Abstract
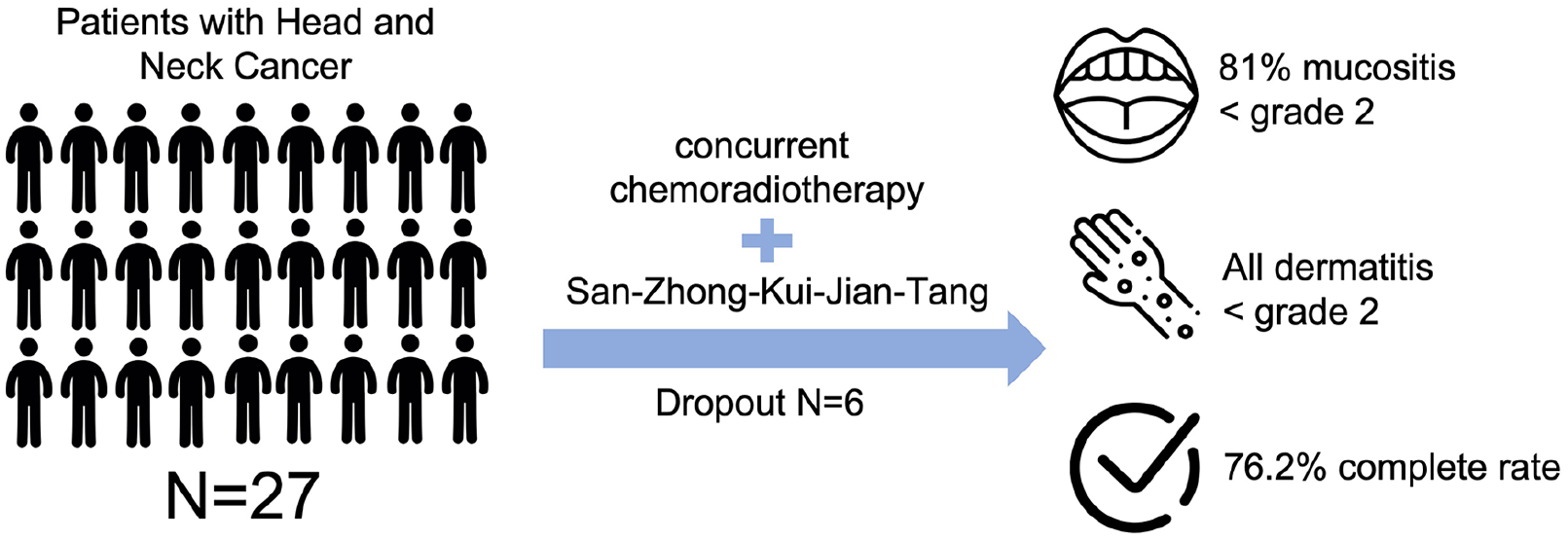
The objective of this study was to evaluate the efficacy of the traditional Chinese herbal medicine formula, San-Zhong-Kui-Jian-Tang (SZKJT), on patients with head and neck cancer who underwent concurrent chemoradiotherapy (CCRT). We performed a single-center, open-label, prospective feasibility study from 2018 to 2020. A total of 27 head and neck cancer patients who received CCRT were recruited for the study. SZKJT was given to patients simultaneously with CCRT for 9 weeks. The primary endpoint was the feasibility and completion rate of CCRT, while the secondary endpoint was occurrence of adverse effects. The quality of life (QoL) and traditional Chinese medicine body constitutions were measured by the QLQ-C30 and the Body Constitution Questionnaire (BCQ), respectively. There were 6 dropouts from the study due to complications, while 21 participants completed the trial. Among those 21 participants, 16 completed the CCRT treatment course, yielding a completion rate of 76.2%. The side effects observed during the CCRT and SZKJT trial included dermatitis in 4.8% (Grade 0), 57.1% (Grade 1), and 38.1% (Grade 2), and oral mucositis in 62.0% (Grade 1), 19.0% (Grade 2), and 19.0% (Grade 3) of participants. A comparison of the pre- and post-treatment QLQ-C30 scores revealed that QoL was unaffected (P = .506). However, the BCQ results showed significant increases in the Yin-Xu, Yang-Xu, and stasis constitutions (P < .001). No serious adverse events were observed due to SZKJT. Preliminary results indicated that additional SZKJT with CCRT was feasible, while noting a high completion CCRT rate (76.2%) among the SZKJT-treated patients. Our study reveals that SZKJT can effectively reduce the severity of dermatitis and oral mucositis associated with CCRT. Larger randomized controlled trials are required to further assess the efficacy and safety of SZKJT.
