Abstract
Gastrointestinal (GI) cancers cause one-third of all cancer-related deaths worldwide. Natural compounds are emerging as alternative or adjuvant cancer therapies given their distinct advantage of manipulating multiple pathways to both suppress tumor growth and alleviate cancer comorbidities; however, concerns regarding efficacy, bioavailability, and safety are barriers to their development for clinical use. Emodin (1,3,8-trihydroxy-6-methylanthraquinone), a Chinese herb-derived anthraquinone, has been shown to exert anti-tumor effects in colon, liver, and pancreatic cancers. While the mechanisms underlying emodin’s tumoricidal effects continue to be unearthed, recent evidence highlights a role for mitochondrial mediated apoptosis, modulated stress and inflammatory signaling pathways, and blunted angiogenesis. The goals of this review are to (1) highlight emodin’s anti-cancer properties within GI cancers, (2) discuss the known anti-cancer mechanisms of action of emodin, (3) address emodin’s potential as a treatment complementary to standard chemotherapeutics, (4) assess the efficacy and bioavailability of emodin derivatives as they relate to cancer, and (5) evaluate the safety of emodin.
Keywords
Introduction
Gastrointestinal (GI) cancers account for a significant proportion of cancer incidence and mortality; indeed, in 2018 total cancer incidence was estimated to be 4.8 million cases worldwide and GI cancers were responsible for 35% of all cancer-related deaths. 1 Currently, the treatment strategies for these cancers may include cytotoxic chemotherapeutics, radiation, immunotherapy, and/or surgery. 2 Although these treatment paradigms have undoubtedly contributed to increased survival, there has been little advancement in counteracting the pervasive off-target effects and acquired drug resistance that occur with these treatments.2-8 The use of natural compounds has shown promise in both suppressing tumor growth and alleviating adverse effects associated with standard chemotherapeutics.9,10 Indeed, many natural compounds boast of very broad pleiotropic effects, making them potentially attractive candidates to target the multiple pathways driving tumorigenesis and treatment side-effects.9,10 As such, natural compounds that increase apoptosis, decrease proliferation, modulate intracellular signaling, and inhibit tumorigenesis have shown the best potential as anti-cancer agents.9,10 The ability of natural compounds to exert anti-cancer and chemoprotective effects may be associated with improved patient tolerability, quality of life, and survival.9,10 Unfortunately, however, their use in clinical oncology remains extremely limited, likely due to inconsistencies in efficacy and toxicity findings across preclinical and clinical studies.
Emodin (1,3,8-trihydroxy-6-methylanthraquinone), isolated from the roots and barks of several plants, is a Chinese herb-derived anthraquinone that has shown promise pre-clinically as a natural anti-cancer agent for the treatment of various cancers including breast, pancreatic, colon, gastric, liver, gallbladder, and lung.5,6,11,12 Recent evidence suggests that emodin exerts its anti-cancer effects through inducing mitochondrial mediated apoptosis, alongside suppressing pathways that promote inflammation, proliferation, angiogenesis, and tumorigenesis (Table 1). Indeed, emodin has been demonstrated to elicit these effects at both low (1-40 mg/kg)13 -20 and higher (50-80 mg/kg)16,18 -22 doses consistently across pre-clinical models of colon, liver, and pancreatic cancer. Given emodin’s multimodal anti-tumor properties, popularity has grown not only in investigating its potential as a stand-alone treatment, but also as a complementary therapy to existent chemotherapeutics as it has been reported to enhance the sensitivity of select anti-cancer drugs.15,21,23,24 Although emodin’s anti-cancer effects have been previously reviewed,11,12,25,26 this review will profile the most recent (2017-2021) advancements in emodin’s efficacy, safety, and mechanisms regulating its anti-cancer properties in GI cancers, including colon, liver, and pancreas. Additionally, emphasis will be placed on emodin dosing as it pertains to the disparity amongst treatment regimens that has contributed to equivocal results and reported toxicities.
Apoptotic and Tumorigenic Effects of Emodin.

Emodin’s Beneficial Effects on Tumorigenesis Outcomes
Over the last 2 decades we have witnessed an increase in the investigation of natural compounds, including emodin, for their potential as anti-cancer agents. To put this in perspective, a search of the National Library of Medicine using the term “emodin and cancer” revealed 2 publications in 2000 and 58 in 2020. In this section, we will focus largely on the most recent literature (2017-2021) documenting emodin’s effects on GI cancers; we will first address in vitro studies and then will report the available in vivo findings. Of note, there are emerging reports on (1) emodin’s potential as a complementary therapy to standard anti-cancer therapies and (2) anti-cancer effects of emodin derivatives; these will be addressed in sections IV and V, respectively.
In vitro studies
Several recent in vitro studies have documented emodin’s ability to suppress tumor growth in colon, liver, and pancreatic cell lines. We will address the recent in vitro studies investigating emodin’s effects on these 3 GI cancers. Mechanisms for emodin’s reported anti-cancer effects will be addressed in section III.
