Abstract
Keywords
Introduction
Nausea and vomiting are among the most prevalent and disturbing side effects of chemotherapy.1,2 These side effects may lead to several adverse impacts in patients receiving chemotherapeutic agents, such as decreased quality of life, anxiety, depression, and lack of compliance to the medication.2,3 Chemotherapy-induced nausea and vomiting (CINV) is classified into 3 types: acute CINV, which occurs during the first 24 hours postchemotherapy; delayed CINV, which begins after24 hours postchemotherapy and may last for up to 6 or 7 days; and finally anticipatory CINV, which affects people who have experienced severe nausea and vomiting in their previous administrations of chemotherapeutic agents. The prevalence of untreated CINV is about 70% to 80%, with delayed CINV being experienced by up to 80% and anticipatory CINV by approximately 14% to 63% of patients.
4
5-HT3 receptor antagonists represent a widely used class of antiemetic agents that are among the first choices for the treatment and prevention of CINV. However, in spite of their effectiveness, still a considerable fraction of patients experience CINV.5-11 For instance, 10% to 30% of patients treated with 5-HT3 receptor antagonists have been reported to experience acute cisplatin-induced emesis,9-11 and even a higher rate of patients experience delayed cisplatin-induced emesis.12,13 There are also other reports indicating a prevalence of 48% to 67% for CINV after receiving conventional antiemetic therapy.2,5,6,8 Besides, unlike acute CINV, delayed and anticipatory types of CINV appear to be resistant to pharmacological treatments, thereby remaining poorly controlled by currently available medications.2,7,12-17 With respect to the above-mentioned problems, there is a great demand for the introduction of new and safe agents that could effectively reduce CINV rate. One of the potential sources for such agents is medicinal plants, and among them, ginger (
Ginger has a widespread use as a spice and flavoring agent and has a long reputation in both Asian (including Indian [Ayuverdic], Chinese, and middle eastern) and Western traditional medicines.18,19 The powdered rhizome of
Methods
Subjects and Design
This was a randomized, open-label, clinical trial that was performed between July 2008 and November 2009 in the Oncology Unit of the Baqiyatallah Hospital. The participants included were women who had cancer (mainly new cases of advanced breast cancer diagnosed by the oncologist and thus undergoing their first experience of chemotherapy). Most of these patients were initially assigned to the components of TEC regimen, including docetaxel, epirubicin, and cyclophosphamide. Docetaxel is associated with intermediate emetic risk, whereas epirubicin and cyclophosphamide are highly emetogenic. 37 Therefore, the patients who were under single or combinational therapy with components of the TEC regimen may be considered to experience chemotherapy with moderate to high emetogenic risk. None of the women were pregnant, lactating, or undergoing concurrent radiotherapy. Exclusion criteria were participation in another study with different drugs; history of bone marrow or stem cell transplantation; presence of respiratory, cardiovascular, liver, renal, metabolic, or gastrointestinal diseases; and history of motion sickness. From the 110 initially evaluated women, 100 were qualified to enter the trial. All these 100 women were randomly assigned to ginger (1.5 g/d in 3 divided doses every 8 hours) plus standard antiemetic regimen (granisetron plus dexamethasone; ginger group; n = 50) or standard antiemetic regimen alone (control group; n = 50). The first dose of ginger was consumed about 30 minutes after the completion of chemotherapy. The participants of this open-label trial were individually and alternatively allocated to ginger or control group with the first allocation being chosen randomly. At the beginning of study and before patient recruitment, blank questionnaires were numbered and alternatively coded as ginger or control. The first code was chosen randomly (by lottery). Then recruited patients were categorized into ginger or control group from questionnaire 1, based on the predefined allocation. Powdered and dried ginger root was prepared and validated by the Iran Herb Medical Society and administered as 500 mg capsules prepared by the Razak Laboratories Corporation (Tehran, Iran). The duration of treatment with ginger was specified as 4 days from the initiation of chemotherapy, to allow the evaluation of ginger’s effects on both acute and delayed CINV. The day of chemotherapy was considered as day 1. Apart from medical interventions, patients were advised to eat easily digestible foods and avoid emetogenic ones.
The study was approved by the institutional ethics committee and conducted according to the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants.
