Abstract
Solanum lycopersicum L. Moench (Tomato) is a rich source of bioactive compounds. This study investigated the anticancer potential of S. lycopersicum roots methanol extract (TMESLR) and their nanocrystals (TMESLR-NCs) against breast (MCF-7), hepatocellular (HepG2), and colon (Caco-2) cancer cell lines, for the first time. TMESLR exhibited significant cytotoxicity against all 3 cell lines, with the nanocrystals demonstrating enhanced activity, Caco-2, MCF-7, and HepG2 cells with IC50 values of 9.69 ± 0.6, 12.52 ± 0.58, and 14.34 ± 0.62 µg/mL, respectively. Whereas, the prepared TMESLR-NCs displayed significantly the highest cytostatic potential against Caco-2 with IC50 value of 5.733 ± 0.29 µg/mL. Metabolomic profiling revealed
Introduction
Cancer is the second greatest cause of death globally and a serious public health concern. 1 It is prompted by abnormal processing of genetic information, due to mutations affecting tumor suppressor genes and oncogenes, or altered epigenetic pathways resulting in chromatin structural abnormalities that are either localized or global. 2 The dysregulated expression of several histone methyltransferases is a major cause of changes in the epigenetic landscapes of cancer cells. The pathogenesis of colorectal, breast, esophageal squamous cell carcinoma, hepatocellular carcinoma, and melanoma has been associated with changes in the expression or activity of G9a, Setdb1, Smyd2, or PR-SET7 methyltransferases. 3
Interestingly, the innovative and developing field of nanobiotechnology is tremendously useful in the biomedical industry. Drug nanocrystals are molecular assemblies that can be combined to generate the drug in a crystalline form enclosed in a thin stabilizer layer. Nanocrystals technology is a remarkable alternative to the existing nanocarrier drug delivery mechanisms in enhancing 4 the bioavailability of medications which are not freely soluble in water. 5 Because of their small size (350-500 nm), they usually exhibit noteworthy biological and physicochemical activities that are different from those of larger particles. 6 They have exceptionally shown significant potential in cancer treatments, 7 orthopedics, 8 and dentistry. 9 Nanocrystal formulation can be used to improve drug delivery, targeting and bioavailability. It is possible to administer the drug orally or intravenously and the limited carrier, which mostly consists of a thin layer of surfactant may greatly reduce any toxicity. 10
Natural products have been evidenced as a source for developing new and effective therapeutic agents. Modern equipment in isolation, structure elucidation and identification of natural products, as well as the use of updated software and databases, give a great opportunity in screening natural products as forceful lead molecules. This has evolutionally confirmed their paramount role in drug discovery.11 -15
Likewise, S. lycopersicum, belonging to family Solanaceae, has shown powerful antioxidant efficacy in addition to cardiovascular protection, anticancer, anti-inflammatory, antimicrobial, antiviral, neuroprotection, antidiabetic, radioprotective and gut modulating activities. 16 The main reason for this biological diversity was attributed to the active metabolites, including caretonoids, alkaloids, flavonoids, and steroidal derivatives. 16 Furthermore, various secondary metabolites comprising flavonoids and phenolic acids, in addition to lycopene and β-carotene are abundant in S. lycopersicum seeds and roots. Tomato is a model vegetable crop with a vast array of valuable phytochemicals and biological uses. 16
In previous investigations, the seeds, fruits as well as the leaves of S. lycopersicum were extensively studied, Whereas the roots, that are considered as plant by-products, are underexplored parts. Therefore, the present research work aimed to investigate the cytotoxic efficacy of the TMESLR and TMESLR-NCs against 3 distinct cancer cell lines (Caco-2, MCF-7, and HepG2). Moreover, the TMESLR chemical profile was investigated through metabolomic analysis. Finally, the study involved protein-protein interaction network construction, gene ontology analysis as well as molecular docking for the dereplicated metabolites, with the purpose to comprehend the gene functions and molecular pathways associated with the bioactive compounds, as potential therapeutic agents against cancer.
Materials and Methods
Plant Material
In March 2022, 500 g of the fresh S. lycopersicum roots were taken from a farm in Maghagha city, Minia, Egypt, with the owner’s consent. Prof. Nasser Barakat (Department of Botany and Microbiology, Faculty of Science, Minia University) identified the plant.
