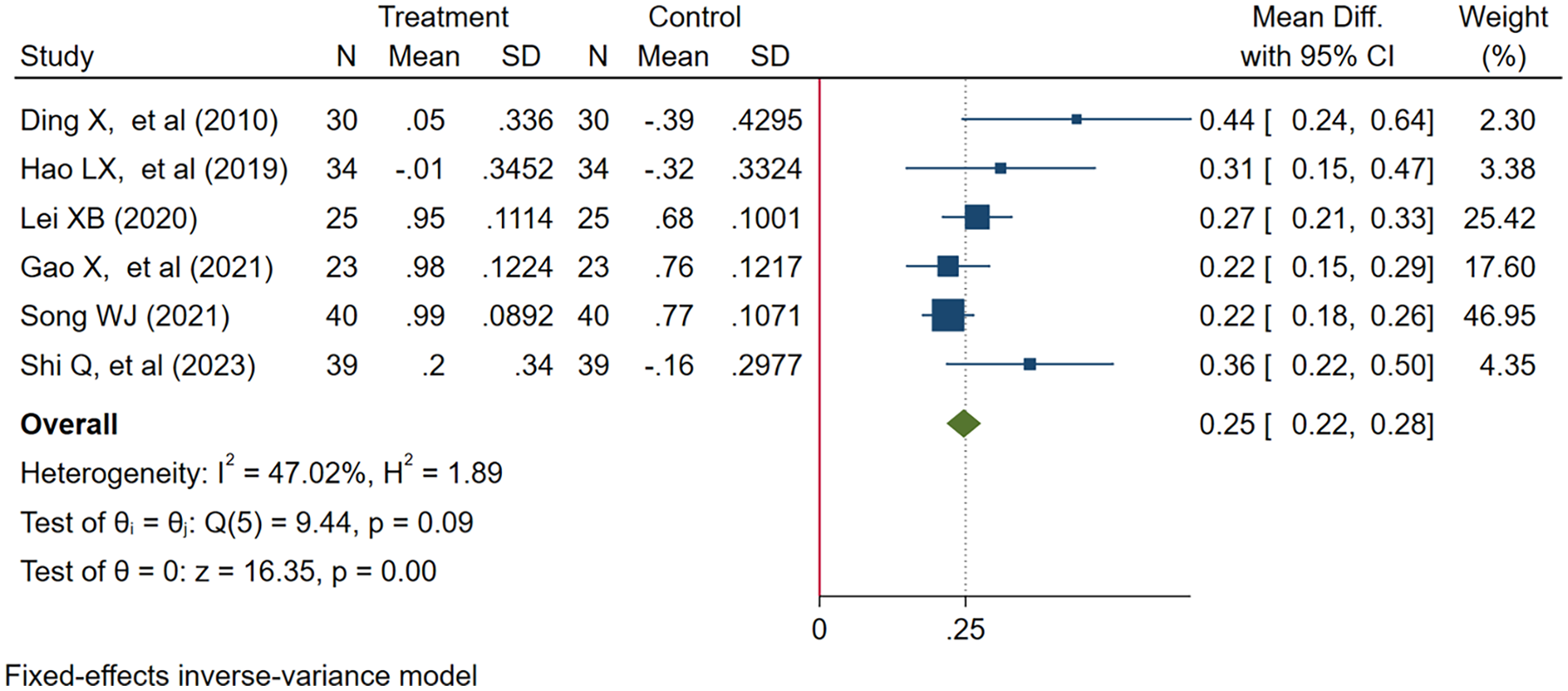
Forest plot of CD4+/CD8+.
Long-term survival outcomes
4 trials26,27,33,38 reported long-term survival outcomes. Ding et al. 27 reported that there were no significant differences between the experimental group and the control group in PFS and mOS (PFS: 9.2 vs 8.7 months; mOS:19.8 vs 18.6 months). But 1 trial 26 found that compared with chemotherapy, CKI + chemotherapy could prolong median survival time (24 vs 15.5 months). Another trial 32 presented that CKI + chemotherapy increased the 1-year survival rate by 15.39% (23.08% vs. 7.69%) with a statistical difference. Another trial 38 observed patients treated with second-line chemotherapy combined with CKI after first-line chemotherapy failed. It was found that the progression-free survival rate (PFSR) of patients treated with RALOX + CKI at 6 and 12 months were significantly higher than those of control group (P < .05).
