Abstract
Background:
The traditional Chinese medicine (TCM) Xiaoliu Pingyi recipe (XLPYR) has been clinically used for several decades, demonstrating favorable therapeutic effects. However, the underlying regulatory mechanisms remain unclear. The aim of this study was to explore the anti-tumor effects of XLPYR and its regulatory role in the vascular microenvironment through in vivo and in vitro experiment.
Materials and methods:
In the in vivo study, a C57BL/6J mouse model of lung adenocarcinoma (LUAD) allografts was established, and various interventions were administered for 14 days (Model group: administered normal saline via oral gavage; Pemetrexed (PEM) group: intraperitoneally injected with a solution of pemetrexed, once every 3d; XLPYR group: administered XLPYR via oral gavage; Combination (COMBI) group: received XLPYR via oral gavage simultaneously with intraperitoneal injection of pemetrexed solution). Tumor volume and weight were then compared among the groups. The impact of XLPYR on the tumor vascular microenvironment was assessed using immunohistochemistry staining. In the in vitro study, XLPYR-containing serum was prepared by oral administration to SD rats. The CCK-8 assay evaluated the effect of the serum on the proliferation of normal lung epithelial BEAS-2B cells and LUAD A549 cells, determining the optimal intervention concentrations. The cell migration and invasion abilities were evaluated using the wound-healing assay and Transwell assay, respectively. Finally, ELISA assay measured VEGF secretion levels in the LUAD cell supernatant, and RT-qPCR and Western Blot were employed to detect differences in HIF-1α, VEGFA, Ang-2, and PI3K/Akt mRNA and protein expression levels in both in vivo and in vitro experiments.
Results:
In the in vivo study, XLPYR significantly inhibited the growth of mice LUAD allografts, with enhanced anti-tumor effects observed with prolonged drug intervention. Immunohistochemistry staining revealed reduced MVD and increased pericyte coverage in all intervention groups. Regarding vascular function, FITC-Dextran extravasation in the tumor tissues of the Model group was significantly higher than in the intervention groups, particularly with lower extravasation in the COMBI group compared to the PEM group. In the in vitro study, XLPYR demonstrated a time- and concentration-dependent inhibitory effect on LUAD cells, and with greater sensitivity in inhibiting LUAD cells compared to BEAS-2B cells. The wound-healing assay and Transwell assay confirmed that XLPYR significantly suppressed the migration and invasion abilities of LUAD cells. ELISA experiments further revealed a significant decrease in VEGF expression in the supernatant of each intervention group. RT-qPCR and Western Blot results showed consistent findings between the in vivo and in vitro experiments. HIF-1α, VEGFA, and Ang-2 mRNA and protein expression levels were significantly downregulated in the PEM group, XLPYR group, and COMBI group. There were no significant differences in the expression of PI3K and Akt mRNA and total protein, but the expression levels of phosphorylated p-PI3K and p-Akt were notably downregulated
Conclusion:
XLPYR significantly inhibited C57BL/6J mouse LUAD allograft growth and improved the vascular microenvironment, thereby intervening in tumor angiogenesis and inducing vascular normalization. It suppressed LUAD cell proliferation, migration, and invasion, while reducing VEGF concentration in the cell supernatant. The regulatory mechanism may involve inhibiting PI3K/Akt protein phosphorylation and downregulating angiogenesis-related factors, such as HIF-1α, VEGF, and Ang-2.
Keywords
Introduction
Lung cancer is one of the most common malignant tumors worldwide, with the highest mortality rate. Its incidence is second only to breast cancer, and both incidence and mortality rates are still on the rise year by year, posing a serious threat to human health. 1 In the pathological classification of lung cancer, non-small cell lung cancer (NSCLC) accounts for approximately 85% of cases, with lung adenocarcinoma (LUAD) comprising 45% of NSCLC cases. 2 In recent decades, despite significant advancements in multimodal treatment strategies, including targeted therapy, radiotherapy, and immunotherapy, the 5-year overall survival rate for LUAD remains approximately 18%.3,4
The tumor vasculature microenvironment is distinguished by the presence of an aberrant vascular architecture and compromised functionality within the tumor tissue. This abnormal vessel structure hampers proper blood perfusion, resulting in insufficient delivery of oxygen and nutrients to the tumor cells, as well as impediments in the clearance of metabolic waste. Consequently, a state of hypoxia and acidosis arises within the tumor microenvironment (TME), thereby facilitating the progression of the tumor. Moreover, the heightened permeability of the vasculature affords the tumor the ability to metastasize to remote organs.5,6 Tumor angiogenesis is regulated by the delicate balance between pro-angiogenic, and anti-angiogenic factors present within the tissue. this equilibrium is inclined toward the pro-angiogenic spectrum, prompting vascular remodeling and the acquisition of an activated angiogenic phenotype. Consequently, this augmented angiogenesis fosters the proliferation and migration of endothelial cells (ECs), thus facilitating their dissemination to distant organs.7,8 These empirical observations substantiate the pivotal role of angiogenesis inhibition as an exceedingly effective therapeutic approach against NSCLC, as evidenced by the favorable therapeutic outcomes of angiogenesis-targeting pharmaceutical agents in clinical settings. 9 While numerous molecules have been discovered to play crucial roles in angiogenesis, the majority of research conducted thus far has still focused on vascular endothelial growth factor (VEGF) and its receptors. 10 In recent years, novel classes of drugs targeting alternative effectors within the angiogenic signaling pathways have been developed. These include inhibitors of vascular endothelial growth factor receptors (VEGFRs), platelet-derived growth factors (PDGFs), fibroblast growth factors (FGFs), and angiopoietins (ANGs). These new types of drugs have been widely used in the treatment of various cancers, including lung cancer.11,12 Bevacizumab and similar drugs exert their effects by inhibiting the VEGF signaling pathway, leading to the pruning of abnormal blood vessels and remodeling of the remaining vasculature, resulting in the establishment of a normalized vascular system. However, conventional anti-angiogenic agents, when administered at high doses or with excessive potency, may unintentionally shift the balance toward anti-angiogenesis. This can lead to excessive pruning of tumor vasculature, exacerbating hypoxia, increasing interstitial pressure, and potentially promoting metastasis. 13 Therefore, the identification of drugs capable of modulating the vascular microenvironment and promoting vascular normalization is an imperative challenge in the field of anti-tumor angiogenesis.
