Abstract
Background:
Gastric cancer is a common cause of global mortality, with significant challenges during treatment due to side effects and complications. Traditional herbal medicine (THM) has emerged as a potential adjuvant therapy to enhance cancer treatment by reducing side effects and bolstering the immune response. This study conducted a meta-analysis to assess the efficacy and safety of THM as an adjuvant therapy in post-surgical gastric cancer patients.
Methods:
PubMed, Cochrane Library, EMBASE, CNKI, CiNii, KMBASE, KISS, OASIS, RISS, and ScienceON databases were searched from inception through December, 2021. The outcomes considered in this analysis encompassed tumor response, quality of life (QoL), side effects, and tumor markers. Additionally, a frequency analysis of the most commonly used herbs in the included studies was conducted. A total of 36 randomized controlled trials (RCTs) were included, and data were extracted according to study design. The analysis compared groups receiving chemotherapy alone with those receiving both chemotherapy and THM treatment.
Results:
The group receiving both chemotherapy and THM showed substantial improvement in tumor response compared to the chemotherapy-only control group (RR 1.25, 95% CI [1.09, 1.45]). QoL also significantly increased in the THM-treated group. Most drug adverse reactions displayed statistical significance, except for platelet reduction. Tumor markers CEA, CA19-9, and CA72-4 exhibited significant improvements, but CA125 did not. The 1, 2, and 3-year survival rates improved, with RR values of 1.08 (95% CI [1.02, 1.14]), 1.32 (95% CI [1.19, 1.47]), and 1.42 (95% CI [1.12, 1.79]) respectively. However, some publication bias was indicated.
Conclusion:
THM may offer potential benefits as a complementary approach to post-surgical anticancer therapy in gastric cancer patients. Improved tumor response, quality of life, and survival rates were reported. However, it is important to exercise caution due to the possibility of publication bias, and further research is needed to confirm these findings.
Registration:PROSPERO CRD 42022354133
Introduction
Gastric cancer (GC) is one of the most common cancers in the digestive system. According to the Global Cancer Observatory (GCO), in 2020, gastric cancer had 1 089 103 new cases and resulted in 768 793 deaths, making it the fourth leading cause of mortality and the fifth most common cancer in terms of incidence. Notably, its incidence in men is twice that of women. 1 The management of gastric cancer involves a comprehensive and multidisciplinary approach to diagnosis and treatment. This requires a carefully planned and coordinated combination of various treatment modalities, such as surgery, radiotherapy, chemotherapy, targeted therapy, endocrine therapy, immunotherapy, and interventional therapy. 2 Adjuvant chemotherapy has been shown to improve overall survival and disease-free survival compared to surgery alone.3 -6
While treatment guidelines vary by country, gastric cancer treatment generally involves surgery, radiation, and chemotherapy. However, surgery and chemotherapy for gastric cancer can cause patients to experience numerous side effects and complications,7,8 and patients can suffer from psychological symptoms related to tumor diagnosis, postoperative complications, and the side effects of treatment. 9 According to Bang et al’s 3 study, more than half of the patients receiving adjuvant cancer treatment after gastric cancer surgery reported experiencing side effects. These physiological and psychological changes can significantly lower the quality of life after surgery, and the side effects and complications still need to be resolved in clinical practice.
Traditional Herbal Medicine (THM) is widely used as a complementary and alternative therapy and can be helpful in the treatment of gastric cancer.2,10 As highlighted by Lu et al’s 11 study, THM is commonly used either on its own or in combination with chemotherapy at various stages of gastric cancer progression. Notably, THM is used to reduce the side effects of chemotherapy, assist in post-surgical recovery, and alleviate symptoms. 12 Liu et al’s 13 study indicates that the group treated with THM in conjunction with chemotherapy experienced fewer side effects from gastric cancer compared to those undergoing chemotherapy alone. Moreover, this study revealed that THM can bolster the immune response in cancer patients, promoting apoptosis and effectively halting the progression of gastric cancer. 13 Fu et al’s 14 study stated that THM effectively reduces toxicity and side effects caused by chemotherapy and improves clinical symptoms and quality of life. Consequently, THM alleviates symptoms in cancer patients and enhances the quality of life and the efficacy of anticancer treatment.
Despite the promising potential of THM in gastric cancer treatment, a significant gap exists in the current literature. Few studies have conducted a comprehensive and standardized review that integrates both THM and GC treatments. Hence, this study aims to provide clinical evidence via a meta-analysis, evaluating the efficacy and safety of combining adjuvant chemotherapy with herbal medicine in the treatment of post-surgical gastric cancer.
Methods
Study Inclusion Criteria
The inclusion criteria were limited to randomized controlled trials (RCTs) that enrolled participants diagnosed with stages I to III gastric cancer. These trials must have allocated patients into 2 distinct groups: an intervention group receiving both THM and adjuvant chemotherapy post-surgery, and a control group receiving adjuvant chemotherapy alone. The primary outcome for inclusion was specified as “Tumor Response,” while secondary outcomes included “Quality of Life (QoL),” “Adverse Drug Reactions (ADRs),” “Tumor Marker Levels,” and “Survival Rate.” Non-randomized trials, observational studies, case reports, and other non-randomized comparisons were excluded to ensure a high level of evidence and homogeneity within the analyzed data.
Objective and Design
The objective of this study is to conduct a meta-analysis to examine the effectiveness and safety of combining THM with adjuvant chemotherapy for the treatment of gastric cancer. The study was carried out in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). The study protocol has been registered on PROSPERO with the registration number CRD 42022354133.
Our approach has included a quantitative synthesis of data through meta-analysis from studies that have met our pre-defined eligibility criteria, specifically those providing sufficient quantitative data and having comparable outcome measures. We have performed meta-analysis when we have found at least 2 studies that are sufficiently homogenous in terms of participants, interventions, comparators, and outcomes, as per our predefined protocol thresholds.
Search Strategy
The literature search was conducted by utilizing international web databases including KMBASE, KISS, OASIS, RISS, ScienceON, EMBASE, Pubmed, CNKI, Cochrane Library, and CiNii. To ensure a comprehensive coverage of the literature, articles published in English, Chinese, Japanese, and Korean were included. The search period for all databases extended through June 2022. The searching strategy of the PubMed database, which was searched in the corresponding database, detailed search strategy is provided in Table S1 (Supplemental Material). This research solely focused on the electronic searches.
