Abstract
Background
Among the different cancers found in women, breast cancer is the most common. Breast cancer-related lymphedema is a serious health complication affecting the quality of life and sleep quality. This study evaluates the quality of life and sleep quality among Saudi women with different stages of lymphedema following the treatment of breast cancer.
Methods
This cross-sectional correlational study included 163 Saudi women with breast cancer-related lymphedema (Stages I-III), aged 28 to 56 years. From the patients identified for this study, women who suffered from mental and psychological dysfunctions or other malignant disorders were excluded. Copies of structured questionnaires were given to each participant during their visits to outpatient physiotherapy clinics. Quality of life was assessed using a valid questionnaire (EORTC QLQ-C30), while sleep quality was assessed using the Pittsburgh Sleep Quality Index (PSQI). The differences between the different lymphedema stages have been assessed.
Results
Of the total number of participants 27 women had been diagnosed with stage I lymphedema, 84 women had been diagnosed with stage II lymphedema, and 52 women had been diagnosed with stage III lymphedema. All participants have shown low scores on both EORTC QLQ-C30 and PSQI. While analyzing the differences between the 3 stages of lymphedema with the Kruskal–Wallis test, noteworthy statistical differences between the 3 stages of lymphedema (P < .05) have been found. The Stage III lymphedema patients have been shown the lowest quality of life values in all scales when compared with the stage I and stage II lymphedema patients. For PSQI scores, the stage III lymphedema patients worse values than the stage I and stage II lymphedema patients (P < .05).
Conclusion and Recommendations
Both quality of life and quality of sleep have significantly decreased in Saudi women with different stages of breast cancer-related lymphedema. Quality of life and quality of sleep are the worst in stage III lymphedema patients. Future research should consider repeat and enlarge these results as well as assess the risk factors that affect the quality of life and quality of sleep among Saudi women suffering from breast cancer-related lymphedema.
Introduction
Breast cancer is a major and widespread cancer affecting women worldwide. 1 Its incidence has increased up to approximately 28.7% in Saudi Arabia and 23% worldwide. 2 Despite the improvements in the treatment protocols, the breast tissue’s eradication (mastectomy) remains the prevalent surgical intervention for 20% to 30% of breast cancer patients. 3 Lymphedema is the most common complication subsequent to breast cancer surgery and is known as breast cancer-related lymphedema. 4 It is a state of accumulation of extreme soft tissue fluids manifested with localized pain, persistent swelling, atrophied skin, and frequent infection that severely affects the physiological and psychological status of the affected women. 3 Lymphedema also affects health-related quality of life as a result of impaired arm functions, disturbed body image, and augmented issues including cellulitis and repeated infections. 5
Breast cancer-related lymphedema following mastectomy accounts for approximately 14.05% of the breast cancer patients among Saudi women. 6 Several predisposing factors are associated with severe lymphedema such as weight gain, infection, dissected lymph nodes, long period of the disease, chemotherapy, radiotherapy, and surgical procedures.7,8 Lymphedema is classified into primary and secondary lymphedema. Primary lymphedema is a disturbed lymphatic function or structure without a known cause yet, while the secondary lymphedema is an acquired interrupted lymphatic compression caused by a tumor or its interventional modality. 9 Lymphedema develops gradually and can be divided into 3 different symptomatic stages. The first stage is a reversible stage distinguished by pitting edema, minor ache, and a sensation of fullness in the arms. The second stage is irreversible and is distinguished by a non-pitting edema, skin thickness, hair loss, and pain. The third stage is also irreversible and is distinguished by severe limb swelling and open wounds, which develop to massive infections. 10
Several treatment protocols have been considered in the management of lymphedema such as pharmacotherapy, tailored education, exercise intervention, and therapeutic compression modalities.4,11-13 However, complete decongestive therapy is the most common treatment protocol.14,15
Women with secondary lymphedema, subsequent to mastectomy, suffer from psychological issues such as depression and anxiety, physical and functional impairments, and poor quality of life.16,17 It was also reported that lymphedema, following mastectomy, may lead to restricted functional performance, chronic disabilities, and disturbed quality of life.18-20 Moreover, it was documented that sleep disturbance may be demonstrated early during the diagnosis of breast cancer and continue for a long period after the intervention.21-23
No previous studies have assessed the quality of life and quality of sleep in the different stages of secondary lymphedema subsequent to breast cancer surgery among Saudi women. This study aims therefore to evaluate the quality of life and quality of sleep among Saudi women with different stages of breast cancer-related lymphedema.