In recent years, with the continuous deepening of research on traditional Chinese medicine (TCM), significant advantages have been discovered in its treatment of LUAD. Compared to traditional anti-tumor treatments, TCM not only ensures therapeutic efficacy but also exhibits lower toxicity. When employed synergistically with conventional anti-tumor therapies, TCM exerts a beneficial impact referred to as “enhanced efficacy and reduced toxicity.”14,15 The understanding of meridians in TCM exhibits certain consistency with the knowledge of vascular microenvironment in modern biology. The Xiaoliu Pingyi recipe (XLPYR), developed based on the theory of meridian disease, has demonstrated promising clinical efficacy in anti-tumor and metastasis-dispersing effects. It is tailored to the characteristics of lung cancer meridian disease, which involves a combination of deficiency and excess, as well as blood stasis and toxin accumulation. This recipe utilizes Chinese herbal medicine and animal-based medicine, including Ginseng radix et rhizoma (Renshen), Astragali radix (Huangqi), Curcumae rhizome (Ezhu), Scolopendra (Wugong), Fritillariae thunbergii bulbus (Zhebeimu), Herba Hedyotis Diffusae (Baihuasheshecao), and Glycyrrhizae Radix et Rhizoma (Gancao), aiming to tonify Qi, promote blood circulation, eliminate blood stasis, and detoxify the body. 16
In the present study, we established a Lewis lung cancer allograft model in C57BL/6J mice and prepared drug-containing serum from rats to observe the anti-tumor effects of the XLPYR in both in vivo and in vitro settings. We aimed to investigate its impact on the tumor vascular microenvironment and explore the underlying regulatory mechanisms. By doing so, we aim to further enrich the modern biological understanding of TCM theory and provide new scientific evidence for the clinical application of the XLPYR.
Materials and Methods
Preparation of XLPYR
XLPYR comprises Ginseng radix et rhizoma (10 g), Astragali radix (30 g), Curcumae rhizoma (15 g), Scolopendra (2 g), Fritillariae thunbergii bulbus (20 g), Herba Hedyotis Diffusae (30 g), and Glycyrrhizae Radix et Rhizoma (6 g), which were obtained from Tianjiang Pharmaceutical Co., Ltd. (Jiangyin, China). The formulation of this recipe utilizes a granule dosage form, and the details of all herbs are shown in Table 1. The XLPYR granules had a total weight of 16.8 g. The daily dose for experimental animals was determined using conversion factors of 9.1 for mice and 6.3 for rats, based on an average adult body weight of 60 kg. The calculated daily dose was 2.548 g/kg for mice and 1.764 g/kg for rats.
The Composition of XLPYR.

Drugs, Reagents, and Antibodies
The pemetrexed (PEM, LY231514, CAS No.: 137281-23-3) used in the in vitro experiments was purchased from MedChemExpress (MCE, Princeton, NJ, USA), while the pemetrexed used in the in vivo experiments was obtained from Qilu Pharmaceutical, Co., Ltd (Lot number: 2G0035B34, Jinan, China).
RPMI-1640 and DMEM were purchased from Hyclone (Logan, UT, USA). Fetal bovine serum (FBS) was purchased from ExCell Bio (Taicang, China). Penicillin-Streptomycin liquid and Trypsin-EDTA liquid were purchased from Gibco (Grand Island, NY, USA). Bovine Serum Albumin (BSA), Cy3 conjugated Goat Anti-Rabbit IgG (#GB21303), Cy3-Tyramide (#G1223), Cy5 conjugated Goat Anti-rabbit IgG (#GB27303), and DAPI staining solution (#G1012) were bought from Servicebio (Wuhan, China). FITC-Dextran (#HY-128868) was bought from MCE. Cell Counting Kit-8 (CCK-8) Assay Kit was purchased from Dojindo (#CK04, Kumamoto, Japan). Matrigel matrix and transwell plates with 8 μm pore were purchased from Corning (#356234, New York, NY, USA). Human Vascular Endothelial Cell Growth Factor (VEGF) ELISA KIT was bought from CUSABIO (#CSB-E11718h, Wuhan, China). SPARKscript II RT Plus Kit (With gDNA Eraser) (#AG0304) and 2×SYBR Green qPCR Mix (With ROX) (#AH0104) used in the RT-qPCR assay were bought from Sparkjade (Qingdao, China). About 10% SDS-PAGE Gel Quick Preparation Kit was bought from Sparkjade. RIPA buffer (high), PMSF Solution, BCA Protein Assay Kit, 5× SDS-PAGE loading buffer (with DTT) and ECL Western Blotting Substrate were purchased from Solarbio (Beijing, China). About 10 to 180 kDa PageRuler Prestained Protein Ladder was bought from Thermo Scientific (Rockford, IL, USA). 0.45 μm PVDF membranes were purchased from Millipore (Bedford, MA, USA).
Primary antibodies against total PI3K (#4249), total Akt (#4691) and phosphor-Akt (p-Akt, #4060), HIF-1α (#36169) were purchased from Cell Signaling Technology (Danvers, MA, USA), phosphor-PI3K (p-PI3K, #bs-5570R) was purchased from Bioss (Beijing, China), Ang-2 (#ab155106) was purchased from Abcam (Cambridge, UK), GAPDH (#10494-1-AP) was purchased from Proteintech (Wuhan, China), VEGFA (#GB11034B), CD31 (#GB11063-2), α-SMA (#GB111364) were purchased from Servicebio. Second antibody HRP conjugated Goat Anti-Rabbit IgG (#GB23303) was purchased from Servicebio.
Analysis of XLPYR by UPLC-Q-Orbitrap-MS/MS
The component analysis of XLPYR was carried out on an UltiMate™ 3000 RS HPLC Systems coupled to a Q Exactive™ Hybrid Quadrupole-Orbitrap™ Mass Spectrometer (Thermo Fisher Scientific, Waltham, MA, USA). 17 The chromatographic separation utilized an Ultimate AQ-C18 column (150 mm × 2.1 mm, 1.8 μm; Welch Materials, Shanghai, China) at 35°C. Mobile phase A was 0.1% formic acid aqueous solution, and mobile phase B was methanol. Gradient elution (0.30 mL/min) proceeded as follows: 0 to 1 minute (2% B), 1 to 5 minutes (2%-20% B), 5 to 10 minutes (20%-50% B), 10 to 15 minutes (50%-80% B), 15 to 20 minutes (80%-95% B), 20 to 27 minutes (95% B), 27 to 28 minutes (95-2% B), 28 to 30 minutes (2% B). Injection volume: 5 μL. MS analysis used ESI source in positive- and negative-ion modes with sheath gas flow rate 40 Arb, auxiliary air velocity 15 Arb, auxiliary gas temperature 350°C, scanning mode: Full mass/dd-MS2, resolution: 70 000 (Full mass); 17 500 (dd-MS2). Data collected from 100.0–1500.0 m/z.
Cell Lines and Cell Culture
The human LUAD cell line (A549, RRID:CVCL_0023) was obtained from the Shandong Academy of Medical Sciences (Jinan, China), and human normal lung epithelial cell line (BEAS-2B, RRID:CVCL_0168) was obtained from the ATCC. Mouse LUAD cell line (Lewis Lung Carcinoma, LLC, RRID:CVCL_4358) was purchased from iCell Bioscience Inc (Shanghai, China). A549 and BEAS-2B cells were cultured in RPMI-1640 supplemented with 10% FBS and 1% penicillin-streptomycin, LLC cells were cultured in DMEM supplemented with 10% FBS and 1% penicillin-streptomycin. All cells were maintained at 37°C and 5% CO2 in a humidified incubator.
Animals Experiment and Preparation of Drug-Containing Serum
The experimental female C57BL/6J mice aged 6 to 8 weeks and female SD rats aged 8 to 9 weeks were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). All animals were reared in a SPF environment at the Animal Experimental Center of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine. The animals were maintained on a 12-hour light-dark cycle with a temperature of 22°C to 26°C and humidity ranging from 40% to 60%. The experimental protocol involving animals was reviewed and approved by the Ethics Committee of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine (Ethical approval number: 2021-82).