With regards to colon cancer, Dai et al 20 reported that emodin (15-60 μg/ml) suppressed the growth, adhesion, and migration of HCT116 cells, a human colon cancer cell line. In another study emodin (15-80 µM) decreased viability of human colon cancer cells (DLD-1 and COLO-20) in a time and dose-dependent manner compared to vehicle-treated control without significantly impacting normal colon epithelial cells (CCD841 CoN). 27 Similarly, Gu et al 17 reported that emodin (5-20 µM) inhibited the invasion and migration abilities of the human colon cancer cell line RKO. Another recent study documented that emodin (15-60 µM) exhibited potent anticancer effects on CACO-2 human colon carcinoma cells. 28 Thus, emodin appears to have anti-tumor efficacy in several colon cancer cell lines.17,20,27,28
The evidence for emodin’s protective effects in liver cancer is just as positive with several in vitro studies documenting beneficial effects of emodin in liver cancer cell lines.13,19,29,30 For example, it was recently reported that emodin can inhibit growth of HepG2 cells in a time- and dose- dependent manner. 29 Using the same cell line, Bai et al 13 reported that emodin (10 and 100 nM, 0-36 hours) time dependently suppressed HepG2 cell proliferation in vitro. Similarly, Dong et al 30 demonstrated that emodin time- and dose- dependently inhibited HepaRG cell growth. Finally, it has been reported that emodin inhibited the proliferation of SMMC-7721 cells, also in a dose- and time-dependent manner 19 These studies support a beneficial effect of emodin as an anti-cancer agent with efficacy against several liver cancer cell lines.13,19,29,30
In pancreatic cancer, emodin significantly inhibited the ability of SW1990 cells to proliferate in a dose- and time-dependent manner. 16 Further, it was reported that emodin increased apoptosis in the pancreatic cell lines, PANC-1 and BxPC-3. 23 Similarly, emodin was found to have efficacy against PANC-1 and MiaPaCa2 given its ability to inhibit cell viability. 31 Although limited, the available studies unequivocally document a beneficial effect of emodin on pancreatic cell lines in vitro.16,23,31
Overall, the available in vitro studies are overwhelmingly positive for a beneficial effect of emodin on colon,17,20,27,28 liver,13,19,29,30 and pancreatic16,23,31 cell lines. However, it must be noted that there remains a dearth of evidence in this area; studies are still parsing out effective dosing and timing for optimal efficacy.
In vivo studies
In vivo studies on emodin as a cancer therapeutic agent for GI cancers are just beginning to emerge in the literature. However, those that are available show great promise for the development of emodin as either a stand-alone anti-cancer therapy or as a combinational therapy (addressed in section IV). We will discuss the most recent available in vivo studies on emodin’s influence on GI cancers.
To our knowledge, prior to 2020, only 2 studies used in vivo models of colon cancer treated with emodin.32,33 The earliest available study (2012) reported that emodin (40 mg/kg administered i.p., twice a week for 4 weeks) effectively suppressed tumor growth in nude mice xenografts bearing LS1034. 32 In contrast, in 2017, Braumann et al 33 reported that emodin (5-10 mg/kg) administered twice a day for a week had no significant tumor suppressive effects in a colon cancer model in which tumor cells were injected in the cecum and back in rats. However, more recently, this same group found that emodin (2.5 and 5 mg/kg) administered to rats for a week inhibited tumorigenesis of established solid colon tumors that were originally implanted intraperitoneally (ip) or subcutaneously (sc). 14 Likewise, emodin (50 mg/kg) prevented colon carcinogenesis in an azoxymethane/dextran sodium sulfate (AOM/DSS) mouse model. 22 Similarly, pretreatment with emodin (40 mg/kg, 1 week) in a xenograft model of colon cancer (RKO) suppressed tumor growth. 17 While the number of studies available remains relatively low, the data consistently showed a beneficial effect of emodin on colon cancer in rodent models.14,17,22
The literature presents fewer studies on the effects of emodin on liver or pancreatic cancer; despite this, the findings are very promising. Indeed, several recent studies have documented emodin’s beneficial effects on liver and pancreatic cancer in vivo (Tables 1 and 2). In 2020, Bai et al 13 reported that daily emodin (1 and 10 mg/kg) administration, initiated 1 day after subcutaneous injection of HepG2 cells, inhibited tumor growth and reduced mortality in a dose-dependent manner. Hu et al 21 implanted MiaPaCa2 cells into athymic mice and reported that emodin (50 mg/kg) inhibited cancer-cell growth when administered as at treatment (i.e. 4 weeks after tumor implantation). Further, emodin (50 mg/kg) also attenuated 2 pathological constituents of cancer cachexia in this model, namely high hepatic gluconeogenesis and skeletal-muscle proteolysis. 21 In a model of pancreatic cancer liver metastasis, emodin (20 and 50 mg/kg) was reported to suppress tumorigenesis. 16 Based on the available studies, emodin (1-50 mg/kg) shows promise as an agent to treat liver cancer and pancreatic cancer.13,16,21
Hepatatoprotective and Hepatotoxic Effects of Emodin and Its Derivatives.

Abbreviations: CMC, sodium carboxymethylcellulose; HO-1, heme oxygenase-1; Gpx2, glutathione peroxidase 2, Gsta1/2: glutathione S-transferases, Gadd45a, growth arrest and DNA-damage-inducible protein.
Agent dissolved in DMSO.
Improved liver and kidney function (↓ ALT, AST, AKP, GGT, Cr and BUN).
Dose-dependent manner.
