Abstract
Gastrointestinal (GI) cancer stands as one of the most prevalent forms of cancer globally, presenting a substantial medical and economic burden on cancer treatment. Despite advancements in therapies, it continues to exhibit the second highest mortality rate, primarily attributed to drug resistance and post-treatment side effects. There is an urgent need for novel therapeutic approaches to tackle this persistent challenge. Scutellaria baicalensis, widely used in Traditional Chinese Medicine (TCM), holds a profound pharmaceutical legacy. Modern pharmacological studies have unveiled its anticancer, antioxidant, and immune-enhancing properties. S. baicalensis contains hundreds of active ingredients, with flavonoids, polysaccharides, phenylethanoid glycosides, terpenoids, and sterols being the principal components. These constituents contribute to the treatment of GI cancer by inducing apoptosis in tumor cells, arresting the cell cycle, inhibiting tumor proliferation and metastasis, regulating the tumor microenvironment, modulating epigenetics, and reversing drug resistance. Furthermore, the utilization of modern drug delivery technologies can enhance the bioavailability and therapeutic efficacy of TCM. The treatment of GI cancer with S. baicalensis is characterized by its multi-component, multi-target, and multi-pathway advantages, and S. baicalensis has a broad prospect of becoming a clinical adjuvant or even the main therapy for GI cancer.
Introduction
The global incidence and mortality rates of gastrointestinal(GI) cancer are on a steady rise, with projected new cases and deaths expected to increase by 73% and 58% respectively, reaching 7.5 million and 5.6 million by the year 2040. 1 GI cancer encompasses a spectrum of cancers originating from various parts of the gastrointestinal tract, including esophageal cancer (EC), gastric cancer (GC), hepatocellular carcinoma (HCC), gallbladder cancer, pancreatic cancer (PC), colorectal cancer (CRC), and others. According to the most recent statistics released by the International Agency for Research on Cancer (IARC) in 2022, lung cancer remains the most prevalent form of cancer globally, accounting for 12.4% of total cases, which is followed by female breast cancer at 11.6%, CRC at 9.6%, prostate cancer at 7.3%, and GC at 4.9%. In terms of mortality, lung cancer contributes to approximately 19% of all cancer-related deaths, followed by CRC at 9.3%, HCC at 7.8%, female breast cancer at 6.9%, and GC at 6.8%. PC and EC rank as the sixth and seventh leading causes of global cancer mortality. 2 It can be seen that GI cancer is at the forefront of global cancer incidence and mortality, which brings a great burden to global cancer treatment. With the evolving lifestyle and the impact of an aging population, the prevalence of GI cancer is on the rise in China, making effective GI cancer treatment crucial for clinical guidance. 3
Over the past two decades, significant advancements have been made in the treatment of GI cancer. While surgery and chemotherapy remain cornerstone therapies, targeted therapy, radiotherapy, and immunotherapy have emerged as important treatment modalities, garnering increasing attention. These novel approaches have contributed to improving the survival rates of patients with GI cancer. However, despite these advancements, GI cancer continues to be a leading cause of cancer-related deaths globally. 4 Chemotherapy, while often the primary treatment for GI cancer, can bring about severe adverse reactions such as liver and kidney damage, bone marrow suppression, digestive tract issues, and cardiotoxicity. Additionally, chemotherapy resistance in later stages can promote tumor recurrence and metastasis, significantly impacting patients’ quality of life. Consequently, many cancer patients seek TCM as an alternative therapy. TCM has shown promise in reducing complications from radiotherapy, chemotherapy, and targeted therapy, while also improving overall quality of life, extending survival time, alleviating immediate tumor symptoms (such as fatigue, depression, chronic pain, anorexia/cachexia, insomnia), and enhancing Karnofsky score. 5 Therefore, in-depth study of the active components and potential anticancer mechanisms of TCM to improve the survival rate and quality of life of tumor patients is the direction and purpose of cancer research.
Scutellaria baicalensis (Figure 1), a perennial herb belonging to the family Labiatae, has been utilized as a medicinal herb for over 2000 years and stands as one of the most extensively used TCM. Its natural habitat spans regions including Japan, North China, Mongolia, and Russia. 6 As an increasingly popular herb with great medicinal value, S. baicalensis has made important research progress in recent years. The modern pharmacology has shown that S. baicalensis has anticancer, antioxidant, anti-inflammatory, antiviral, neuroprotective, hepatoprotective, and immune-enhancing pharmacological effects. 7 To date, more than 40 compounds have been isolated from S. baicalensis, of which flavonoids and flavonoid glycosides are the mainstay of S. baicalensis in exerting its pharmacological effects, which have exerted significant antitumor activity in both cancer cell lines and animal models. 8 Studies have found that compounds in S. baicalensis can prevent and treat GI cancer in a variety of ways. 9 In this review, we will summarize the main active components of S. baicalensis, and its molecular mechanisms against GI cancer.

Pictures of S. baicalensis plants and herbal slices.
Active Ingredients of S. baicalensis
S. baicalensis contains a diverse array of bioactive constituents beyond flavonoids. These include terpenoids, polysaccharides, phenylethanoid glycosides, and various other compounds. It is through the collective action of these active ingredients that S. baicalensis is able to exert a broad spectrum of pharmacological effects. 10
Flavonoids
Flavonoids present in S. baicalensis encompass various subclasses, including flavones, flavanones, flavonols, dihydroflavonoids, dihydroflavonols, dihydrochalcones, and numerous others. Among these, the four most extensively studied flavonoids are baicalin, baicalein, wogonoside, and wogonin. 11 Li et al identified 15 flavonoids from the above-ground parts of S. baicalensis. Among them, eight flavonoids were found to constitute 75% of the total flavonoid content. These high-content flavonoids, listed in descending order of abundance, were as follows: baicalein, baicalin, scutellarin, apigenin 7-O-β-D-glucoside, chrysin 7-O-β-D-glucuronide, carthamidin 7-O-β-D- glucuronide, apigenin, and chrysin. Among these, baicalein, baicalin, and scutellarin were identified as the primary active constituents of S. baicalensis and played a pivotal role in its antitumor activity. 12 Indeed, flavonoids exhibit anticancer activity through various mechanisms. They induce apoptosis of cancer cells, inhibition of cancer cell proliferation, metastasis and angiogenesis, regulation of the tumor microenvironment, Additionally, flavonoids regulate cancer-related signaling pathways such as AKT/mTOR, WNT/β-catenin, PTEN/PI3K/AKT/mTOR, JNK/ERK/p38 MAPK, etc. 13
Polysaccharides
Olennikov identified five polysaccharides from the above-ground parts of S. baicalensis, designated as WSPS-1, WSPS-2, WSPS-3, WSPS-4, and WSPS-5. 10 Polysaccharides have been shown to exert anti-tumor effects directly by inducing cell cycle arrest and apoptosis. Moreover, they indirectly inhibit tumors by modulating the host immune system, activating nonspecific or specific immune responses. Additionally, certain polysaccharides can regulate the tumor microenvironment, inhibiting the proliferation and metastasis of tumor cells. 14
Phenylethanoid glycosides
Nine phenylethanoid glycosides have been identified in S. baicalensis, including plantainoside C, calceorioside B, isomartynoside, acteoside (verbascoside), desrhamnosyl acteoside, isocrenatoside, eutigoide A, verbascoside, and osmanthuside B6. 6 Phenylethanoid glycosides possess the capability to inhibit the proliferation of tumor cells by blocking the cell cycle, promoting cell differentiation, inducing apoptosis or autophagy, and regulating signaling pathways. 15
Terpenoids
S. baicalensis is rich in terpenoids, primarily diterpenoids, with over 100 diterpenoids isolated to date. These compounds exhibit a range of pharmacological effects, including antifungal, anticancer, and anthelmintic properties. 10 The anticancer mechanism of diterpenoids is multi-faceted and includes induction of apoptosis in cancer cells, inhibition of the cell cycle, enhancement of the anti-tumor activity of lymphocytes, suppression of angiogenesis, inhibition of the anti-inflammatory effects mediated by NF-κB, and blockade of various carcinogenic and anti-apoptotic signaling pathways. 16
Other components
The volatile oil constituents of S. baicalensis primarily include acetophenone, l-phenyl-1,3-butanedione, palmitic acid, and oleic acid. 17 Additionally, fourteen amino acids have been reported in S. baicalensis, with proline accounting for up to 80% of the total amino acid content. Sterols found in S. baicalensis mainly include α-spinasterol and beta-sitosterol. Furthermore, the plant contains various carotenoids such as β-carotene, lutein, phytoene, zeaxanthin, and ξ-carotene. 18
Mechanism of Action of Bioactive Compounds From S. baicalensis in Anti-Tumor Activity
Induction of tumor cell apoptosis
Caspases are pivotal regulators of the apoptotic process, orchestrating cell death through various pathways. Three key pathways capable of inducing tumor cell apoptosis via caspase activation include the intrinsic (or mitochondrial) pathway, the extrinsic (or death receptor) pathway, and the endoplasmic reticulum (ER) pathway. 19 In the extrinsic apoptosis pathway, death ligands bind to death receptors, culminating in the formation of the death-inducing signaling complex (DISC). This complex activates pro-caspase-8, initiating downstream caspase cascades that trigger apoptosis. 20 Under conditions such as hypoxia, elevated cytoplasmic Ca2+ levels, and oxidative stress, the intrinsic mitochondrial pathway is activated. This involves the modulation of pro- and anti-apoptotic proteins within the Bcl-2 family, ultimately leading to the release of pro-apoptotic factors from mitochondria and the initiation of caspase cascades, culminating in apoptosis. 21 When cellular stress damages the ER, the dissociation of TNF receptor-associated factor 2 (TRAF2) from procaspase-12 occurs, activating procaspase-12, and subsequently inducing apoptosis. 22