Methods
Study Design, Settings, and Ethics
The current study was a cross-sectional correlational study. It was conducted in multiple outpatient physiotherapy clinics in Saudi Arabia between November 2020 and February 2021. The ethical approval for this study was received from the local review board of the physiotherapy department at PSAU (RHPT/020/066). After being explained the objectives and procedures of the study, each participant signed a written consent form before joining the study program.
Sample Size
Due to the deficiency of data availability, the sample size was estimated in accordance with the traditional power analysis for a descriptive design (α = .5, effect size = 0.4, and confidence interval = 80%). The minimum required sample size was 158 participants. Therefore, 163 women were enrolled in the study to compensate for the withdrawal rate.
Participants
A total of 163 Saudi women with breast cancer-related lymphedema (Stages I-III), aged 28 to 56 years were included this cross-sectional study. Women suffering from mental and psychological dysfunctions or other malignant disorders were excluded from this study. Copies of structured questionnaires were given to each of the study participants during their visits to the outpatient physiotherapy clinics covered by this study.
Assessment
Demographics, quality of life, and quality of sleep questionnaires were administered for this observational study. Quality of life was assessed using a valid Arabic version of the breast cancer questionnaire known as the European Organization for Research and Treatment of Quality of Life Cancer 30 (EORTC QLQ-C30) that has been validated to evaluate the quality of life in women with breast cancer-related lymphedema.24,25 This questionnaire contains 30 items under 3 main scales, and it measures the 2-item global health status, 15-item functional status, and 13-item symptoms scales. The functional scales comprise 5 domains including role-related, physical, social, cognitive, and emotional functions, while the symptoms scales comprise 9 domains including loss of appetite, constipation, dyspnea, diarrhea, insomnia, nausea/vomiting, fatigue, and financial difficulties.24,25
Quality of sleep was evaluated by using the validated and reliable Arabic version of the 19-item PSQI.26,27 These 7 domains of this instrument include daytime dysfunction, sleep duration, sleep latency, habitual sleep efficiency, subjective sleep quality, sleep disturbance, and use of sleep medications during the previous month. This questionnaire shows how much time (in minutes) is ordinarily taken each night to fall asleep, and how the patients rate their overall quality of sleep. The score of habitual sleep efficiency is estimated as a percentage of the time taken from going to bed till falling sleep. Clinically, a score of above 5 indicates noteworthy sleep disturbances, 26 and the score of sleep efficiency below 85% suggests poor sleep quality. 27 PSQI is a valid and reliable instrument that has been used to assess quality of sleep in breast cancer survivors. 28
Statistical Analysis
All data were collected and analyzed using SPSS v. 25 (IBM SPSS, Armonk, NY, USA). A descriptive analysis was conducted to explain how the sample relates to demographics and clinical features concerned. Categorical variables were analyzed using the Kruskal-Wallis test and the unpaired student’s t-test. The frequencies were demonstrated in percentages. The Spearman coefficient correlation was used to determine the relationship between the lymphedema stages and the domains of quality of life and quality of sleep. Statistically, a P-value below .05 was considered significant.