•More effective than ursodeoxycholic acid (UDCA: anti-cholestasis drug) and dexamethasone (DXM: immunosuppresspressive agent).
#↑ALT, AST.
↓TC, alanine transaminase, aspartate transaminase, alkaline phosphatase, γ-glutamyltransferase, total protein, LDL-c, and MDA.
Overall, the available in vivo studies assessing emodin’s anti-cancer effects in GI cancers are strong; in almost all the reported studies emodin shows promise as a stand-alone therapy in the treatment of colon, liver, or pancreatic cancer.13,14,16,17,21,22 Additional studies to confirm these findings as well as studies that address optimal timing and dosing of emodin administration are needed to support the development of emodin for clinical use. Further, it is worth noting that the majority of studies to date appear to be focused on prevention of tumorigenesis. Thus, pre-clinical evidence to support a therapeutic effect of emodin are warranted.
Mechanisms of Emodin as an Anti-Cancer Therapy
It is clear from section II above that emodin shows promise as an anti-cancer agent for GI cancers.13,14,16,17,21,22 However, a clear understanding of its mechanisms is essential to its development for clinical use. This is complicated by the fact that many natural compounds, like emodin, boast of very broad pleiotropic effects. In the succeeding section we will address the known mechanisms of action of emodin as they relate to GI cancers.
Emodin Targets Mitochondrial Mediated Apoptosis
Direct regulation of mitochondrial mediated apoptosis
It has been well documented that emodin induces apoptosis in GI cancers (Table 1); however, the proposed mechanisms of action have not been well described, resulting in inconsistencies across the literature. While apoptosis can be initiated through several regulatory pathways, recent evidence has emerged showing emodin’s ability to induce apoptosis by targeting mitochondria.27,28,30,34 In order for apoptosis to occur, a family of Cysteine proteases named caspases must become activated to cleave several substrates to result in cellular degradation and death. 35 While mitochondrial mediated apoptosis activates caspases through the release of cytochrome c from the mitochondria, the intracellular signaling mechanisms regulating this process are diverse.35,36 While emodin has been demonstrated to induce mitochondrial mediated apoptosis, these conclusions have been made largely from simple outcome measures with little mechanistic insight. This review will attempt to streamline the findings of several papers on emodin induced apoptosis by examining their reported proteins and apoptotic readouts.
Mitochondrial mediated apoptosis is regulated by maintaining the balance between pro- and anti- apoptotic proteins within the mitochondrial membrane. 36 Interestingly, emodin (50 and 100 µM) has been found to primarily localize in the mitochondria of GI cancer cells. 37 Mitochondrial mediated cell death occurs through the inhibition of the anti-apoptotic B-cell lymphoma 2 (Bcl-2) enabling pro-apoptotic Bax, Bak, and PARP to disrupt the mitochondrial membrane potential (MMP) (Figure 1). 36 The loss of MMP triggers the release of cytochrome c to activate caspase -9 and -3 resulting in apoptosis (Figure 1). 36 To this end, emodin and its derivatives have been shown to reduce Bcl-2, disrupt MMP, and increase Bax, PARP, cytochrome c release, and cleaved caspase -9 and -3 to result in apoptosis of colon,27,28,38,39 pancreatic,23,40 and liver19,30,41-43 cancer cells (Table 1).
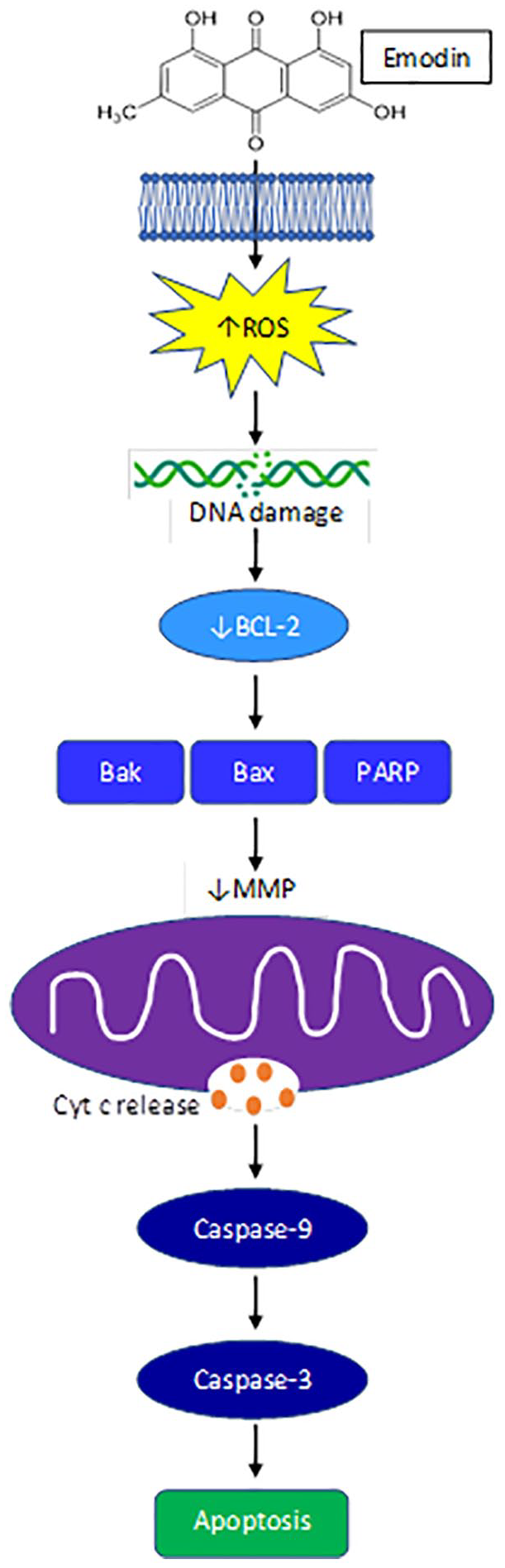
Emodin reduces ROS mitochondrial mediated apoptosis.