Selection of Studies
The search results were collected using the reference management program Endnote version 20 and duplicates were removed. 15 Two independent researchers (SDK and JHK) evaluated the titles and abstracts of the studies; and excluded those that did not meet the criteria. The full texts were then reviewed and studies meeting the criteria were included, after consultation between the 2 researchers. If there was a disagreement, the issue was resolved by a third researcher (DHK). Excluded studies were recorded with reasons.
Data Extraction
The data were extracted by 2 independent researchers (SDK and JHK). The extracted data included the first author, study period, year of publication, participants, comparison, interventions in treatment and control groups, outcomes, and adverse events. If any data were missing from the material, the group could discuss the issue among themselves and reach out to the first author by email.
Quality Assessment
To assess the quality of the selected literature data, we utilized the Cochrane Risk of Bias (ROB) tool. 16 Two researchers (SDK and JHK) used ROB to assess the risk of bias. The risk of trial bias was assessed through a total of 7 items: random sequence generation and allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. If there was a disagreement, the issue was resolved by a third researcher (DHK).
Building upon this foundation, our quality of evidence assessment applied the GRADE system to categorize evidence levels as high, moderate, low, or very low. RCTs initially offer high-quality evidence, which was then downgraded based on the presence of serious limitations like risk of bias, inconsistency, indirectness, and imprecision, and evaluated for potential publication bias. Upgrading of evidence was considered for factors such as a large effect size and dose-response relationship. The quality of evidence for each outcome was then summarized in a GRADE evidence profile, providing a clear and systematic assessment of the evidence strength and areas needing further research.
Statistical Analysis
Review Manager Software 5.4 was used to generate data results. 17 In this study, the risk ratio (RR) with 95% confidence interval (CI) was used to measure dichotomous data. For continuous data, 95% CI with mean differences (MD) were used when measuring treatment outcomes on the same scale. Heterogeneity was calculated using Higgins’ I2 in this study. An I2 value greater than 50% has been considered indicative of substantial heterogeneity. In cases of substantial heterogeneity, we have explored potential sources through subgroup analyses and sensitivity analyses, based on predefined study characteristics such as population demographics, different types of herbal medicines, and chemotherapy regimens. Should substantial heterogeneity have remained unexplained, findings have been cautiously interpreted, and we have provided a narrative synthesis discussing the plausible reasons for these discrepancies. The meta-analysis, if conducted, has utilized a random-effects model to account for any identified heterogeneity among the included studies.
Frequency Analysis
To identify the predominant herbal interventions in the treatment of gastric cancer, a frequency analysis was conducted on the included studies. The frequency analysis aimed to determine the most commonly used herbs in the studies included in the meta-analysis. This analysis was carried out by tallying the occurrences of various herbs across the studies.
Results
Study Selection
An initial search of 10 electronic databases yielded 3709 studies. After removing 483 duplicates, the remaining 3226 studies were evaluated based on their abstracts. This resulted in the further exclusion of 3129 studies, leaving 97 articles for full-text review. The eligibility criteria were then applied, a total of 61 studies were excluded for the following reasons: 14 studies included patients without a history of surgery or those who did not undergo surgery for gastric cancer, 4 studies had participants other than gastric cancer patients, 28 studies were not randomized controlled trials, and 15 studies had different study designs from the intended outcome. Consequently, a total of 36 randomized controlled trials were included in this analysis (Figure 1).
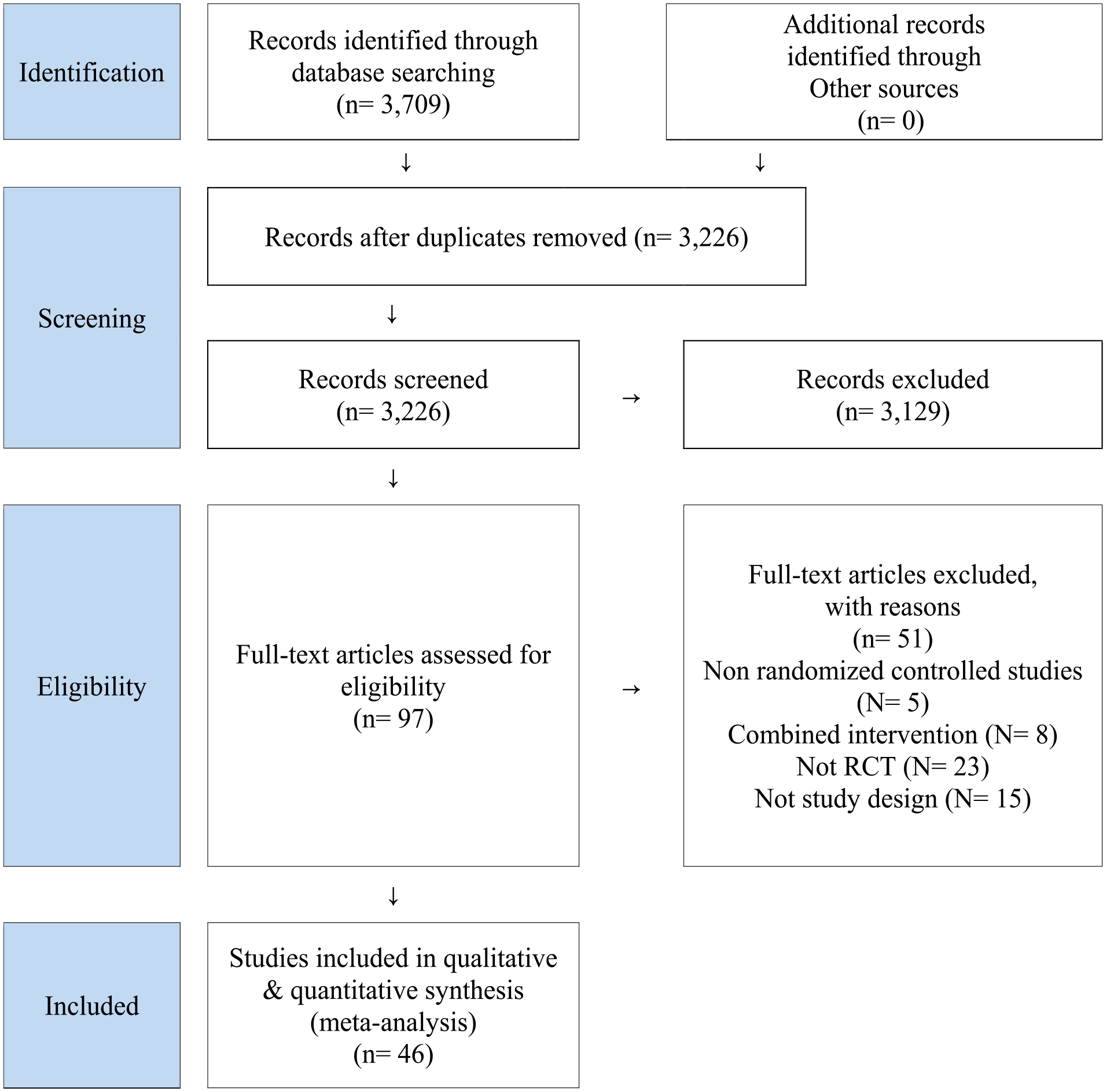
PRISMA flowchart of systematic review.