Results
Demographics and Clinical Characteristics
Out of a total of 192 women, 163 women completed the structured questionnaires. Their mean age was 42.6 ± 8.3 years, mean body mass index (BMI) was 32.5 ± 4.7 kg/m2, and mean duration after mastectomy was 18.6 ± 7.5 months. As for their level of education, it was found that 2.45% had not have any formal education, 28.22% had have elementary education, 52.15% had have higher education, and 17.18% had have tertiary or other education. About 31.29% of the participants were working women, and 68.71% were not employed. Further, 83.4% were married, and 16.6% were not married. Regarding surgical interventions, 59.51% had undergone only breast surgery, and 40.49% of the participants had undergone breast surgery plus axillary surgery. As many as 61.96% of the participants had undergone chemotherapy, and 38.04% underwent chemotherapy and radiotherapy. As for the 3 different stages of the disease, 16.56% of the women were at stage I, 51.54% were at stage II, and 31.9% were at stage III. The affected arm was the dependent arm in the case of 60.74% of the women and the independent arm in the case of 39.26% of them (Table 1).
Demographics and Clinical Features of the Study Participants.
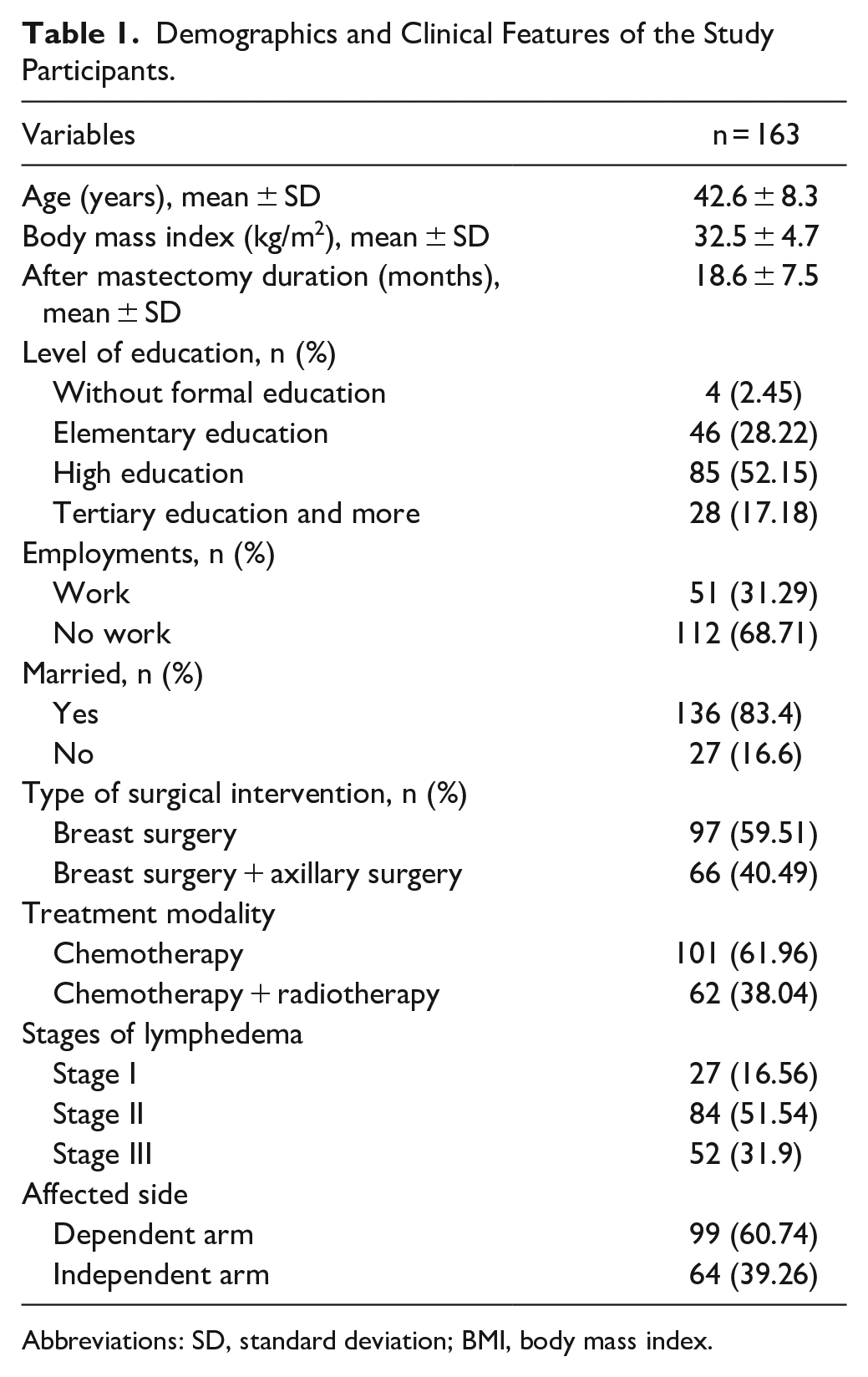
Abbreviations: SD, standard deviation; BMI, body mass index.