Publication Bias
We conducted a publication bias analysis on both ORR and DCR. Egger’s test found that ORR had no publication bias (P = .1770), but DCR showed evidence of publication bias (P = .0196). To address this, the nonparametric trim and fill method was employed. After the data of 5 virtual studies were included, meta-analysis of all studies was performed again according to stata software. The adjusted results indicated a RR of 1.059, with a 95% CI ranging from 1.017 to 1.104. This suggested that even after incorporating the additional 5 studies, the findings remained statistically significant, with no reversal in effect, thus confirming the robustness of the combined outcomes. The ORR findings were graphically represented in a funnel plot (Figure 16A), while the DCR results were depicted using the funnel plot adjusted by the nonparametric trim and fill method (Figure 16B). With the addition of the 5 references, the funnel plot exhibited approximate symmetry, indicating a balanced distribution of study results.

Publication bias analysis:(A) ORR; (B) DCR was treated by the nonparametric trim and fill method.
Quality of Evidence and Sensitivity Analyses
Based on the GRADE criteria, the quality of the evidence for ORR and leukopenia was moderate. Other results were evaluated as having low or very low quality of evidence, mainly due to risk of bias, small study sample size, statistical heterogeneity, and invalid confidence interval. A summary of these findings is presented in Table 3.
Summary of Evidence.

Abbreviations: CI, confidence interval; MD, mean difference; RR, risk ratio.
Blind method is not reported, and most information is from studies at unclear risk of bias.
Small sample size.
Statistical heterogeneity exists.
The confidence interval crosses the clinical decision threshold.
In the sensitivity analysis, each individual study was omitted sequentially to assess its impact. The pooled estimate remained consistent with the overall observed estimate, indicating that no single study had a large influence on the pooled results. The primary outcomes of the sensitivity analysis estimates are provided in Figure 17, and the other outcomes are depicted in Supplemental Material 3.
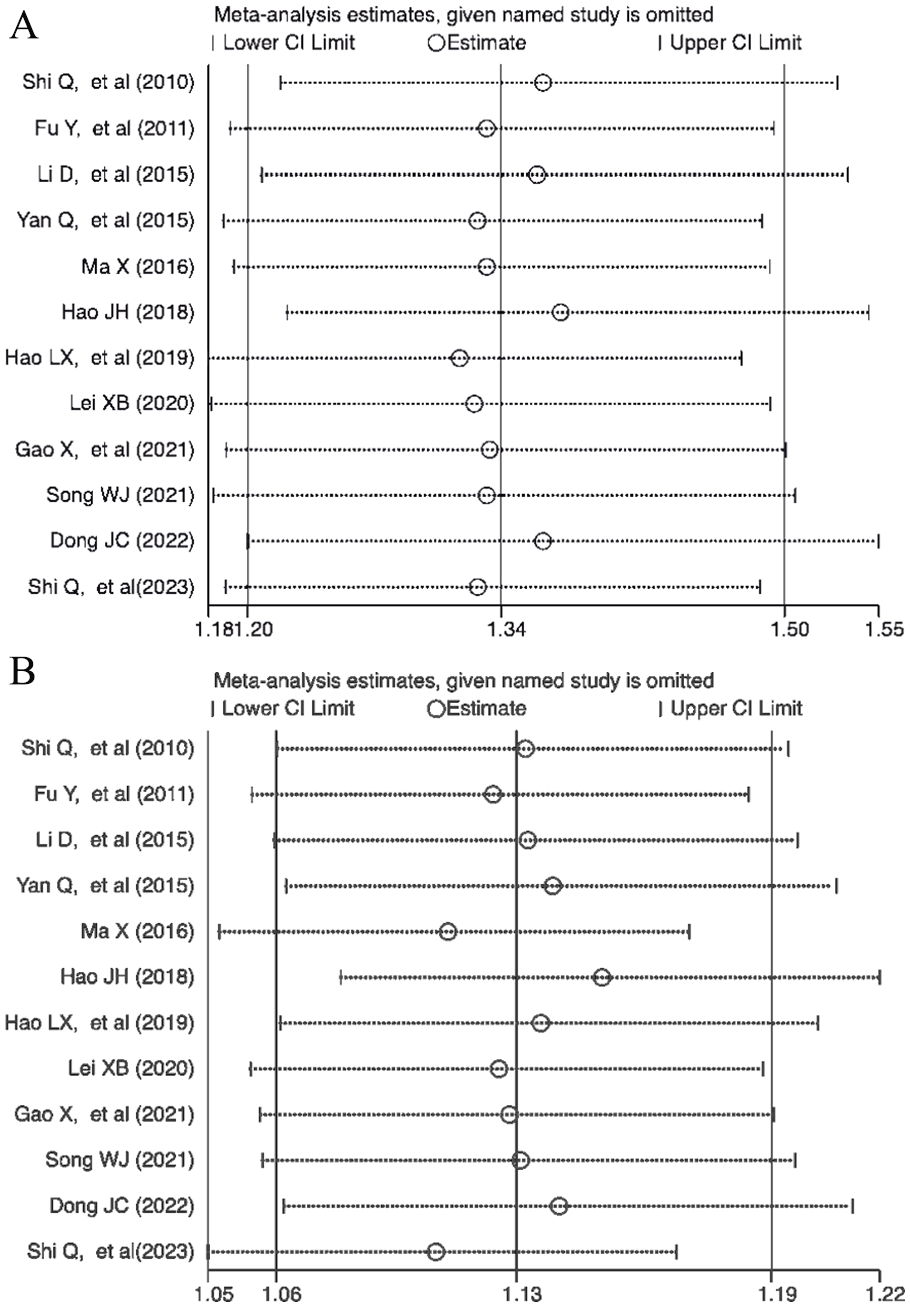
Sensitivity analyses of primary outcomes: (A) ORR; (B) DCR.
Trial Sequential Analyses