Chemicals and Reagents
Ethanol, Formic acid (Merck, Germany), petroleum ether (Merck, Germany), methanol (99.8%) and DMSO (El-Nasr Company for Pharmaceuticals and Chemicals, Egypt), Insulin, acetonitrile, and 1% penicillin-streptomycin, Staurosporine® (Sigma-Aldrich, Germany), 10% FBS (Hyclone, USA), and DMEM high glucose (Invitrogen/Life Technologies, USA).
Extraction
The collected S. lycopersicum roots were dried in shade for 2 weeks. Afterward, they were grinded into fine powder resulting in a total amount of 55 g. The resulted fine powder was extracted using 99.8% methanol (1 L, 3×, 2 weeks interval). TMESLR was then evaporated using rotary evaporator (Heidolph®, Germany). The yielded viscous pale green TMESLR (5 g). It was kept in the refrigerator until further processing.
Cytotoxic Activity Assay
The antiproliferative potential of TMESLR and their prepared nanocrystals was evaluated, implementing the MTT assay.17,18 The cancer cell lines utilized in this test were previously obtained from the American Type Culture Collection (Manassas, VA, USA). They comprised Caco-2 (RRID: CVCL_0025), MCF-7 (RRID: CVCL_0031) and HepG2 (RRID: CVCL_0027) cell lines. First, the cells were cultured at 37°C and 5% CO2 in DMEM high glucose (Invitrogen/Life Technologies, USA) with 10% FBS (Hyclone, USA), 1% penicillin-streptomycin, and 10 mg/mL of insulin. After that, they were transferred to 96-well plates and incubated for the entire night at densities of 2.2 and 104 cells/cm2. Subsequently, the growing cells were exposed to TMESLR at diverse concentrations (20, 30, 40, 50, and 60 mg/mL) dissolved in DMSO. The following day, the cell viability was evaluated using the MTT assay, as formerly described by Hamed et al. 17
Preparation of TMESLR-NCs
TMESLR-NCs were prepared by dissolving a specified amount of TMESLR in absolute ethanol and petroleum ether mixture 25:75 ratio. The final amount achieved 50 mg/5 mL solution was well sonicated in an ultrasonicate bath at a frequency of 50 kHz (Branson® Ultrasonic Bath,). Tween 80 surfactant (the reported toxicity effect of tween 80 according to previous literature is 1 mg/mL while the concentration used in this formulation is 0.02 mg/mL that had not cytotoxic effect) 19 was added. After that, the mixture was stirred for 15 min at 1000 rpm. For solvent evaporation, the resultant solution was put on a BUCHI Rotavapor™ R-300 rotary evaporator. The resultant powder was kept at −20°C after being collected as TMESLR-NCs.7 -9
Particle Size (nm) and Size Distribution
Photon correlation spectroscopy was utilized to ascertain the size distribution and particle size of the prepared particles in terms of average volume diameters and polydispersity index. Dynamic Light Scattering (DLS) particle size analyzer (Zetasizer Nano ZN, Malvern Panalytical Ltd, UK) was used at a fixed angle of 173° at 25°C. Three examinations of the sample were conducted. 5
Scanning Electron Microscopy (SEM)
High-resolution SEM was performed to investigate the nanocrystals morphology. The samples were diluted (1:10) in ultrapure water, then 20 µL of slurries were spread on amorphous polycarbonate grids and left to dry at 25°C. They were extra dried using CO2, sputter coated with gold in a metallizer and examined under a SEM with accelerating voltage at 200 kV. 20
The produced nanocrystals’ morphology was examined using a SEM device (SEM, TESCAN, Warrendale, PA). This was accomplished by placing the nanoparticle powder on stubs and covering them with a layer of gold.