While emodin has been shown to modulate several proteins associated with mitochondrial mediated apoptosis in GI cancer cells, the initial cellular stress response required to initiate mitochondrial mediated apoptosis is not as heavily investigated. It has been suggested that the chemical structure of quinones such as emodin, can interact with intracellular molecular oxygen to become superoxide anions, generating reactive oxygen species (ROS).37,44 Although it is possible that emodin could induce other cellular stresses, besides ROS, that are known to result in mitochondrial mediated apoptosis, including ER stress, nutrient deficiencies, hypoxia, etc., 45 this has not been reported for emodin in GI cancers. Emodin has been reported to elicit oxidative stress, primarily via ROS, which was suggested to initiate mitochondrial mediated apoptosis in GI cancers.30,32,37,39,46 Indeed, increased ROS can target structures susceptible to oxidative damage and can disrupt the mitochondrial membrane potential (MMP) and increase cytochrome c release to induce apoptosis. 45 Emodin (20-80 μM) increased intracellular ROS and promoted DNA damage to separately induce mitochondrial dysfunction and subsequent mitochondrial mediated apoptosis, as well as cell cycle arrest through increased p53, p21, and other cyclin E proteins resulting in reduced proliferation in human HepaRG hepatocellular carcinoma (HCC) cells 30 and colon cancer cell lines HCT116 and LS1034 in a dose-dependent manner.32,38 In an in vitro model of HCC, emodin (50 and 100 μM) dose-dependently increased the expression of redox sensitive-mitochondrial matrix protein, cyclophilin D (CypD) that induced MMP loss to further accumulate ROS resulting in mitochondrial-mediated cell death. 37 Emodin (5-40 μM) 46 and aloe-emodin (AE) (10-80 μM) 40 were also shown to promote cell death through inducing autophagy via mitochondrial ROS and the activation of LC3-2. Importantly, emodin does not induce oxidative stress (ROS) nor apoptosis 27 in normal GI cells, 47 or at the very least emodin does not induce ROS in great enough quantities needed to initiate apoptosis as seen in GI cancer cells. An increasing body of evidence supports this notion that the manipulation of ROS by emodin can be selective between cancerous and normal cells. 11 Although not well distinguished in the literature, emodin appears to mediate GI cancer cell death through the promotion of ROS and subsequent mitochondrial damage via the mitochondrial pathway30,32,37,39 (Table 1); however, further work is needed to firmly establish these actions of emodin.
Indirect regulation of mitochondrial mediated apoptosis
The use of natural compounds has the distinct advantage of manipulating numerous aberrant signaling pathways in cancers. Oxidative stress induced by emodin can negatively regulate multiple signaling pathways associated with cell growth, apoptosis, and differentiation to result in GI cancer cell death (Table 1; Figure 1). Emodin at low doses (15-50 μM) was sufficient to induce mitochondrial mediated cell death by suppressing phosphoinositide 3-kinase (PI3K)/Akt,13,19,21,27,28,48,49 mitogen activated protein kinases (MAPK), 27 and extracellular signal-regulated kinase (ERK1/2) signaling (Table 1).13,21,37
The PI3K/Akt and MAPK/ERK signaling pathways are commonly activated in cancers as they promote cancer cell survival, proliferation, and growth. Emodin or its derivatives have been shown to induce liver,13,19 colon,20,27,28,48,49 and pancreatic21,49 cancer cell apoptosis dose dependently at concentrations between 15-200 μM by downregulating PI3K/Akt and AKT-ERK1/2 signaling. Emodin (20-200 μM) also dose dependently decreased the phosphorylation of Akt and Erk1/2 to inhibit pancreatic cancer cell growth and hypoxia, as indicated by decreased HIF-1a. 21 Stress induced activation of these cell regulating pathways, specifically the PI3K/AKT pathway, strains mitochondria to produce energy for survival and provide protection from Redox. 50 This strain eventually results in mitochondrial damage and downregulation of these pathways to promote mitochondrial mediated apoptosis. Altogether, emodin can induce oxidative stress to inhibit numerous signaling pathways which converge in the activation of mitochondrial mediated apoptosis.