For ORR, the cumulative Z curve crossed both the conventional and the trial sequential monitoring boundary. This indicated that a sample size of 288 patients was required to draw stable conclusions (Figure 18A). For DCR, a sample size of 729 patients was determined to be necessary for drawing stable conclusions. The cumulative Z-curve for DCR also crossed both the conventional and the trial sequential monitoring boundary (Figure 18B).
Thus, the findings suggested that the current trials for ORR and DCR have achieved the required information size. The results were both conclusive and robust, indicating that additional trials were unlikely to change the established conclusions.

Outcomes of TSA. The blue cumulative Z-curve was constructed by using a fixed-effects model. The green horizontal dotted lines on the plot indicated the conventional boundaries for benefit, while the horizontal red lines signified the TSA boundaries for benefit. (A) TSA of ORR: A required information size of 288 patients was calculated, based on an event proportion of 47.8% observed in the control group. (B) TSA of DCR: A required information size of 729 patients was calculated, based on an event proportion of 78.2% observed in the control group.
Discussion
Principal Findings
We searched for as many trials as possible and conducted a meta-analysis with the aim of addressing our 2 questions. The results suggested that (1) Short-term efficacy outcomes–ORR and DCR could be improved by 30% and 8%, respectively, when CKI combined with chemotherapy. The results were confirmed by TSA, and they were reliable and stable. (2) Both 15 and 20 ml CKI/day combined with FOLFOX were effective in improving ORR. Furthermore, the ORR could be improved by 23% when the accumulated CKI dose reached 280 ml per course and by 26% when the accumulated CKI dose reached 420 ml in total. 7 days/course as well as 14 days/course CKI combined with FOLFOX are effective duration in ORR. Regarding DCR, CKI 7 days/course combined with FOLFOX could improve efficacy. In addition, we found that a higher ORR and DCR were associated with receiving at least 4 courses of combination therapy (CKI + FOLFOX). Furthermore, we suggested that CKI combined with chemotherapy could improve performance status with a better KPS improvement rate, increase CD4+ ratio and CD4+/CD8+ ratio, reduce CD8+ ratio and the incidence of ADRs (leukopenia, thrombocytopenia, liver damage, and gastrointestinal reactions).