The LLC cell density was adjusted to 1 × 107 cells/mL using cooled PBS. Subsequently, the cell suspension was subcutaneously injected into the right axilla of each mouse at a dose of 100 μL per 10 g body weight to establish LLC tumor-bearing mice. After 7 days of injection, subcutaneous tumors were palpable in mice, indicating successful tumor modeling. LLC tumor-bearing mice were randomly divided into 4 groups and subjected to a 14 days consecutive intervention: the model group (administered normal saline via oral gavage at a dose of 0.1 mL per 10 g body weight, n = 6); the pemetrexed group (PEM group, intraperitoneally injected with a solution of pemetrexed at a concentration of 15 mg/mL, at a dose of 0.1 mL per 10 g body weight, once every 3 days, n = 6); the XLPYR group (XLPYR granules was dissolved in normal saline and administered via oral gavage at a dose of 0.1 mL per 10 g body weight, n = 6); the combination group (COMBI group, received XLPYR via oral gavage simultaneously with intraperitoneal injection of pemetrexed solution, n = 6). The tumor volume was measured and recorded daily following the initiation of drug intervention, according to the following formula: tumor volume = 0.5 × length (mm) × width 2 (mm). At the end of the 14 days intervention period, all mice were sacrificed, and LUAD tissues were excised.
Drug-containing serum was prepared using SD rats in this study. Thirty rats were randomly assigned to the XLPYR group (n = 20) and the control group (n = 10). The XLPYR group received oral gavage of XLPYR at a dose of 10 mL/kg of body weight, while the control group received normal saline. Administration was conducted daily for 7 days, with a 12 hours fasting period before the last administration, but with free access to water. Blood samples were collected through abdominal aortic puncture 2 hours after the final administration. The samples were kept at room temperature for 2 hours, then centrifuged at 3000 rpm for 15 minutes. The supernatant was heat-inactivated at 56°C for 30 minutes and filtered through a 0.22 μm microporous filter for sterilization. The filtered serum was stored at −80°C until further use.
Immunohistochemistry Staining
After excising the LUAD tissues from the mice, they were fixed in 4% paraformaldehyde solution for more than 24h. Paraffin sections were prepared (approximately 4 μm thick), and immunofluorescent double staining was performed using CD31 and α-SMA markers to observe microvessel density (MVD) and pericyte coverage within the tissues. 18 Briefly, after incubating with a 3% H2O2 for 25 minutes, the samples were washed 3 times with PBS and air-dried. Then, 3% BSA was added for 30 minutes to block nonspecific binding sites. After removing the blocking solution, CD31 primary antibody (dilution ratio 1:1000) was added and incubated overnight at 4°C. The next day, the slides were taken out, washed 3 times with PBS, and incubated with the secondary antibody (dilution ratio 1:500) at room temperature in the dark for 1h. After completion of the incubation, the slides were washed 3 times with PBS and air-dried. CY3-Tyramide was added and incubated in the dark for 10 minutes, followed by 3 washes with TBST. Using EDTA (pH 8.0) antigen retrieval solution, the bound primary antibodies were removed, and then α-SMA primary antibody was added for overnight incubation. The following day, after washing the slides 3 times with PBS, Cy5-labeled goat anti-rabbit IgG (dilution ratio 1:400) was added and incubated in the dark for 1h. After completion of the incubation, the slides were air-dried, stained with DAPI for 10 minutes, and mounted with antifade mounting medium. Fluorescence images were captured under a fluorescence microscope (Nikon, Tokyo, Japan).
Thirty minutes before sacrificing the mice, FITC-Dextran was intravenously injected via the tail vein at a dose of 10 mg/kg. After excising the LUAD tissues, they were rapidly frozen in liquid nitrogen and used to prepare frozen sections (approximately 8 μm thick). CD31 and FITC-Dextran fluorescence markers were utilized to observe vascular permeability within the tissues. Briefly, the sections were blocked with 3% BSA for 30 minutes. After completing the blocking step, the excess liquid was removed, and CD31 primary antibody was added, followed by overnight incubation at 4°C. The next day, the sections were washed 3 times with PBS, and then Cy3-labeled goat anti-rabbit IgG was added and incubated at room temperature in the dark for 1h. After DAPI staining for cell nuclei, the sections were mounted with antifade mounting medium to prevent fluorescence quenching. Fluorescence images were captured and recorded under a fluorescence microscope.
Cell Viability Assay
Cell viability was assessed using the CCK-8 Assay Kit according to the manufacturer’s instructions. 19 A549 and BEAS-2B cells were adjusted to a density of 5 × 104 cells/mL in cell suspension and seeded at 100 μL per well in a 96-well plate, incubated overnight. The next day, after observing cell adhesion, the original culture medium was discarded, and the cells were washed twice with PBS. The drug-containing serum was then diluted with RPMI-1640 to different concentration gradients (2.5%, 5%, 10%, 20%), with a control group of blank rat serum at the same concentration gradients. Each group had 5 replicates, and RPMI-1640 medium was added around the 96-well plate for zero calibration. After incubating for 24, 48, and 72 hours, 10 μL of CCK-8 reagent was added to each well and incubated for 2 hours at 37°C. The optical density (OD) values of each group were measured at 450 nm to calculate the relative cell proliferation rate.
Wound-Healing Assay
A549 cells were seeded at a density of 5 × 105 cells/mL in 6-well plates (3 mL per well) and incubated overnight. Once the cells reached >80% confluence, the culture medium was removed, and a scratch was made vertically using a 200 μL pipette tip. After washing the cells twice with PBS, the initial scratch width was recorded under an inverted microscope. The cells were then treated with drugs according to the groups and further incubated for 24 hours. After incubation, images were captured (Carl Zeiss, Oberkochen, Germany), and the cell migration rate was calculated using the ImageJ software with the formula: Cell migration rate = (initial scratch area − 24 hours scratch area)/initial scratch area × 100%.
Transwell Invasion Assay
Matrigel matrix gel (diluted 8 times with serum-free RPMI-1640 medium) was added to each Transwell upper chamber (50 μL per chamber) and solidified overnight at 4°C. About 60 μL RPMI-1640 was added to the upper chamber and equilibrated in the cell culture incubator for 30 minutes. Next, the culture medium was removed, and cells from each group were resuspended in their respective medium to achieve a cell density of 1 × 105 cells/mL. About 200 μL of the cell suspension were then added to each Transwell upper chamber with 3 replicates per group. The lower chambers were filled with 700 μL of RPMI-1640 medium containing 20% FBS, and the plates were incubated in the cell culture incubator for 24 hours. After incubation, the liquid in both the upper and lower chambers was discarded, and any remaining cells and Matrigel in the upper chamber were gently removed. The upper chamber was fixed with 4% paraformaldehyde for 30 minutes. Following fixation, the upper chamber was washed twice with PBS and stained with 0.1% crystal violet solution for 10 minutes. After staining, the upper chamber was washed twice with PBS, and cells on the Transwell membrane were observed and counted under a microscope. 20
ELISA Assay
After 24 hours of drug intervention, cell culture supernatants were collected from A549 cells. Prior to the experiment, the Human VEGF ELISA Kit was taken out from the refrigerator and allowed to equilibrate at room temperature for at least 30 minutes. ELISA assay was performed according to the manufacturer’s instructions. 21 After the color reaction was terminated, the OD values of each group were measured at a wavelength of 450 nm within 5 minutes. VEGF levels in the cell culture supernatants of each group were calculated based on the OD values.