Emodin’s Impact on Mechanisms Regulating Tumor Invasion, Metastasis, and Angiogenesis
Epithelial-mesenchymal transition (EMT) and angiogenesis are 2 critical factors that aid in tumor growth, invasion, and metastasis. Tumor EMT is a process in which epithelial cells acquire a mesenchymal, stem-cell-like phenotype possessing increased generation of extracellular (ECM) components and enhanced cell migratory abilities to enhance growth and metastasis. 51 Additionally, sustained growth and metastasis requires the formation of new blood vessels via vascular endothelial growth factor (VEGF)-mediated angiogenesis. 51 Emodin (1-80 mg/kg) has been proposed to inhibit tumor growth through its suppression of angiogenesis and inhibition of EMT.13,16-18,52
The transforming growth factor (TGF)-β/Smad signaling has an established role in tumorigenesis, angiogenesis, and metastasis through ECM remodeling and EMT. 53 Emodin has been shown to reduce TGF-β signaling. 54 Indeed, emodin (20, 40, and 80 mg/kg) was shown to dose dependently inhibit TGF-β1 and upregulate TGF-β1 receptors (TβRI and TβRII) and this was associated with repression of angiogenesis in a pancreatic cancer model (xenograft model using SW1990 human pancreatic cancer cells). 18 Additionally, emodin’s inhibition of TGF-β1 repressed the secretion of Angptl4 by pancreatic tumor cells via Smad4, which is suggested to prevent angiogenesis. 18
Emodin’s inhibition of tumor EMT and promotion of tumor cell migration also has been attributed to its involvement in both Wnt/β-catenin and VEGFR2-AKT-ERK1/2 signaling.13,17 Emodin (40 mg/kg, p.o.) significantly suppressed Wnt/β-catenin signaling in vivo sufficiently to block colon cancer EMT and cell mobility. 17 Emodin (5-20 μM) also reduced colon tumor cell invasion and migration in vitro through reduced VEGF. 17 In addition, it was reported that emodin effectively upregulated the expression of tumor-suppressing miR-34a in cancerous liver tissue, and suppressed VEGFR2-induced activation of AKT, ERK1/2, SMAD2 and SMAD4. 13 Lastly, emodin (20-80 mg/kg, i.p., 3 weeks, 3x/week) suppressed angiogenesis in pancreatic cancer by reducing expression levels of oncogenic and angiogenesis-associated miRNAs, miR-155, and miR-210, and increasing anti-angiogenic miR-20b expression. 18 Drugs that can affect both molecular signaling pathways and miRNAs are of current therapeutic interest as many of them mediate tumorigenesis and cancer progression through the regulation of oncogenes and/or tumor suppressor genes. 55
Emodin as a Complementary Dietary Anti-cancer Therapy
In section II and III above we addressed the effects of emodin as a stand-alone therapy against colon, liver, and pancreatic cancers. Excitingly, there also is an emerging body of literature on emodin’s use as a complementary anti-cancer therapy. In this section we will discuss the available literature on emodin’s efficacy as a complementary therapy in GI cancers along with relevant mechanisms.
Emodin (10-50 mg/kg) has been reported to combat tumorigenesis in HCC and pancreatic cancer models when given in combination with chemotherapeutics.15,21,23,24 Currently, the first-line treatment for advanced HCC is sorafenib, a kinase inhibitor that effectively mitigates tumor growth, angiogenesis, and metastasis. 56 While sorafenib (5 mg/kg, i.p) and emodin (10 mg/kg, i.p.) alone significantly inhibited tumor growth and cell proliferation in HCC cells including HepG2, Hep3B, Huh7, SK-HEP-1, and PLC/PRF5, emodin synergistically increased sorafenib efficacy to result in greater tumor inhibition. 15 Consistently, in the same report, animal models xenografted with HepG2 or SK-HEP-1 cells showed that the combination of emodin and sorafenib, administered following establishment of tumors, was sufficient to inhibit tumor growth suggesting that emodin given as a complementary therapy has the potential to improve efficacy of standard therapies. 15 Mechanistically, the authors associated these benefits of emodin (10 mg/kg, i.p.) with its ability to inhibit sterol regulatory element-binding protein-2 (SREBP-2) transcriptional activity, which suppresses cholesterol biosynthesis and oncogenic protein kinase B (AKT) signaling. 15 Furthermore, attenuated cholesterol synthesis and oncogenic AKT signaling inactivated the oncogenic transcript factor, signal transducer and activator of transcription 3 (STAT3). 15 Additionally, emodin (10 mg/kg, i.p.) synergistically increased cell cycle arrest in the G1 phase and apoptotic cells in the presence of sorafenib (5 mg/kg, i.p). 15
Likewise, to counteract drug resistance of EGFR inhibitors in the treatment of pancreatic cancer, emodin (0-90 μM) was examined in combination with the EGFR inhibitor, afatinib (20 nM), and was reported to reduce proliferation of PANC-1 and BxPC-3 cells. 23 Consistently, the same study reported that the combination of emodin (50 mg/kg) with afatinib (50 mg/kg), administered following tumor establishment, exhibited a synergistic anti-cancer effect in a xenograft pancreatic mouse model. 23 Mechanistically the authors attributed this to emodin’s (50 mg/kg) ability to promote afatinib-induced apoptosis by inhibiting the Stat3 signaling pathway. 23 Several studies have reported benefits when emodin is administered in combination with gemcitabine—a widely utilized first-line drug for advanced pancreatic cancer.24,31 In gemcitabine-resistant PANC-1 pancreatic cancer cell xenografts, the combination of gemcitabine (125 mg/kg) and emodin (40 mg/kg) was shown to significantly reduce xenograft volume and tumor growth in mice compared with treatment with gemcitabine or emodin only. 24 In addition, emodin (40 mg/kg) treatment reduced resistance to gemcitabine, which was attributed to the downregulation of P-glycoprotein, MRP1 and MRP5—drug resistance factors—expression in the group receiving combination treatment. 24 Similarly, in 2 pancreatic cell lines, Panc-1 and MIA-PaCa-2, that were either normal or gemcitabine-resistant, emodin (40 μM) and gemcitabine (20 μM) alone significantly increased the apoptosis rate, but the combination of the 2 drugs further increased the apoptosis rate above the individual treatments in both the normal pancreatic cell lines and the gemcitabine-resistant lines. 31 The authors associated these effects with emodin’s (40 μM) ability to reduce the mRNA and protein expression levels of Survivin, XIAP, NF-κB, and IKKβ, and significantly increase the mRNA and protein expression levels of Caspase-3/9 and IκB-α. 31
Although the data are limited, emodin (10-50 mg/kg) shows promise as a combinational therapy to improve efficacy and reduce resistance of standard anti-cancer drugs.15,23,24,31 This can be attributed to the broad and pleiotropic effects of emodin. Future work in this area should continue to improve our understanding of emodin’s potential as a complementary cancer therapy so that it can be integrated into clinical treatment paradigms.