Study Characteristics
This meta-analysis includes 36 randomized controlled trials published up until 2021, with a total of 2176 participants diagnosed at different stages of gastric cancer, ranging from TNM stage Ⅰ to stage Ⅲ.18 -53 The experimental groups received chemotherapy combined with different TCM formulas as an adjuvant treatment, while the control groups received standard chemotherapy regimens, such as FOLFOX, XELOX, SOX, OLF, DC, and others. The treatment duration varied across the studies, ranging from 2 weeks to 12 months (Table 1). The primary outcomes evaluated in these studies included disease control rate (DCR), overall response rate (ORR). DCR is defined as the sum of complete response, partial response, and stable disease. This measure provides a comprehensive assessment of the control over the disease progression. ORR is calculated as the sum of complete response and partial response, offering a focused view on the effectiveness of the treatment in reducing tumor size and progression. The secondary outcomes were set as Karnofsky Performance Status (KPS), ADR, Tumor Marker Levels, and Survival rate. Each of these outcomes offers a unique perspective on the overall impact of the treatment, encompassing both the efficacy and safety profile of combining Traditional Herbal Medicine with chemotherapy in Gastric Cancer treatment.
Characteristics of Included Trials.

Abbreviations: FOLFOX, folinic acid, fluorouracil, and oxaliplatin; XELOX, capecitabine and oxaliplatin; OLF, oxaliplatin, leucovorin and fluorouracil; SOX, S-1(tegafur, gimeracil and oteracil) and oxaliplatin; DC, docetaxel and cyclophosphamide; OXA, oxaliplatin; TS, thymidylate; 5-FU, fluorouacil; CF, calcium folinate; L-OHP, oxaliplatin; DPF, docetaxel, cisplatin and fluorouracil; TX, taxotere (docetaxel); DCF, docetaxel, cisplatin and fluorouracil; FAM, fluorouracil, adriamycin (Doxorubicin) and mitomycin.
Risk of Bias in Included Trials
Based on the Cochrane risk of bias tool, the trials included in this review exhibited a moderate to high risk of bias due to insufficient methodological reporting (Figure 2). Most studies employed proper random sequence generation, while one study was considered to have a high risk of bias in this regard. 52 Allocation concealment information was not provided in 18 trials,19 -22,25-27,29,31,32,34,40,42,46,47,49,50,52 leading to an unclear risk of bias assessment. Blinding participants or personnel was unfeasible since THM was administered solely to the experimental group.

Risk of bias summary.
Detection bias was deemed to be at high risk in 20 studies,20 -22,24,26,29-32,34,35,37,42,43,46,47,49,50,52,53 and no relevant information was reported in 4 studies.19,40,41,44 Reporting bias was assessed as unclear risk in all studies due to the inability to locate the registered protocols. All studies were assessed to have a low risk of attrition and other biases (Figure 3).
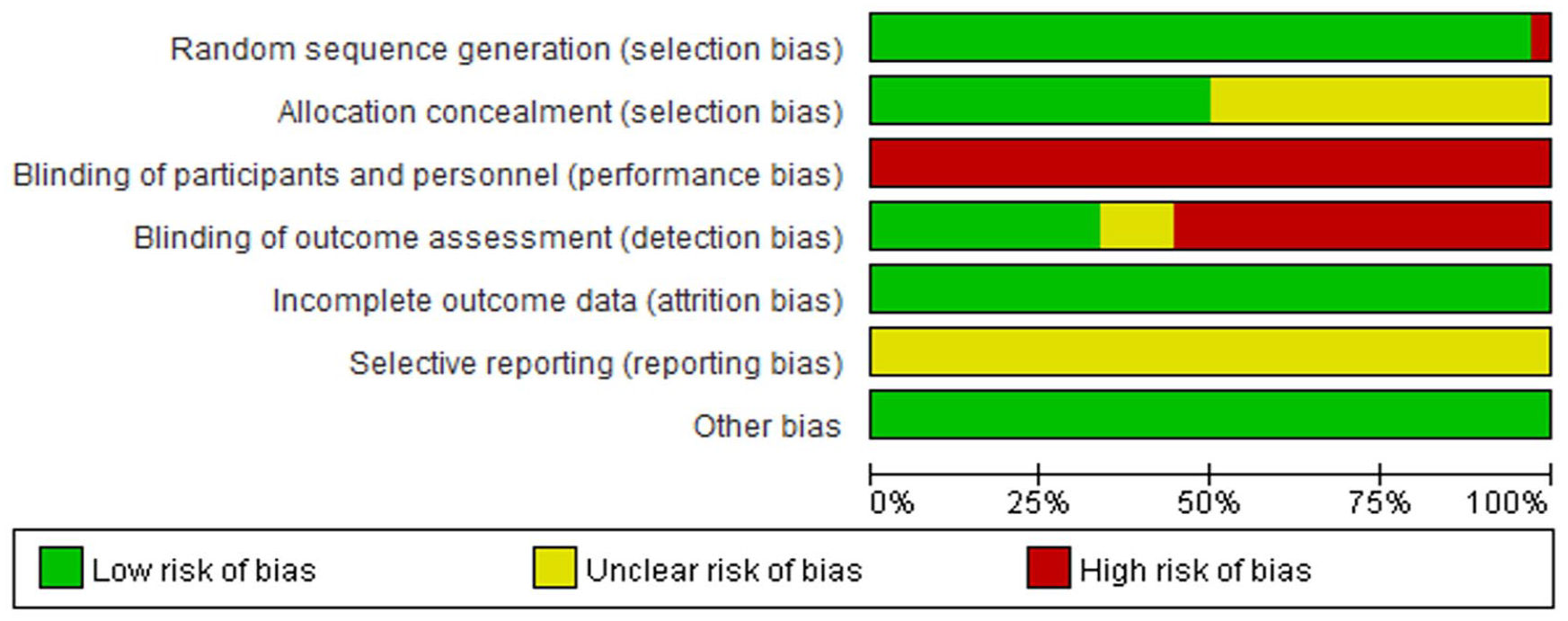
Risk of bias graph.