The trial registration number is NCT05590650 on ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT05590650
Graphical Abstract
Background
The incidence and death rates of head and neck cancer are increasing in Taiwan on an annual basis. According to National Health Administration statistics, head and neck cancer was ranked sixth among the top 10 cancer-related causes of death in 2020. 1 Meanwhile, in the ranking of the top 10 cancer-related deaths among males in 2020, head and neck cancer ranked fourth behind liver cancer, lung cancer and colorectal cancer. 1 Perhaps more alarmingly, it has the highest incidence rate of cancers among young males between the ages of 25 and 40 in Taiwan, with an average mortality age of 54, more than 10 years earlier than that of other cancers.
The primary treatments for head and neck cancer patients include surgery, radiotherapy (RT), and chemotherapy. Although radical surgery alone can cure early head and neck cancer, high-risk patients have a higher recurrence rate associated with pathologic features of positive or close margins and extra-nodular extension. The addition of concomitant cisplatin to postoperative RT improves local-regional control and disease-free survival. According to previous clinical trials, a high dose of cisplatin at 100 mg/m2 every 3 weeks (3-weekly cisplatin) concurrent with RT is considered the standard treatment regimen.2 -5
An optional treatment modality for patients with locally advanced or unresectable head and neck cancer is definitive chemoradiotherapy (CRT)/concurrent chemoradiotherapy (CCRT). In terms of organ preservation, the 3-weekly cisplatin and RT treatment provides a better laryngeal preservation rate. In addition, it has been reported that the overall survival (OS) rate was significantly better in the 3-weekly cisplatin and RT treatment than that of RT alone for head and neck patients with unresectable disease.6,7
Xerostomia and oral mucositis are 2 common side effects noted in head and neck cancer patients after RT and chemotherapy. Statistics show that the incidence of ulcerative mucositis may be up to 80%, while at least 40% of patients have stomatitis with a severity score of 3 to 4. 8 Several studies have reported that up to 77% of patients may suffer from severe side effects due to CCRT, 3 thereby reducing patients’ quality of life (QoL) and increasing the hospitalization rate 8 and potentially requiring the suspension of the RT course, 9 resulting in a prolonged treatment time. Meanwhile, the effect of local treatment deteriorates,10 -12 which subsequently affects the survival and recurrence rates in head and neck cancer patients.13,14
Current approaches to prevent or reduce the symptoms and severity of mucositis induced by cancer treatment include the combined use of cetuximab or amifostine during radiotherapy, modifying the radiotherapy course, prophylactic feeding line placement, benzydamine mouthwash, pilocarpine, and cryotherapy. However, no definitively effective treatment strategy is currently available.9,15
In traditional Chinese medicine (TCM), San-Zhong-Kui-Jian-Tang (SZKJT) is an herbal formula first identified in the Secret Treasure of the Orchid Chamber, a text written by Gao Li (AD1180-1251). SZKJT contains 16 herbs and is commonly used to treat abnormal swelling or nodules. Based on TCM theory, oral mucositis induced by cancer treatment is typically characterized by swelling, erythema, and ulceration of the oral mucosal tissues, resulting from an accumulation of heat toxins. SZKJT is believed to possess heat-clearing and detoxifying properties that may help alleviate inflammation, reduce swelling, and disperse nodules, potentially offering benefits in the treatment of mucositis.
The aim of this study was to explore the clinical efficacy and safety of SZKJT in improving the completion rate of CCRT in head and neck cancer patients, while evaluating the effectiveness of SZKJT in ameliorating the adverse side effects associated with CCRT and improving QoL.
