Abstract
Background:
The current standard treatment for chemotherapy-induced nausea and vomiting (CINV) with standard antiemetics is insufficient. Rikkunshito, a Japanese traditional herbal medicine, has been shown to improve cisplatin-induced anorexia and functional dyspepsia, and our exploratory study found that rikkunshito has an additive beneficial effect on CINV in patients with uterine corpus and cervical cancer receiving cisplatin containing chemotherapy (JORTC KMP-02).
Methods:
One hundred eighty patients with uterine corpus or cervical cancer who were scheduled to receive treatment with a cisplatin based regimen as initial chemotherapy were enrolled across 17 Japanese institutions. Patients were randomized with a 1:1 equal allocation ratio to the rikkunshito group or placebo groups and given oral administration on days 1 to 5 with standard antiemetics (granisetron, aprepitant, and dexamethasone). The primary endpoint was complete response (CR; no vomiting or rescue medication) during the delayed phase (24-120 hours after cisplatin treatment). The secondary endpoints were complete control (CC; CR without significant nausea) and total control (TC; CR without nausea) rates during the overall (0-120 hours), acute (0-24 hours), and delayed phases, as well as the CR rate during the overall and acute phases, time to treatment failure, degree of nausea and appetite during the overall phase, and adherence to the intervention.
Results:
The CR rate in the delayed phase was similar between the rikkunshito group and control groups (50.6% vs 58.9%, P = .2631), as were the secondary endpoints: CR rates in the overall and acute phases, CC and TC rates in the overall, acute, and delayed phases, degrees of nausea and appetite, and time to treatment failure.
Conclusion:
Rikkunshito had no additive effect on CINV prevention in patients with uterine corpus or cervical cancer who were treated with a cisplatin based regimen and standard antiemetics.
Clinical Trial Registration:
https://jrct.mhlw.go.jp/re/reports/detail/66957, identifier jRCT1011190007
Keywords
Introduction
Chemotherapy induced nausea and vomiting (CINV) is a serious side effect of anticancer chemotherapy. International guidelines recommend the use of serotonin (5-hydroxytryptamine) type 3 (5-HT3)-receptor antagonists, neurokinin-1-receptor antagonists, and corticosteroids in patients receiving highly emetogenic chemotherapy such as cisplatin.1,2 However, the effectiveness of these treatment approaches remains unsatisfactory, with reported rates of complete response (CR), defined as no vomiting and no use of rescue medication, ranging from 44% to 90%. 3 Recently, olanzapine, a potent antipsychotic medication, has been recommended for preventing CINV by highly emetogenic chemotherapy4,5; however, mild-to-moderate sedation and hyperglycemia are potential side effects.6,7
Rikkunshito, a Japanese traditional herbal (kampo) preparation, 8 comprises 8 herbs including Atractylodis lanceae rhizoma, Ginseng radix, Pinelliae tuber, Poria, Zizyphi fructus, Aurantii nobilis pericarpium, Glycyrrhizae radix, and Zingiberis rhizoma, and is widely prescribed (7.5 g/day) in Japan to treat gastrointestinal disorders such as anorexia, functional dyspepsia, and gastroesophageal reflux disease.9 -12 In mice, rikkunshito was shown to improve cisplatin-induced anorexia. This effect was due to the antagonism of the 5-HT2B and 5-HT2C receptors by the flavonoids in rikkunshito, resulting in a suppression of the decrease in plasma levels of acylated-ghrelin, an orexigenic peptide released from the stomach. 13
We recently conducted a randomized exploratory trial to evaluate the additive beneficial effect of rikkunshito on CINV and anorexia in patients with uterine cervical or corpus cancer who were treated with cisplatin and paclitaxel (JORTC KMP-02). 14 This study found that CR rates during the overall (0-120 hours after cisplatin treatment) and delayed phase (24-120 hours after cisplatin treatment) were significantly higher in the rikkunshito group than in the control group (84.2% vs 52.9%; P = .042, 84.2% versus 52.9%; P = .042, respectively), implying that rikkunshito may have an additive beneficial effect in patients with CINV.
We therefore conducted a multicenter, randomized, double-blind, placebo-controlled confirmatory study to assess the additively beneficial effect of rikkunshito on CINV in patients with uterine cervical or corpus cancer treated with cisplatin.
Materials and Methods
This is a multicenter, randomized, double-blind, placebo-controlled phase III trial that was conducted in 17 institutions across Japan (JORTC-KMP03). This trial was registered with the Japan Registry of Clinical Trials (jRCT) as jRCT1011190007 (https://jrct.niph.go.jp/en-latest-detail/jRCT1011190007) before patient enrollment. The trial design was described previously, 15 and the study flow is depicted in Figure 1. The study ran from December 2019 to April 2022.