LC-MS Metabolomic Analysis
A 6530 Q-TOF LC-MS (Agilent Technologies, Japan) equipped with an autosampler (G7129A), a Quat pump (G7104C), and a Column Comp (G7116A) for chromatographic separation were used at the Faculty of Pharmacy, Fayoum University, to perform metabolomic profiling of TMESLR. An Agilent Technologies Zorbax RP-18 column (150 mm × 3 mm, dp = 2.7 μm) was used to separate the analytes. The mass spectra were obtained using ESI in both positive and negative ionization modes with a capillary voltage of 4500 V. They were captured between 50 and 3000 m/z. The temperature of the drying gas was 200°C, and the flow rate was 8 mL/min. The collision energy was set at 10 V, while the fragmentator and skimmer voltages were set at 130 and 65 V, respectively. The Phenomenex Kinetex 2.6 mm XB-C18 150 mm × 4.6 mm column, which was maintained at 30°C and connected to a guard column, was filled with 10 mL of samples (1 mg/mL in methanol). For the mobile phase, a mixture of acetonitrile (B) and LC-MS grade water (A) with 0.1% formic acid each was used. The gradient elution proceeded from 5% to 20% B in 2 min, 20% to 98% B in 18 min, 98% B in 5 min, and finally 98% to 5% B in 2 min, all while maintaining a flow rate of 500 µL/min. Capillary temperature (320°C), spray voltage (+3.5 or −2.7 kV), sheath gas (57.50 Pa), sweep gas (3.25 Pa), auxiliary gas (16.25 Pa), probe heater (462.50°C), AGC target (1e6), S-Lens RF (50 cm) resolution (70.000), and microscans (1) were the MS parameters used for the HPLC-HESI-HRMS analysis. The raw data was converted into positive and negative files in the mz/mL format using ProteoWizard after a variance analysis of the MS data was obtained using Mzmine 2.12. Lastly, metabolite identification was accomplished by referring to the metabolite database (METLIN 2020) and Dictionary of Natural Products (DNP 2020) databases.21,22
In Silico Study
Protein-Protein Interaction (PPI) Network Analysis
To explore the interactions between the bioactive secondary metabolites tentatively identified in our study and their protein targets, we utilized the STITCH database. 23 This platform facilitated the investigation of compound-protein interactions, integrating data from sources such as GEO and PharmGKB. This study was able to better understand the molecular mechanisms behind the metabolites effects by mapping these interactions, which also highlighted the metabolites’ potential as therapeutic agents in the treatment of cancer. 24 To better recognize the cytotoxic and anticancer effects of the bioactive compounds identified in TMESLR, the STRING database was utilized for constructing (PPI) networks. 25 Only interactions with a confidence score above 0.4 were included to ensure robustness and reliability. The networks were analyzed and visualized using Cytoscape software, with the CytoHubba plugin was employed to identify central molecular targets relevant to anticancer activity. 26 This analysis highlighted critical proteins and molecular pathways, providing insights into the therapeutic potential of these compounds for cancer management.
Gene Ontology (GO) and KEGG Pathway Analysis
Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were performed in order to obtain a better understanding of the molecular mechanisms linked to the cytotoxic effects of TMESLR compounds. These analyses elucidated 3 critical dimensions: Biological Processes (BP), focusing on cancer progression; Cellular Components (CC), pinpointing the loci of activity within cells, and Molecular Functions (MF), detailing specific molecular interactions. The Shiny GO platform (v0.80.) was utilized for this analysis, applying a False Discovery Rate (FDR) threshold of less than 0.05 to ensure statistical significance. 27 Results were visualized using enrichment bubble plots created by SRplot (v1.0.). 28 This comprehensive approach provided valuable insights into the gene functions and pathways associated with the bioactive compounds, underscoring their potential as therapeutic agents against cancer.
Molecular Docking Studies
The binding relationships between bioactive tentatively secondary metabolites from TMESLR and important proteins implicated in cancer processes were examined using molecular docking simulations. These investigations sought to investigate putative molecular anticancer mechanisms and validate the projected molecular targets. The RCSB Protein Data Bank (https://www.rcsb.org/) provided pertinent protein structures, preprocessed to remove non-essential molecules, and optimized with necessary modifications such as the addition of hydrogen atoms and charges. Docking experiments were conducted using the Discovery Studio Client platform (v.16.), 29 revealing promising binding interactions between the tentatively identified compounds and target proteins involved in cancer cell proliferation and survival. This analysis provided a foundation for further exploration of these compounds as potential anticancer agents.
Statistical Analyses
The mean ± standard error of mean was used to display the data. Tukey-Kramar post-analysis testing is performed after one-way analysis of variance (ANOVA). Graph Pad Prism 7 (Graph Pad Software, San Diego, California, USA) was used for statistical calculations. The results were estimated significant when P < 0.05.
Results
Cytotoxic Activity Assay
The MTT viability assay was used to examine the cytotoxic activity of TMESLR, with Staurosporine®, an anticancer medication, serving as the positive control. Several cancer cell lines (Caco-2, MCF-7, and HepG2) were used in this assay. The findings demonstrated that TMESLR had a significant level of inhibitory activity against Caco-2, MCF-7, and HepG2 cells, with respective IC 50 values of 9.69 ± 0.6, 12.52 ± 0.58, and 14.34 ± 0.62 µg/mL. While, the TMESLR-NCs showed higher cytotoxic activity especially against Caco-2, and HepG2 with IC50 value of 5.733 ± 0.29, and 12.08 ± 0.56 µg/mL, respectively, demonstrating an enhanced cytotoxic activity for the TMESLR. While, the growth of MCF-7 cell line was inhibited at IC50 value of 20.91 ± 1.22 µg/mL as described in Table 1.