Methods
Participants
The patients included in this study were recruited from the Department of hematology at China Medical University Hospital, Taichung City, Taiwan. The protocol was reviewed and approved by the Research Ethics Committee of China Medical University Hospital (CMUH 106-REC3-149). All participants were informed as to the purpose and procedures of the trial, and written informed consent was obtained prior to the trial.
The inclusion criteria of participants were: (1) age from 20 to 65 years; (2) initially diagnosed with head and neck cancer of stage II, III, or IV; (3) post-surgical adjuvant therapy with Cisplatin chemotherapy, or combined with radiotherapy; (4) or Cisplatin chemotherapy for those without surgery; (5) willingness to sign informed consent. The exclusion criteria were: (1) pregnancy; (2) non-Squamous-cell carcinoma; (3) second cancer or Carcinoma in situ in the past 5 years; (4) any evidence of metastasis; (5) abnormality of liver, kidney or bone marrow functions prior to treatments; (6) unstable vital signs; (7) episode of acute infection; (8) unclear consciousness for informed consent; (9) receiving other herbal, complementary, or acupuncture therapy.
The trial received IRB approval on March 12, 2018. The first participant was enrolled in May 2019, and the last participant completed the trial in June 2020 with follow-up to December 2020. The authors confirm that all ongoing and related trials for this intervention are registered in October 2022 (NCT05590650 at ClinicalTrials.gov).
Study Design
This was an open-label, non-randomized clinical trial. Patients diagnosed with head and neck cancer for the first time and willing to take SZKJT were recruited into this study. Under the standard treatment protocol for head and neck cancer, SZKJT was given for 9 weeks concurrently with CCRT. Evaluations were performed prior to CCRT and SZKJT and at the end of the treatment course. The evaluation index included basic information, side effect assessment with Common Terminology Criteria for Adverse Events (CTCAE), QoL with EORTC QLQ-C30 (Chinese Mandarin (Taiwan) version. Retrieved from [https://qol.eortc.org/questionnaire/eortc-qlq-c30/].), TCM Body Constitution Questionnaire (BCQ),16 -20 and monitoring of liver and kidney functions. The assessment of CTCAE adverse events was performed by cancer case managers. We used CTCAE Version 5.0, published on November 27, 2017. Due to budgetary constraints, we did not perform other immunochemistry analyses or additional follow-ups aside from those described in the original study protocol.
The CCRT in our study was carried out according with the principles listed here. The 3 courses of 3-weekly cisplatin 100 mg/m2 were arranged for the first, fourth and seventh week. A cumulative dosage of cisplatin at 200 mg/m2 was recognized as completion of the course. The dosage of cisplatin was adjusted according to variables shown in Table S1. RT was performed according to surgery status. For post-surgical patients, RT was performed with at least 60 Gy to the primary tumor bed, 60 to 70 Gy to the involved nodes, and 44 to 56 Gy to the non-invasive but potentially risky lymphatic area once a day, five times a week with 1.8 to 2.2 Gy each time. For those patients without surgery, RT was modified as 66 to 72 Gy to the primary tumor and involved nodes, and 44 to 56 Gy to the non-invasive but potentially risky lymph nodes. The entire course of CCRT took 7 to 8 weeks.
The SZKJT was given for 9 weeks concurrent with the start of CCRT. Participants took 5 g of concentrated herbal medicine powder containing SZKJT twice daily. Cessation of herbal medicine intake for over 10 days was considered as discontinuity.
The primary endpoint of the study was the completion rate of CCRT with concurrent SZKJT treatment. The secondary endpoints were adverse effects according to CTCAE, and the BCQ and QLQ-C30 questionnaires. Liver and kidney functions were monitored for adverse effects associated with SZKJT. Participants were not charged any fees during the study period.