CONSORT flow diagram.
Patient Eligibility
Patients who met the following inclusion criteria were enrolled: ≥20 years old, histologically diagnosed with uterine cervical or corpus cancer by their doctor, voluntarily signing the Certified Review Board-approved written informed consent, having an Eastern Cooperative Oncology Group (ECOG) performance status grade 0 to 2, having good oral intake, having an expected prognosis of ≥3 months; and patients with no history of chemotherapy who are planned to receive chemotherapy including ≥50 mg/m2 cisplatin. In addition, patients with the following laboratory test values was qualified: white blood cell count, 3000 to 12 000/μL; neutrophil count, ≥1500/μL; platelet count, ≥100 000/μL; hemoglobin, ≥8.0 g/dL; aspartate aminotransferase, ≤100 IU/L; alanine aminotransferase, ≤100 IU/L; total bilirubin, ≤2.0 mg/dL; potassium, ≥3.0 mEq/L; creatinine clearance estimated using the Cockroft–Gault equation, ≥60 mL/min; and no abnormalities on electrocardiography. The following illnesses precluded participants from the study: brain metastasis or peritoneal dissemination; double cancer; seizure requiring treatment; unconsciousness; gastrointestinal obstruction; vomiting or nausea greater than grade 2 as described by the National Cancer Institute Common Toxicity Criteria for Adverse Events (CTCAE) version 4.0; registry to a clinical trial for another medication; uncontrollable hypertension; uncontrollable diabetes mellitus or that treated with insulin; treatment with any medicine that increases appetite such as corticosteroids, androgen, progesterone, and other herbal medicines; treatment with opioids; pregnancy or breast-feeding or inability to use contraception; any uncontrollable infectious disease; body temperature ≥38°C; psychosis; alcoholism; and patients who are scheduled to receive radiotherapy.
Treatment Plan
Patients were randomly assigned to either the rikkunshito or the placebo group (Figure 1). After written informed consent was obtained, randomization of the patient was performed in the web-based VIEDOC 4 system (PCG Solutions, Sweden) using the minimization method by balancing the groups for age (<55 or ≥ 55 years), amount of alcohol intake (<5 or ≥5cups/week), chemotherapy (adjuvant chemotherapy, neoadjuvant chemotherapy, or chemotherapy only), and study site. The investigators, patients, and pharmacists were blinded to the allocation of each patient. 15 All patients received chemotherapy via intravenous (IV) cisplatin (≥50 mg/m2) for 2 hours on day 1. They also received standard antiemetic therapy according to the Japanese antiemetic guideline by the Japan Society of Clinical Oncology which included the following: IV granisetron, 3 mg on day 1; oral aprepitant, 125 mg on day 1 and 80 mg on days 2 and 3; and IV dexamethasone, 9.9 mg on day 1 and oral dexamethasone, 8 mg on days 2 to 4. The rikkunshito or placebo (2.5 g granules) were administered orally before each meal on days 1 to 5. Additional concomitant anticancer drugs could be administered on day 1. In patients who received additional concomitant anticancer drugs on day 0, IV granisetron (3 mg) on day 0 was permitted. Paclitaxel could also be administered on day 0; in these patients, an IV dexamethasone (16.5 mg) on day 0 was allowed for anaphylaxis prevention. Rescue medication was permitted to treat refractory and persistent CINV; however, these treatments were deemed treatment failure. The investigator determined the timing and choice of rescue treatment.
Assessment
Before enrollment, the investigators determined the eligibility of potential participants based on the inclusion and exclusion criteria. The screening tests were conducted after the informed consent form was signed. The detailed screening items for evaluating potential participants are described below. The patients’ clinical characteristics included a physical examination and vital signs, blood sampling for serological tests like complete blood count and biochemical tests, and electrocardiography. The subjective and objective outcome measures included a numerical rating scale (NRS) for nausea and appetite, the frequency and duration of vomiting, the time until rescue medication was necessary, and adherence to rikkunshito. Each patient was asked to keep a paper-based diary on the intensity of nausea, number of vomiting episodes, appetite, and treatment compliance daily from day 1 to day 6. The frequency of recording was set as follows: appetite and study medication adherence were recorded at every meal, the NRS for nausea was recorded once daily at its worst value, and the number of vomiting episodes was recorded each time vomiting occurred.