EORTC QLQ-C30
As presented in Table 2, analyzing the quality of life using EORTC QLQ-C30 showed that the stage I lymphedema patients (n = 27) reported the best values concerning the functional status scales including physical, role-related, emotional, cognitive, and social scales (64.2 ± 9.4, 66.3 ± 10.2, 71.6 ± 11.4, 68.5 ± 10.8, and 65.4 ± 9.8 respectively), followed by the symptoms status scales including appetite loss, fatigue, dyspnea, nausea/vomiting, pain, insomnia, diarrhea, constipation, and financial problems (74.5 ± 10.6, 70.6 ± 9.7, 73.2 ± 10.4, 68.4 ± 9.5, 71.5 ± 9.8, 74.7 ± 10.2, 45.3 ± 7.5, 53.7 ± 8.6, and 51.6 ± 7.7 respectively), and then the global health quality of life scales (38.7 ± 7.4). The stage II lymphedema patients (n = 84) showed lower mean values when compared with those in stage I in the functional status scales including physical, role-related, emotional, cognitive, and social scales (61.4 ± 9.1, 63.5 ± 9.8, 69.6 ± 10.5, 65.2 ± 9.4, and 62.3 ± 8.7 respectively), followed by the symptoms status scales including appetite loss, fatigue, dyspnea, nausea/vomiting, pain, insomnia, diarrhea, constipation, and financial problems (72.2 ± 10.1, 67.8 ± 9.4, 70.6 ± 9.8, 65.7 ± 9.1, 68.8 ± 9.4, 72.7 ± 9.8, 42.5 ± 7.2, 50.6 ± 8.5, and 50.2 ± 7.4 respectively), and then the global health quality of life scales (36.1 ± 6.8).The stage III lymphedema patients (n = 52) showed the lowest mean values when compared with the other 2 stages in the functional status scales including physical, role, emotional, cognitive, and social scales (57.3 ± 8.5, 59.6 ± 8.7, 65.2 ± 9.4, 61.6 ± 8.3, and 58.5 ± 7.9 respectively), followed by the symptoms status scales including appetite loss, fatigue, dyspnea, nausea/vomiting, pain, insomnia, diarrhea, constipation, and financial problems (68.2 ± 9.5, 63.7 ± 8.7, 67.1 ± 9.6, 62.5 ± 8.8, 64.3 ± 8.4, 69.4 ± 9.7, 40.1 ± 6.2, 48.1 ± 7.5, and 48.5 ± 6.8 respectively), and then the global health quality of life scales (32.4 ± 5.6).
Quality of Life and Quality of Sleep of the Study Participants According to the Stages of Lymphedema.