Anti-cancer Properties of Emodin Derivatives and Formulations
Circumventing the pitfalls of natural compounds like emodin, including poor bioavailability, solubility, and selective cytotoxicity in vivo can be achieved using emodin derivatives and/or formulations. Indeed, there are now several reports of the development of novel emodin-containing drugs to improve its anti-cancer efficacy.34,42,57,58 This section will address the anti-cancer properties of emodin derivatives and/or formulations as they relate to colon, liver, pancreatic cancer.
Several studies have documented improved efficacy of emodin derivatives in liver cancer.34,42,57,58 In one study, a novel series of emodin derivatives were designed and synthesized via the introduction of an amino acid using linkers of varying lengths and composition. 34 In vitro anti-proliferation tests revealed that these derivatives exhibited moderate to potent anti-proliferative activity against HepG2 cells. 34 The authors noted that one specific compound, known as 7a (IC50: 4.95 μM), indicated better selective anti-proliferative activity and specificity than emodin alone. 34 The potential mechanism for this was attributed to a significant effect in inducing cell cycle arrest at G0/G1 phase and inducing cell apoptosis in HepG2 cells via release of cytochrome c and subsequent activation of caspase-9 and caspase-3. 34 In another study, the novel drug compound, β-dihydroartemisinin-emodin (β-DHA-emodin) showed high suppressive activity against the proliferation of HepG2, by inhibiting Ki-67 expression. 42 Further, through modification of emodin’s structure, the derivative emodin succinyl ester (5-20 μM) strongly inhibited HCC cell proliferation and migration in vitro. 58 Another group used emodin-loaded N-acetylaminogalactosyl-poly(lactide-co-glycolide)-succinyl-D-α-tocopherol polyethylene glycol 1000 succinate (GalNAc-PLGA-sTPGS) nanoparticles (EGPTN) to examine efficacy against primary liver cancer; EGPTN was reported to achieve improved anti-cancer efficacy compared to groups without emodin. 57 Similarly, emodin succinyl ester (50-200 mg/kg) dose-dependently suppressed tumorigenicity in xenograft and diethylnitrosamine (DEN)-induced HCC mouse models. 58
With regards to colon cancer and pancreatic cancer fewer reports exist; in fact, we noted just 2 recent reports for colon cancer39,59, and one for pancreatic cancer 40 — all of which used aloe-emodin (AE)—a natural compound extract from Aloe Vera. The first colon cancer study examined the impact of AE on SW620 and HT29 cell lines in vitro; AE (10-40 μM) suppressed cell viability and induced cell apoptosis in SW620 and HT29 cell lines. 39 Furthermore, both cell lines generated ROS when exposed to AE (10-40 μM) supporting its ability to induce apoptosis. 39 The second colon cancer study utilized a genetic model of colorectal cancer, ApcMin/+ mice, treated with or without dextran sodium sulfate (DSS), to induce intestinal mucosa inflammation and multiple neoplasms, that were administered dietary AE. 59 In this study, low dose dietary AE (5 ppm in diet) reduced the number of colorectal tumors in both tumor models. 59 Similarly, AE (0-100 μM) induced dose-dependent cytotoxicity in 2 different pancreatic carcinoma cell lines with MiaPaCa2 cells being more susceptible than PANC-1. 40
In addition to the development of emodin derivatives and formulations to improve efficacy, recent papers also have sought to overcome emodin’s poor solubility observed in water, 10% ethanol, and DMSO. 60 Indeed, a thermoreversible poloxamer gel containing emodin possessed 100-fold greater solubility enhancement and decreased fibroblast cell viability compared to emodin in water or 10% ethanol. 60 In another approach, emodin’s poor solubility may be overcome by using carrier systems such as mesoporous silica nanomaterial (SNM) labeled with fluorochromes. 48 Indeed, nanomaterials loaded with emodin were applied against human colon carcinoma cells HT-29 and found to enter HT-29 cells and release emodin in higher amounts within 48 hours compared to naked emodin. 48 This led to improved bioavailability and selectivity, as well as enhanced autophagy (increased LC3A/B) and mitochondrial mediated apoptosis through increased pro-apoptotic proteins (compared to naked emodin), phosphorylated-AKT downregulation, PI3K/Akt pathway inhibition, and increased phosphorylated-ERK in HT-29 cells at concentrations of 20-80 μM. 48
Overall, there is great potential for emodin derivatives and or formulations to be used in the treatment of GI cancers. In most, if not all cases, the available studies on emodin derivatives and/or formulations show improved efficacy over emodin alone.34,39,40,42,57-59 Further, research is underway to improve the solubility of emodin, which also has resulted in improved efficacy.48,60 These data underscore the potential of this natural compound in the cancer treatment domain.