Real-Time Quantitative PCR (RT-qPCR)
Total RNA was extracted from mice LUAD tissues or A549 cells using the Total RNA Kit I (OMEGA bio-tek, GA, USA) following the manufacturer’s protocol. The concentration of RNA was determined by measuring the absorbance at 260/280 nm using a Nanodrop 2000 spectrophotometer (Nanodrop, DE, USA). Reverse-transcribed into cDNA with SPARKscript II RT Plus Kit (With gDNA Eraser), and RT-PCR was performed with 2×SYBR Green qPCR Mix (With ROX) and Roche LightCycler® 480 Instrument II system (Roche, Mannheim, Germany). 22 The Sequence of primers used for RT-qPCR are listed in Table 2.
Sequence of Primers Used for RT-qPCR.

Western Blotting (WB)
Total protein was extracted from the mice LUAD tissues and A549 cells using RIPA buffer supplemented with PMSF and phosphatase inhibitors. The protein concentration was determined using the BCA Protein Assay Kit following the manufacturer’s instructions. The total protein was separated by 10% SDS-PAGE and transferred to PVDF membranes. After that, the membranes were blocked with 5% milk at room temperature for 1 hours and then incubated with primary antibodies at 4°C overnight. The PVDF membranes were washed 3 times with TBST on the following day. Then, they were incubated with the secondary antibody (1:5000) at room temperature for 1 hours, followed by another 3 washes with TBST. The protein bands were finally visualized using the ECL Western Blotting Substrate. 23
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 7 (GraphPad Software, CA, USA). The data are presented as mean ± SD, and the differences between groups were compared using One-way ANOVA. A significance level of P < .05 was considered statistically significant.
Results
Components Analysis of XLPYR
UPLC-Q-Orbitrap-MS/MS analysis revealed 33 significant active compounds in both positive and negative ion modes within XLPYR. Figure 1 depicts the extracted ion chromatograms, while Table 3 provides details on these 33 compounds.

The extracted ion chromatograms of the XLPYR by UPLC-Q-Orbitrap-MS/MS in negative- (above, black) and positive-ion modes (Below, red).
The Information of 33 Compounds in XLPYR.

XLPYR Significantly Inhibited the Growth of Subcutaneous Transplanted Tumors in C57BL/6J Mice
After the initiation of drug intervention, there were no significant differences in the daily average food intake and water consumption among the groups of mice. All mice in each group displayed agile responses and good mental states, and their fur appeared normal without any obvious abnormalities. In the later stages of the experiment, as the tumors gradually grew larger, mice in the model group exhibited mild lethargy and reduced activity. Throughout the experimental intervention, no mice deaths occurred due to procedural errors or drug-related effects.
The study results revealed that there were no significant differences in tumor volume among the groups on the first and third days. However, starting from the sixth day, both the COMBI group and PEM group exhibited significantly smaller tumor volumes compared to the Model group. From the ninth day onward, the XLPYR group also showed significantly smaller tumor volumes than the Model group. As the duration of drug intervention increased, the differences in tumor volume between the PEM, XLPYR, and COMBI groups compared to the Model group became more pronounced, indicating a progressively evident tumor-inhibitory effect of each drug group (Figure 2A and B).
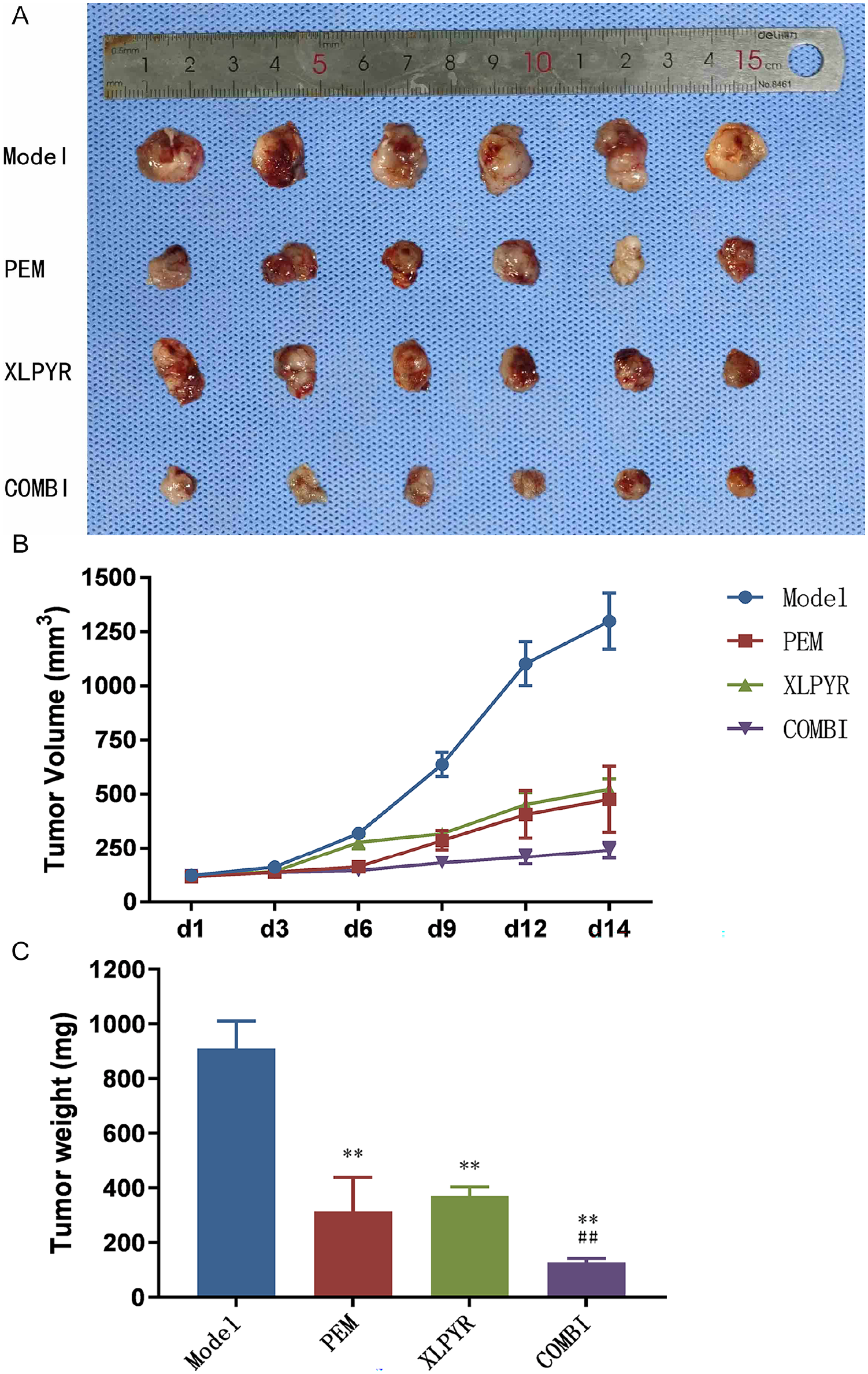
XLPYR suppressed growth of LLC-allograft tumors. (A) Tumor tissue images of LLC allograft mouse model in each group. (B) Tumor volume changes in mice from each group at different intervention time points. (C) Tumor weights of each group after mice were sacrificed at 14 days of treatment.