Cytotoxic activities of TMESLR, and its nanocrystals.
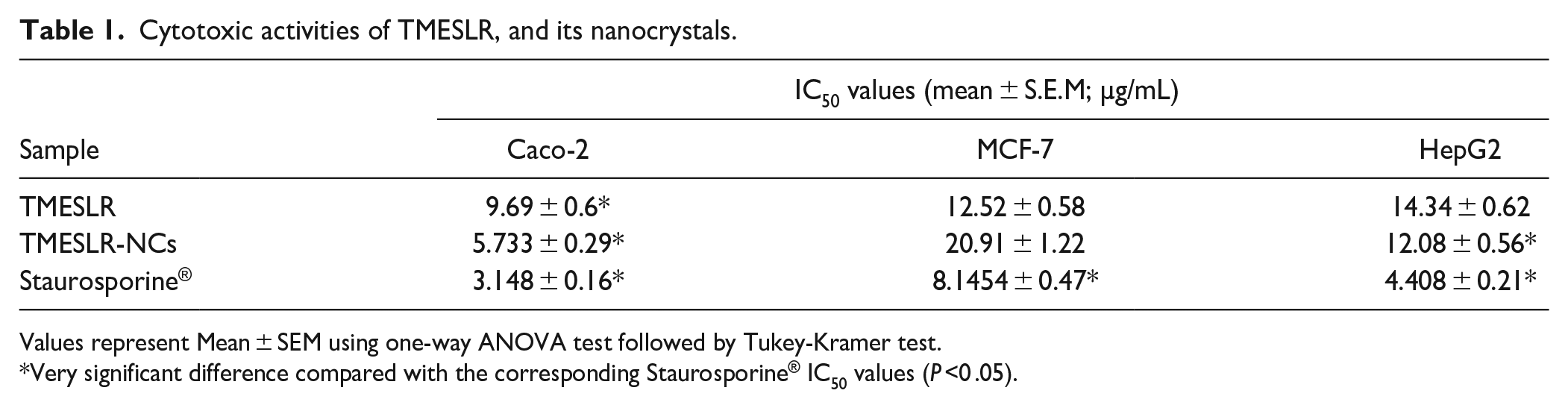
Values represent Mean ± SEM using one-way ANOVA test followed by Tukey-Kramer test.
Very significant difference compared with the corresponding Staurosporine® IC50 values (P ˂0 .05).
Characterization of Nanocrystals
The TMESLR-NCs displayed a mean particle size of 570 ± 25 nm and a poly dispersity index value of 0.2 to 0.5 indicating a narrow size distribution. The data of particle size distribution relative to their PI were presented in Figure 1 that enhance the bioavailability, in addition to zeta potential −23 ± 4.9 mV indicating high stability.30,31,53 SEM results Figure 2 approved different particle sizes distribution and showed that the nanocrystals had grouped quasispheroidal within the sample. 32

Mean particle size (left) and zeta potential of nanocrystals suspensions (right).

SEM images of TMESLR nanocrystals with power of magnification (15 KV×3.500).
Metabolomic Study
The metabolomic profile of TMESLR revealed different varieties of chemical classes. Supplemental Table S1 as well as Figures 3-5 are illustrating the secondary metabolites that were tentatively identified through metabolic profiling of TMESLR utilizing the untargeted HPLC-HESI-HRMS metabolomics technique. In reference to (DNP 2020) and (METLIN 2020) databases,21,22 the mass ion peak at m/z 165.0792 [M+H]+ for the anticipated molecular formula C10H16 was dereplicated as coumaric acid (

Chemical structures of the tentatively characterized metabolites from TMESLR.

Positive total ion chromatogram of TMESLR.

Negative total ion chromatogram of TMESLR.