Safety of Emodin
Evaluation of the safety of dietary compounds is principal to their clinical development and emodin is no exception. Despite the positive effects on tumorigenesis, several studies have reported side effects of emodin that may preclude its development beyond pre-clinical studies. Given the focus of this review on GI cancers, in this section we will address side effects of emodin as related to the GI system. On the contrary, there also is a literature base supporting a protective effect of emodin on the GI system and therefore this will be addressed.
Toxicity of Emodin
There are now a number of reports of hepatotoxic effects of emodin that appear to be dose dependent (Table 2). In one recent study, rats were administered 150, 500, or 1500 mg/kg of emodin daily for 4 weeks and liver damage was assessed. 61 The findings revealed that as the dose of emodin increased, the damage was accelerated, and this was also time dependent. 61 Specifically, inflammation was considered minimal when administered at 150 mg/kg, but was more elevated in the 500 mg/kg group, and was rated as serious in the 1500 mg/kg group and associated with the presence of fat vacuoles. 61 These findings were consistent with alanine transaminase (ALT) and aspartate transaminase (AST) levels supporting a toxic effect of emodin in the liver. 61 In a similar study, rats administered 150 mg/kg of emodin daily for 4 weeks exhibited significant increases in levels of ALT and AST indicating damage to the liver. 62
The mechanisms responsible for emodin-induced hepatotoxicity continue to be unearthed but several studies implicated mitochondria and oxidative stress in playing a role.61 -64 Indeed, 1500 mg/kg daily dose of emodin for 4 weeks inhibited proton transport and subsequently induced the activation of the mitochondrial apoptosis pathway. 61 A role for mitochondria also was implicated in another study, suggesting emodin (160 μM) to induce oxidative stress in the liver by directly targeting acadvl/complex IV and inhibiting fatty acid β-oxidation, citric acid cycle, and oxidative phosphorylation in mitochondria. 62 It is well known that redox homeostasis is maintained by antioxidant production as antioxidants help in degrading free radicals such as ROS. Emodin at very high doses (150-10 000 mg/kg) down-regulates antioxidant proteins thus suppressing antioxidant defenses that may increase susceptibility to prolonged oxidative stress and excessive ROS production.62,64,65 Oxidative stress resulting in a prolonged imbalance between the production of ROS with their elimination by antioxidants in the liver can contribute to liver inflammation leading to fibrosis and HCC. 66 Specifically, high dosage of emodin (150 mg/kg, daily, 4 weeks) induced hepatotoxicity by inhibiting antioxidants within the mitochondria, including peroxiredoxins (Prdx), glutathione peroxidases (Gpx), thioredoxin (Trx), and glutaredoxin (Glrx), all of which help in the degradation of free radicals such as ROS. 62 Emodin’s inhibition of these antioxidants allowed for excessive ROS accumulation that inhibited the activity of all mitochondrial respiratory chain complexes (oxidative phosphorylation) that led to prolonged oxidative stress, mitochondrial damage, chronic inflammation, and liver injury. 62 Similarly, high dose aloe-emodin (800 or 1,600 mg/kg) resulted in oxidative redox imbalance leading to increased accumulation of ROS, mitochondrial dysfunction, and cell death. 64 In summary, the inhibition of mitochondrial pathways along with the downregulation of anti-oxidant proteins helps to explain high dose emodin’s (150-10,000 mg/kg) ability to promote prolonged redox imbalance, excessive ROS accumulation, and oxidative stress resulting in liver damage.61 -64
In addition to reports of hepatoxicity, there have been some studies that have documented laxative effects of emodin.44,67 In one study in mice, treatment with emodin (1, 2, and 3 g/kg) increased the fecal water content in the colon of mice and evaluation index of defecation in a dose-dependent manner; this was associated with upregulation of aquaporin 3—a protein that plays an important role in regulation of water transfer in the colon. 67
While it is clear that emodin does exhibit some toxicity, some perspective is needed when interpreting these findings. The hepatotoxicity and laxative effects that have been documented were following extremely high doses of emodin. For example, the liver damage documented in rats was following a daily dose of emodin 100x the clinical equivalent of rhubarb. 61 Similarly, the laxative effects have been documented following 1-3 g/kg of emodin daily. 67 Of note, this is much higher than the dose range (1-50 mg/kg) that boasts of anti-cancer effects discussed in section II. Finally, in a sub-chronic (12 week) toxicity study using 3 different doses of emodin (~20 mg/kg, 40 mg/kg, and 80 mg/kg) it was reported that emodin did not cause pathophysiological perturbations in major organs nor did it cause an increase in ALT or AST. 68 While assessing toxicity is certainly an important aspect for the development of emodin as an anti-cancer agent; it should not preclude its progress in this regard, given that most every current cancer drug has potent dose-dependent toxicities. Indeed, just as there are reports of toxicity with emodin, there also is a body of evidence that documents a protective role of emodin that is discussed below.