Assessment of the Prevalence and Severity of CINV
Participants were asked to record their symptoms in each day of trial using a simplified form of the Rhodes Index of Nausea, Vomiting, and Retching (RINVR).38,39 The RINVR is a self-reporting 8-item, 5-point Likert-type tool that measures the frequency and duration as well as the distress caused by the symptoms of nausea, vomiting, and retching. The modified form of RINVR, which was used in the present study, contained 8 questions asking about the prevalence and duration of nausea, vomiting, and retching; volume of vomit; and the distress caused by these symptoms. For each of the 8 questions, lack of symptoms was given 0 points, whereas the most severe or prevalent symptoms were given 3 points. Thus, the total score for each subject ranged from 0 to 24, with higher scores indicating more symptoms. A total score of 0 was considered as lack of nausea and vomiting, whereas scores 1 to 8, 9 to 16, and 17 to 24 were considered as mild, moderate, and severe forms of CINV, respectively. Prevalence (number of episodes) score and severity of nausea, vomiting, and retching were assessed in the first 6 hours postchemotherapy, between 6 and 24 hours postchemotherapy, and days 2, 3, and 4 of the trial. Postchemotherapy nausea and vomiting data were collected by a self-report diary approach using the aforementioned questionnaire. The participants were asked to fill the questionnaire in each of the days of the trial at home. Assessment of adverse effects was based on self-reports. Patients were asked to report the adverse effects in their questionnaire. They were also asked for adverse effects while returning questionnaires.
Statistical Analysis
Statistical analyses were performed using SPSS software for Windows (version 13.0; SPSS Inc, Chicago, IL). Values were presented as number (%) or mean ± standard deviation. Between-group (ginger vs control) comparisons were performed using independent-samples
Results
From the 110 initially screened women with advanced breast cancer, 100 met the eligibility criteria and entered the trial (age range = 35-74 years, mean age = 51.83 ± 9.18 years). Of these 100 women, 78 completed the trial (37 in the ginger group and 41 in the control group) with filled questionnaires and were included in the final analysis (Figure 1). The reasons for dropping out were mainly being lost to follow-up and lack of compliance (based on self-report). The difference in dropout rate between the 2 groups was not found to be significant (

Flowchart of the trial
Effect of Ginger on the Prevalence of Nausea, Vomiting, and Retching
Addition of ginger to the standard antiemetic therapy was found to be associated with a significant reduction in the prevalence of nausea, but not vomiting and retching, at 6 to 24 hours postchemotherapy in the ginger group compared with the control group (
Prevalence of Nausea, Vomiting, and Retching in Ginger and Control Groups

Pearson χ2 values with 1 degree of freedom.
Values are presented as number (%). Comparisons were performed using χ2 or Fisher’s exact tests with 1 degree of freedom. Power calculations were performed using the PS software (version 3.0), with α = .05.
Effect of Ginger on the Rhodes Index Scores of Nausea, Vomiting, and Retching
Overall, the mean scores for nausea, vomiting, and retching were not significantly different between ginger and control groups and this was true for all assessed intervals—the first 6 hours, 6 to 24 hours, and second, third, and fourth days postchemotherapy (
Rhodes Index Scores of Nausea, Vomiting, and Retching in Ginger and Control Groups
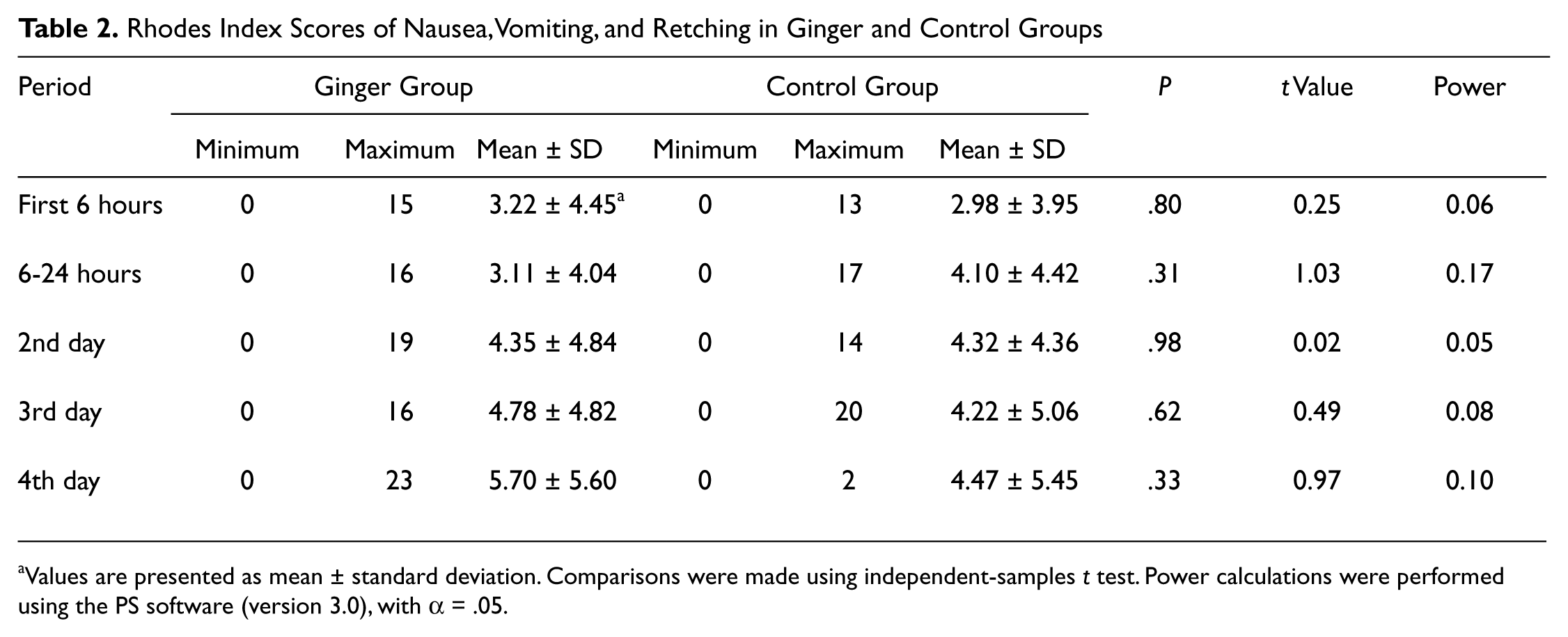
Values are presented as mean ± standard deviation. Comparisons were made using independent-samples