In Silico Studies
Identifying Therapeutic Targets for Cancer Treatment
Protein targets were identified using NCBI-GEO and PharmGKB databases to ensure relevance to Caco-2, MCF-7, and HepG2 cancer models.40,41 NCBI-GEO datasets with at least 50 tumor and 50 normal samples were selected, and differential gene expression analysis was performed using GEO2R and the limma package, applying a cutoff of log2FC ≥ ±1.5, and FDR < 0.05. 42 Only genes consistently altered across at least 3 independent datasets were included. PharmGKB was used to filter cancer-related protein targets based on gene-drug interactions, prioritizing those involved in apoptosis, p53 signaling, TNF, NF-κB, and epigenetic regulation (eg, HDAC2, CASP3, BCL2, FASLG, and TNF). The final 73 (Supplemental Table S2) selected protein targets were mapped using STRING for protein-protein interaction (PPI) analysis, 43 followed by Gene Ontology (GO) enrichment and molecular docking studies to assess their interactions with bioactive compounds tentatively identified in TMESLR. 44
STITCH database analysis of TMESLR compounds and cancer targets
To explore the interactions between the key bioactive compounds tentatively identified from TMESLR; such as coumaric acid (

STITCH analysis illustrating the interactions between key protein targets associated with cancer and the principal bioactive compounds identified in the TMESLR.
In order to build the Protein-Protein Interaction (PPI) network for this investigation, the STRING database was used (accessed on Sep. 10, 2024), version 12.0 (https://string-db.org/), to examine proteins associated with cancer pathways and the bioactive chemicals found in the TMESLR. This analysis enabled the identification of direct and functional associations between the selected proteins and compounds. The resulting PPI network was visualized using Cytoscape software, version 3.10.1, allowing for comprehensive exploration of the interactions. The Cytoscape analyzer tool was utilized to generate a detailed interaction network consisting of 67 nodes and 1306 connections, with an average node connectivity of 38.98 (Supplemental Figure S1). This network provides a deeper understanding of the complex interplay between bioactive compounds and protein targets, revealing potential pathways involved in the cytotoxic activities of TMESLR.
Identification of Key Hub Genes in the PPI Network
Important hub genes in the PPI network linked to the anticancer properties of TMESLR compounds were found using the CytoHubba plugin. The highly connected hub genes include CASP3, CASP8, CASP9, FADD, BCL2, BCL2L1, CYCS, APAF1, TNF, FASLG, and XIAP, which are central to apoptosis regulation and cancer cell signaling pathways Figure 7. These genes represent potential molecular targets and provide valuable insights into the mechanisms driving the cytotoxic effects of the bioactive compounds. Their strong connectivity in the network underscores their significance in mediating the therapeutic potential of the compounds.

Key hub genes identified in the PPI network related to the anticancer activity of TMESLR compounds. The highlighted genes, including CASP3, CASP8, CASP9, FADD, BCL2, BCL2L1, CYCS, APAF1, TNF, FASLG, and XIAP, play critical roles in apoptosis and cancer-related signaling pathways, emphasizing their importance as central nodes in the interaction network.
Analysis of Overrepresented Gene Ontology (GO) Terms
GO enrichment study for the current study on the anticancer properties of TMESLR compounds was accompanied using ShinyGO v0.80. Our results highlight the main protein involvement across key categories Figure 8, providing insights into their functional roles in cancer-related pathways. Apoptotic processes, the control of apoptosis-related cysteine-type endopeptidase activity, intrinsic apoptotic signaling pathways, and the activation of apoptotic signaling through death domain receptors are among the enriched GO items in the BP category. Other notable terms involve the positive regulation of cytochrome c release from mitochondria and the regulation of necroptosis and autophagy. These findings indicate that the bioactive compounds are heavily involved in processes central to the induction of apoptosis and cancer cell death mechanisms, underscoring their therapeutic potential. In the CC category, significant GO terms include the death-inducing signaling complex, caspase complex, apoptosome, ripoptosome, and the CD95 death-inducing signaling complex. Additional terms such as the tumor necrosis factor receptor superfamily complex, and inflammasome complex highlight the involvement of these proteins in apoptotic and inflammatory signaling. These cellular locations suggest that the compounds target critical components within signaling complexes, leading to the activation of programed cell death, and immune-modulatory effects. For the MF category, enriched terms include BH domain binding, death receptor binding, tumor necrosis factor receptor binding, TRAIL binding, and kinase binding. Additionally, terms such as protein homodimerization activity, ubiquitin-protein ligase binding, and enzyme inhibitor action were recognized. These molecular functions propose that the compounds exert their effects through interactions with receptors, and enzymes critical for cancer cell survival, and apoptosis regulation. This comprehensive GO analysis reveals that TMESLR compounds target multiple pathways, and mechanisms central to cancer progression, highlighting their potential as multifunctional therapeutic agents. The visualized data provide a deeper understanding of their molecular roles, and possible applications in anticancer therapy.