Baicalin is one of the most abundant flavonoids found in the dried roots of S. baicalensis, and has been shown to have anti-inflammatory, anti-viral, anti-tumor, anti-bacterial, anti-oxidant, hepatoprotective and neuroprotective effects. 23 Baicalin decreased the expression of Bcl-2 and increased the levels of caspase-3, caspase-9 and Bax, leading to apoptosis in BGC-823 and MGC-803 cells in GC. 24 It was found that the incidence of HCC was significantly higher within 5 years of the diagnosis of Type 2 diabetes (T2D) compared with healthy individuals, and that hyperglycemia resulted in increased HKDC1 protein levels and apoptotic pathways JAK2/STAT1/caspase-3 was inhibited, which was reversed by application of baicalin thereby promoting apoptosis in T2D-HCC. 25 After treatment with baicalein, CRC cells HCT116 and SW480 exhibited increased expression of apoptotic markers, while the level of senescence-associated β-galactosidase (SA-β-Gal), a marker of cell senescence, was significantly elevated following treatment with baicalin. The regulation of apoptosis and senescence in CRC cells induced by baicalein and baicalin involves the MAPK ERK and p38 signaling pathways. 26 Indeed, specificity protein 1 (Sp1) acts as an oncogene, promoting the proliferation, invasion, and metastasis of tumor cells. In CRC SW480 cells, baicalin has been shown to down-regulate Sp1 expression. This down-regulation of Sp1 results in the upregulation of cleaved caspase-3 (C-caspase-3) and cleaved poly (ADP-ribose) polymerase (C-PARP), ultimately triggering apoptosis. 27
Scutellarein has anti-inflammatory, antioxidant, antiviral, neuroprotective, hypoglycemic, hypolipidemic, anticancer and cardiovascular protective properties. 28 The tumor suppressor p53 and the inhibitor of apoptosis proteins (IAPs) both play a role in regulating the process of apoptosis. MDM2 interacts with the p53, leading to its degradation. Scutellarein has been found to promote apoptosis in GC cells AGS and SNU-484 by down-regulating MDM2 and members of the IAPs family (cIAP1, cIAP2, and XIAP), as well as activating p53. 29 Scutellarein has been demonstrated to upregulate the expression of Bax and Cytochrome C while downregulating Bcl-2, Wnt1, cytoplasmic β-catenin, and basal cytoplasmic β-catenin levels in GC cells. Inhibition of the Wnt/β-catenin pathway promotes apoptosis in GC cells. 30 Scutellarein treatment promoted the ubiquitination of the receptor for advanced glycosylation end products (RAGE) protein in CRC SW480 cells, resulting in a decrease in RAGE protein expression. Overexpression of RAGE significantly inhibited scutellarein’s ability to increase cleaved Caspase-3/7 levels, and CDC4 facilitated the degradation of RAGE protein. Thus, scutellarein induced apoptosis of CRC cells by upregulating CDC4 to mediate RAGE ubiquitination. 31
Oroxylin A is a flavonoid obtained from the plants Oroxylum indicum, S. baicalensis, and S. lateriflora with anti-inflammatory, antibacterial, antiviral, and anticancer potential. It has broad prospects in the treatment of cancer, cardiovascular diseases, inflammation, nervous system diseases, osteoarthritis, and so on. 32 The induction of apoptosis in gallbladder cancer cells by oroxylin A is primarily accomplished through the inhibition of the PTEN/PI3K/AKT signaling pathway. 32 The compound oroxylin A induces apoptosis in HCC cells by activating caspase-8 and promoting proteolysis of p62/SQSTM1. 33 CDK9 is frequently overexpressed in HCC tissues, where both MDM2 and SIRT1 serve as substrates for phosphorylated CDK9. The restoration of wild-type (wt)-p53 function has been shown to inhibit HCC growth. Oroxylin A treatment reduces the expression of CDK9, phosphorylated SIRT1 and MDM2 proteins. Conversely, the level of wild-type p53 (wt-p53) proteins is significantly increased, leading to the promotion of apoptosis in HCC cells. 34
Salidroside mainly exists in the root of Rhodiola spp, and has pharmacological effects such as anti-cancer, anti-oxidation, anti-aging, anti-diabetes, anti-depression, anti-hyperlipidemia, anti-inflammation, and immune regulation. 35 Salidroside is capable of inducing both endogenous and exogenous cell apoptosis in HCC HepG2 cells by activating caspase-3, caspase-8, and caspase-9, up-regulating Bax and Cytochrome c, as well as decreasing Bcl-2 levels. Additionally, salidroside also up-regulates the expression of p-PERK, eIF2a, ATF-6, and CHOP, thereby promoting apoptosis of HCC cells through ER stress. 36 The compound acteoside, a phenylethanol glycoside isolated from many dicotyledonous plants, has antioxidant, anti-inflammatory, anti-cancer, neuroprotective, cardiovascular protective, liver protective, and anti-microbial effects. 37 Acteoside downregulates the expression of Bcl-2 while upregulating phosphorylated c-Jun N-terminal kinase (p-JNK) and light chain 3 (LC3). This modulation of the JNK signaling pathway promotes both autophagy and apoptosis in HCC cells. 38 Kallikrein-related peptidase (KLK) serves as a biomarker for both the diagnosis and prognosis of HCC. Elevated levels of KLK are associated with tumor cell growth, invasion, and angiogenesis. Treatment with acteoside significantly reduces the mRNA expression of KLK. Additionally, p53, a tumor-suppressor protein, induces apoptosis in tumor cells. Acteoside exerts anti-HCC effects by upregulating p53 expression and inhibiting KLK expression, thereby suppressing angiogenesis and tumor progression. 39
Wogonin has a variety of pharmacological activities, including antiviral, anti-inflammatory, antioxidant, neuroprotective, and anti-diabetic related to disease prevention. 40 After treatment with wogonin, the levels of cleaved caspase 3 and cleaved caspase 9 increased significantly in CRC HCT-116 cells. Additionally, wogonin induced ER stress, which led to the localization of p53 to the cytoplasm and an increase in the total level of p53. This modulation promoted apoptosis and inhibited autophagy in CRC HCT-116 cells. 41 Wogonin downregulates phosphorylated IκB (p-IκB) and phosphorylated p65 (p-p65), thereby inhibiting the NF-κB/Bcl-2 pathway. This inhibition results in reduced levels of the anti-apoptotic protein Bcl-2 and induces apoptosis in HCC Bel7402 cells. 42 Iron-induced cell death can trigger apoptosis in tumor cells. Wogonin increases the level of Fe2+ and reduces the expression of iron death suppressor genes. The downregulation of Nuclear factor E2-related factor 2 (Nrf2) makes cancer cells more susceptible to iron-induced cell death. Given that GPX4 is a downstream target of Nrf2, wogonin could induce iron-induced cell death in PC cells by inhibiting the Nrf2/GPX4 axis. 43
Chrysin is found in many plants, honey and propolis and has antioxidant, anti-inflammatory, anticancer and antiviral activities. 44 Hexokinase 2 (HK2), a crucial glycolytic enzyme, serves as a marker of poor prognosis in HCC. Treatment with chrysin results in decreased expression of HK2, facilitating the binding of Bax to VDAC. This interaction forms the VDAC-Bax complex, which subsequently elevates the levels of cytochrome C and other pro-apoptotic proteins. Consequently, chrysin inhibits glycolysis in HCC by targeting HK2 and induces apoptosis in HCC cells. 45 Chrysin treatment significantly upregulates the expression of Ten-eleven translocation 1 (TET1) in GC cells. Moreover, overexpression of TET1 has been shown to promote apoptosis. 46 Chrysin initiates the intracellular caspase signaling cascade by activating ERK, JNK, and P38 signaling molecules in the MAPK signaling pathway. This activation ultimately leads to typical apoptotic morphology changes and PARP cleavage. Thus, chrysin promotes apoptosis in HCC SMMC-7721 cells through the MAPK signaling pathway. 47 Apigenin, which is found in vegetables, fruits and herbs, can be used to treat diseases such as inflammation, autoimmune, and cancer. 48 Apigenin induces apoptosis of GC HGC-27 cells by regulating the Akt/Bad/Bcl2/Bax axis. 49 Additionally, in HCC Hep3B cells, apigenin induces apoptosis by upregulating TNF-α, Bax, cleaved PARP, and caspase 3, while downregulating Bcl-xl, p-p65, and IκB. Moreover, apigenin activates the apoptotic markers RIP3, p-RIP3, and p-MLKL. 50
Other components of S. baicalensis can also induce apoptosis. For instance, Scutellaria. barbata polysaccharide (SBP) extracted from S. baicalensis has the ability to upregulate p53 and Bax/Bcl-2, thereby promoting apoptosis in HCC cells. 51 Stigmasterol is an unsaturated phytosterol that is found in a variety of natural sources and has powerful pharmacological effects, such as anti-cancer, anti-inflammatory, anti-diabetic, immunomodulatory, anti-parasitic, antibacterial, antioxidant and neuroprotective properties. 52 In HCC HepG2 cells, stigmasterol upregulates Bax and p53 while downregulating the level of Bcl-2 protein. Moreover, it reduces mitochondrial membrane potential and increases intracellular Ca2+ flow, ultimately leading to apoptosis induction. 53 Activation of AMPK can up-regulate p53 and p21 proteins, activate caspase to promote apoptosis, and phosphatase and tensin homolog (PTEN) can inhibit phosphorylation of PI3 kinase. β-sitosterol exists widely in plants and has biological effects such as anti-anxiety, sedation, analgesia, immune regulation, antibacterial, anticancer, and liver protection. 54 β-sitosterol induces apoptosis in GC AGS cells by up-regulating the expression of PTEN and phospho-AMPK (p-AMPK), while down-regulating Hsp90 protein. 55
Autophagy serves to maintain cellular homeostasis by facilitating lysosomal degradation and the removal of damaged organelles, thereby promoting cell survival. However, under conditions of excessive cellular stress, autophagy can promote cell death. As such, autophagy possesses dual roles in cancer biology. On one hand, it can inhibit apoptosis and promote tumor progression. On the other hand, it can promote apoptosis to exert an anti-tumor effect. 56 The total flavonoid aglycone extracted (TFAE) induces apoptosis and autophagy by inhibiting the PI3K/Akt/mTOR pathway. Interestingly, the pro-apoptotic effect of TFAE is enhanced after the application of the autophagy inhibitor 3-MA. This suggests that TFAE-stimulated autophagy hinders apoptosis in PC cells. 57 Salidroside triggers autophagy and apoptosis in HCC via the PI3K/Akt/mTOR signal transduction. Chloroquine diphosphate (CQ) inhibits autophagy and promotes mitochondrial apoptosis induced by salidroside. Therefore, salidroside-mediated autophagy may be a protective mechanism. 58 Scutellarin is an herbal flavonoid glucuronide with a variety of beneficial effects such as antioxidant, anti-inflammatory, antiplatelet, anticancer, and cardioprotective properties, which is clinically used in the treatment of stroke, myocardial infarction and cancer. 59 Scutellarin promotes up-regulation of LC3II, Beclin 1 autophagy protein and apoptosis-related proteins Caspase 3, 8, 9 and Bax expression, decreases Bcl-2 to induce apoptosis in human CRC cells. 60 Apigenin can induce autophagic cell death in AGS cells of GC through three mechanisms: up-regulation of LC3-II, ATG5, AMPK, and ULK1; activation of protein kinase R-like endoplasmic reticulum kinase (PERK) signal transduction; down-regulation of HIF-1α and Ezh2 expression under hypoxia. 61 Apigenin upregulates Beclin-1 and LC3-II while downregulating p62. Additionally, it inhibits the mTOR/PI3K/AKT pathway, ultimately leading to apoptosis of CRC cells. 62