Primary Outcome
Tumor response assessment
A comprehensive review of 12 studies demonstrated a statistically significant increase in both DCR and ORR for those patients receiving a combination of THM and chemotherapy,21,23,29,32,34,37,39 -41,44,47,52 compared to those receiving chemotherapy alone (RR 1.25, 95% CI [1.09, 1.45], I2 = 79%, n = 835) and (RR 1.54, 95% CI [1.31, 1.81], I2 = 0%, n = 835, Figure 4). To address the substantial heterogeneity in the DCR outcomes (I2 = 79%), 3 studies were excluded due to their notably high risk ratios and wide confidence intervals, which were identified as potential outliers.23,47,52 The recalculated data from the remaining studies still demonstrated an enhancement in DCR due to THM (RR 1.18, 95% CI [1.07, 1.29], I2 = 42%, n = 646, Figure 5). However, the decrease in the I2 value to 42% suggests the initial heterogeneity may have been driven by reporting bias.

ORR of THM + chemotherapy.
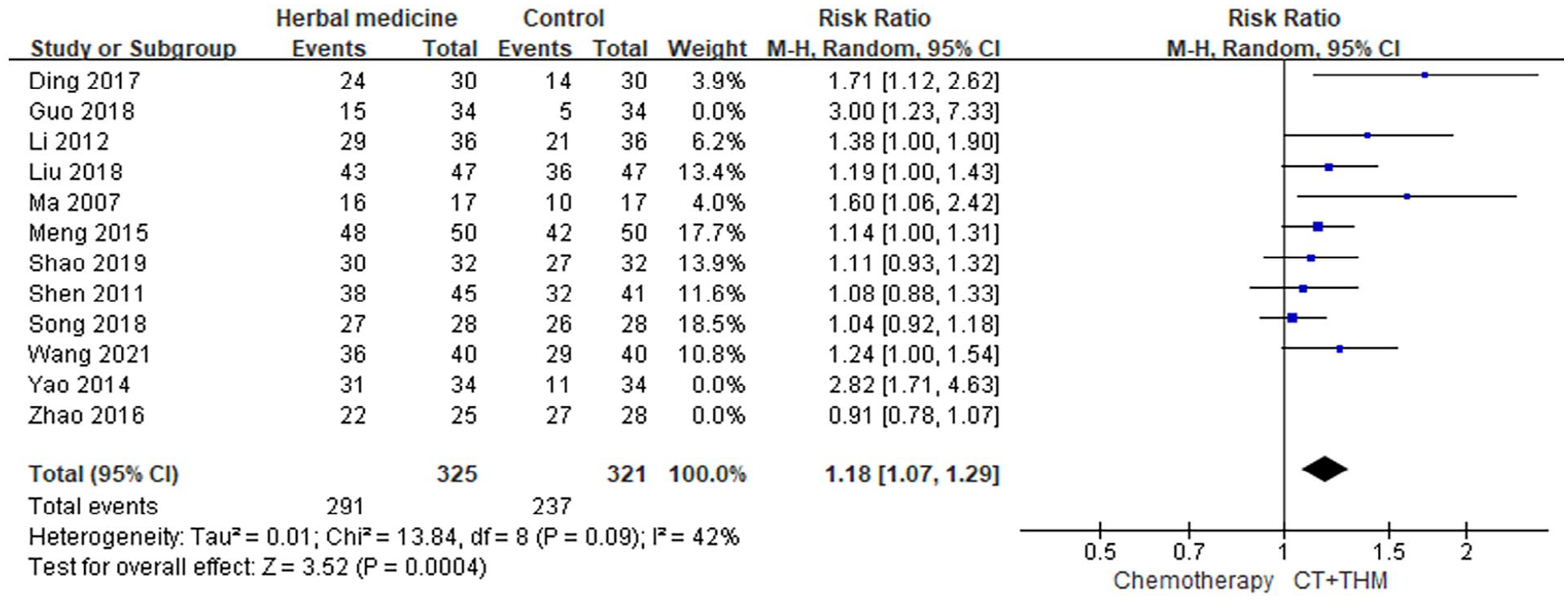
DCR of THM + chemotherapy.
Secondary Outcomes
Quality of life assessment
In line with the established criteria for evaluating KPS score enhancement, 14 studies measured QoL through KPS score improvement. A combined analysis of these 14 studies revealed a significantly greater rate of KPS score enhancement in the group receiving both THM and chemotherapy compared to the group undergoing chemotherapy alone. This difference was statistically significant (MD of 6.84, 95% CI [5.77, 7.92], I2 = 49%, n = 922, Figure 6).21,22,25,28,30,31,34,36,42,44,47,51 -53

KPS Score of THM + chemotherapy.
Adverse drug reactions assessment
The findings from the meta-analysis revealed that the group receiving both THM and chemotherapy exhibited a reduced likelihood of experiencing adverse drug reactions compared to the chemotherapy-only group. This included lower risks of leukocyte reduction (RR 0.62, 95% CI [0.50, 0.76], I2 = 49%, n = 1284, 16 trials),18,21,24,25,28,30,31,34,39,40,42 -44,47,50,51 platelet reduction (RR 0.86, 95% CI [0.72, 1.01], I2 = 10%, n = 1198, 16 trials),21,24,25,28,30 -32,34,39,40,42 -44,47,50,51 liver injury (RR 0.59, 95% CI [0.42, 0.82], I2 = 29%, n = 951, 13 trials),18,19,21,25,26,28,30,35,40,42,47,51 renal injury (RR 0.46, 95% CI [0.28, 0.75], I2 = 0%, n = 589, 7 trials),18,20,25,26,28,40,51 nausea and vomiting (RR 0.68, 95% CI [0.56, 0.84], I2 = 51%, n = 1038, 14 trials),24,25,28,30,31,34,40,42-45,47,50,51 diarrhea (RR 0.74, 95% CI [0.61, 0.88], I2 = 4%, n = 900, 12 trials),24,25,28,30,31,34,40,42,43,47,50,51 and neurotoxicity (RR 0.63, 95% CI [0.48, 0.84], I2 = 0%, n = 651, 9 trials).24,25,28,30,31,34,35,51,52 Except for platelet reduction (P = .07), all differences were statistically significant, with further details presented in Table 2.