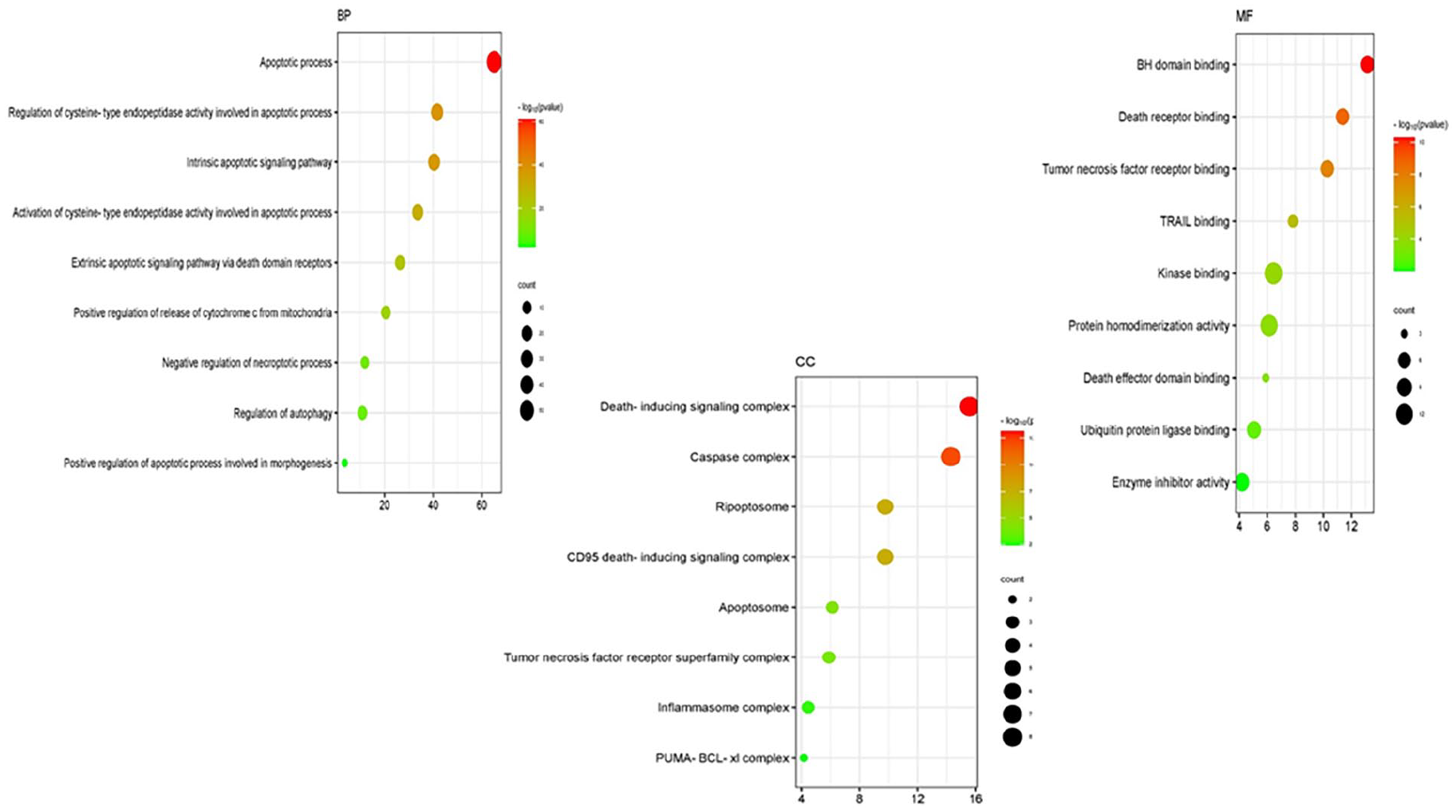
The Gene Ontology (GO) classifications for the anticancer properties of compounds found in TMESLR are depicted in a bubble chart. The enriched categories Biological Processes (BP), Cellular Components (CC), and Molecular Functions (MF) are highlighted in the chart, with particular attention paid to their roles in apoptosis, signaling cascades, and protein interactions that are essential for cancer treatment.