Preparation of San-Zhong-Kui-Jian-Tang (SZKJT)
San-Zhong-Kui-Jian-Tang (SZKJT) contains 16 herbs: Scutellaria baicalensis Georgi, Gentiana scabra Bge., Trichosanthes kirilowii Maxim., Phellodendron chinense Schneid., Anemarrhena asphodeloides Bge., Platycodon grandiflorum Jacq., Laminaria japonica Aresch., Bupleurum chinense DC., Glycyrrhiza uralensis Fisch., Sparganium stoloniferum Buch.-Ham., Curcuma phaeocaulis Val., Forsythia suspensa (Thunb.) Vahl, Pueraria lobata (Willd.) Ohwi, Paeonia lactiflora Pall., Angelica sinensis (Oliv.) Diels, Coptis chinensis Franch., and Cimicifuga heracleifolia Kom. Every 9 g of concentrated SZKJT granules consisted of the 16 herbs with an original total weight of 30.5 g. The herbal product was analyzed for heavy metals, microorganisms, and pesticides according to the standards established by the government of Taiwan. The SZKJT samples with the lot number K2019023247 were made and packaged by Kaiser Pharmaceutical Co. Ltd., a firm that meets the good manufacturing practice (GMP) certification requirements of Taiwan. Each 5 g of concentrated SZKJT was sealed in an aluminum foil sachet.
Assessment
The BCQ is used by TCM physicians to assess patient body constitutions. The questionnaire consists of 44 questions with a 5-point Likert-type response scale. The results of the BCQ are used to categorize patients into 1 of 3 types of body constitution: Yang-Xu, Yin-Xu, and phlegm-stasis constitution. A higher score implies more significant deviations in terms of specific body constitution. Subjects who fail to be categorized into 1 of the 3 body constitution types are considered healthy. The BCQ was approved as an assessment by Professor Yi-Chang Su., Ph.D., Director of the National Research Institute of Chinese Medicine, Ministry of Health and Welfare, Taiwan.
The QoL score in this study was assessed using the European Organisation for Research and Treatment of Cancer (EORTC) Quality of Life of Cancer Patients questionnaire, QLQ-C30. A higher score in the 5 functioning scales of QLQ-C30 implies better functioning, while a higher symptomatic score indicates more severe symptoms.
The BCQ questionnaire was assessed by TCM physicians, while the QLQ-C30 and adverse events were documented and assessed by an oncologist and radiologist, respectively.
Statistics
On the QLQ-C30 questionnaire, the Cronbach’s confidence α were .906 and .891 before and after CCRT, respectively. The statistical analysis ran with type I error α = .05 using the statistical software package, SAS Version 9.4 (SAS Institute, Inc, Cary, NC). In addition, the paired sample t-test was used in this study.
The primary outcome of this study was the completion of the CCRT course of treatment in 7 weeks. According to previous reports, the completion rate of CCRT is approximately 70%, while the completion rate for the group receiving SZKJT in our study was expected to be 15% higher. By using the t-test with an alpha error of .05, a beta error of .05, and a standard deviation of 25%, at least 26 subjects were required for study participation with a dropout rate of 10%.
Results
We recruited 27 patients with an initial diagnosis of head and neck cancer. Throughout the study period, 4 participants dropped out due to the bitter taste of the SZKJT. Additionally, 2 patients interrupted the CCRT course due to complications; among them, 1 participant interrupted the CCRT due to an episode of pneumonia, while the other patient ceased the treatment due to adverse effects of CCRT. Thus, 21 participants (77.8%) completed the trial with concurrent treatments (Figure 1).