Organ Protective Functions of Emodin
With regard to the liver, based on a review of the literature it is likely that emodin has bidirectional potential; thus, in addition to the reported hepatoxicity described above, liver protection also has been documented (Table 2).68 -77 In a model of alcohol-induced fatty liver injury, emodin ameliorated liver steatosis, lowered ALT and AST, and decreased hepatic oxidative stress. 69 This finding was supported in vitro where emodin (10-100 μM) suppressed ethanol-induced cytotoxicity in Hep2G cells. 70 In another study, the role of emodin (20 mg/kg) in protection against fibrogenesis caused by carbon tetrachloride was examined in rats. 71 Emodin (20 mg/kg), was able to counteract the hepatic fibrosis induced by carbon tetrachloride. 71 Further, levels of ALT and AST and hydroxyproline content were significantly reduced following the carbon tetrachloride insult in rats administered emodin (20 mg/kg). 71 Consistent with this, another study used the same insult (carbon tetrachloride) and administered water extracts from fresh Polygonum multiflorum, containing emodin (3 mg/kg), to rats for 56 days. 72 It was found that the extract containing emodin reduced ALT and AST, decreased inflammatory mediators and malonaldehyde, and rescued glutathione S-transferase and catalase activity. 72 In addition, fatty degeneration and necrosis were found to be reduced following treatment with the emodin containing extract. 72 In a study of acetaminophen-induced toxicity, treatment with emodin (30 and 40 mg/kg, p.o.) conferred hepatoprotective ability. 73 Using a model of concanavalin A (Con A)-induced hepatitis in mice, emodin (12.5-50 mg/kg) was examined for its protective properties. 75 Consistent with other reports, emodin (12.5-50 mg/kg) decreased circulating ALT and AST and reduced liver necrosis. 75 In addition, there was a marked decrease in pro-inflammatory mediators with emodin (12.5-50 mg/kg) and this was consistent with a decrease in activation of p38 MAPK and NF-κB. 75
Studies utilizing lower doses (1-100 mg/kg) overwhelmingly support a hepatoprotective role of emodin following various insults (Table 2). Thus, it is safe to conclude that emodin possesses bidirectional potential with regards to liver toxicity; indeed, low doses of emodin (1-100 mg/kg) offer hepatoprotection whereas higher doses (500-3000 mg/kg) promote toxicity. The mechanisms responsible for emodin-induced hepatoprotection and hepatotoxicity are also bidirectional in their modulation of oxidative stress, ROS, and antioxidants which is dosage dependent (Table 2). At lower doses (10-20 uM in vitro and 10-40 mg/kg in vivo), emodin protects against non-cancerous hypoxic- and chemical- induced liver damage and hepatic oxidative stress by reducing ROS, oxidative stress (↓MDA), and hepatocellular apoptosis as well as upregulating antioxidants (↑SOD and ↑GSH)47,78 (Table 2). While at higher doses (150-10,000 mg/kg), emodin results in liver damage by promoting prolonged redox imbalance, excessive ROS accumulation and oxidative stress, as well as by down-regulating antioxidant proteins to further increase ROS.61 -64 This underscores the importance of integrating dose response studies into the development of natural compounds. Further, it supports the safety of emodin, at doses below 100 mg/kg, for potential use as an anti-cancer agent.
Conclusions
Given the global burden of GI cancers, treatment options that target multi-aspects of tumorigenesis or increase the efficacy of currently approved drugs are continually being sought after. Through the manipulation of multiple pathways, plant-derived agents have been suggested to elicit their tumoricidal effects without displaying the high toxicity exhibited by traditional chemotherapeutics. Emodin is a promising alternative anti-cancer drug that has documented efficacy in pre-clinical models of colon, liver, and pancreatic cancers. While the data continue to accumulate, we can conclude the most, if not all, reports of emodin’s anti-cancer efficacy against GI cancers are positive and using doses that are attainable in humans. Indeed, even at the higher effective pre-clinical dose range of 50-80 mg/kg16,18 -22 the human equivalent dose would be 4-6.5 mg/kg. 79 Evidence suggests that emodin exerts its anti-cancer effects through inducing mitochondrial mediated apoptosis, alongside suppressing pathways that promote inflammation, proliferation, angiogenesis, and tumorigenesis. Equally exciting is the ability of emodin to enhance the efficacy of anti-cancer drugs including sorafenib, afatinib, and gemcitabine, supporting its potential as a complementary treatment to standard chemotherapeutics. As with any natural compound there are concerns surrounding bioavailability; emodin is no exception. To counteract this there are studies that have successfully improved bioavailability of emodin. Finally, there is a body of literature that has addressed toxicity of emodin. Based on a review of the literature emodin possesses bidirectional potential; lower doses of emodin exhibit hepatoprotection whereas higher doses are hepatotoxic. In conclusion, emodin offers great potential as either a stand-alone treatment or a complementary treatment to existing chemotherapeutics that target GI cancers, and should be further developed in this regard.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