Recent evidence suggested that CKI can inhibit proliferation, metastasis, and invasion of cancer cells by inducing cell cycle arrest, accelerating cell apoptosis, and inhibiting angiogenesis 39 which may explain the mechanism why CKI combined with chemotherapy can play a synergistic role in CRC. One experiment concluded that CKI effectively induced cell-cycle arrest of CRC cells in vitro and suppressed development of CRC in vivo by downregulating the expression of p53 and CHEK1. CKI synergistically enhanced the ability of cisplatin to mediate anti-tumor activity in mutant CRC cells of p53-R273H/P309S through the induction of extrinsic apoptosis pathway by specifically increasing the expression of DR5. 40 It provided evidence for the potential application of CKI combined with cisplatin in the treatment of p53-mutated CRC. Furthermore, our subgroup analysis showed that CKI combined with chemotherapy had to reach a certain dose and course to be effective in ORR and DCR. Wang et al. 41 suggested that CKI may play an anti-tumor effect through anti-angiogenesis and with an increase in CKI dose, the tumor blood supply was reduced, thus inhibiting tumor growth . Additionally, our results found that CKI may regulate immune function by CD4+, CD4+/CD8+, and CD8+ ratio. CD4+ T cells can target tumor cells in various ways, either directly by eliminating tumor cells through cytolytic mechanisms or indirectly by modulating the tumor microenvironment. CD4+ helper T cells promote priming and both effector functions and memory functions of cytotoxic T lymphocytes, which provides an opportunity to amplify the T cell response against tumor-associated antigens without deleterious autoimmunity. 42 CD8+ T cells represent inhibitory T cells. The tumor immunity cycle is a sequence of events ending with CD8+ T cell-directed tumor cytolysis. The interactions between dendritic cells and CD8+ T cells are mediated in a receptor–ligand-related way and CD4+ T cells provide help (stimulus) for the priming of CD8+ T cells. 43 Maintenance of immune balance depends on mutual coordination of the T cell subsets.
Strengths and Comparison with Other Studies
Our findings are similar to results from previously published systematic reviews. A network meta-analysis comparing the efficacy and safety of 8 types of TCM injections in combination with chemotherapy for the treatment of CRC showed that CKI performed the best in improving the ORR of patients when combined with chemotherapy. 44 CKI is a potential effective adjuvant drug for the treatment of tumor diseases. However, due to the current low quality of research methodology and evidence, its effectiveness needs to be confirmed by more high-quality evidence-based medical evidence. 45 In this systematic review and meta-analysis, we incorporated evidence from the most recent studies and had some strengths over previous reviews. (1) We performed several predefined subgroup analyzes for ORR and DCR according to the retrieved data, finding that both 15 and 20 ml/day of CKI combined with FOLFOX were effective in ORR. Considering that 20 ml/day is advised in the medicine specification, this may bring advantages in health economics if 15 ml/day is effective, too. But 15 ml/day of CKI needs more high-quality clinical evidence to support. (2) We concluded the effective dose of CKI per course and the total accumulated dose combined with FOLFOX in ORR, which can provide some guidance for the clinical use of CKI in advanced CRC. (3) More than 4 courses of CKI + FOLFOX and 7 days of CKI per course were effective in ORR and DCR, indicating that a sufficient treatment course was necessary. (4) All available data from the collected trials were applied without intentional selection and we defined the randomization process explicitly to bring more convincing eligible trials, which was beneficial for the quality of the evidence. (5) GRADE criteria was performed to synthesize the quality of outcomes and a sensitivity analysis was adopted to assess the robustness of the results. We further applied TSA to obtain more stable evidence, and the results indicated that current trials have reached the required information size in ORR and DCR. So our conclusion that CKI combined with chemotherapy was effective in ORR and DCR was confirmed by TSA. Based on the above, we believed that the features of our current meta-analysis would provide more accurate and reliable evidence.