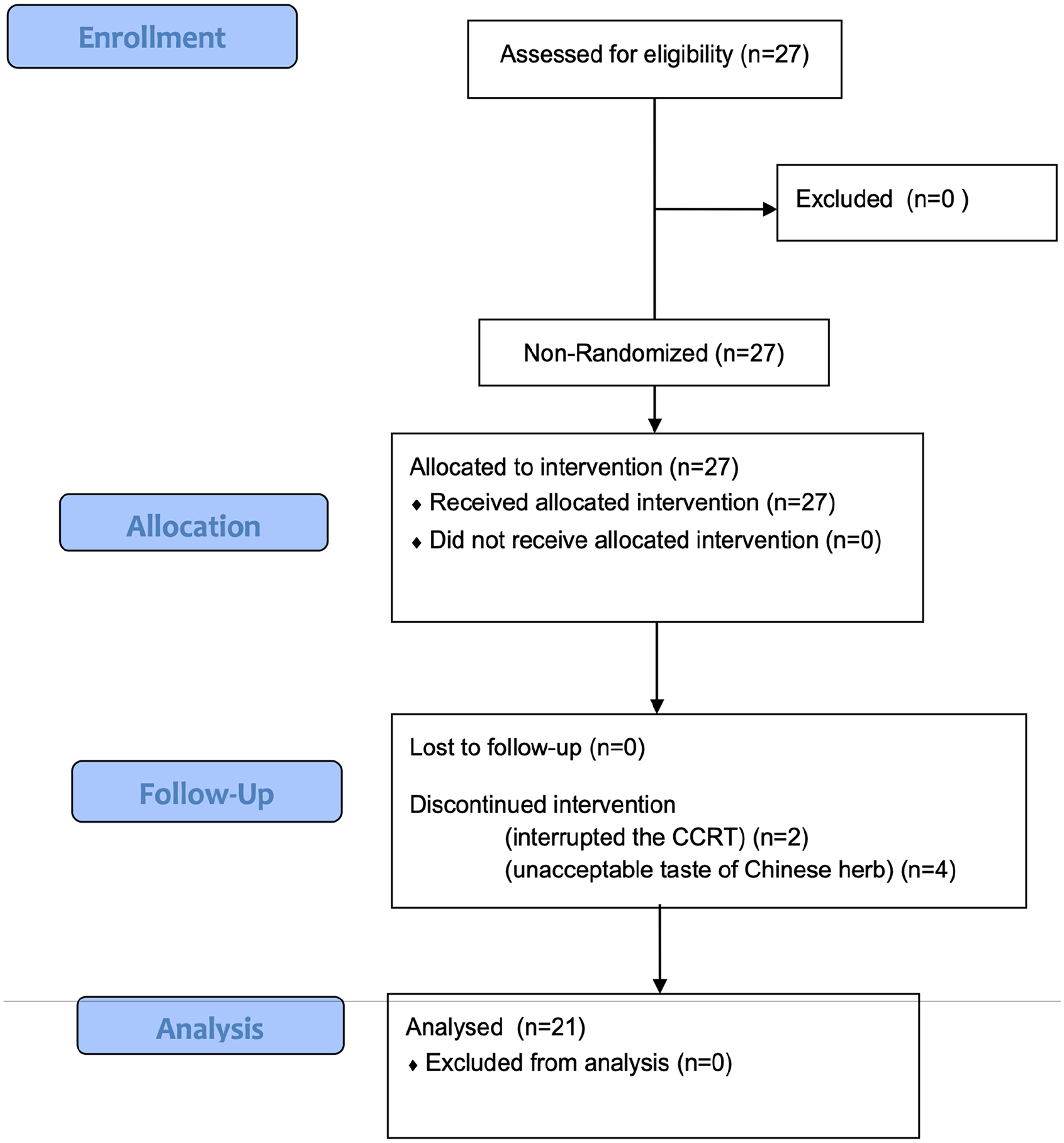
The CONSORT flow diagram. Flow diagram of the open-label, non-randomized clinical trial in head and neck cancer patients receiving concurrent CCRT and SZKJT.
The basic demographic characteristics of the study participants are shown in Table 1. A majority of the study population (95.2%) was male. In terms of education level, 71.4% of our participants had completed junior and/or senior high school. Approximately half of the participants were below a normal Body Mass Index (BMI), while 33.3% of subjects were defined as overweight or obese. Most participants recorded a good performance on the Eastern Cooperative Oncology Group (ECOG) evaluation, ranging from 0 to 1 (95.3%). A vast majority of the participants were former or current smokers (90.5%), alcohol consumers (85.7%) or betel nut users (81.0%). In terms of the specific cancer diagnosis, 81.1% of the participants were oral cavity cancer patients, while 90.5% of the participants were metastatic with clinical stage IV cancer. In addition, 4 (19%) of the subjects received the therapy as an adjuvant treatment due to high-risk pathology features. Among the 21 study participants, 16 received a cumulative dosage of cisplatin over 200 mg/m2 and completed the CCRT treatment course. Among the 5 subjects whose cumulative dosage of cisplatin was lower than 200 mg/m2, 4 of them completed the CCRT treatment course. Hence, the CCRT completion rate was 76.2% (16/21).