Objectives
The primary endpoint is the rate of complete response (CR), which is defined as no vomiting and no need for rescue medication during the delayed phase. The secondary endpoints are as follows: rates of complete control (CC), defined as CR with no more than mild nausea (NRS ≤ 3), 16 and total control (TC), defined as CR with no nausea (NRS = 0) during the delayed phase; rates of CR, CC, and TC during the overall phase; rates of CR, CC, and TC during the acute phase (0-24 hours after cisplatin treatment); time to treatment failure, defined as the time to the first emetic episode or rescue medication, during the overall phase; NRS of nausea and appetite during the overall phase and adherence to intervention.
Statistical Analysis
Based on the findings of our previous study, 14 we estimated CR rates of 80% and 60% in the rikkunshito and placebo groups, respectively, during the delayed phase. Assuming an exclusion rate of 10%, a sample size of 90 evaluable patients in each group is required to achieve 80% power and a 2-tailed significance level of 0.05 for comparisons. The primary endpoint of the CR rate during the delayed phase, the superiority of the rikkunshito group over the placebo group was evaluated using the chi-squared test. A 2-tailed P < .05 will indicate a significant difference in the planned analysis. Point estimates with 95% confidence intervals (CI) for both groups, as well as differences between them, were calculated. The secondary endpoints, which included CC and TC rates during the delayed phase, CR, CC, and TC rates during the overall phase, and CR, CC, and TC rates during the acute phase, were assessed using the same approach as the primary endpoint. The statistical analysis done on an intention to treat basis. The difference in time to treatment failure between the groups was calculated using Kaplan–Meier estimates and the log-rank test. Longitudinal changes in NRS for nausea and appetite were assessed using mean scores and 95% CIs. Summary statistics of each group were measured for adherence to the intervention. The distributions of adverse events of different grades and the incidence rates of ≥grade 3 and 4 adverse events were determined. All analyses were performed with SAS version 9.4 (SAS Institute, Cary, NC, USA). A comprehensive statistical analysis plan was developed prior to evaluating the data. Objective outcome measures were scored using standard methodology.
Results
Patient Characteristics
From January 2020 to April 2022, 180 patients were enrolled across 17 institutions. One patient from the rikkunshito group was excluded due to treatment refusal after allocation, leaving 89 patients in the rikkunshito group and 90 patients in the placebo group to be evaluated (Figure 1). The patient characteristics are summarized in Table 1. The baseline characteristics of the study subjects were comparable across groups. The average patient age was 53.8 (range 27-76 years), and all had a good ECOG PS (0-1). The most common cancer type was cervical cancer (55.0%), and the majority of patients (77.2%) were treated with postoperative chemotherapy. The following chemotherapeutic agents were combined with cisplatin: paclitaxel (42.8%), doxorubicin (44.4%), and bevacizumab (3.3%).
Patients’ Features.

Abbreviations: SD, standard deviation; PS, performance status; BMI, body mass index.
Efficacy
The CR rates in the delayed phase were 50.6% (95% CI, 40.2-61.0) in the rikkunshito group and 58.9% (95% CI, 48.7-69.1) in the placebo group, which did not reach the primary endpoint (Table 2). Furthermore, rikkunshito did not improve CR rates in either the overall or delayed phases. Rikkunshito administration also did not increase CC or TC rates in the acute, delayed, or overall phases. The NRS for nausea and appetite were similar in the rikkunshito and placebo groups (Figure 2). The time to treatment failure was also similar between these groups (Figure 3). Both groups demonstrated good adherence to the intervention (Table 3).
Outcomes of the Main Endpoint.

Abbreviations: CR, complete response; CC, complete control; TC, total control. CI; confidence interval.

Longitudinal variations in numerical rating scale (NRS) for nausea (A) and appetite (B).
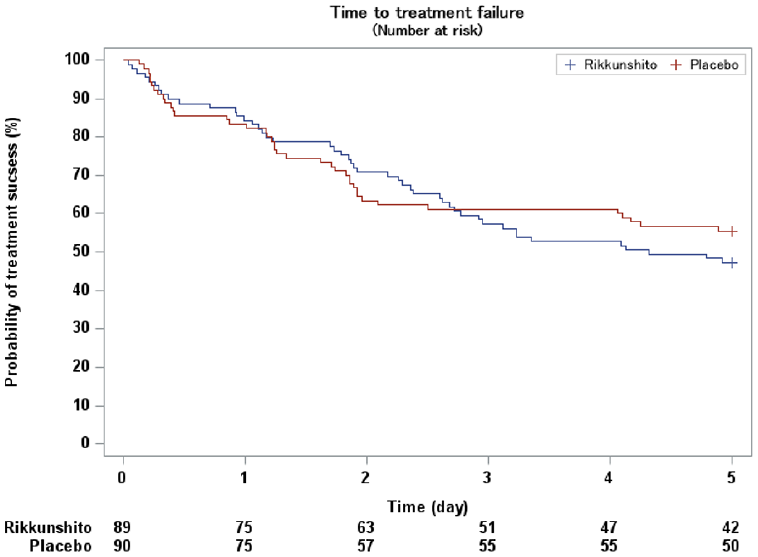
Duration to treatment failure.