The reactive oxygen species (ROS) molecule can serve as a signaling factor to trigger autophagy, while elevated levels of ROS may result in cellular senescence or apoptosis. 56 The production of ROS serves as a crucial indicator of oxidative stress. Wogonoside induces apoptosis in GC cells by elevating intracellular ROS levels. Additionally, wogonoside treatment upregulates ER stress-related proteins such as GRP94, phosphorylated eIF2α(p-eIF2α), and CHOP. Therefore, wogonoside promotes apoptosis in GC cells through the induction of ROS accumulation and ER stress. 63 Scutellarein can induce apoptosis of CRC HCT116 cells by increasing intracellular ROS production, enhancing caspase-3 activity and Bax protein expression, downregulating Bcl-2, and promoting the mitochondrial release of cytochrome c. 64
Inhibition of tumor cell proliferation
Inhibition of tumor cell proliferation is crucial in impeding tumor growth. Baicalein is an important flavonoid present in the root of S. baicalensis, which has antioxidant, anti-inflammatory, anti-hepatotoxic, antiviral and anti-tumor properties. 8 Baicalein treatment decreased the expression levels of p-mTOR, p-Akt, p-IκB and NF-κB proteins, and suppressed GC cells by inhibiting the PI3K/Akt. 65 Baicalin reduces HCC proliferation and metastasis by inhibiting the ROCK1/GSK-3β/β-catenin signaling pathway. 66 β-catenin, survivin, GSK3B, and Bax are potential targets of the Wnt/β-catenin pathway. Wogonin downregulates CTNNB1, BIRC5, and GSK3B while upregulating the expression of Bax. Inhibition of the Wnt/β-catenin pathway suppresses the proliferation of CRC SW480 cells. 67 Wogonin inhibits the proliferation of GC SGC7901 cells by inhibiting Wnt/β-catenin and decreasing the levels of β-catenin, C-myc and Cyclin D1 proteins. 68 EGFR, functioning as an oncogene, is capable of promoting the proliferation of HCC cells. Wogonin primarily exerts its inhibitory effects on the proliferation and invasion of HCC cells by targeting EGFR and its downstream factors EGFR/AKT,EGFR/ERK/MMP2 and EGFR/cyclin D1. 42 Focal adhesion kinase (FAK) is capable of promoting the proliferation and metastasis of esophageal squamous cell carcinoma (ESCC). The oncogenic protein diacylglycerol kinase α (DGKα) can interact with FAK to form the DGKα/FAK complex. Chrysin inhibits ESCC proliferation by disrupting the DGKα/FAK complex and suppressing the FAK/AKT signaling pathway. 69
The knock-down of serine and arginine-rich splicing factor 9 (SRSF9) in HCC Hep3B and Huh-7 cells resulted in a significant decrease in the protein expression levels of Wnt signaling pathway components (DVL2 and β-catenin) as well as cell cycle pathway regulators (Cyclin D and Cyclin E). Apigenin inhibits HCC cell proliferation by targeting SRSF9. 70 Salidroside reduces the proliferation and invasive ability of human PC PANC1 and SW1990 cells by inhibiting AKT and ERK signaling pathways. 71 Lymphoid enhancer binding factor 1 (LEF-1) is upregulated as an oncogene in CRC tissues, and LEF-1 can activate the Wnt/β-catenin pathway. Beta-sitosterol significantly downregulates LEF-1 and inhibits the Wnt/β-catenin pathway. Consequently, it downregulates its downstream targets such as C-myc, Survivin, and ND1, thereby inhibiting CRC cell proliferation. 72 Both High Mobility Group Box 1 (HMGB1) and RAGE are highly expressed in ESCC. The HMGB1/RAGE axis promotes tumorigenesis and development, leading to malignant behavior of ESCC cells. Acteoside reduces the expression of HMGB1 and RAGE, and inhibits HMGB1/RAGE-induced CDC42 activation in ESCC cells. CDC42 inactivation can inhibit ESCC cell proliferation. 73
Inducing tumor cell cycle arrest
Dysregulation of the cell cycle leads to enhanced cell division and abnormal proliferation, ultimately resulting in tumor formation. The cell cycle comprises four main phases: G1 (gap 1), S (DNA replication), G2 (gap 2), and M (mitosis—cell and nuclear division). Cyclins, cyclin-dependent kinases, and other core components of the cell cycle drive cell division. 74 S. baicalensis exerts its antitumor effects by inhibiting cell cycle transitions and the survival of certain checkpoints. 75
AKT, a serine/threonine kinase, plays a crucial role in promoting the growth of ESCC cells. Scutellarin binds to AKT1/2 proteins, inhibiting their activity. This action induces G2 phase cell cycle arrest and increases p27 expression. Consequently, scutellarin significantly inhibits ESCC tumor growth by suppressing the AKT signaling pathway. 76 The extract of S. baicalensis significantly down-regulated the expressions of CDK2, CDK4, CDK6, Cyclin D1 and Cyclin E1 in a dose-dependent manner, and inhibited the growth of HCC cells in G1/S phase by inhibiting CDK4/CDK6. 77 Scutellarein induces G2/M phase arrest in HCC Hep3B cells by reducing the levels of CDC25C, CDK1, and Cyclin B1 proteins. 78 Apigenin 7-O-glucoside induces G2/M phase arrest in HCC HepG2 cells by modulating the levels of Cyclin B1 and CDK1. 79 Baicalein induces S-phase arrest in gallbladder cancer cells by down-regulating Cyclin B1 and Cyclin D1 in gallbladder cancer BGC-SD and SGC996 cells while up-regulating Cyclin A. 80 Ezrin facilitates the proliferation and growth of CRC. Following baicalein treatment, there is a down-regulation of Ezrin, CyclinD1, and CDK4, as well as an up-regulation of p53 and p21 protein levels, thereby leading to the induction of CRC HCT116 cell cycle arrest. 81 Baicalein suppresses the PI3K/Akt pathway by up-regulating p21/CDKN1A and p27/CDKN1B, leading to cell cycle arrest in the S phase and G2/M phase of HCC Bel-7402 cells. 82
Overexpression of CyclinD1 promotes uncontrolled cell division, leading to cancer development. Wogonin induces CyclinD1 degradation and reduces CyclinD1 protein levels through the activation of glycogen synthase kinase-3 beta (GSK3β), thereby inhibiting proliferation of HCC MH97L and HepG2 cells. 83 Apigenin induces cell cycle arrest at the G2/M phase in human CRC HCT116 cells by inhibiting CyclinB1 and its activating partners CDC2 and CDC25C, while also up-regulating p53 and p21. 84 Apigenin induces G1 phase cell cycle arrest in HCC HepG2, SMMC-7721, and Huh-7 cells by up-regulating CyclinD1 and down-regulating CDK4. 85 Baicalein treatment down-regulates CyclinD1 in SMMC-7721 cells, inducing cell cycle arrest of HCC. 86 Wogonin significantly reduced the levels of CyclinD1, CyclinE and CDK4/6 proteins to induce HCC cells to stall in the G1-S phase. 42 Baicalin treatment significantly reduces the levels of CyclinB1, CyclinE1, CyclinD1, and p-Akt in CRC RKO and HCT116 cells, inducing CRC cell arrest in the G1 phase. 87 S. barbata polysaccharide(SBP)-2A down-regulates the expression of CyclinD1 and CDK4, inducing HCC cells to block in the G0/G1 phase. 51
Inhibition of tumor cell invasion and metastasis
Cancer begins as a localized disease, which can be effectively treated by removing the primary tumor when diagnosed early. However, metastatic tumors, being systemic and largely resistant to drugs, represent a significant challenge, accounting for over 90% of cancer-related deaths. 88 The metastasis of cancer cells consists of the following steps: (1) invasion of epithelial cells in the primary tumor through surrounding extracellular matrix (ECM), (2) endocytosis into the vascular lumen, (3) survival in vasculature systemic transport, (4) retention in a distant organ, (5) exocytosis into the parenchyma of a distant tissue, (6) adapting to the foreign microenvironment and forming micrometastases, (7) initiating proliferation at the metastatic site to form macroscopic, clinically detectable tumor growth. Moreover, interactions between cancer cells and non-tumorigenic stromal cells also play a crucial role in the overall invasion-metastasis cascade response. 89
Epithelial-mesenchymal transition (EMT)
EMT is a biological process wherein epithelial cells undergo molecular changes, losing their characteristic features and adopting mesenchymal cell properties. This transformation enables cells to acquire motile and invasive characteristics. Aberrant activation of EMT is closely associated with enhanced tumor cell migration and invasiveness, as well as resistance to chemotherapy and immunotherapy. 90 The EMT program is mainly executed by EMT-activating transcription factors (EMT-TFs), which include SNAI1, SNAI2, TWIST1, TWIST2, ZEB1, and ZEB2. EMT-TFs inhibit epithelial genes such as cytokeratins and E-cadherin, and activate genes associated with mesenchymal phenotypes, including vimentin, fibronectin, and N-cadherin to promote EMT Additionally, various signaling pathways such as TGF-β, Wnt, Notch, and PI3K-AKT also exert influence on the EMT process. 91
Salidroside has been shown to up-regulate the expression of E-cadherin while down-regulating the protein levels of N-cadherin, MMP-2, MMP-9, and heat shock protein 70 (HSP70). Additionally, it inhibits the phosphorylation of Src, Akt, ERK, and FAK, thereby suppressing the metastasis of GC BGC-823 cells. 92 Salidroside has been found to significantly down-regulate the expression of Notch1, snail, COX-2, MMP-2, and MMP-9, while concurrently up-regulating E-cadherin. Moreover, salidroside reduces the metastasis of HCC cells by specifically inhibiting the Notch1 signaling pathway. 93
Activation of the JAK/STAT pathway has been associated with the progression of EMT in cancer cells, facilitating cancer cell invasion and metastasis. Scutellarin treatment has been shown to significantly decrease the levels of phosphorylated JAK2 and STAT3 while increasing the expression of E-cadherin and reducing the levels of snail and vimentin proteins. Consequently, scutellarin inhibits invasion and metastasis of HCC by suppressing the JAK2/STAT3 pathway and EMT. 94 PTEN, functioning as a lipid phosphatase, exerts its tumor-inhibitory role by antagonizing PI3K signaling. The PI3K/Akt pathway, in turn, activates nuclear factor-κB (NF-κB) via phosphorylated IκB kinase, thereby promoting cancer cell metastasis. Scutellarein has been shown to inhibit the PI3K/Akt/NF-κB pathway by up-regulating PTEN expression. Furthermore, scutellarein up-regulates the expression of MMP extracellular inhibitors, such as TIMP2, while down-regulating MMP-2 and MMP-9. This regulation of the EMT pathway contributes to the inhibition of metastasis in HCC HepG2 cells. 95
After baicalin treatment, there is an observed upregulation in the expression of E-cadherin, cytokeratin 18, and claudin1 in CRC cells. Conversely, N-cadherin, vimentin, snail, and twist exhibit downregulation. Notably, baicalin demonstrates an inhibitory effect on EMT by blocking the TGFβ/Smad signaling pathway. 96 MMPs facilitate the degradation of the extracellular matrix, thereby promoting tumor cell metastasis. Baicalein exerts inhibitory effects on CRC invasion and metastasis by targeting ERK and down-regulating MMP-2 and MMP-9. 97 Zinc finger protein X-linked (ZFX) promotes tumor cells metastasis; baicalein was able to inhibit the metastasis of gallbladder cancer cells by down-regulating ZFX, MMP-2 and MMP-9. 80 Snail serves as a crucial transcription factor in EMT. Baicalein treatment effectively inhibits the snail-induced EMT process in CRC HT29 and DLD1 cells, thereby impeding the metastasis of CRC cells. 98