Examination of Predominant KEGG Pathways
The KEGG pathway analysis conducted in the present study highlights the molecular mechanisms by which the bioactive compounds from TMESLR exerts anticancer effects. The results are visualized in Figure 9, reveal important enhancement in key pathways such as apoptosis, the p53 signaling pathway, TNF signaling, NF-kappa B signaling, and necroptosis. These pathways are central to cancer progression, and cell survival, illustrating the ability of the bioactive compounds to regulate apoptotic mechanisms, and disrupt cancer cell proliferation. Additionally, pathways associated with drug resistance (eg, platinum drug resistance), and specific cancer types (eg, small cell lung cancer) were notably enriched, suggesting the therapeutic potential of these compounds. This analysis provides a comprehensive understanding of the targeted pathways, offering insights into how TMESLR compounds interact with molecular networks.
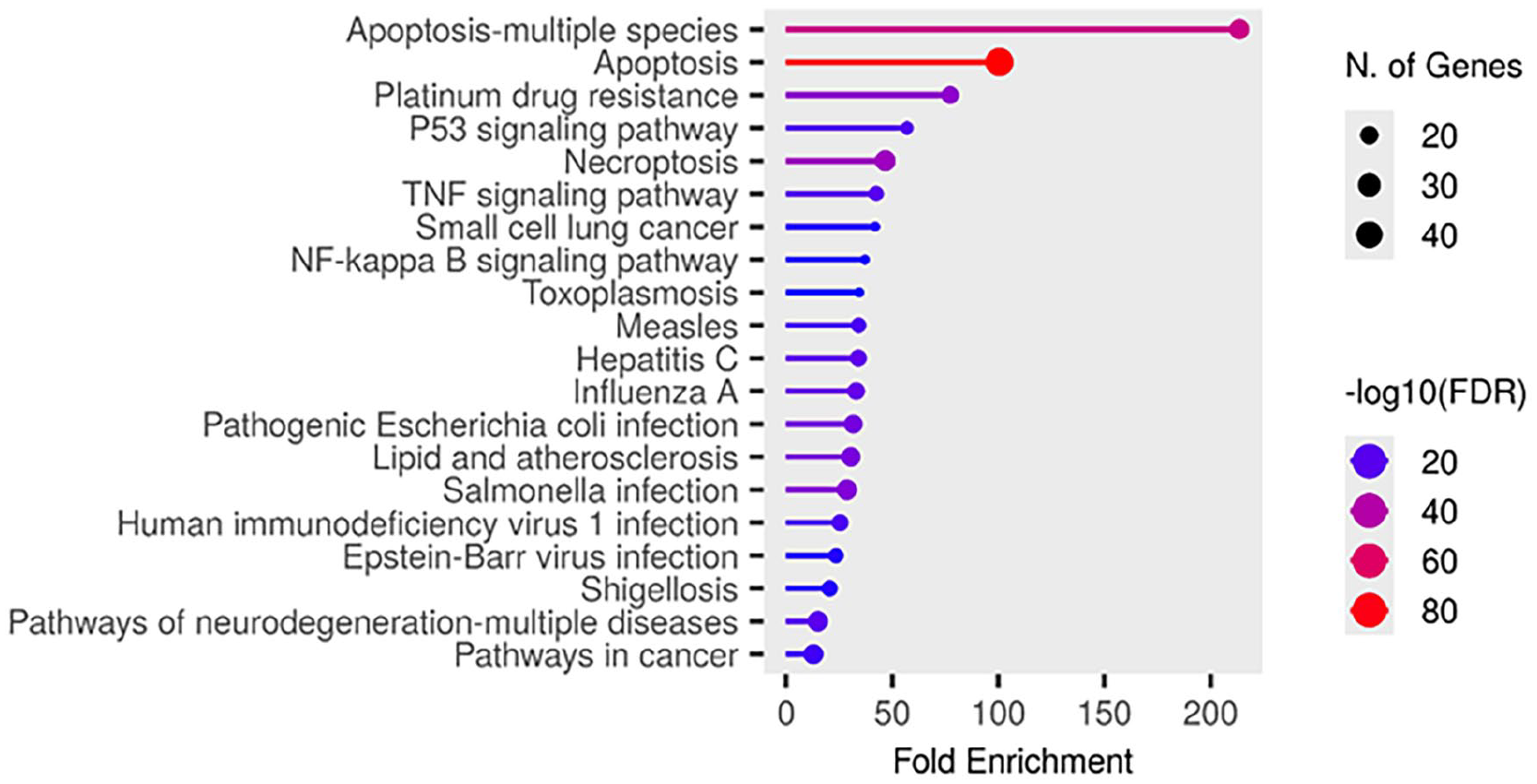
Bar plot illustrating the significantly enriched KEGG pathways targeted by TMESLR compounds, highlighting their potential anticancer effects. The pathways include apoptosis, p53 signaling, TNF signaling, and NF-kappa B signaling, showcasing the role in disrupting cancer progression and promoting therapeutic outcomes.