Adherence to Intervention.

Abbreviations: Obs, observations; SD, standard deviation.
Discussion
In this study, we investigated the efficacy of rikkunshito on CINV in patients receiving a cisplatin-containing regimen, to confirm our previous exploratory study. The CR rate in the delayed phase was comparable between the rikkunshito and placebo groups, as were the secondary endpoints: CR rates in the acute and overall phases, CC and TC rates in the acute, delayed, and overall phases, degrees of nausea and appetite during the overall phase, and time to treatment failure. The CR rates in the derayed phase of both groups in this study were relatively low compared with other studies.17,18 However, the CR rate in the delayed phase for the control group in the previous exploratory trial was 52.9%, which was not significantly different from the results of both groups in the current study. The major differences between this trial and the previous exploratory trial in patient characteristics are the inclusion of patients with endometrial cancer and the use of different regimens. Nonetheless, all patients in both studies were female and relatively young, which remains consistent. The lower delayed-phase CR rate compared to previous reports on triplet antiemetic prophylaxis may be explained by the study population’s characteristics, as both female and younger age have been reported to be associated with heightened CINV severity.19,20
Our previous exploratory study found that rikkunshito significantly improved CINV in patients with uterine cervical or corpus cancer receiving cisplatin and paclitaxel; however, the previous study was conducted without blinding. 14 Meanwhile, Harada et al conducted a similar exploratory study in lung cancer patients treated with a cisplatin based regimen, finding that rikkunshito had no additive effect on CINV. 21 The majority of the lung cancer patients in their study were relatively old men; however, our previous study was conducted on middle-aged women. Therefore, we hypothesized that rikkunshito could be effective for CINV in relatively young women, as CINV is known to be more severe in women and younger patients. However, the findings of our previous study, combined with our current study, indicate that information bias, particularly participant expectation bias, had a significant impact on the results of our previous study. 22 To provide a more detailed explanation for the significant differences in the CR rate observed between the exploratory study and the current trial in the group receiving rikkunshito, it is important to note that in the prior exploratory study, patients in the rikkunshito group were consistently assigned to the active drug. This likely cultivated an expectation that the medication would effectively alleviate nausea. This so-called expectation bias might have contributed to the observed better outcomes. In contrast, in the current trial, the participants were unaware of whether they were assigned to the active drug or the placebo, which might explain the diminished expectation bias and the resulting different outcomes. Considering that CINV is significantly influenced by psychological factors, we hypothesize that this expectation bias might have played a substantial role in trials evaluating the efficacy of antiemetic agents.
It has been reported that rikkunshito suppresses cisplatin-induced decreases in food intake and plasma acylated-ghrelin levels in mice, with this effect mediated by antagonism of 5-HT2B and 5-HT2C receptors. 13 Furthermore, rikkunshito has been shown to enhance ghrelin receptor signaling. 23 Our previous clinical trial found that the visual analog scale for appetite was superior in the rikkunshito group from day 2 to day 6, but serum acyl ghrelin levels did not decrease after cisplatin administration, and there was significant variability among the patients. Although we did not investigate serum acyl ghrelin levels in the current study, NRS for appetite was comparable between the rikkunshito and placebo groups. Therefore, rikkunshito would not affect anorexia in patients receiving cisplatin. More placebo-controlled studies are needed to confirm other situations, such as body weight loss and gastrointestinal symptoms following esophagostomy and gastrectomy (Yoshizumi et al 24 ; Nakamura et al 25 ).
Limitation
This study has several limitations. First, it aimed to evaluate the efficacy of Rikkunshito in combination with a 3-drug antiemetic regimen; however, no additional benefit was observed. As a 4-drug regimen including olanzapine has recently become the standard, the effectiveness of Rikkunshito in this setting remains unknown. Second, the intervention period was limited to 5 days, which may not fully capture its potential benefits in managing delayed or prolonged CINV. Finally, since this study was conducted exclusively in Japanese institutions, its generalizability to populations with different genetic backgrounds or dietary habits remains uncertain.
Conclusion
Rikkunshito had no additive effect on the prevention of CINV in patients with uterine corpus or cervical cancer who were treated with a cisplatin based regimen and standard antiemetics.