Tumor weights in the PEM, XLPYR, and COMBI groups were significantly lower than the Model group. The tumor inhibition rates were 65.47% (PEM), 59.36% (XLPYR), and 86.02% (COMBI), demonstrating a synergistic effect of XLPYR combined with PEM (Figure 2C).
XLPYR Regulated the Vascular Microenvironment by Improving the Structure and Function of Tumor Blood Vessels
Immunofluorescence staining revealed that the PEM, XLPYR, and COMBI groups had significantly reduced MVD compared to the Model group. Notably, the COMBI group exhibited the lowest MVD among all groups. pericyte coverage was significantly increased in all intervention groups compared to the Model group, with the COMBI group showed the highest increase over the PEM group. These findings demonstrated that both PEM and XLPYR inhibited tumor angiogenesis and enhance pericyte coverage, promoting tumor vessel maturation and improving vascular structure. The combination of XLPYR and PEM showed a superior effect in inhibiting tumor regression and metastasis (Figure 3A-C).
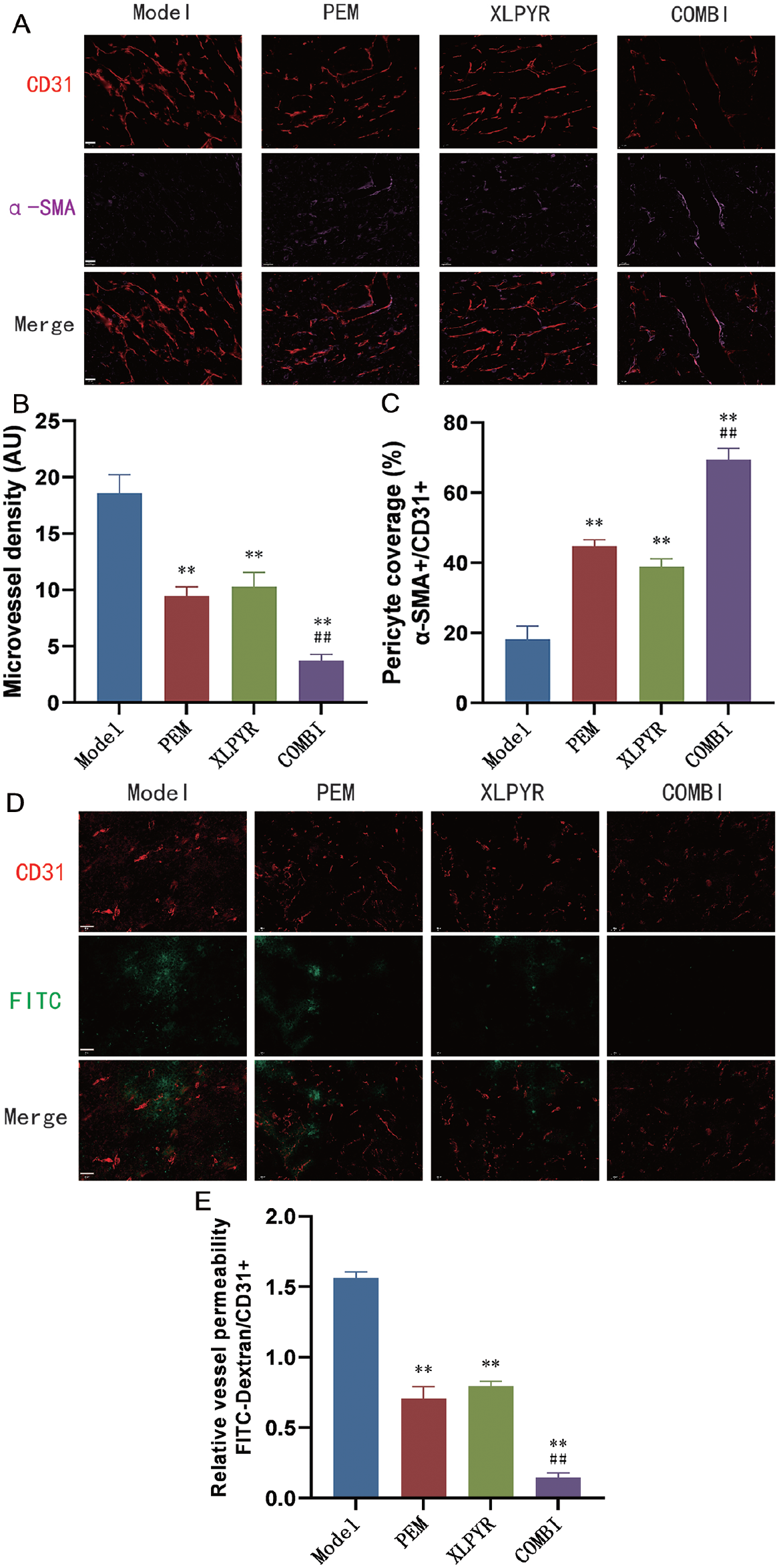
XLPYR modulates the tumor vascular microenvironment by enhancing the structural and functional properties of tumor blood vessels. (A) Immunohistological staining of pericyte coverage (α-SMA, purple) surrounding tumor vessels (CD31, red) in LLC tumor tissues, and comparison of MVD (B) and pericyte coverage (C) among different groups of mice. (D) Changes in vascular permeability in tumor tissues of mice from different groups (CD31, red; FITC-Dextran, green), and comparison among the groups (E). Scale bars, 50 μm.
FITC-Dextran extravasation in the Model group was significantly higher than in the PEM, XLPYR, and COMBI groups. In the COMBI group, FITC-Dextran extravasation was significantly lower than in the PEM group. These findings suggested that both PEM and XLPYR improved tumor vascular function by reducing vascular leakage. The combined application of both drugs further enhanced this effect in reducing vascular leakage and improving tumor vascular function (Figure 3D and E).
XLPYR Regulated the Expression of HIF-1α, VEGFA, Ang-2, and PI3K/Akt In Vivo
The RT-qPCR results demonstrated a significant downregulation of HIF-1α, VEGFA, and Ang-2 mRNA expression levels in the PEM, XLPYR, and COMBI groups compared to the Model group. The differences were statistically significant. However, there were no significant differences in the mRNA expression levels of PI3K and Akt among the groups (Figure 4A-E).