The combination of apigenin (25 μM) and chrysin (25 μM) leads to the downregulation of MMP2, MMP9, snail, and twist proteins, while also reducing P38-MAPK/AKT activity, thereby impeding the metastasis of CRC cells. 99 Daucosterol, as a phytosterol, plays a key role in inflammation suppression, immunomodulation and anticancer effect. 100 Wnt/β-catenin promotes hypoxia-induced EMT in HCC cells, and daucosterol impedes HCC SMMC-7721 cell metastasis by hindering Wnt/β-catenin signaling. 100 TGF-β1 triggers EMT during the later stages of tumor development. Following treatment with TGF-β1, E-cadherin levels decline while N-cadherin, Vimentin, and Twist1 levels increase. Additionally, non-steroidal anti-inflammatory drug-activated gene-1 (NAG-1) has been shown to inhibit TGF-β1 signaling. Oroxylin A has been found to suppress HCC cells by up-regulating NAG-1 to counteract the TGF-β1/Smad signaling pathway. 101
Wogonin upregulates E-cadherin expression and downregulates vimentin, ZEB2, N-cadherin, and SMAD3 expression to regulate the EMT process in CRC. Dysregulation of the Hippo signaling pathway promotes aberrant cell proliferation, with YAP serving as an effector molecule of this pathway, while IRF3 acts as an agonist of YAP. Wogonin reduces YAP1 and IRF3 levels in CRC cells, thereby regulating the Hippo signaling pathway to inhibit the EMT process in CRC cells. 102 Forkhead box M1 (FOXM1) is upregulated in HCC tissues, and beta-sitosterol-induced downregulation of FOXM1 not only inhibits EMT of HepG2 cells but also suppresses the Wnt/β-catenin signaling pathway and its downstream targets, thereby impeding the growth and metastasis of HCC cells. 103 Acteoside inhibits CRC HT29 cell metastasis through inhibition of the Rac-1, hypoxia-inducible factor 1 alpha (HIF-1α) and Zeb-1 signaling pathways. 104
Angiogenesis
Tumor angiogenesis is critical for providing essential nutrients and oxygen to rapidly growing malignant tissues. It promotes the survival and growth of tumor cells by facilitating their access to the vascular system, thereby increasing the risk of metastasis. 105
Girders of actin filaments (Girdin) is an actin-binding protein implicated in invasion and angiogenesis across diverse cancers. Upregulation of Girdin expression has been associated with heightened metastatic potential in HCC. Treatment with scutellarin has been shown to downregulate STAT3 and Girdin expression, thereby inhibiting HCC cell metastasis through disruption of the STAT3/Girdin/Akt pathway. 106 Overexpression of Girdin in PC cells enhances cancer cell migration induced by epidermal growth factor (EGF), and Girdin has been found to upregulate VEGF-A expression. Scutellarin inhibits metastasis and angiogenesis in PC by suppressing Girdin phosphorylation. 107 Scutellarin is able to inhibit angiogenesis and human umbilical vascular endothelial cells (HUVECs) migration in CRC cells by targeting ephrinb2 signaling, which in turn reduces CRC cell metastasis. 108 The ruthenium baicalein complex effectively suppresses Akt, mTOR and vascular endothelial growth factor (VEGF) in CRC HT-29 cells, leading to inhibition of invasion and metastasis in CRC cells. 109 Baicalein inhibits VEGF by downregulating HIF-1α, a crucial regulator of angiogenesis. Additionally, baicalein hinders VEGF-induced angiogenesis by upregulating p53/Rb and downregulating VEGFR/ERK pathways. These actions collectively inhibit cancer cell proliferation and migration. 109
Cancer stem cells (CSCs)
The presence of CSCs is associated with tumor recurrence, metastasis, and drug resistance. Baicalin has demonstrated the ability to significantly reduce the expression levels of stem cell markers such as CD133, CD44, and SOX2 in CRC RKO and HCT116 cells, thereby inhibiting the invasion and migration of CRC. 87 The Sonic Hedgehog (Shh) signaling pathway is aberrantly activated in CSCs. Following baicalein treatment, the expression of key components of the Shh pathway, including SHH, SMO, and Gli-2, is downregulated. Consequently, baicalein inhibits the metastasis of PC by impeding the Shh pathway. 110
Regulation of the tumor microenvironment
The tumor microenvironment (TME) comprises non-tumor cells and components present within the tumor, along with the molecules they produce and release. The ongoing interaction between tumor cells and the tumor microenvironment significantly influences tumor genesis, progression, and metastasis. 111 Studies have demonstrated that S. baicalensis can remodel the immunosuppressive, inflammatory, hypoxic, and acidic aspects of the TME. 112
Regulation of the tumor-associated immune microenvironment
Tumor cells engage in interactions with host immune cells within the TME. Various immune cells, such as B cells, T cells, natural killer cells (NK), regulatory T cells (Tregs), dendritic cells (DCs), macrophages, tumor-associated macrophages (TAMs), and myeloid-derived suppressor cells (MDSCs), among others, play crucial roles in regulating cancer cells within the TME. 109 First, the immune system inhibits tumor growth through immune responses. Second, the immune system can induce an inflammatory response that promotes tumor cell growth, survival, and angiogenesis. Finally, tumor cells exploit immune regulatory mechanisms to create an immunosuppressive microenvironment. This microenvironment not only suppresses host immune responses but also forms a barrier that blocks anti-tumor immunotherapy. 113
The repolarization of TAMs to the M1 phenotype fosters an immune microenvironment conducive to tumor regression. Baicalin has been shown to induce the transformation of macrophages from the M2 type to the M1 type. This induction is accompanied by upregulation of pro-inflammatory factors such as TNF-α and IL12, along with downregulation of the anti-inflammatory factor IL10, ultimately inhibiting HCC expansion. Additionally, baicalin treatment of TAMs leads to increased mRNA expression of RelB and continued phosphorylation of p100 and p52, indicating the activation of the RelB/p52 pathway mediating TAMs repolarization. 109 Baicalin treatment significantly downregulates the protein levels of Toll-like receptor 4 (TLR4) and p-IκBα in the NF-κB pathway. By inhibiting the NF-κB pathway, baicalin downregulates programed cell death ligand 1 (PD-L1) and MDSCs in CRC CT26 cells. Furthermore, baicalin upregulates CD4 and CD8 T cells, thereby improving the tumor immunosuppressive microenvironment and enhancing anti-tumor immunity. 114 Baicalin and baicalein down-regulate PD-L1 expression induced by IFN-γ by reducing STAT3 activity. This process enhances T cell sensitivity to eliminate tumor cells and inhibits the growth of HCC cells. 115 CD24 exhibits an anti-phagocytic effect, and inhibiting CD24 can bolster anti-tumor immunity. In highly metastatic HCC cell lines and recurrent tumor tissues, CD24 is notably overexpressed. Baicalein counteracts this by down-regulating CD24 in HCC cells, thus impeding HCC progression. 116
Chrysin increased the proportion of CD4/CD8 positive T cells in tumor tissue of HCC H22 xenografted mice, and decreased the expression of PD-L1 by blocking STAT3 and NF-κB pathways, thus enhancing the anti-tumor immunity of mice. 117 Apigenin is demonstrated to bolster the proliferation of NK cells through the up-regulation of Bcl-2 and the down-regulation of Bax expression. Moreover, it enhances the cytotoxic capabilities of NK cells against PC cells by activating the JNK and ERK pathways. 118 The Src Homology-2 (SH2) domain-containing Inositol 5′-Phosphatase-1 (SHIP-1) plays a pivotal role in the regulation of immune cell activities, including those of macrophages and DCs. Down-regulation of SHIP-1 is associated with fostering a pro-tumorigenic microenvironment. However, treatment with apigenin has been shown to increase SHIP-1 expression, leading to a decrease in the immunosuppressive M2-type TAMs and a shift toward the immunogenic M1-type TAMs. This phenotypic conversion consequently supports the regression of PC. 119 In HCC cells expressing HIF-1α, the cytotoxicity of NK cells was significantly reduced, and apigenin augmented the cytotoxicity of NK to HIF-1α-expressing HCC cells by up-regulating the expression of CD95L on NK cells. 120
Palmitic acid is a common fatty acid that plays a regulatory role in diseases such as metabolic syndrome, cardiovascular disease, cancer, and inflammation. 121 The combination of palmitic acid and γ-interferon (γ-IFN) has been shown to upregulate M1-type TAMs and downregulate M2-type TAMs via the TLR4 pathway, thereby inhibiting GC by modulating macrophage polarization. 122 After treatment with ceramide and palmitic acid, macrophages showed an increase in the secretion of the M1-type TAMs markers CD68 and IL-12 and a decrease in the secretion of the M2-type TAMs markers CD163 and IL-10, palmitic acid and ceramide can also inhibit M2-type TAMs by inhibiting the IL-10-STAT3-NF-κB signal transduction axis. 123 liposomal encapsulated beta-sitosterol by increasing IL-12, IL-18, and IFN-γ expression levels and CD4/CD8 T-cell subsets in small intestinal tissues in order to enhance anti-CRC immunity. 124
Modulation of the inflammatory microenvironment
Tumors are often likened to wounds that do not heal, with inflammation playing a pivotal role in cancer development, progression, and metastasis. Predominant inflammatory cytokines found within the tumor milieu include tumor necrosis factor α (TNF-α), interferon-gamma (IFN-γ), interleukin (IL)-6, IL-8, IL-1, and transforming growth factor-beta (TGF-β), among others. 125 The transcription factor NF-κB has been identified as a central player in orchestrating the inflammatory response, catalyzing the production of inflammatory mediators that contribute to cancer advancement. 126
TLR4 is overexpressed in CRC and is associated with inflammation. Baicalein directly binds to TLR4, leading to its inactivation. HIF-1α and VEGF, which are downstream targets of TLR4, are also affected by this interaction. Consequently, baicalein can inhibit CRC growth and angiogenesis by disrupting the TLR4/HIF-1α/VEGF signaling pathway. 127 After administration of baicalein, a notable reduction in the number of intestinal tumors is observed, alongside a decrease in the intestinal index and an increase in the spleen index. Furthermore, baicalein therapy significantly diminishes the levels of pro-inflammatory cytokines such as interleukin-1 beta (IL-1β), IL-2, IL-6, and GM-CSF. These effects collectively contribute to baicalein’s anti-CRC activity by modulating intestinal inflammation. 128 Chronic pancreatitis can lead to acinar-to-ductal metaplasia (ADM) and eventually pancreatic ductal adenocarcinoma (PDAC). The research team led by Li Kang Sun discovered that baicalein ameliorates the inflammatory microenvironment by attenuating TNF-NF-κB signaling. This inhibition of the NF-κB pathway results in the suppression of ADM in pancreatic exocrine cells. Consequently, baicalein may serve as a preventative agent against PDAC by mitigating the underlying pancreatitis. 129