Demographic Data of Patients Receiving SZKJT with CCRT (n = 21).

Abbreviations: BMI, body mass index; ECOG, Eastern Cooperative Oncology Group.
The adverse effects observed in study participants during the CCRT and SZKJT treatments were monitored and categorized according to the CTCAE grading, as shown in Table 2. Over 60% of subjects had oral mucositis (n = 13) or dermatitis (n = 13) of Grade 0 to 1; in addition, 19.0% (n = 4) of subjects had oral mucositis of Grades 2 and 3. Of note, no oral mucositis of Grades 4 or 5 were observed in our study. Dermatitis of Grade 2 was noted in 38.1% (n = 8) of subjects; however, no severe dermatitis of Grade 3 or higher was observed. Other adverse effects noted in our study included renal dysfunction (9.5%), hyponatremia (9.5%), anemia (9.5%), acid regurgitation (14.3%), and hiccup (4.8%). No subjects had events of infection, elevation of transaminase, leukopenia, or febrile neutropenia. The most common side effects noted in our study were oral mucositis and dermatitis, although these cases were mild to moderate in severity.
Adverse Events Noted in Patients Treated with CCRT and SZKJT.

We also used questionnaires to monitor and assess the QoL and body constitutions of study participants both before and after the CCRT and SZKJT treatment, as shown in Table 3. For statistical analysis, we applied the paired sample t-test. In the BCQ, the scores in the 3 types of body constitutions were significantly higher after CCRT and SZKJT. Prior to CCRT, 25% (n = 6), 4.2% (n = 1), and 8.3% (n = 2) of participants were categorized in the Yin-Xu, Yang-Xu and stasis constitutions, respectively. However, after the CCRT and SZKJT treatment, the proportion of participants categorized in the Yin-Xu, Yang-Xu, and stasis constitutions increased to 70.8% (n = 17), 29.2% (n = 7), and 41.7% (n = 10), respectively. As for the QoL scores, no significant change was noted in the global QoL scores after the CCRT and SZKJT treatment. However, significant decreases were observed in the Mental Health assessment and Role Functioning in the QLQ-C30. Scores for fatigue (P < .001), pain (P < .001), insomnia (P = .030), appetite loss (P = .001), and constipation (P = .017) significantly increased after CCRT and SZKJT. We further compared liver and kidney functions before and after CCRT and SZKJT, as shown in Table 3. There were no significant changes noted in serum aspartate aminotransferase (AST) and serum alanine transaminase (ALT) activities. However, a decrease in the glomerular filtration rate (GFR) was observed, while no accompanying increase in blood urea nitrogen (BUN) was noted. Hence, the decrease in GFR may not be clinically relevant. During the entire course of treatment, no adverse effects related to SZKJT were observed.
Comparison Between Pre-treatment and Post-treatment CCRT and SZKJT.

Abbreviations: BCQ, body constitution questionnaire; QLQ-C30, EORTC quality-of-life questionnaire; BUN, blood urea nitrogen; GFR, glomerular filtration rate; AST, serum aspartate aminotransferase; ALT, serum alanine transaminase.
These results suggest that the combined CCRT and SZKJT induced significant disturbances to the participants’ body constitutions. However, the global QoL scores and certain functioning scales of the participants were unaffected after the treatment.