Adverse Drug Reactions of THM + chemotherapy.

P < .01. ***P < .001.
Tumor marker level
In the THM plus chemotherapy group, there was a greater decrease in CEA compared to the chemotherapy-alone group, as demonstrated by the pooled results of 16 trials (MD −1.55, 95% CI [−2.23, −0.87], I2 = 51%, n = 967).19,22,26-28,30,31,34-36,42,45-47,51,52 Moreover, the pooled results of 11 trials revealed a higher CA19-9 decrease in the THM plus chemotherapy group than in the chemotherapy group (MD −2.50, 95% CI [−3.91, −1.09], I2 = 18%, n = 748).19,22,27,28,30,31,36,46,47,51,52 Conversely, the pooled results of 8 trials indicated no significant change in CA125 between the THM plus chemotherapy group and chemotherapy group (MD 0.12, 95%, CI [−1.47, 1.70], I2 = 59%, n = 596).18,19,22,27,28,35,46,52 Additionally, the pooled results of 6 trials showed a greater CA72-4 decrease in the THM plus chemotherapy group than in the chemotherapy group (MD −1.80, 95% CI [−2.59, −1.01], I2 = 43%, n = 393).22,28,30,31,35,52 The detailed information has been presented in Table 3.
Tumor Markers of THM + Chemotherapy.

P < .001.
One-year survival rate
The meta-analysis of 10 studies assessed the 1-year survival rate and found that THM plus chemotherapy group demonstrated a statistically significant improvement in the 1-year survival rate (RR 1.08, 95% CI [1.02, 1.14], I2 = 0%, n = 868, 10 trials, Figure 7).20,24,33,37,38,40,41,43,49,50 These findings provide strong evidence that the combination of THM and chemotherapy has a positive effect on the 1-year survival rate.

One-year survival rate of THM + chemotherapy.
Two-year survival rate
The meta-analysis of 8 studies indicated that the combination of THM and chemotherapy was effective in improving the 2-year survival rate (RR 1.32, 95% CI [1.19, 1.47], I2 = 0%, n = 712, 8 trials, Figure 8), providing robust evidence for the effectiveness of the THM and chemotherapy combination in enhancing the 2-year survival rate.
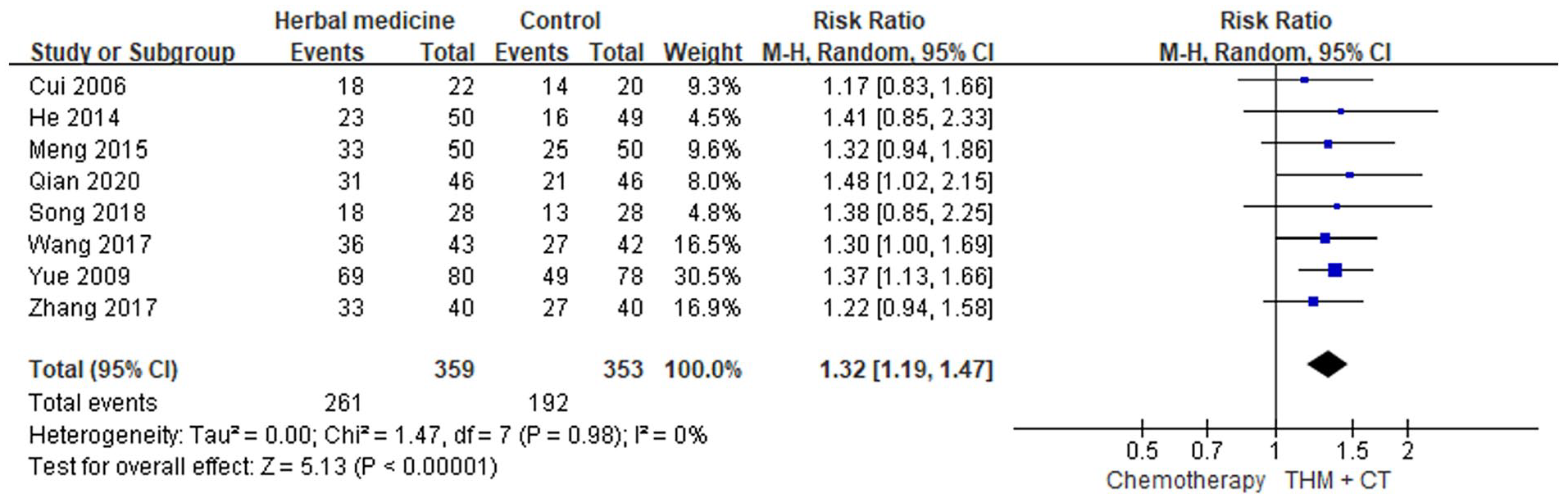
Two-year survival rate of THM + chemotherapy.
Three-year survival rate
The statistically significant improvement observed in the 3-year survival rate (RR 1.42, 95% CI [1.12, 1.79], I2 = 0%, n = 343, 3 trials, Figure 9) further reinforces the efficacy of combining THM and chemotherapy as a promising approach. These findings provide valuable insights into the potential long-term benefits of this treatment strategy in improving patient outcomes.

Three-year survival rate of THM + chemotherapy.
Publication Bias
To investigate publication bias, funnel plots were created using RR, MD, and 1/(standard error: SE) values extracted from trials that measured ORR, KPS score, and 1-year survival rate. The funnel plot analysis for the ORR study exhibited a symmetrical shape (Figure 10), indicating a balanced distribution of studies across a range of effect sizes and their corresponding precision measures. This symmetry suggests the absence of significant publication bias or small study effects, reinforcing the reliability and robustness of the study’s findings regarding ORR. However, the funnel plots generated based on the KPS score and 1-year survival rate displayed an asymmetrical distribution (Figures 11 and 12), suggesting the potential presence of publication bias in these analyses.