Lycopene, which is mainly derived from tomatoes, contains many vitamins, fiber, and minerals and has the potential to regulate blood lipids, endothelial function, blood pressure, antioxidant and anti-cancer potential. 130 Lycopene effectively reduced the expression of inflammation-related proteins such as NF-κB, JNK, TNF-α, IL-6, IL-1β, inducible nitric oxide synthase (iNOS), and cyclooxygenase-2 (COX-2) in CRC SW480 cells. By inhibiting the NF-κB pathway, lycopene may contribute to the suppression of CRC progression. 131 TRAF6 serves as a regulator of NF-κB signaling and is found to be overexpressed in PC tissues. Wogonoside is a bioactive flavonoid with anti-inflammatory, anti-angiogenic and anticancer activities. 132 Wogonoside has been shown to decrease the levels of TRAF6, p-P65/P65, and VCAM1 proteins, while also reducing the expression of IL-6, TNF-α, and IL-1β in PC cells. Therefore, it can be concluded that wogonoside effectively treats PC by targeting the TRAF6/NF-κB/p65 signal transduction. 133 In CRC HT-29 cells, scutellarin reduces the concentration of TNF-α and IL-6 by inhibiting Wnt/β-catenin to treat colitis-related CRC. 134 Apigenin suppresses Helicobacter pylori (H. pylori) by upregulating the expression of IκBα, leading to the inactivation of NF-κB and reduction in levels of inflammatory factors ICAM-1, IL-6, IL-8, and COX-2. ROS activate NF-kB, causing extensive gastric mucosal injury and progression to intestinal metaplasia and GC. Apigenin inhibits inflammation-associated GC by decreasing H. pylori colonization levels, blocking NF-kB activation, and reducing ROS. 135
Regulation of the hypoxic microenvironment
Due to rapid proliferation, high metabolic activity, and elevated oxygen consumption, tumor cells often experience inadequate oxygen supply, leading to a hypoxic TME. Normally, the partial pressure of oxygen in human tissues is around 40 mmHg, whereas in most TMEs, it drops below 7.5 mmHg. In such hypoxic conditions, levels of HIF rise, consequently up-regulating VEGF. This up-regulation promotes the formation of new blood vessels, aiding in the maintenance of oxygen homeostasis within the tumor microenvironment. 135
Lysyl oxidation-like protein 2 (LOXL2) is a target gene of HIF-1α. Hypoxia can up-regulate LOXL2 and induce PC. LOXL2 promotes the metastasis of PC by increasing MMP2/9 levels and decreasing E-cadherin expression. Salidroside has been found to down-regulate the levels of HIF-1α and LOXL2 in PC BxPC-3 cells and ameliorate hypoxia-induced PC by inhibiting the HIF-1α/LOXL2 pathway. 136 HIF-1α promotes the development and metastasis of PC under hypoxic conditions. Accompanied by increased levels of HIF-1α, downstream VEGF and GLUT-1 are activated. Treatment with apigenin has shown to reduce the levels of GLUT-1, HIF-1α, and VEGF, suggesting its potential as a therapeutic drug for PC. 84
Regulation of acidic microenvironment
Under hypoxic conditions, most cancer cells adopt aerobic glycolysis to support their rapid growth and proliferation, a phenomenon known as the Warburg effect. This metabolic shift leads to increased production and secretion of lactic acid. Consequently, the accumulation of lactic acid causes acidification of the TME, which in turn fosters the development of an immunosuppressive TME. 137
After treatment with wogonin, the levels of lactate dehydrogenase (LDH), succinate dehydrogenase (SDH), adenosine triphosphate (ATP), and HIF-1α proteins were significantly reduced in GC SGC-7901 cells. This inhibition of energy metabolism, proliferation, and angiogenesis was observed in GC cells. 138 In CRC cells expressing wild-type p53 but not mutant p53, wogonin demonstrated inhibitory effects on glycolysis while increasing the expression of p53 and TP53-inducible glycolysis and apoptosis regulator (TIGAR). Concurrently, wogonin decreased glucose transporter 1 (GLUT1) and glycolytic enzymes, thereby inhibiting CRC. 139
Oroxyloside, a novel PPARγ/α dual agonist, functions by inhibiting glucose catabolism and promoting fatty acid oxidation. This metabolic modulation leads to the production of acetyl-CoA for the tricarboxylic acid cycle and oxidative phosphorylation. The resulting metabolic switch induced by oroxyloside significantly increases the level of ROS and induces cell-cycle arrest in the G1 phase. These findings suggest that oroxyloside holds promise as a novel therapeutic agent for exerting anti-HCC effects. 140 Oroxylin A acts as a GLUT1/mitochondrial fusion inhibitor, restricting glucose catabolism in HCC cells. It accomplishes this by inhibiting mitochondrial fusion through interference with the SIRT1/PDK2/PARL axis. Inhibition of mitochondrial fusion disrupts mitochondrial metabolism and enhances cancer cell susceptibility to glucose-restricted therapy. Thus, the combined application of GLUT1 inhibitors with mitochondrial fusion inhibitors may have a synergistic effect in cancer therapy. 141
HK-2 plays a crucial role in the Warburg effect across various cancers. Administration of chrysin reduces HK-2 expression in HCC cells, leading to decreased glucose uptake and lactate production. Furthermore, HK-2 forms a complex with VDAC-1, preventing apoptosis of HCC cells. Reduction of HK-2 disrupts the HK-2/VDAC-1 interaction, inducing apoptosis in HCC. 45 Pyruvate kinase M2 (PKM2) acts as a rate-limiting enzyme in aerobic glycolysis. HIF-1 induces the nuclear translocation of PKM2, enhancing the expression of GLUT1 and LDHA to promote cancer progression. Apigenin exerts an anti-CRC effect by targeting PKM2, thereby inhibiting glycolysis in CRC LS-174T cells. 142 Salidroside has demonstrated an ability to inhibit glycolysis and decrease the levels of glycolysis-related enzymes such as PKM2, enolase 1 (ENO1), and GLUT1. This action plays a significant role in the metabolism of GC cells, particularly GC SGC-7901 and MKN-45 cells. 143 Baicalein, by reducing the expression levels of HIF-1A and PKM2, can inhibit the glycolysis process in ESCC cells. This inhibition leads to the blockade of energy supply, reduction in the expression levels of CyclinD1/CDK4, arrest of ESCC cells in the G1 phase, and enhancement of sensitivity to radiotherapy. 144
Epigenetic regulation
The dysregulation of the epigenome plays a pivotal role in driving abnormal transcriptional programs, thereby promoting the occurrence and progression of cancer. Deficiencies in gene regulation not only impact tumor carcinogenesis and inhibitory factors but also affect the immunogenicity of tumors. Changes in the epigenome can influence immune cells involved in anti-tumor responses. 145
Mutations in long non-coding RNAs (lncRNAs) contribute to the progression of malignant tumors. These lncRNAs can exhibit either tumor suppressor or oncogenic effects. Furthermore, lncRNAs hold promise as potential cancer biomarkers and therapeutic targets. 146 One such example is NF-κB Interacting lncRNA (NKILA), which functions by inhibiting IκBα phosphorylation and nuclear translocation of NF-κB, thereby suppressing NF-κB activity. The combination of NKILA and baicalein has been shown to enhance the inhibition of NF-κB signaling, presenting a potential therapeutic strategy for treating HCC. 147 The lncRNA H19 plays a crucial role in mediating the occurrence and progression of HCC. Apigenin has been found to inhibit HCC growth by down-regulating H19 RNA, which in turn suppresses the Wnt/β-catenin signaling pathway. 148 GPC3 and SULF2 are identified as potential targets for HCC treatment. Elevated levels of GPC3, SULF2, and lncRNA-AF085935 are associated with increased HCC proliferation. Chrysin attenuates HCC proliferation by inhibiting the GPC3/SULF2 axis and reducing the expression of lncRNA-AF085935. 149 Studies have demonstrated that decreased expression of lncRNAs H19 inhibits tumor growth, while decreased expression of let-7 promotes tumor progression. The interaction between H19 and let-7 regulates the expression of COPB2. Decreased expression of COPB2 leads to increased expression of p53 and E-cadherin. Consequently, chrysin promotes GC cell apoptosis by regulating the H19/LET-7A/COPB2 axis. 150
The expression pattern of microRNAs (miRNAs) is closely associated with cancer type and stage. MiRNAs have emerged as valuable tools for cancer diagnosis and prognosis assessment due to their dynamic roles. These molecules can exhibit oncogenic or tumor-suppressive effects, influencing various aspects of tumor biology including cell proliferation, apoptosis, invasion/metastasis, and angiogenesis. 151 MicroRNA (miR)-139-3p interacted with CDK16, baicalin increased miR-139-3p expression while decreasing the level of CDK16, which blocked the cell cycle and inhibited the cell proliferation of CRC cells, suggesting that baicalin inhibits CRC cells by regulating the miR-139-3p/CDK16 axis. 152 Baicalein treatment increases the level of miR-3178 and decreases HDAC10 expression, resulting in the inactivation of the AKT signaling pathways. This modulation leads to the inhibition of growth in HCC MH-97H and SMMC-7721 cells by regulating the miR-3178/HDAC10 pathway. 153
MiR-520b, identified as a tumor suppressor, exhibits significant reduction in expression levels in the doxorubicin(ADM)-resistant HCC cell line BEL-7402/ADM. Overexpression of miR-520b enhances the sensitivity of BEL-7402/ADM cells to ADM. Furthermore, miR-520b exerts chemosensitization effects by targeting ATG7. Consequently, apigenin sensitizes BEL-7402/ADM cells to ADM by modulating the miR-520b/ATG7 pathway. 154 MiR-101 has been found to enhance the sensitivity of the human ADM-resistant HCC cell line BEL-7402/ADM to ADM. Upregulation of NRF2 expression has been associated with increased resistance to ADM. MiR-101 exerts its chemosensitizing effect by targeting Nrf2. Consequently, apigenin sensitizes BEL-7402/ADM cells to ADM by inhibiting the miR-101/Nrf2 pathway. 154 Furthermore, up-regulation of miR-215-5p by apigenin results in direct targeting of E2F1/3 transcription factors and subsequent down-regulation of their expression. This cascade leads to cell cycle arrest in the G0/G1 phase of CRC HCT116 cells. 155
Studies have revealed that miR-107 and tropomyosin alpha-1 (TPM1) play crucial roles in regulating cancer cell proliferation. Skullcapflavone I (SFI) down-regulates miR-107 and upregulates TPM1 in CRC HCT116 cells. This modulation inhibits the MEK/ERK and NF-κB signaling pathways, consequently suppressing the proliferation of CRC cells. 156 Additionally, SFI significantly inhibits Panc-1-induced proliferation, migration, and invasion of PC cells by down-regulating miR-23a and inhibiting the JAK/STAT and MAPK pathways. 157 Acteoside increases the radiosensitivity of HCC cells by upregulating the expression of miR-101-3p and decreasing the expression of WEE1 in HCC cells. 158
Combined Use and Reversal of Drug Resistance
Chemotherapy is a widely used approach for treating malignant tumors, but the development of chemoresistance often occurs with repeated administration, limiting the efficacy of the drugs. Drug-resistant cancer cells demonstrate uncontrolled proliferation and reduced susceptibility to cell death. 159 The active ingredient of S. baicalensis has been found to reverse chemoresistance through several mechanisms. When combined with chemotherapeutic agents, it can enhance their therapeutic effects.