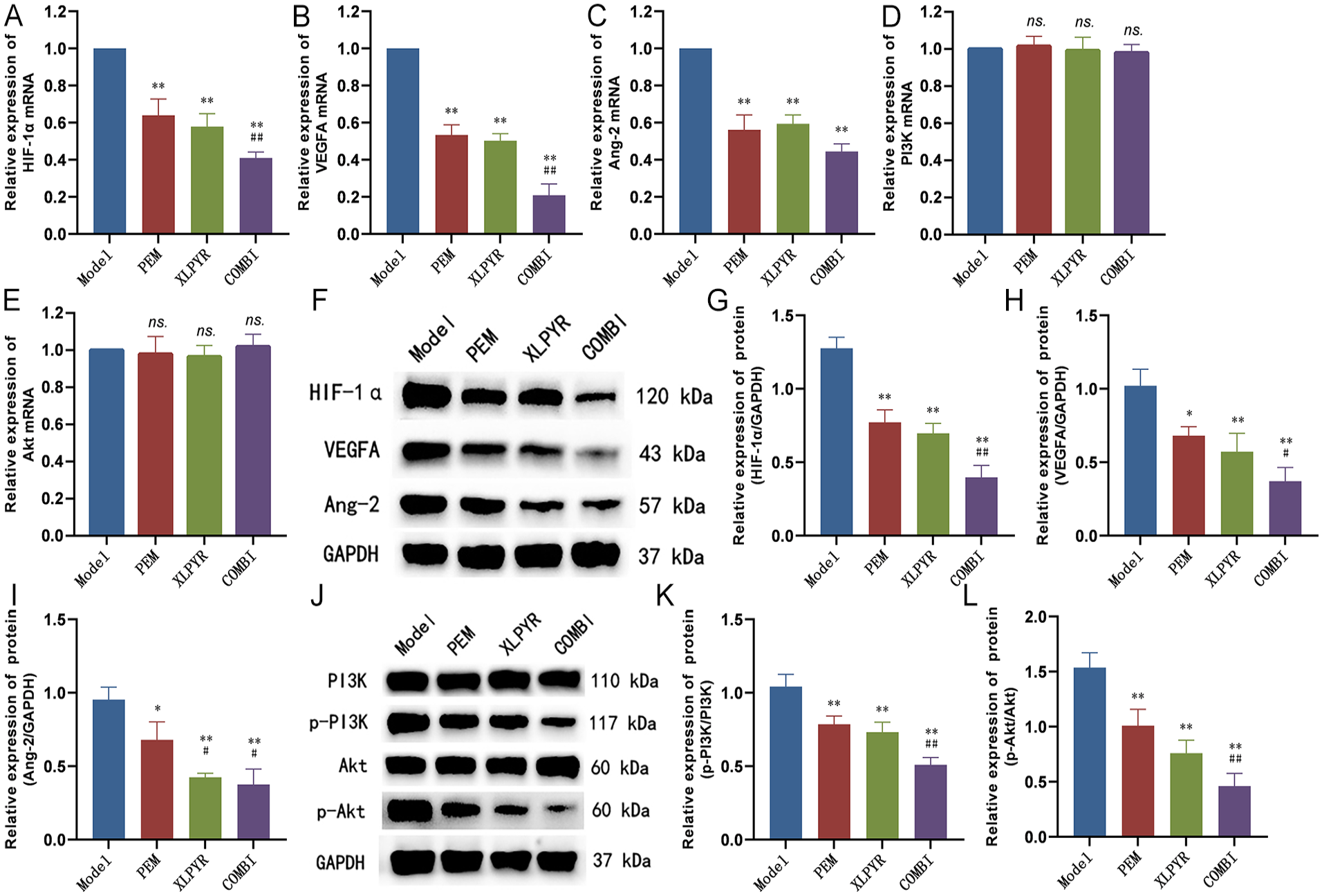
Inhibition of angiogenesis-related factors expression and PI3K/Akt phosphorylation by XLPYR in vivo. RT-qPCR results showed significant downregulation of HIF-1α (A), VEGFA (B), and Ang-2 (C) mRNA expression, while no significant difference in PI3K(D) and Akt (E) mRNA expression was observed. (F and J) Western blot band images. Relative quantitative analysis revealed a significant downregulation of HIF-1α (G), VEGFA (H), and Ang-2 (I) protein expression in all intervention groups. No significant difference was observed in total protein expression of PI3K and Akt among the intervention groups. Nevertheless, the expression levels of p-PI3K(K) and p-Akt (L) were significantly decreased.
The WB results showed a significant downregulation of HIF-1α, VEGFA, and Ang-2 protein expression in the PEM, XLPYR, and COMBI groups compared to the Model group. The differences were statistically significant. There were no significant differences in total protein expression levels of PI3K and Akt among the intervention groups. However, p-PI3K and p-Akt levels were significantly reduced in the XLPYR group, indicating that XLPYR inhibited the phosphorylation levels of PI3K and Akt proteins in vivo, further suppressing downstream pathway activation (Figure 4F-L).
The Effect of XLPYR-Containing Serum on Cell Viability and the Selection of the Optimal Intervention Concentration
The CCK-8 assay compared the effects of different concentrations of XLPYR-containing serum and blank serum (2.5%, 5%, 10%, 20%) on BEAS-2B cells. XLPYR-containing serum at concentrations of 2.5%, 5%, and 10% did not significantly affect cell proliferation rate compared to the blank serum group (P > .05). However, at a concentration of 20% XLPYR-containing serum, there was a significant decrease in cell proliferation rate (P < .05) (Figure 5A).

XLPYR-containing serum inhibited the proliferation of LUAD cells in vitro. Effects of different drugs on the relative proliferation rates of human normal lung epithelial BEAS-2B cells (A) and human lung cancer A549 (B) cells in each group.
The subsequent study evaluated the impact of XLPYR-containing serum on the relative proliferation rate of lung cancer A549 cells. Under the same intervention time, the 2.5% XLPYR-containing serum group did not show a statistically significant difference in cell proliferation rate compared to the blank serum group (P > .05). However, significant differences were observed in the 5%, 10%, and 20% XLPYR-containing serum groups, as well as the PEM group, compared to the blank serum group (P < .01). No significant difference was found between the 10%to 20% XLPYR-containing serum groups (P > .05). The study results indicated that the inhibitory effect of XLPYR on lung cancer cells depends on time and concentration. Nevertheless, under the same intervention time, cell proliferation rate did not significantly decrease when the XLPYR-containing serum concentration exceeded 10%. A549 cells were found to be more sensitive to the inhibitory effect of XLPYR-containing serum compared to BEAS-2B cells. Thus, based on the overall results from both cell types, the concentration of 10% was selected as the optimal intervention concentration of XLPYR-containing serum for subsequent experiments. Additionally, we investigated the combined effect of 10% XLPYR-containing serum and PEM on the proliferation rate of lung cancer A549 cells. The results demonstrated a significant reduction in cell proliferation rate in the COMBI group compared to using PEM or XLPYR-containing serum alone, indicating a synergistic effect of the combination (Figure 5B).
XLPYR-Containing Serum Significantly Inhibited the Migration and Invasion of LUAD Cells
By using ImageJ software to calculate the cell migration rate in the wound-healing assay after 24 hours of drug intervention, significant reductions in cell migration rate were observed in the PEM, XLPYR, and COMBI groups compared to the Control group (P < .05). Furthermore, the COMBI group showed a significant decrease in cell migration rate compared to the PEM group (P < .05) (Figure 6A and B).