Discussion
Traditional Chinese medicine (TCM) is covered by the National Health Insurance system of Taiwan, while it remains a common form of alternative and adjunctive therapy with a utility rate of approximately 30% among the general population. 21 In terms of cancer treatments, a retrospective cohort study reported a higher survival outcome in patients with head and neck cancer who used TCM as an adjunctive therapy. 22 In addition, several systematic reviews have found that TCM combined with Western medicine can prolong the survival rate in nasopharyngeal cancer patients, improve the QoL, 23 reduce the oral and skin-related side effects caused by radiotherapy, regulate immunity, 24 and reduce distant metastasis. 25
According to TCM theory, CCRT causes a type of heat/fire evil, wastes Qi and depletes Yin in the patient’s body. Accordingly, a study by Hsu et al 26 of 30 patients with head and neck cancer used different herbal formulas aimed at treating the side effects associated with CCRT. Although the severity of xerostomia decreased, between 50% and 60% of subjects had moderate to severe mucositis and dermatitis in their study. By contrast, we here used the SZKJT formula, which can not only clear heat and resolve toxins but also disperse swelling and binds. In our single-arm study, approximately 60% of subjects had no instance or only a mild degree of mucositis and dermatitis (Table 2). Meanwhile, a separate study by Tsan et al 27 reported a greater level of toxicity in subjects receiving a low weekly dose of cisplatin treatment in comparison to those receiving a high dose every 3 weeks. More specifically, severe mucositis (Grade 3 or higher) ranged from 38.5% (high dose) to 75% (low dose), while severe dermatitis (Grade 3 or higher) was observed in 7.7% (high dose) to 8.3% (low dose) of patients. In a study involving patients with head and neck carcinomas of stage III and IV with a protocol of high-dose cisplatin every 3 weeks plus radiotherapy, 32.4% of patients had severe mucositis and 8.6% had severe dermatitis. 28 In our study, we applied protocols of low- and high-dose cisplatin (30-40 mg/m2 per week vs 100 mg/m2 every 3 weeks) plus RT. With similar cumulative dosage of cisplatin and radiation, our study showed that only 19% of subjects had severe mucositis while no cases of severe dermatitis occurred (Table 2). These results may reasonably be attributed to the effects of SZKJT.
SZKJT is a TCM formula comprised of 16 herbs in which several active ingredients have previously been identified, including baicalin, berberine, glycyrrhizin, gentiopicroside, palmatine, and mangiferin. 29 Several previous studies have reported on the potential therapeutic efficacy of SZKJT in breast, colon and hepatic carcinomas. In one study, SZKJT exhibited effective cell growth inhibition by inducing breast cancer cells to undergo G0/G1 phase arrest and apoptosis through induction of the p21/WAF1 pathway. 30 In addition, the proliferation of human HepG2 cells was inhibited by SZKJT by induction of apoptosis and autophagy.31,32 Furthermore, a retrospective cohort study reported that among breast cancer patients, those taking SZKJT had a 50% reduced risk of chronic hepatitis compared with non-SZKJT users. 33 Indeed, it has been hypothesized that certain herbal medicines may act to regulate the tumor microenvironment and cancer cell proliferation. 34 This is the first study demonstrating the adjuvant potential of SZKJT to alleviate the side effects induced in head and neck cancer patients undergoing CCRT.
In clinical practice, patient compliance with the treatment plan is a critical factor underlying therapeutic effectiveness. In our study, 76.2% of subjects completed the course of CCRT, which is similar to previous reports indicating compliance rates ranging from 62.5% to 88.5%.3,27 Although the original assumption was that SZKJT would improve the completion rate by 15%, the actual result only reached 76.2%. Our findings suggest that while SZKJT may reduce the side effects of CCRT, its bitter taste appears to introduce a new challenge, impacting patient adherence and contributing to an increased dropout rate. This ultimately resulted in a slight shortfall from the anticipated target completion rate.
It has been reported that the BCQ used in this study may accurately identify certain diseases including hypertension, diabetes, and stroke. 35 It is also reported that Yang-Xu and Qi-Xu are significantly associated with cancer-related fatigue.36,37 Our study further demonstrates increased rates of Yang-Xu, Yin-Xu, and stasis constitutions after CCRT (Table 3). Yang-Xu and Yin-Xu means the deficiency of Yang and Yin in the body. Meanwhile, although our study indicates no significant changes to certain items of the QLQ-C30, the factor related to the relatively small sample size should be taken into consideration.