Treatment with baicalein has been shown to increase the intracellular concentration of epirubicin compared to untreated cells. This suggests that baicalein reverses P-glycoprotein (P-gp)-mediated resistance in multidrug-resistant HCC (Bel7402/5-FU) cells by reducing the levels of P-gp and Bcl-xl. 160 Zhang et al ’s research revealed that baicalein induced S-phase cell cycle arrest in PC CAPAN-2 cells by reducing cyclin-B1 expression. Additionally, baicalein induced apoptosis in cisplatin-resistant PC cells by decreasing caspase-3 and Bcl-2 expression while increasing cleaved caspase-3 and Bax expression. 161 Treatment with wogonin has been shown to significantly reduce the levels of p-Akt and Bcl-2 proteins, while upregulating Bad expression levels. This action inhibits Akt signaling and promotes apoptosis in gemcitabine-resistant PC line Panc-1. 162 Carbonyl reductase 1 (CBR1), highly expressed in PC tissues, plays a protective role against oxidative damage. Chrysin binds to CBR1, inhibiting its activity, which in turn increases intracellular levels of ROS. Elevated ROS levels induce ROS-dependent autophagy and raise intracellular free iron levels, thereby enhancing the sensitivity of PC cells to gemcitabine. 163 Activation of the PI3K/AKT pathway has been found to promote glycolysis in HCC HepG2 cells. Similarly, activation of GSK-3β promotes glucose uptake and lactate production, along with up-regulation of glycolytic enzymes. Salvigenin is a trimethoxylated flavonoid derived from S. baicalensis, which has the properties of lipid reduction, mitochondrial function enhancement and anti-tumor activity. 164 Salvigenin has been shown to impede aerobic glycolysis and chemotherapy resistance to 5-FU in HCC cells by inhibiting the PI3K/AKT/GSK-3β pathway. 164 In CRC, treatment with 5-FU leads to a significant increase in the p-AKT/total AKT ratio, rendering CRC resistant to 5-FU. However, when combined with acteoside, 5-FU significantly reduces the levels of PI3K and p-AKT/total AKT. This combination inhibits the PI3K/AKT pathway, thereby reducing the resistance of CRC to 5-FU. 165 Activation of HIF-1α has been linked to multidrug resistance and the development of EMT in HCC. In oxaliplatin-resistant cells with high expression of HIF-1α, treatment with salidroside promotes HIF-1α degradation, reduces the expression levels of Twist1 and Zeb1 (markers of EMT), and increases E-cadherin expression. By inhibiting the HIF-1α signaling pathway, salidroside significantly enhances the sensitivity of HCC to oxaliplatin and inhibits hypoxia-induced EMT. 166 ATP-driven multidrug resistance (MDR) efflux transporter proteins, such as MDR1 and MRP, play a role in multidrug resistance. Overexpression of HIF-1α can upregulate MDR1 and MRP. Co-treatment with apatinib and salidroside reduces the expression levels of HIF-1α, VEGF, MRP, and MDR1 in GC MKN-45/MDR cells. This combined treatment enhances the efficacy of chemotherapy and induces apoptosis in GC cells by modulating the hypoxic tumor microenvironment. 167 The co-treatment of chrysin and sorafenib synergistically enhances the anti-tumor activity of sorafenib by inhibiting the ATP-binding cassette super-family G member 2 (ABCG2) transporter. Additionally, chrysin induces sustained phosphorylation of ERK1/2, leading to the inhibition of HCC cell growth and increased sensitivity of HCC cells to sorafenib. 168
Combining the active components of S. baicalensis with chemotherapy treatment has been shown to enhance the anti-tumor effect and reduce toxic side effects. For instance, chrysin combined with cisplatin induces apoptosis in HCC Hep G2 cells through several mechanisms, including activation of ERK1/2, promotion of p53 phosphorylation, up-regulation of Bax and DR5, and down-regulation of Bcl-2 expression. 169 Furthermore, the apoptosis rate of PC PNCA-1 cells induced by 10 nM docetaxel was 76.07 ± 9.61%, while the apoptosis rate of 10 nM docetaxel combined with 10 μM baicalein was 87.63 ± 5.19%. Baicalein combined with gemcitabine/docetaxel promotes apoptosis of PC cells by activating the caspase-3/PARP signaling pathway. 170 Treatment with baicalein in combination with epirubicin has been shown to have significant effects in HCC Hep-G2 cells. This combination resulted in a notable increase in the concentrations of beclin 1, Bax, and ATG-7 while down-regulating the expression of TGFβ1. Consequently, baicalein significantly enhanced the cytotoxicity of epirubicin in HCC and increased its safety profile by promoting apoptosis and autophagy. 171 The combination of baicalin and 5-Fu has been observed to increase intracellular ROS levels in GC cells. Ferrostatin-1 (Fer-1), an inhibitor of iron death, reversed baicalin-induced intracellular ROS production in GC cells. Baicalin enhances the efficacy of 5-Fu in GC by promoting ROS-mediated iron death. 172 Additionally, wogonin has been found to enhance the cytotoxic effects of oxaliplatin on GC BGC-823 cells. This enhancement is achieved by regulating the levels of phospho-JNK, phospho-ULK1, and LC3II, exacerbating the dissipation of mitochondrial membrane potential (ΔΨm), and promoting the formation of peroxynitrite. 173 Combining bleomycin with scutellarin treatment has been found to significantly increase the levels of cleaved-caspase-3 and cleaved-caspase-8, enhance p53 and miR-29b expression, down-regulate TGF-β1 expression, and induce apoptosis in HCC H22 cells through activation of the p53-mediated apoptotic pathway. 174 Additionally, after 12 hours of treatment with chrysin and cisplatin, apoptosis in HCC HepG2 cells was induced by up-regulation of cleaved caspase-3, -8, and -9, and PARP, as well as down-regulation of PARP, cFLIP, and caspase-3 proteins. 175 The combined application of salidroside and paclitaxel has been shown to have several effects on CRC cells. It up-regulates E-cadherin protein expression while down-regulating N-cadherin and vimentin protein expression. Additionally, this combination blocks the PI3K/AKT pathway in CRC cells. These actions collectively inhibit CRC proliferation, migration, and invasion by mediating the EMT pathway. 176
The chemical constituents of S. baicalensis have demonstrated a synergistic effect when combined with ablation, radiotherapy, and targeted therapies. While nanosecond pulsed electric fields (nsPEFs) are effective in ablating liver tumors, they may cause damage to normal liver tissues and lead to hepatic insufficiency. However, low concentrations of baicalin have been found to synergistically enhance the necrosis of HCC cells induced by nsPEFs. Importantly, baicalin can also protect normal hepatocytes from ablation by increasing mitochondrial transmembrane potential and decreasing ROS production. 177 Escherichia coli DH5α-lux/βG was able to activate baicalin and increase the cytotoxic effect of baicalin on the CC C26 cell line. 178 LY294002 is a specific inhibitor of PI3K, and the combination of baicalein and LY294002 further reduced the levels of Akt, ERK1/2, and GSK-3βmRNA in HCC SMMC-7721 cells compared to single baicalein treatment, inducing HCC cells apoptosis through inhibition of PI3K/Akt signaling pathway. 86 The active ingredient of S. baicalensis has shown promise in improving the sensitivity of targeted therapy. In a study combining sorafenib with apigenin therapy, decreased levels of IL-6, IL-1β, TNF-α, AFP, Nanog, TLR4, c-Myc, CD-10, and β-catenin were observed in HCC tissues. Conversely, there was an up-regulation of p53 expression. Apigenin was found to enhance the drug sensitivity and cytotoxic effects of sorafenib, suggesting a potential synergistic effect of this combination therapy. 179 The combination of S. baicalensis and other herbal components has demonstrated an enhanced antitumor effect through multiple pathways. For instance, when combined with wogonin, artesunate significantly increased the levels of TNFα, TNF receptor-associated factor 3 (TRAF3), and DNA-damage-inducible, alpha (GADD45A). These molecules are all associated with apoptosis signaling pathways, suggesting that the combination therapy inhibits HCC through the apoptotic pathway. 180
Prevention of Precancerous Lesions
The development of malignant tumors follows a complex and gradual process, often progressing from “normal epithelium” to “simple hyperplasia,” “dysplasia,” “carcinoma in situ,” and finally “invasive carcinoma.” This evolution is driven by various carcinogenic factors and is considered the classical pattern of cancer development. 181 Most tumors are preceded by precancerous lesions, which serve as intermediate stages in the progression to cancer. Identifying and targeting these precancerous lesions offers valuable opportunities for early diagnosis and effective treatment of tumors. Detecting and intervening at the precancerous stage can potentially prevent the development of cancer, making it a crucial aspect of cancer prevention and management.