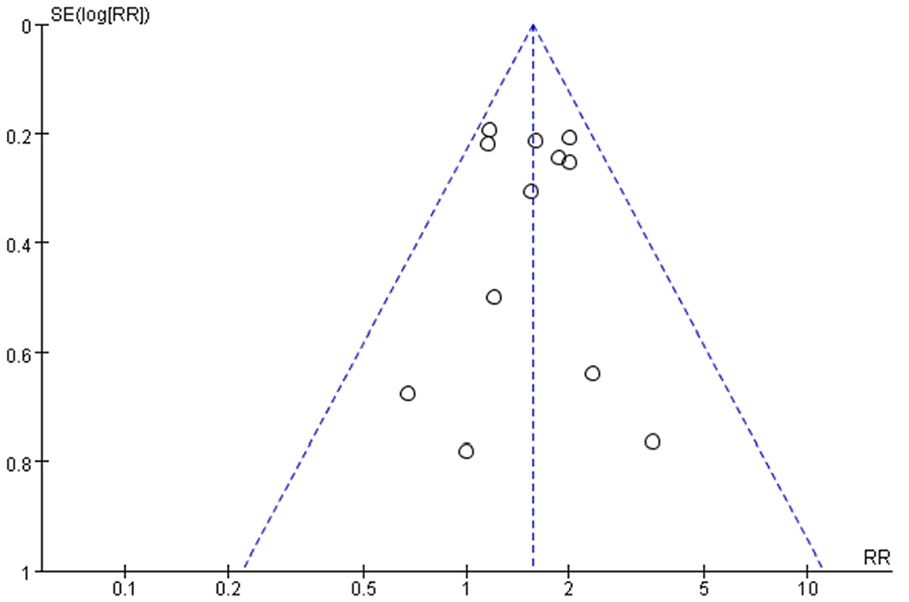
Funnel plot assessing publication bias (ORR in 12 trials).
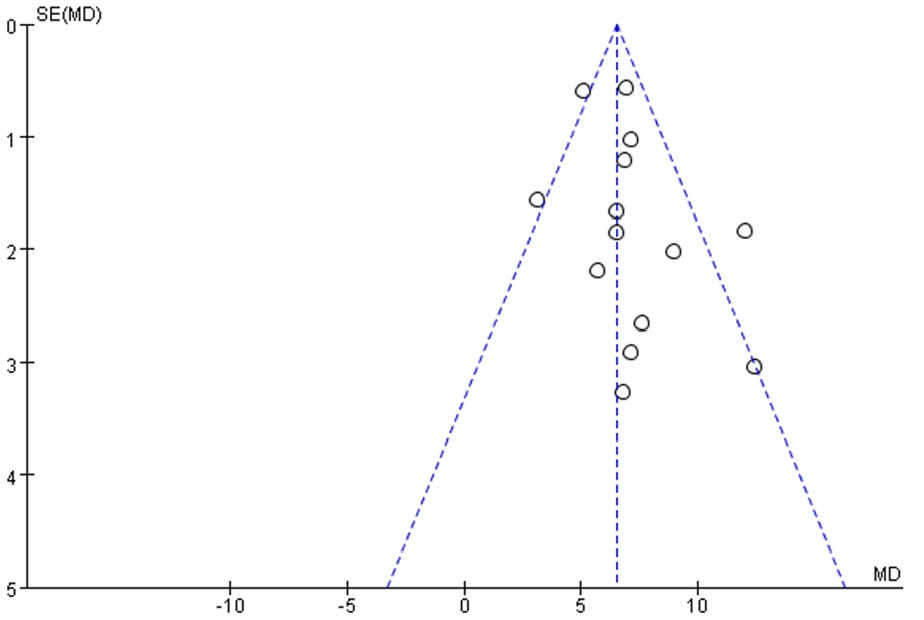
Funnel plot assessing publication bias (KPS in 14 trials).
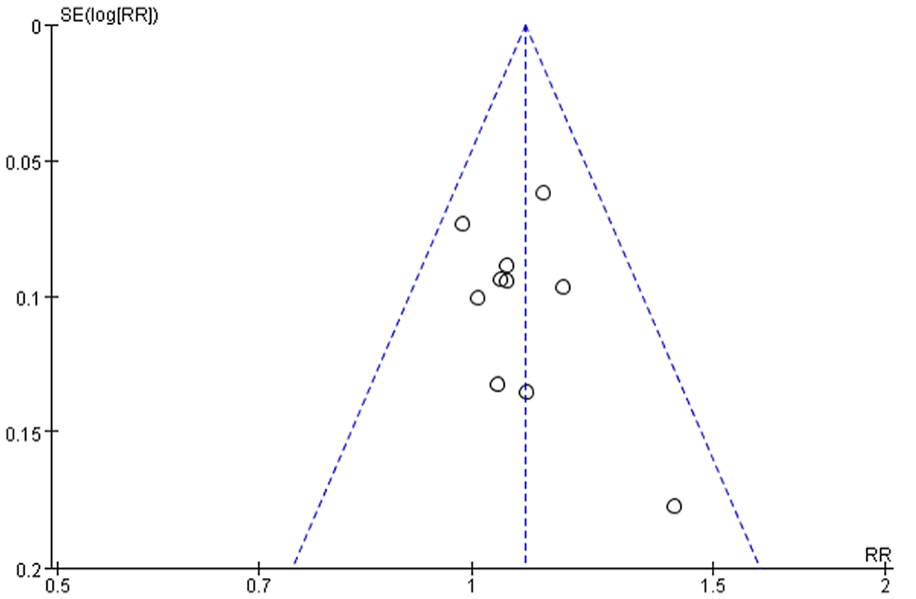
Funnel plot assessing publication bias (1-year survival rate in 10 trials).