Effect of Ginger on the Severity of Nausea, Vomiting, and Retching
There was no significant difference between the ginger and control groups in the number of individuals in each of the 4 assessed subclasses based on the severity of symptoms (lack of symptoms, mild, moderate, and severe symptoms). The lack of significant difference was observed at all assessed intervals—the first 6 hours, 6 to 24 hours, and second, third, and fourth days postchemotherapy (
Severity of Nausea, Vomiting, and Retching in Ginger and Control Groups a

Severity of symptoms were assessed based on the Rhodes index.
Pearson χ2 values with 1 degree of freedom.
Values are presented as number (%). Comparisons were performed using χ2 or Fisher’s exact tests with 1 degree of freedom. Power calculations were performed using the PS software (version 3.0), with α = .05.
Discussion
The results of the present study indicated a significantly lower prevalence of nausea in the ginger group during 6 to 24 hours postchemotherapy. Despite this effect, no other significant additional benefit from ginger (1.5 g/d) was observed against prevalence or severity of acute or delayed chemotherapy-induced nausea, vomiting, and retching in patients with advanced breast cancer. Noteworthy, there were ~17% and 10% reduction in the prevalence of nausea during the first 6 hours and second day of chemotherapy, respectively. Although these reductions were not found to be significant from the statistical point of view (probably because of insufficient power), they might be of clinical relevance and thus deserve to be verified by larger trials.
Unlike nausea/vomiting related with other situations such as pregnancy, motion sickness, and surgery, the efficacy of ginger to reduce the CINV remains relatively unknown with only few studies in this regard. Therefore, further investigations in this regard appear necessary and might be useful to become available to the scientific community. To our knowledge, there have been 6 previous reports on the efficacy of ginger against CINV,31-36 of which 3 are available only in the abstract form.31,32,36 The study by Pace 31 investigated the effectiveness of ginger compared with placebo in a group of 41 leukemic patients who were under chemotherapy plus antiemetic agent prochlorperazine. The results indicated a significant decrease in the acute nausea symptom score in the ginger versus placebo group. 31 However, in the mentioned study (published in the abstract form), the effect of ginger on delayed CINV was not assessed. In the second study, by Pecoraro et al, 32 a greater complete treatment response of acute CINV was reported in subjects receiving ginger compared with placebo. This was again a small study (12 participants) with limitation regarding examination of ginger effect on delayed CINV.
The third study, by Sontakke et al, 33 which had a randomized, prospective, crossover, and double-blind design, evaluated the effects of ginger in comparison with metoclopramide and ondansetron among 60 patients receiving low-dose cyclophosphamide in combination with other chemotherapeutic agents causing mild emesis. The results implied the effectiveness of ginger in controlling nausea and vomiting, with its antiemetic efficacy being equal to that of metoclopramide but lower than that of ondansetron. The study of Sontakke et al 33 is not directly comparable to the present work, as they administered ginger instead of the standard antiemetic therapy such as a 5-HT3 antagonist, whereas in the current work ginger was coadministered with the standard regimen (granisetron plus dexamethasone). In addition, the latter study did not investigate the effect of ginger on the prevalence or severity of delayed CINV.