Liver diseases such as viral hepatitis, cirrhosis, liver fibrosis, liver nodules, non-alcoholic fatty liver disease (NAFLD), and non-alcoholic steatohepatitis (NASH) are known risk factors for the development of HCC. S. baicalensis has shown promise in mitigating the progression of these liver diseases and reducing the risk of HCC. S. baicalensis can reduce serum aminotransferase levels and attenuate the degree of hepatocellular injury in hepatitis animal model through anti-oxidative stress, anti-inflammatory, anti-apoptotic and antiviral pathways, and can also fight liver fibrosis through reducing extracellular matrix, antioxidant, anti-inflammatory, and inducing apoptosis of hepatic stellate cells (HSC). 182 NAFLD is a significant contributor to the development of cirrhosis and HCC. Huangqin decoction has been shown to ameliorate lipid metabolism disorders and insulin resistance in NAFLD by modulating the Sirt1/NF-κB pathway. 183 Furthermore, baicalin has demonstrated efficacy in the treatment of NASH. Baicalin treatment can reduce the expression of NLR pyrin domain containing 3 (NLRP3), gasdermin D (GSDMD), and IL-1β in NASH. By blocking the NLRP3-GSDMD signaling pathway, baicalin reduces pyroptotic cell death of hepatocytes, providing a potential therapeutic approach for NASH. 184 Baicalin has demonstrated significant efficacy in reducing hepatitis B surface antigen (HBsAg) and hepatitis B virus (HBV) DNA levels in HCC HepG2 cells. This effect is mediated by up-regulation of TRIM25, activation of the JAK/STAT pathway, and inhibition of the NF-κB pathway. 185 Treatment with oroxylin A has been shown to reduce collagen deposition and induce senescence in HSC by promoting ferritin autophagy. This mechanism significantly inhibits the progression of liver fibrosis. 186
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis (UC), has the potential to trigger the development of CRC. 187 Huangqin decoction significantly decreased the expression of serum IL-1β, IL-6, TNF-α, p-p65, and p-IκBα in mouse models of UC, inhibited TNF-α-induced NF-κB activation, significantly alleviated UC and promoted epithelial repair. 188 Treatment with baicalin significantly reduced the expression of pro-inflammatory cytokines TNF-α and IL-1β in CRC HT-29 cells, restored the levels of autophagy-related genes of LC3, ATG5, and BECN1, inhibited NF-κB activation, increased the level of Claudin 1 protein, and restored the integrity of intestinal epithelial cells in order to treat IBD to prevent its malignant evolution. 189 Baicalein significantly reduced the mRNA levels of iNOS, ICAM-1, MCP-1, COX-2, TLR4, TNF-α, and IL-1β in mouse models of colitis, and inhibited the activity of NF-κB and MAPK to treat colitis. 190 Colorectal adenomatous polyps may progress to colorectal adenocarcinoma, and cancer lesions are frequently observed in larger adenomas (>1 cm). Apigenin can promote p53 phosphorylation and reduce the number of colorectal polyps. 191 Baicalin treats NAFLD/NASH, UC and cholestasis by mediating PI3K/Akt/NRF2, Keap-1, NF-κB, and HO-1; regulates IL-6, IL-1β, TNF-α, MIP-2 inflammatory factor and TGF-β1/ Smads, STAT3 and NF-κB pathways to treat NAFLD/NASH and liver fibrosis; and affects PD-1/PDL-1 and TLR4-NF-κB to alleviate HCC, UC and CRC. 192
PC is often associated with an increased incidence of obesity and pancreatitis. Baicalein reduces the levels of NF-κB p65 and phosphorylated p38 MAPK, ERK, and STAT3 in acute pancreatitis (AP) models, thereby treating AP by inhibiting NF-κB, MAPK, and STAT3 signal transduction. 193 Chronic pancreatitis (CP) promotes ADM and increases the risk of PDAC. Baicalein inhibits rTNFα-induced NF-κB activation in pancreatic AR42J cells. Additionally, it suppresses the secretion of pro-inflammatory mediators such as TNFα and nitric oxide (NO), further inhibiting ADM in pancreatic AR42J cells by ameliorating the inflammatory microenvironment. 194
GC can be induced by H. pylori infection, chronic gastritis, and atrophic gastritis. In a mouse model of H. pylori infection, high doses of baicalin and baicalein inhibited the growth of H. pylori, while decreasing serum levels of IL-1β, H. pylori IgM and IgA. 195 Baicalin down-regulated the expression of TNF-α, IL-8, Akt, p-Akt, IκBα, p-IκBα, NF-κBp65, and NF-κBp-p65, thereby inhibiting Akt/NF-κB activation in the treatment of chronic gastritis. 196 Furthermore, baicalin treats chronic gastritis by reducing levels of IL-2, IL-8, and TNF-α, while increasing the expression of EGFR and Bcl-2. 197
New Drug Delivery Technologies
Traditional drug delivery systems face numerous limitations, including limited targeting, low therapeutic index, poor water solubility, and induction of drug resistance. However, the application of new drug delivery systems as carriers can address these shortcomings and enhance therapeutic efficacy. 198 Nano-preparation, for instance, has emerged as a promising approach to improve the antitumor activity of TCM. By promoting intracellular concentration and facilitating slow and sustained release of drugs, nano-preparation can reduce toxicity to normal cells and overcome targeted delivery barriers to malignant cells. Consequently, it significantly enhances the bioavailability of TCM. 169
Liposomes, composed of phospholipids with additives like cholesterol, sphingolipids, and detergents, offer a versatile platform for encapsulating and delivering both water-soluble and insoluble drugs. Given the inherent hydrophobicity of lipid components, lipid compounds exhibit potential therapeutic value but often suffer from low bioavailability. Through the application of liposomes, lipid compounds can be encapsulated, enhancing their solubility and thereby improving their biological effects. 199 Baicalein (BAI) has demonstrated the ability to increase the Bax/Bcl-2 ratio, thereby promoting apoptosis in PADC, and to decrease the expression of Mcl-1 protein, thus reducing PADC chemoresistance. However, baicalein suffers from low water solubility and susceptibility to degradation by the digestive system. Utilizing a liposomal formulation of BAI (BAI-L) addresses these challenges by protecting baicalein from degradation and enhancing its distribution in cancer tissues. 200 Dual-loaded liposomes containing apigenin and 5-fluorouracil demonstrate anti-CRC effects by targeting multiple pathways. They reduce angiogenesis, inhibit cell proliferation, and induce apoptosis. Furthermore, they up-regulate the activity of pAMPK and downstream targets, thereby disrupting glycolytic metabolism. 201 Apolipoprotein A1 (ApoA1)-modified liposomes significantly enhance the cellular uptake and specific distribution of baicalin (BA) in the liver. The ApoA1-modified BA liposome (BAA1) demonstrates notable inhibitory effects on HBsAg, HBeAg, HBV RNA, and HBV DNA. BAA1 enhances baicalin bioavailability and regulates the HNFs-ER axis, thereby aiding in the inhibition of HBV transcription and replication. 202 Encapsulation of baicalein into liposomal bilayers exhibits a therapeutic efficacy close to 90% for PDAC. 200
Baicalin and baicalin-loaded nanoliposomes (BA-NL) were found to down-regulate TLR4 in methionine and choline deficient diet (MCD)-induced NAFLD mouse models, reducing the production of inflammatory mediators. BA-NL demonstrated greater efficacy compared to baicalin alone. 203 Nanostructured lipid carriers (HA-GEM-BCL NLCs) were developed using gemcitabine (GEM) and baicalein (BCL) prodrugs via the nanoprecipitation technique. These carriers exhibited high drug loading capacity and stability. In nude mice with subcutaneous grafts of PC AsPC-1 cells, HA-GEM-BCL NLCs showed substantial intratumor accumulation and significant anti-tumor effects. 204
Scutellarin (SCU) was employed as an inducer of immunogenic cell death (ICD) in HCC immunotherapy. Aminoethyl anisamide-targeted polyethylene glycol-modified poly(lactide-co-glycolide) (PLGA-PEG-AEAA) was utilized to enhance the in vivo delivery efficiency of SCU. The PLGA-PEG-AEAA.SCU preparation reversed the immunosuppressive TME and bolstered the immunotherapeutic effect, resulting in a significant inhibition of HCC cell growth. 205 Nanoparticles loaded with apigenin (ApNnp) exhibited a sustained drug release pattern and effectively reached HCC cells in vitro as well as in the livers of oncogenic animals. Following treatment with ApNnp, the potency of apigenin in both the blood and liver significantly increased. The delivery of ApNnp enhanced the therapeutic efficacy of apigenin against HCC. 206 β-sitosterol-assisted silver nanoparticles (BSS-SNPs) significantly inhibited the proliferation of HCC HepG2 cells and induced the expression of ROS and Nrf-2, up-regulated Bax, p53, cytochrome c, and caspases-9, -3 pro-apoptotic markers, and down-regulated Bcl-2 expression induced apoptosis in HCC. 207 Encapsulation of chrysin into poly (d, l-lactic-co-glycolic acid) poly (ethylene glycol) (PLGA-PEG) nanoparticles resulted in enhanced effects compared to free chrysin. PLGA-PEG-chrysin complexes demonstrated increased induction of apoptosis and significantly down-regulated mRNA expression of miR-18a, miR-21, and miR-221 in GC AGS cells. Therefore, utilizing PLGA-PEG as a nanocarrier for chrysin could improve drug efficacy. 208 Chrysin-loaded PLGA-PEG exhibited significantly lower IC50 values compared to chrysin alone, along with greater up-regulation of the expression of miR-22, miR-34a, and miR-126, resulting in inhibition of GC AGS cells. Encapsulation by PLGA-PEG retained all the biological functions of chrysin and improved therapeutic efficiency. 209
β-sitosterol exhibits ameliorative effects on HCC; however, its poor water solubility, low bioavailability, and short elimination half-life limit its application. β-sitosterol-loaded PEGylated niosomes demonstrated greater cytotoxicity potential and enhanced cellular uptake against HCC HepG2 cells. 210 Baicalein (BAI)-micelles (BAI-M) exhibited a sustained release pattern compared to free BAI, thereby improving drug solubility. Additionally, BAI-M showed an approximate 3-fold increase in oral bioavailability, enhancing the therapeutic efficacy against CRC Caco-2 cells. 211
Clinical Research