Quality of Evidence Assessment
In the assessment of tumor response, the DCR and ORR were analyzed from 9 and 12 RCTs respectively, showing evidence of “Low” and “Moderate” certainty. Concerning the QoL, the KPC indicator from 14 RCTs demonstrated “Moderate” certainty. Adverse drug reactions showed varied results: leukocyte decrease, liver injury, nausea & vomiting, diarrhea, tumor marker levels (CEA, CA19-9, CA125, CA72-4) were all assessed with “Low” certainty, while platelet decrease, renal injury, and neurotoxicity held a “Moderate” certainty. For survival rate evaluations, the 1-, 2-, and 3-year survival rates all were categorized with “Moderate” certainty. The reasons for downgrading the evidence quality across these evaluations primarily included risk of bias and inconsistencies. Various outcomes also suffered from imprecision, often attributed to limited sample sizes. Detailed outcomes, effects, and absolute effects can be cross-referenced in the provided Table 4.
A Summary of Comprehensive Assessment.

Abbreviations: CI, confidence interval; MD, mean difference; RR, risk ratio; seriousᵃ, Moderate heterogeneity (30% ≤ I2 ≤ 75%); seriousᵇ, Wide confidence intervals; seriousᶜ, Low heterogeneity (I2 ≤ 30%).
Frequency Analysis
The frequency analysis of the most commonly used herbs in the included studies unveiled significant trends in herbal prescriptions for post-surgical gastric cancer therapy. Among the 122 herbs observed over 36 prescriptions, the top 10 herbs are presented in Table 5. Atractylodes macrocephala exhibited the highest frequency, present in 75.0% of the analyzed prescriptions. Subsequently, Poria cocos showed a frequency of 63.9%, while Glycyrrhiza uralensis, Citrus reticulata, Astragalus membranaceus, and Hedyotis diffusa all shared a frequency of 61.1% across the prescriptions. Codonopsis pilosula and Pinellia ternata were also frequently prescribed, each appearing in 58.3% of the prescriptions. Angelicae Sinensis Radix and Coix lacryma-jobi were similarly common, noted in 38.9% of the prescriptions. These findings provide insight into the prevalent herbal interventions employed in the prescriptions analyzed, showcasing a selection of herbs frequently prescribed in post-surgical gastric cancer therapy involving THM.
Herbal Medicine Frequencies in Post-Surgical Gastric Cancer Therapy.

Total prescription numbers, n = 36.
Discussion
This meta-analysis and systematic review investigated the efficacy and safety of combining THM with chemotherapy, specifically for GC, across 36 randomized controlled trials involving 2176 patients. A critical evaluation of the included studies was performed using the RoB tool, which helped in assessing the potential biases in these trials. We identified that the trials exhibited a moderate to high risk of bias due to gaps in methodological reporting. Notably, a significant number lacked details on allocation concealment, and blinding of participants was challenging due to the nature of the intervention with THM. Additionally, a high risk of detection bias was observed in many studies, and the reporting bias remained uncertain for all included trials due to the absence of registered protocols. Primary outcomes revealed a positive association between THM and tumor response, with both DCR and ORR indicating statistically significant improvements. The RoB assessments underscore the need for further rigorously designed RCTs in this domain to strengthen the evidence base. A re-examination, after excluding 3 studies, resolved initial heterogeneity in DCR findings. Regarding the outcomes reported in this study, it is important to note that the outcome of “cancer recurrence prevention rate” was included in our initial registration record but was not reported in the final manuscript. 54 This decision was due to the observed inconsistencies in how this outcome was measured across various studies, which could potentially compromise the reliability and comparability of our findings. Conversely, the outcome of “tumor markers,” while not initially included in the registration record, was later reported in our study. This inclusion was driven by a deeper analysis and understanding of the review’s objectives, where we recognized the significance of tumor markers in providing valuable insights into treatment efficacy and cancer progression. 55 Secondary outcomes demonstrated that patients receiving both THM and chemotherapy had improved QoL scores and fewer adverse drug reactions compared to those on chemotherapy alone. Notable reductions were observed in specific reactions such as leukocyte and platelet reduction, liver and renal injuries, nausea, vomiting, diarrhea, and neurotoxicity, though platelet reduction was an exception with no significant statistical difference. One aspect that demands further discussion is the reporting of tumor markers. Our analysis underscores the importance of these biomarkers as they often serve as critical indicators of tumor burden and treatment response. Tumor markers can provide valuable insights into the pathophysiology of cancer, guide therapy decisions, and help in monitoring disease progression or response to treatment. 55 In this review, we observed that combining THM with chemotherapy led to significant reductions in certain markers, except CA125. Detailed and consistent reporting of tumor markers would contribute substantially to the understanding of the mechanisms through which THM may potentiate the efficacy of chemotherapy. Additionally, survival rate analyses showcased remarkable enhancements in 1-, 2-, and 3-year survival rates with the THM and chemotherapy combination. To further validate our findings, particularly in light of the wide range of treatment durations in the included studies, we conducted a sensitivity analysis. This analysis confirmed that the duration of herbal medication, whether short-term or long-term, did not significantly influence the overall effectiveness of the treatments. This consistent finding across various studies indicates that differences in treatment durations did not introduce notable biases or affect the results, thus affirming the robustness and reliability of our conclusions, despite the diversity in treatment lengths. However, research is needed to determine whether the size of the effect varies depending on the composition and administration period of herbal medicine.