There are several limitations to the current study. First, this single-arm study with a limited number of cases is only suitable for evaluating the feasibility of applying SZKJT during CCRT. Due to the lack of an appropriate control group and sufficient sample size, it is challenging to clearly determine whether the treatment effects are derived from the intervention itself, the natural course of the disease, placebo effects, or other potential factors, making it difficult to establish direct causality. Furthermore, the study results cannot ascertain whether the treatment is more effective compared to other therapies. The absence of a control group also increases the risk of selection bias, which may affect the reliability of the results. Large-scale randomized controlled trials are needed in the future to confirm the efficacy of SZKJT. Second, whether SZKJT induces or aggravates other adverse effects related to CCRT is unclear. Third, this study lacks long-term follow-up data, making it difficult to assess whether the therapeutic effects of SZKJT are sustained. Additionally, due to potential delayed or long-term adverse effects, the long-term safety of SZKJT cannot be assured, and the results should therefore be interpreted conservatively. Insight into the long-term effects of SZKJT on QoL and the survival rate warrant further investigation. Fourth, the dropout rate in our study was 22.2% (6 out of 27 participants), with most dropouts (4 out of 6) due to an unwillingness to tolerate the bitter taste of SZKJT. After adjusting for the reduced sample size, the recalculated effect size was 0.636, resulting in a statistical power of approximately 0.79. Although this is slightly below the recommended threshold of 0.8, it still offers a reasonable basis for detecting meaningful effects within the study.
Conclusions
We herein demonstrate that SZKJT may be an effective adjunctive therapy to ameliorate adverse side effects and improve treatment tolerance in head and neck cancer patients receiving CCRT. Further double-blinded randomized controlled trials are warranted to more accurately evaluate the clinical effects of SZKJT.
Supplemental Material
sj-pdf-1-ict-10.1177_15347354251326946 – Supplemental material for Chinese Herbal Medicine for Concurrent Chemoradiation-Induced Adverse Events in Head and Neck Cancer Patients: A Prospective Feasibility Study
Supplemental material, sj-pdf-1-ict-10.1177_15347354251326946 for Chinese Herbal Medicine for Concurrent Chemoradiation-Induced Adverse Events in Head and Neck Cancer Patients: A Prospective Feasibility Study by Hsuan-Ting Tang, Shi-Chen Ou, Chien-Lun Chu, Ai-Lin Lee, Hung-Jen Lin, Ying-Chun Lin, Yao-Ching Wang, Shih-Neng Yang and Ming-Yu Lien in Integrative Cancer Therapies
Supplemental Material
sj-pdf-2-ict-10.1177_15347354251326946 – Supplemental material for Chinese Herbal Medicine for Concurrent Chemoradiation-Induced Adverse Events in Head and Neck Cancer Patients: A Prospective Feasibility Study
Supplemental material, sj-pdf-2-ict-10.1177_15347354251326946 for Chinese Herbal Medicine for Concurrent Chemoradiation-Induced Adverse Events in Head and Neck Cancer Patients: A Prospective Feasibility Study by Hsuan-Ting Tang, Shi-Chen Ou, Chien-Lun Chu, Ai-Lin Lee, Hung-Jen Lin, Ying-Chun Lin, Yao-Ching Wang, Shih-Neng Yang and Ming-Yu Lien in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors would like to thank James Waddell for the critical reading and revision of our manuscript.
Author Contributions
Conceptualization: S-C Ou, A-L Lee, Y-C Wang, M-Y Lien. Data curation: H-J Lin, S-N Yang. Formal analysis: C-L Chu. Investigation: S-C Ou, A-L Lee, Y-C Lin, Y-C Wang, M-Y Lien. Methodology: S-C Ou, A-L Lee, M-Y Lien. Supervision: H-J Lin, M-Y Lien. Writing–original draft: H-T Tang, S-C Ou, C-L Chu. Writing–review & editing: M-Y Lien.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported and funded by China Medical University Hospital [DMR-107-158 and DMR-110-009] and National Science and Technology Council, Taiwan [NSTC113-2320-B-039-01].
Ethical Considerations
The protocol was reviewed and approved by the Research Ethics Committee of China Medical University Hospital (CMUH 106-REC3-149).
Consent to Participate
All participants were informed as to the purpose and procedures of the trial, and written informed consent was obtained prior to the trial.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