Limitations
However, this study had several limitations. Owing to limited resources, we had to exclude studies not published in English and Chinese. Furthermore, due to the lack of enough trials, our subgroups of chemotherapy courses, doses of CKI, and duration were confined to the FOLFOX regimen alone. Additionally, all trials did not adopt blinding methods, which reduced the quality of our evidence, especially in some subjective outcomes such as the KPS improvement rate. Notably, original studies depicted long-term survival outcomes that were scarce in our study, so we could not investigate whether CKI could bring benefits to patients in long-term efficacy. In the part of publication bias, there was publication bias in the DCR results. After addressing this with the nonparametric trim and fill method, the results remained robust. This bias may be attributed to the fact that this study focused on a population that had failed first-line chemotherapy and subsequently received second-line chemotherapy, all of whom had distant metastasis. The prognosis for such patients tends to be poorer compared to the majority of patients in other studies who were treated with first-line chemotherapy regimens.
Implications for Research and Clinical Practice
Based on these limitations, future trials should consider that the need for large-sample, multi-center RCTs that investigate long-term efficacy is urgent, with rigorous prospectively designed methods warranted to guarantee the quality of the outcomes. This study is predominantly focused on the Chinese population, which raises questions about the generalizability of the results to other diverse global populations. To achieve a more comprehensive understanding of CRC and to devise more effective public health strategies, it is essential to extend the applicability of our findings to a wider array of global demographics. We advocate for the initiation of future global, multicenter trials to broaden the scope and impact of our research. Investigators should also turn their attention to CKI combined with immunotherapy or targeted therapy, and related trials in CRC may be warranted. Furthermore, we found that the dose and duration of CKI and the number of chemotherapy courses may influence the efficacy in ORR and DCR when the experimental group adopted CKI + FOLFOX in CRC. However, we lacked sufficient data from RCTs to conclusively prove our hypotheses in CKI combined with other chemotherapy treatments, and additional RCTs that include subgroup analyses are needed to compare the efficacy of different chemotherapy treatments in CRC.
Conclusion
In conclusion, in this systematic review and meta-analysis of 15 trials comprising 1378 patients with advanced CRC, compared with chemotherapy alone, CKI combined with chemotherapy provides a statistically significant and clinically important effect in the improvement of ORR, DCR, performance status, ADR reduction, and immune function. Future studies should examine long-term efficacy indicators, and more original high-quality RCTs including CKI combined with a variety of treatment regimens are necessary.
Supplemental Material
sj-doc-1-ict-10.1177_15347354241258458 – Supplemental material for Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis
Supplemental material, sj-doc-1-ict-10.1177_15347354241258458 for Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis by Jingyuan Wu, Xinyi Ma, Xinmiao Wang, Guanghui Zhu, Heping Wang, Ying Zhang and Jie Li in Integrative Cancer Therapies
Supplemental Material
sj-doc-2-ict-10.1177_15347354241258458 – Supplemental material for Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis
Supplemental material, sj-doc-2-ict-10.1177_15347354241258458 for Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis by Jingyuan Wu, Xinyi Ma, Xinmiao Wang, Guanghui Zhu, Heping Wang, Ying Zhang and Jie Li in Integrative Cancer Therapies
Supplemental Material
sj-docx-3-ict-10.1177_15347354241258458 – Supplemental material for Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis
Supplemental material, sj-docx-3-ict-10.1177_15347354241258458 for Efficacy and Safety of Compound Kushen Injection for Advanced Colorectal Cancer: A Systematic Review and Meta-Analysis of Randomized Clinical Trials with Trial Sequential Analysis by Jingyuan Wu, Xinyi Ma, Xinmiao Wang, Guanghui Zhu, Heping Wang, Ying Zhang and Jie Li in Integrative Cancer Therapies
Footnotes
Author Contributions
JL and YZ conceived this study. JW, XM, and XW collected the data and wrote the original draft. GZ and HW contributed to the revision of the manuscript. All authors have read and approved the final manuscript.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Innovation Team and Talents Cultivation Program of National Administration of Traditional Chinese Medicine: ZYYCXTD-C-202205.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