Given these promising results, our research may offer new insights into the global challenge of treating gastric cancer, a disease fraught with complex risk factors and significant impacts on patients’ quality of life as detailed above. Gastric cancer is one of the commonly diagnosed diseases worldwide and is physically and psychologically distressing. 1 Risk factors for gastric cancer include unavoidable variables such as race, age, and gender, as well as other risk factors such as infection with Helicobacter pylori bacteria, diets high in nitrates and nitrites, and smoking. Additionally, factors like genetics, medical history, and pernicious anemia also contribute to the onset of stomach cancer. 56 Gastric cancer is commonly treated through surgery, radiation, and chemotherapy, but recurrence and side effects often occur.7,8 Furthermore, patients can suffer from psychological symptoms related to tumor diagnosis, postoperative complications, and the side effects of treatment. 9 Chemotherapeutic drugs commonly used in gastric cancer treatment, such as Oxaliplatin, typically cause side effects like nausea, vomiting, diarrhea, and loss of appetite. 57 Also, adjuvant chemotherapy does not unequivocally improve survival rates. 58 The treatment of gastric cancer, including surgery and chemotherapy, still involves high costs, is painful, imposes a significant burden on society, and most importantly, greatly reduces the quality of life for patients. Therefore, the treatment of gastric cancer remains an important challenge. 59
One of the most researched options for gastric cancer is herbal medicine. THM can help in cancer treatment by reducing the side effects of conventional treatments or improving the quality of life. 60 According to a meta-analysis of several RCTs, THM has been shown to improve cancer-related symptoms, enhance quality of life, and reduce the side effects of chemotherapy. This demonstrates the effectiveness of combining anticancer drugs with THM in the treatment of gastric cancer. 61 According to the tumor response results of this paper, in the RCTs used for DCR and ORR research, medicines such as Astragalus membranaceus, Atractylodes macrocephala koidz, Radix angelica sinensis, Poria cocos, and Panax ginseng were widely used. Among them, Buzhong Yiqi decoction was used as an intervention in 3 studies. In the study by Xu, 62 it was found that Buzhong Yiqi decoction promotes the proliferation of T lymphocytes, strengthens immunity, and prevents cancer cells from evading immune responses by reducing the expression of PD-1 and PD-L1. It also inhibits the expression of PD-L1 by suppressing the PI3K/AKT pathway. In the study 32 that combined Buzhong Yiqi decoction with adjuvant chemotherapy, the OFL anticancer regimen used was a combination of oxaliplatin, leucovorin, and fluorouracil. Oxaliplatin in this anticancer drug promotes apoptosis of cancer cells, and fluorouracil interferes with the synthesis components of DNA and RNA in cancer cells. Leucovorin is used to enhance the effect of fluorouracil. 63 Although the roles of OFL and Buzhong Yiqi decoction are different, from this perspective, it is considered that some anticancer drugs interact with the mechanism of THM and show better results than single adjuvant chemotherapy. Not only Buzhong Yiqi decoction, but also other THM with similar properties are being studied. In a study by Li, 64 Yiqi Jianpi Huaji decoction, which includes herbal ingredients such as Astragalus membranaceus, Atractylodes macrocephala koidz, Radix angelica sinensis andCodonopsis pilosula, was found to inhibit cancer cell proliferation and help overcome multiple drug resistance. Chemotherapeutic drugs like fluorouracil inhibit the proliferation of cancer cells, promote cell death, interfere with the synthesis of DNA and RNA, and suppress the tumor microenvironment. Therefore, the use of THMs may help to prevent the progression of cancer and enhance the therapeutic effects in patients with gastric cancer. Similar results are being demonstrated in other types of cancer that use similar chemotherapeutic agents. It has been reported that combining THM with chemotherapy (combined with cisplatin and paclitaxel, gemcitabine, vinorelbine, etoposide) in the treatment of non-small cell lung cancer (NSCLC) is more effective. 65 Compared to chemotherapy treatment alone, combining THM with chemotherapy improved QoL, clinical efficacy, KPS scores, and reduced toxicity (such as leukopenia, hemoglobin reduction, thrombocytopenia, myelosuppression, diarrhea, nausea and vomiting, and liver damage).
The findings from this systematic review align with the findings from a previously presented study.60,61,66 A frequency analysis conducted within this review highlighted prevalent herbal medicines used in GC treatment, offering a deeper understanding of herbal medicine’s role in GC therapeutic strategies. Despite the evident benefits of combining THM with adjuvant chemotherapy for GC, there remain knowledge gaps in THM’s specifics, like discerning essential components and probing potential interactions between herbal medicines and standard treatments. 67 With the varied nature of THM prescriptions, a meticulous evaluation of individual prescriptions is crucial. The absence of serious side effects from the combination therapy underscores the potential of this synergistic treatment. This study underscores the need for continued research to fathom the complex interactions between adjuvant chemotherapy and THM, aiming to identify specific elements boosting THM’s therapeutic advantages. In summation, the combined approach of THM and chemotherapy emerges as a promising, effective, and safe therapeutic pathway, potentially redefining future cancer treatments.
Limitations
This study has some limitations. Firstly, blinding participants or personnel was unfeasible since THM was administered solely to the experimental group. The absence of blinding icould introduce placebo effects, especially in subjective areas like quality of life. Secondly, there were many types of chemotherapy, making it difficult to perform subgroup analysis with the same chemotherapeutic agent. Thirdly, the THM of the studies was diverse, which may lead to variation. Finally, the funnel plot showed unbalanced studies, indicating the possibility of publication bias. Furthermore, all studies included in our analysis were published in China; there is a possibility of geographical publication bias which could affect the outcomes, as findings or perspectives from other regions or languages might vary. However, the meta-analysis results indicate that THM is an effective treatment method for gastric cancer, as it improves patients’ QoL, reduces side effects, and helps prevent the progression of GC. Overall, integrating THM into gastric cancer treatment regimens provides a promising strategy to enhance treatment outcomes and improve patient quality of life. However, more comprehensive and standardized research is needed for broader clinical application.
Conclusions
The systematic review of studies on the combined use of adjuvant chemotherapy and THM in gastric cancer yields promising results. The combined therapy has shown significant improvements in tumor response, quality of life, and survival rates of patients. Furthermore, THM seems to synergize with adjuvant chemotherapy drugs to enhance their anticancer effects while also mitigating the side effects such as leukocyte reduction, liver and renal injuries, nausea, vomiting, diarrhea, and neurotoxicity. Tumor markers CEA, CA19-9, and CA72-4 displayed statistically significant results in the improvement of the quality of life and survival rates. However, the tumor marker CA125 and platelet reduction were not found to be statistically significant. Overall, the review suggests that the combination of adjuvant chemotherapy and THM could potentially be an effective treatment method for gastric cancer. However, considering the methodological limitations in the included studies, further well-designed research, particularly with placebo controls and more rigorous blinding and allocation concealment, is crucial to confirm these findings.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231226256 – Supplemental material for Comprehensive Evaluation of Traditional Herbal Medicine Combined With Adjuvant Chemotherapy on Post-Surgical Gastric Cancer: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ict-10.1177_15347354231226256 for Comprehensive Evaluation of Traditional Herbal Medicine Combined With Adjuvant Chemotherapy on Post-Surgical Gastric Cancer: A Systematic Review and Meta-Analysis by Soo-Dam Kim, Jong-Hee Kim, Dong-Hyun Kim, Ji-Hye Park, Yabin Gong, Chengbing Sun, Hwa-Seung Yoo and So-Jung Park in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HF20C0204 and HF23C0093).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