S. baicalensis, a key component of Chinese patented anti-tumor drugs like Scrophulariae Capsule, Gegen Qinlian Pill, and Dahuang Worm Pill, has found extensive application in cancer treatment, showcasing notable therapeutic efficacy. 212 Kong conducted a study involving 67 cases of malignant obstructive jaundice caused by pancreatic cancer. The control group received conventional therapy supplemented with ERCP biliary stent placement, while the experimental group received Da Bupleurum Decoction (consisting of Chaihu, Rhubarb, Scutellaria baicalensis, Pinelliae, Semixia, Citrus aurantium, Paeoniae lactiflora, etc.) in addition to the treatment received by the control group. The experimental group exhibited a remarkable effective rate of 90%, which was significantly superior to that of the control group. 213 Muhammad Wasif Saif et al conducted a clinical study involving 25 patients with advanced PC who had previously undergone gemcitabine monotherapy or combination therapy. The study investigated the efficacy of PHY906, a preparation comprising S. baicalensis, Glycyrrhiza uralensis Fisch, Ziziphus jujuba Mill, and Paeonia lactiflora Pall, in combination with capecitabine. The results showed that the median progression-free survival (mPFS) was 10.1 weeks, and the median overall survival (mOS) was 21.6 weeks for all patients receiving capecitabine combined with PHY906. Among the 18 patients who completed at least 2 cycles of treatment, the mPFS increased to 12.3 weeks, and the mOS extended to 28 weeks, with a 6-month survival rate of 44%. Furthermore, patients experienced an improvement in quality of life, and the incidence of diarrhea was reduced. The therapeutic effects of PHY906 may be attributed to its ability to inhibit multi-drug resistance, regulate NF-kB activity, down-regulate MMP expression, and inhibit angiogenesis. 214 Shivaani Kummar et al discovered that PHY906 effectively prevented weight loss commonly associated with irinotecan treatment for CRC. Additionally, when combined with irinotecan, 5-FU, and leucovorin for the treatment of advanced CRC, PHY906 not only enhanced therapeutic efficacy but also decreased the incidence of adverse effects such as vomiting, nausea, and grade 3/4 diarrhea. 215 In a study conducted by Zhao and Yj, 60 primary HCC patients were selected. The control group received transcatheter arterial chemoembolization (TACE), while the observation group received TACE with the addition or modification of Huangqin Decoction (comprising S. baicalensis, Jujubae, Paeonia, and licorice). Following treatment, the observation group exhibited a total effective rate of 63.33%, surpassing that of the control group (36.67%). Furthermore, serum levels of HIF-1α, VEGF, and NF-κB were reduced in both groups, with notably lower levels observed in the observation group. The adjunctive use of Huangqin Decoction in TACE for primary HCC showed remarkable efficacy, contributing to improvements in inflammatory response, immune function, liver function, and overall safety. 216
Discussion
Malignant tumors are commonly classified as complex, chronic systemic diseases, with GI cancer being one of the most prevalent types, posing a significant threat to human health and quality of life. S. baicalensis, a traditional natural product, has shown remarkable advantages in treating GI cancer due to its diverse pharmacological activities and flexible application methods. This paper reviews the primary active compounds in S. baicalensis and elucidates their mechanisms of action in treating GI cancer, including the induction of tumor cell apoptosis, inhibition of proliferation, cell cycle arrest, suppression of invasion and metastasis, modulation of the tumor microenvironment, and reversal of chemoresistance. Among the more than 40 compounds isolated from S. baicalensis, flavonoids are the most significant, providing notable pharmacological effects. Other active components, such as terpenoids, polysaccharides, and sterols, also demonstrate anticancer properties, although further study is required. The anticancer mechanisms of S. baicalensis are complex and involve regulating various proteases, genes, proteins, cytokines, immune cells, and associated signaling pathways. Combining S. baicalensis with conventional treatments, such as surgery, radiotherapy, chemotherapy, immunotherapy, and targeted therapy, has been shown to reduce drug toxicity and related side effects, improve immune indices, tumor markers, and chemotherapy tolerance, and significantly enhance patients’ quality of life. This combined approach emphasizes both the physiological and psychological well-being of patients while controlling tumor progression and shrinking lesions.
This paper summarizes the active components of S. baicalensis and their mechanisms of action against GI cancer, as detailed in Table 1, with component-target-pathway information provided in Table 2. The main molecular structural formula of S. baicalensis is shown in Figure 2.217 -231 The active ingredients of S. baicalensis exert their therapeutic effects on GI cancers through multiple pathways, as shown in Figure 3.
Active Components and Antitumor Types of S. baicalensis.
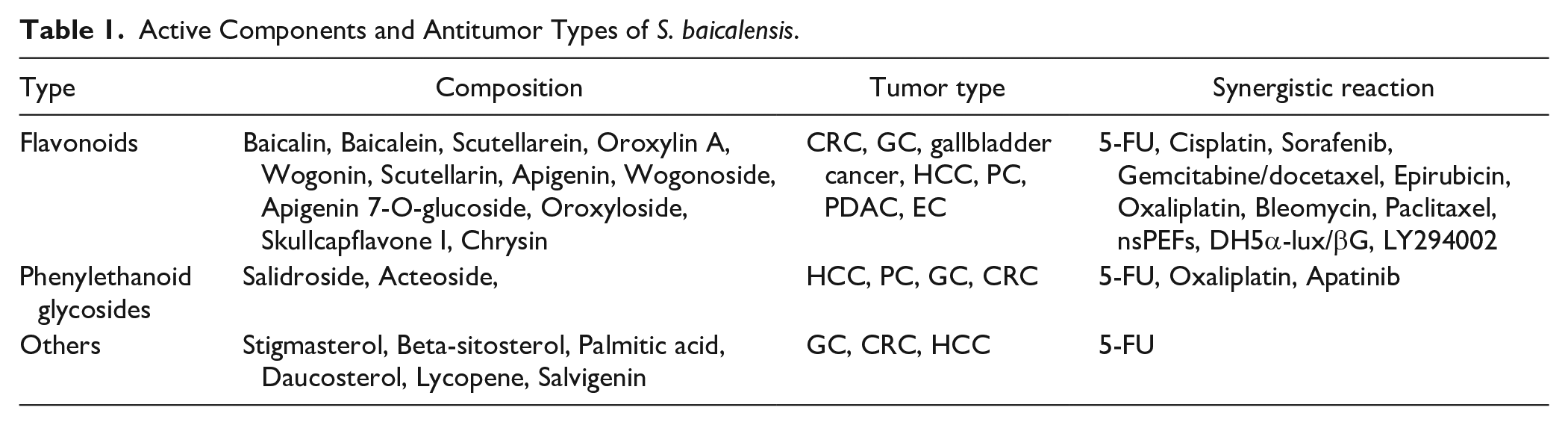
Composition-Target-Pathway of S. baicalensis Against GI Cancer.


Molecular structural formula of the main active ingredients of S. baicalensis. Phenylethanoid glycosides: (A) Acteoside (B) Salidroside; Flavonoid glycosides: (C) Baicalin; Free flavonoids: (D) Scutellarein (E) Oroxylin A (F) Wogonin (G) Chrysin (H)Apigenin (I) Baicalein (J) Skullcapflavone I; Flavonoid glycosides: (K) Apigenin 7-O-glucoside (L) Oroxyloside (M) Wogonoside (N) Scutellarin; Others: (O) Stigmasterol. (Image credit: National Library of Medicine - National Center for Biotechnology Information https://pubchem.ncbi.nlm.nih.gov/).

Component-pathway-organ diagram of S. baicalensis against GI cancer (https://www.figdraw.com).
TCM therapies, with their long history and rich clinical experience, play a crucial role in complementary and alternative medicine, particularly in cancer treatment. Although the therapeutic efficacy of S. baicalensis in treating GI cancer has been studied from various angles, and its multi-targeting characteristics have been confirmed, the scientific evidence supporting these therapies remains limited. Most studies on the anticancer mechanisms of S. baicalensis and its active ingredients focus on in vitro experiments, with in vivo studies on animal models and clinical trials still in their early stages. The complexity of the in vivo environment leaves many mechanisms unclear. Moreover, challenges related to extraction, stability, and drug formulation suggest that S. baicalensis and its components still have a long way to go before becoming widely used clinical drugs. One promising approach is combining S. baicalensis with novel drug delivery systems to address issues of low stability, poor solubility, and bioavailability. While this approach has proven effective for a few key active ingredients, fewer studies have explored nanodelivery technologies for other components of S. baicalensis. Additionally, while combining S. baicalensis with modern drugs holds the potential to enhance efficacy and reduce side effects, more high-quality clinical trials are needed to fully understand its safety, effectiveness, and scope of application.
Given the current state of research, several key challenges need to be addressed: (1) Expanding the multi-target advantages of S. baicalensis using modern molecular biology techniques, conducting in-depth research on its molecular mechanisms against GI cancer, and validating these findings through in vivo studies to provide a solid theoretical foundation for its clinical application; (2) Moving beyond individual molecular mechanisms or signaling pathways to explore the broader relationships between various anti-GI cancer pathways and mechanisms; (3) Continuing to develop the anticancer potential of the various components in S. baicalensis; (4) Extensively evaluating the clinical efficacy of S. baicalensis’s active ingredients in reducing toxicity and enhancing efficacy, supported by clinical trials, especially when used in conjunction with modern drugs; (5) Combining the active ingredients of S. baicalensis with advanced drug delivery technologies to overcome challenges such as low bioavailability and poor pharmacokinetics, exploring more efficient targeting strategies and formulation vehicles, and accelerating the clinical application of these new technologies to improve the targeting and efficacy of cancer treatments.
TCM is an important part of complementary and alternative medicine and serves as a valuable supplement to mainstream medical approaches. It holds broad prospects for improving the clinical outcomes of conventional cancer therapies by addressing issues such as multidrug resistance, alleviating adverse effects, and reducing the mental burden of anticancer treatments. TCM therapies can improve patients’ quality of life, prolong survival, and offer a humanistic perspective on cancer treatment. It is hoped that this review will provide a new theoretical foundation for the clinical application of S. baicalensis in GI cancer treatment, offer promising directions for the development of new anticancer drugs, and contribute to safer and more effective treatment regimens in the future.
Conclusion
S. baicalensis harbors a rich array of anti-tumor active ingredients and mechanisms, promising a bright future as a candidate for anticancer adjuvant therapy. Furthermore, these components have shown potential to mitigate adverse effects and augment therapeutic efficacy when combined with modern medicine. Future endeavors should focus on the development of additional active components of S. baicalensis, paving the way for safe and effective natural agents in the clinical management of malignant tumors. It is hoped that this review can provide new theoretical basis and effective therapeutic regimens for the clinical treatment of S. baicalensis against GI cancer, and provide research directions and ideas for the development of new drugs for S. baicalensis against GI cancer.
Footnotes
Acknowledgements
The authors would like to thank all the reviewers who participated in the review.
Author Contributions
The article presented here was carried out in collaboration between all the authors. Yufeng LYU proposed the research direction and overall guidance; Lan-kang Wang constructed the framework of the paper; Baoyi Ni and Jia Wang wrote the manuscript; Jilai Zhou and Junyi Wang conducted literature analysis and summary; Jiakang Jiang and Yutong Sui conducted a literature search; Feng Gao and Yaoyao Tian contributed to the revision of the article and provided new insights. All authors read and approved the final version of the manuscript.
Data Availability
The data used to support the findings of this study are included within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Not applicable.