Inhibition of LUAD cell migration and invasion by XLPYR-containing serum (50×). (A) The migration of LUAD cells was detected by wound-healing assay, and (B) the cell migration rate was quantified. (C) The invasion of LUAD cells was detected by Transwell assay, and (D) quantification of invaded cells through the membrane.
After 24 hours of drug intervention, the Transwell chambers were processed, and the cells that migrated through the chambers were photographed and counted under a microscope. The results showed a significant decrease in the number of transmembrane cells in the PEM, XLPYR, and COMBI groups compared to the Control group (P < .05). Additionally, the COMBI group exhibited a significant reduction in the number of transmembrane cells compared to the PEM group (P < .05). Combined with the results of the wound-healing assay, it can be concluded that XLPYR-containing serum significantly inhibits the invasion and migration ability of LUAD cells (Figure 6C and D).
XLPYR-Containing Serum Regulated the Expression of HIF-1α, VEGFA, Ang-2, and PI3K/Akt In Vitro
ELISA assay results revealed a significant decrease in VEGF expression in the culture supernatant of LUAD cells in the PEM, XLPYR, and COMBI groups compared to the control group (P < .05) (Figure 7A).

Inhibition of angiogenesis-related factors expression and PI3K/Akt phosphorylation by XLPYR-containing serum in vitro. (A) ELISA assay showed a significant decrease in VEGF expression in the supernatant of cells from all intervention groups. RT-qPCR results showed significant downregulation of HIF-1α (B), VEGFA (C), and Ang-2 (D) mRNA expression, while no significant difference in PI3K (E) and Akt (F) mRNA expression was observed. (G and K) Western blot band images. Relative quantitative analysis revealed a significant downregulation of HIF-1α (H), VEGFA (I), and Ang-2 (J) protein expression in all intervention groups. No significant difference was observed in total protein expression of PI3K and Akt among the intervention groups. Nevertheless, the expression levels of p-PI3K (L) and p-Akt (M) were significantly decreased.
RT-qPCR results indicated a significant downregulation of HIF-1α, VEGFA, and Ang-2 expression levels in the PEM, XLPYR, and COMBI groups compared to the Control group (P < .05). However, there were no significant differences in the expression levels of PI3K and Akt mRNA among the intervention groups (Figure 7B-F).
WB results revealed a significant downregulation of HIF-1α, VEGFA, and Ang-2 protein expression levels in the PEM, XLPYR, and COMBI groups compared to the Control group (P < .05). Although there were no significant differences in the total protein expression levels of PI3K and Akt among the intervention groups, the levels of p-PI3K and p-Akt were significantly downregulated. These research findings were consistent with the in vivo experiments (Figure 7G-M).
Discussion
In the past decades, significant efforts have been devoted to exploring novel therapeutic approaches for lung cancer. With the continuous application of targeted therapy and immunotherapy, the prognosis of advanced NSCLC patients has markedly improved, leading to significant extensions in survival. 24 Anti-angiogenic therapy, as one of the essential treatment modalities for NSCLC, has shown significant clinical benefits when combined with chemotherapy in first-line or second-line treatment.25,26 Moreover, in recent times, the combination of angiogenesis inhibitors, immune checkpoint inhibitors (ICIs), and chemotherapy has emerged as a first-line treatment option for NSCLC. 27 Due to the critical role of VEGF/VEGFR in the process of tumor angiogenesis, there has been a strong focus on developing targeted therapies against the VEGF/VEGFR pathway. By inhibiting tumor angiogenesis and depriving tumors of nutrients, these therapies aim to exert anti-tumor effects. 28 Although anti-angiogenic drugs initially showed promising efficacy, the issue of drug resistance that follows should not be ignored. Besides tumor endothelial heterogeneity, vessel co-option or vasculogenic mimicry (VM) also contribute significantly to anti-angiogenic therapy resistance in NSCLC. Moreover, tumors activate compensatory pathways in the absence of certain signaling molecules, serving as an alternative resistance mechanism. For instance, inhibiting VEGF/VEGFR has been demonstrated to induce tumors to sustain angiogenesis through the secretion of substitute factors like PDGF, bFGF, and Ang-2,29-31 or by recruiting mast cells that secrete proteases, thereby conferring resistance to single-target therapies. 32 The timing and dosage of anti-VEGF therapy play crucial roles in promoting tumor vascular normalization.5,13 The normalization window leads to increased tumor oxygenation, enhanced drug delivery, and improved efficacy when combined with VEGF inhibitors and cytotoxic drugs. However, high doses of VEGF inhibitors can cause toxicity in normal tissues, leading to tumor blood vessel degeneration, increased tumor hypoxia, and tumor progression. 33 Therefore, the search for effective and safe multi-target drugs to improve the tumor vascular microenvironment and promote vascular normalization is a key focus and challenge in anti-angiogenic therapy for NSCLC.
In recent years, an increasing number of studies have demonstrated the importance of TCM in anti-tumor treatments. Compared to conventional therapies, TCM has advantages in its diverse target actions and novel pharmacological mechanisms, ensuring a relatively lower level of toxicity and promising therapeutic efficacy. 34 Moreover, the rich and comprehensive theoretical system of TCM provides a reference for the discovery of specific drugs. For example, Nobel laureate Tu Youyou discovered the anti-malarial properties of the Chinese herb “Qinghao” (Artemisia annua L.) from Ge Hong’s A Handbook of Prescriptions for Emergencies, which led to the identification of artemisinin, a highly effective anti-malarial drug. 35 The Theory of meridian disease in TCM can be traced back to the “Huangdi Neijing” (Yellow Emperor’s Inner Canon). It was further supplemented and improved in the “Shanghan Zabing Lun” (Treatise on Cold Damage and Miscellaneous Diseases). During the Qing Dynasty, medical scholars such as Yu Jiayan, Ye Tianshi, and Wu Jutong advanced the theory of collaterals, bringing it to a mature stage. According to TCM, the meridians of the lungs are considered the foundation for their normal physiological functions, and meridian disease is one of the main causes of lung cancer. This fundamental transformation process of meridian disease in lung cancer bears a striking resemblance to the modern medical understanding of how abnormalities in the structure and function of the vascular microenvironment contribute to tumor progression and metastasis. Previous studies have demonstrated that TCM can effectively inhibit the growth of LUAD by intervening in tumor angiogenesis.36,37
CD31, also known as Platelet Endothelial Cell Adhesion Molecule-1 (PECAM-1), is a transmembrane glycoprotein belonging to the immunoglobulin superfamily. It is commonly expressed on both early and mature vascular endothelial cells. CD31 can effectively and accurately assess the number and density of microvessels within tumor tissues. 38 Pericytes, located around endothelial cells in the microvascular system, play a critical role in maintaining the non-proliferative state of endothelial cells, thereby preserving microvascular density. However, in tumor vessels, the loss of pericyte surveillance leads to excessive activation of endothelial cells, promoting abnormal angiogenesis in the tumor vasculature. 39 Invasion and metastasis are the primary reasons for the progression of malignant tumors. Studies have demonstrated that in tumor microvessels, there is a significant reduction in pericyte coverage around endothelial cells, leading to abnormal tumor vascular structure and facilitating the entry of tumor cells into the bloodstream. Therefore, pericytes play a crucial role as an essential component of the microvascular system in maintaining the structure and morphology of microvessels and promoting vascular maturation. 40 Pericytes can express various markers such as α-SMA, CD13, PDGFR-β, among others. 41 In this study, we selected α-SMA as the marker for pericytes and used CD31 to localize endothelial cells to calculate the pericyte coverage rate within the tumor tissue. We evaluated tumor vascular permeability by intravenously injecting FITC-Dextran through the tail vein and concurrently using CD31 to label blood vessels. The amount of FITC-Dextran extravasated from the blood vessels was observed to assess tumor vascular permeability. Altered tumor vascular permeability not only facilitates tumor cell leakage, leading to metastasis and progression, but also impedes the efficient delivery of chemotherapy drugs and immune cells to the tumor site, leading to treatment failure. Therefore, observing tumor vascular permeability is a vital indicator for assessing tumor vascular function and normalization.42,43 In the present study, XLPYR significantly suppressed subcutaneous LUAD allograft growth in mice through drug intervention. Treatment with XLPYR resulted in reduced MVD, increased pericyte coverage, and suppressed vascular leakage within the tumor tissues, leading to improved microvascular structure and function and effective intervention in tumor angiogenesis for tumor vascular normalization. Additionally, the combination of XLPYR with PEM showed a synergistic effect, enhancing the therapeutic outcome.