Molecular Modeling With Human HDAC2
Human Histone Deacetylase 2 (HDAC2) is a crucial enzyme implicated in the regulation of gene expression, and epigenetic modifications. Its overexpression has been linked to the development, and progression of various cancers, including Caco-2, MCF-7, and HepG2.45-47 Targeting HDAC2 can restore normal acetylation levels, leading to cell cycle arrest, apoptosis, and reduced cancer cell proliferation.
48
This makes HDAC2 a promising therapeutic target for developing effective anticancer agents. In this study, molecular docking simulations were performed to evaluate the interaction of compounds derived from TMESLR (

Binding interactions with Human HDAC2. (A and B) Interaction maps of inosine (
Discussion
Prior research showed that the ethanol extract of S. lycopersicum fruits exhibited an antiproliferative effect against HepG2 cell line with an IC50 value of 48.0 µg/mL. 51 The present study reveals, for the first time, the significant in vitro cytotoxic activity of the waste product, S. lycopersicum roots, against the cancer cell lines Caco-2, MCF-7, and HepG2.
Interestingly, our results evidenced that the prepared TMESLR-NCs notably enhanced the cytotoxic activity of TMESLR, particularly against Caco-2 (41%) and HepG2 (16%) cancer cell lines. A result that indicates the cytotoxic potential of such an ignored plant part, commonly considered as a vegetable waste product.
LC-MS metabolomic analysis characterized the presence of various chemical classes in the studied extract, including flavonoids, terpenes, ionones, and phenolic acids are illustrated in Supplemental Table S1 and Figure 3. This metabolic profile distinctly explicated the cytotoxic potential of TMESLR. For instance, phytol (
TMESLR and its prepared-NCs demonstrated significant cytotoxic effects, involvement in apoptosis-related pathways, and strong interactions with key proteins like HDAC2.
56
The identification of hub genes such as CASP3, BCL2, and TNF, along with enriched pathways including apoptosis and p53 signaling, highlights their molecular mechanisms of action.
57
Molecular docking results further confirm the promising role of inosine (
Conclusions
The findings of this study revealed that the TMESLR revealed significant cytotoxicity against Caco-2, MCF-7, and HepG2 cells. Also, TMESLR-NCs displayed superior cytotoxic activity, particularly against Caco-2 and HepG2 cancer cell lines. Metabolomic profiling using LC-MS metabolomic analysis identified
Supplemental Material
sj-docx-1-ict-10.1177_15347354251335599 – Supplemental material for Cytotoxic Potential and Metabolomic Profiling of Solanum lycopersicum Roots Extract and Their Nanocrystals: An In Silico Approach
Supplemental material, sj-docx-1-ict-10.1177_15347354251335599 for Cytotoxic Potential and Metabolomic Profiling of Solanum lycopersicum Roots Extract and Their Nanocrystals: An In Silico Approach by Marwa A. M. Abdel-Razek, Miada F. Abdelwahab, Soad A. Mohamad, Hesham A. Abou-Zied, Usama R. Abdelmohsen and Ashraf N. E. Hamed in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354251335599 – Supplemental material for Cytotoxic Potential and Metabolomic Profiling of Solanum lycopersicum Roots Extract and Their Nanocrystals: An In Silico Approach
Supplemental material, sj-docx-2-ict-10.1177_15347354251335599 for Cytotoxic Potential and Metabolomic Profiling of Solanum lycopersicum Roots Extract and Their Nanocrystals: An In Silico Approach by Marwa A. M. Abdel-Razek, Miada F. Abdelwahab, Soad A. Mohamad, Hesham A. Abou-Zied, Usama R. Abdelmohsen and Ashraf N. E. Hamed in Integrative Cancer Therapies
Footnotes
Author Contributions
Marwa A. M. Abdelrazek and Miada F. Abdelwahab: writing original draft, visualization, validation, investigation, formal analysis, and data curation. Soad A. Mohamed: methodology, and conceptualization. Hesham A. Abo Zeid: validation, software, methodology, and conceptualization. Usama R. Abdelmohsen and Ashraf N. E. Hamed: reviewing, supervision, methodology, investigation, data curation, and conceptualization.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
This article and its supplemental information files contain all of the data generated during this investigation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