The fourth investigation was a randomized, double-blind, crossover study that evaluated the efficacy of ginger (1 g/d) against both acute and delayed types of cisplatin-induced emesis in 48 patients with gynecologic (ovary or cervix) cancer. Based on the results, addition of ginger to the standard antiemetic regimen of patients under cisplatin therapy had no significant benefit against acute CINV. However, in the delayed phase of chemotherapy (days 2-5), the efficacy of ginger in controlling nausea and vomiting was not found to be significantly different from that of metoclopramide (40 mg/d). 34 These results are almost in line with the findings of the fifth study by Zick et al, 35 who conducted a large randomized and double-blind trial among 162 patients. The patients were randomized to receive 1 g/d ginger, 2 g/d ginger or placebo for 3 days, and all were receiving a 5-HT3 receptor antagonist and/or aprepitant as standard antiemetic therapy. The results implied that ginger, at either dose, had no significant additional benefit in decreasing the prevalence or severity of acute or delayed CINV. In the same study, high-dose ginger (2 g/d) was associated with more severe episodes of delayed nausea compared with low-dose ginger (1 g/d) or placebo. In addition, subjects under both ginger (at either dose) and aprepitant were found to have more severe delayed nausea compared with those who took only aprepitant.
Finally, the most recent report on this topic pertains to a presentation by Ryan et al 36 at the 2009 ASCO Annual Meeting. This was a well-designed multi-site, phase II/III randomized, placebo-controlled, double-blind clinical trial which aimed at the evaluation of ginger effectiveness (0.5, 1.0 or 1.5 g daily) against acute CINV in 644 patients, as well as determination of its most effective dose. The results indicated that all doses of ginger significantly reduced nausea with doses of 0.5 g and 1 g being the most effective. However, the effect of ginger on vomiting was not found to be significant.
With respect to acute CINV, the results of the current study is consistent with those of Ryan et al, 36 indicating that addition of ginger to standard antiemetic therapy causes additional reduction in chemotherapy-induced nausea but not vomiting during the first day of chemotherapy. With respect to delayed CINV, the findings of this work and that of Zick et al 35 implied that addition of ginger does not reduce the prevalence or severity of delayed CINV beyond what is achieved by standard therapy with granisetron plus dexamethasone. However, it must be noted that the findings of the current study may not be directly comparable to some of the previous reports because of differences in the source and dose of ginger, supplementation design, and chemotherapeutic regimen (Table 4). Among the 5 studies with adequately published information in this field (taking into account the present trial), 3 started ginger before the initiation of chemotherapy33,34,36 and 2 (including this study and that of Zick et al 35 ) started ginger after the initiation of chemotherapy. In either type of trials, there have been both positive and negative findings. Nevertheless, it appears that initiation of ginger supplementation before or after chemotherapy is an influential factor on ginger’s efficacy against CINV.
Comparison of Clinical Trials on the Efficacy of Ginger Against CINV

Abbreviation: CINV, chemotherapy-induced nausea and vomiting.
Regarding the type of cancer and administered chemotherapy regimen, it is worth noting that the participants of this study probably constituted a more homogenous group compared with previous studies.33,35,36 Nevertheless, the present study had limitations that should be taken into consideration. The main issue is lack of blindness. This lack of blindness was because of the pilot nature of the study and difficulties in placebo preparation and successful blinding as ginger possesses unique and familiar aroma. 40 However, this lack of blindness and patients’ awareness of the treatment might have caused a so-called reverse placebo effect, 41 thereby leading to a more severe evaluation of CINV symptoms and consequent underestimation of ginger efficacy. Besides, it must be noted that the relatively fewer number of participants and thereby insufficient power could be a reason for the negative findings in the present study. An example of this latter issue is the prevalence of nausea in the first 6 hours postchemotherapy, which is about 17% lower in the ginger group, but not statistically different from that in the control group. As another limitation, the effect of ginger on the rate of chemotherapy-associated adverse events as well as efficacy of chemotherapy were not evaluated. In addition, although ginger is generally safe, accurate assessment of its potential adverse events and interactions in patients undergoing chemotherapy is needed. Finally, the relatively low prevalence and severity of CINV in both ginger and control groups raise the possibility of a “floor effect” that might be responsible for the lack of significant differences between the groups. Therefore, caution must be applied in considering the implications of the current results.
In summary, the data from the current study indicated that addition of ginger (1.5 g/d) to standard antiemetic therapy (granisetron plus dexamethasone) in patients with advanced breast cancer who were under chemotherapy was associated with decreased prevalence of nausea during 6 to 24 hours postchemotherapy. Nevertheless, no other significant effect was observed against prevalence of nausea in the first 6 hours postchemotherapy, severity of acute CINV, and prevalence or severity of delayed CINV. Future larger scale double-blind trials are recommended to verify the results of the current pilot trial. Prospective studies and longer periods of follow-up are also helpful to assess the efficacy of ginger supplementation on the prevalence of CINV symptoms in patients receiving multiple cycles of chemotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:
This work was financially supported by the Baqiyatallah University of Medical Sciences.