Furthermore, through in vitro studies, we demonstrated that XLPYR-containing serum significantly inhibited the proliferation, migration, and invasion of LUAD cells. XLPYR, as a traditional Chinese herbal compound, contains various components with anti-angiogenesis and anti-tumor properties among its constituent drugs. Studies have indicated that ginsenoside Rg3, an active component of the herbal medicine Rhizoma Ginseng, reduced the migration, invasion, and angiogenesis of LUAD cells by inhibiting the expression of COX2 and VEGF. 44 The active component curcumin in the traditional Chinese herb Curcumae rhizome inhibits c-Met-dependent PI3K/Akt/mTOR signaling activation, regulates the expression of VEGF and EMT-related markers, and effectively suppresses the processes of EMT and angiogenesis both in vitro and in vivo. 45 The active ingredient APS (Astragalus polysaccharide) in the traditional Chinese herb Astragali radix downregulates NF-κB activity, inhibiting the proliferation of LUAD cells in vitro, and retarding the growth of LUAD allografts in vivo. 46
Hypoxia is one of the main characteristics of the tumor microenvironment and a major physiological stimulus for inducing angiogenesis. Hypoxia-inducible factors (HIF) are crucial regulatory genes in response to cellular hypoxia. Studies have demonstrated that upregulation of HIF-1α expression can be detected in 90% of lung cancer tissues, whereas adjacent normal tissues do not exhibit abnormal expression of HIF-1α. 47 When the rapid growth of tumors exceeds the limit of oxygen diffusion, local hypoxia enhances the transcriptional activity of HIF-1α. HIF-1α directly activates the expression of VEGF and its downstream angiogenic factor Angiopoietin (Ang) by binding to the hypoxia response element (HRE) located upstream of the VEGF transcription start site. This induces the migration of endothelial cells to the hypoxic area, promoting vascular neovascularization to supply oxygen to the avascular hypoxic region.48-50 Research has shown that blocking HIF-1α transcription leads to a cessation of VEGF secretion by tumor cells, thereby inhibiting the process of tumor angiogenesis. 51 The upregulation of VEGF expression increases tumor vascular permeability, leading to plasma and extracellular fluid leakage, which increases the risk of metastasis. High expression of VEGF in various tumors is indicative of poor prognosis. 52 VEGF-regulated Ang-1 and Ang-2 play antagonistic roles in the process of vascular development. Ang-1 acts as a natural inhibitor of vascular permeability, preventing plasma leakage, while Ang-2 induces vascular dysfunction by increasing smooth muscle cell detachment and permeability.53,54
The PI3K/Akt pathway serves as an important upstream regulator of HIF-1α. Upon activation, the PI3K/Akt pathway upregulates HIF-1α expression, leading to increased secretion of VEGF and subsequent promotion of angiogenesis. 55 Activation of PI3K by upstream stimuli induces conformational changes in its catalytic subunit, resulting in the phosphorylation of PIP2 to form PIP3. 56 As a second messenger, PIP3 provides binding sites for Akt signal proteins with PH domains, facilitating Akt translocation from the cytoplasm to the cell membrane. This exposes the phosphorylation sites Ser473 and Thr308, which are essential for activating downstream pathways. The activation of Akt requires the involvement of Phosphoinositide-Dependent Kinases (PDK), including PDK1 and PDK2, which phosphorylate Ser473 and Thr308 to generate p-Akt with kinase activity, further activating downstream pathways. 57 Activated p-Akt then triggers the phosphorylation of tyrosine and serine residues, inducing the expression of downstream HIF-1α. 58 Activated HIF-1α binds to the HRE located at the 5′ end of VEGF, initiating VEGF transcription, and promoting tumor angiogenesis. 59
The results of this study demonstrated that XLPYR significantly downregulated the expression levels of p-PI3K and p-Akt both in vitro and in vivo. This indicated that XLPYR could inhibit the activation of downstream HIF-1α, VEGF, and Ang-2 by reducing the phosphorylation of PI3K and Akt proteins. Consequently, XLPYR improved the tumor vascular microenvironment and promoted tumor vascular normalization, achieving the anti-tumor effects. However, this study still has some limitations. Due to XLPYR being a traditional Chinese medicine compound with complex chemical components, further research is needed to identify specific effective anti-tumor ingredients. Therefore, in future investigations, we plan to delve deeper into the mechanisms to gain a more comprehensive understanding.
Conclusion
In conclusion, this study found that XLPYR significantly inhibited the growth of C57BL/6J mouse LUAD allografts. It achieved this by reducing MVD within the tumor tissues, increasing pericyte coverage, and suppressing vascular leakage, thereby improving the structure and function of the microvascular system. These effects led to the intervention in tumor angiogenesis, improved the vascular microenvironment, and induced tumor vascular normalization. In vitro experiments also confirmed that XLPYR-containing serum significantly inhibited the proliferation, migration, and invasion of LUAD cells while reducing VEGF concentration in the cell supernatant. The mechanisms underlying these anti-tumor effects may be related to the inhibition of PI3K/Akt protein phosphorylation and downregulation of HIF-1α, VEGF, and Ang-2, which are factors involved in angiogenesis.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
Fei-ran Yang and Hui-jie Li conceived and designed the study. Fei-ran Yang and Hong-lin Li performed the experiments. Fei-ran Yang, Xi-wen Hu and Rong Fu prepared tables and figures and drafted the manuscript. Hui-jie Li and Xiu-rong Li revised the manuscript; Hui-jie Li has given final approval of the version to be published. All authors read and approved the final manuscript.
Data Availability Statement
The data underlying this article are available in the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Natural Science Foundation of Shandong Province (Grant no. ZR2021LZY029).
Ethics Statement
The experimental protocol involving animals was reviewed and approved by the Ethics Committee of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine (Ethical approval number: 2021-82).