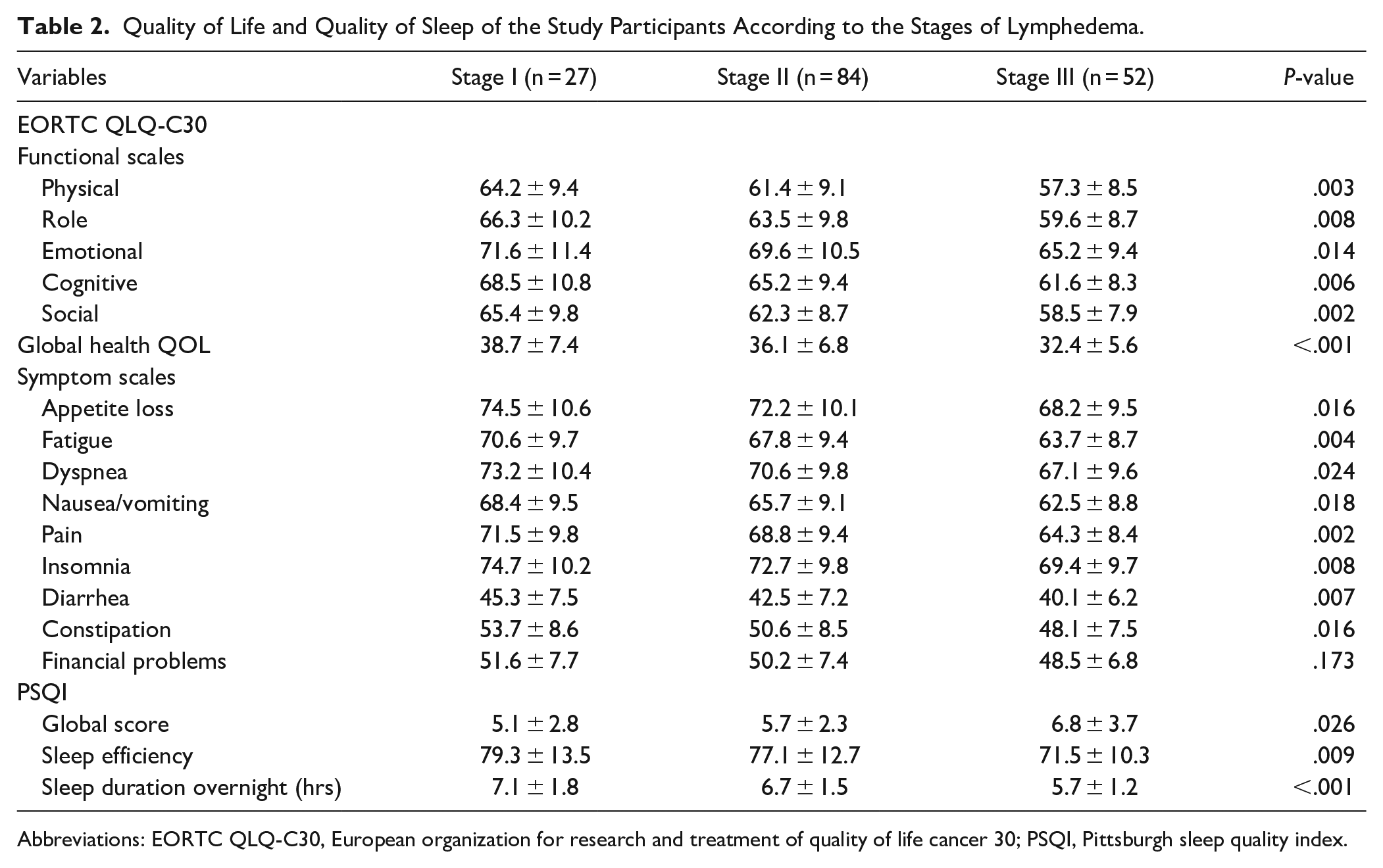
Abbreviations: EORTC QLQ-C30, European organization for research and treatment of quality of life cancer 30; PSQI, Pittsburgh sleep quality index.
PSQI
Analyzing the quality of sleep using PSQI showed that the stage I lymphedema patients suffered from minimal sleep disturbances with a global score mean of 5.1 ± 2.8, sleep efficiency mean of 79.3 ± 13.5, and sleep duration overnight mean of 7.1 ± 1.8 hours. The stage 2 lymphedema patients suffered from moderate sleep disturbances with a global score mean of 5.7 ± 2.3, sleep efficiency mean of 77.1 ± 12.7, and sleep duration overnight mean of 6.7 ± 1.5 hours. The stage III lymphedema patients showed the worst mean values of PSQI when compared with mean values of the patients in the other 2 stages (global score = 6.8 ± 3.7, sleep efficiency score = 71.5 ± 10.3, and sleep duration overnight score = 5.7 ± 1.2 hours) as detailed in Table 2.
Differences Between Lymphedema Stages
Upon analyzing the differences between the 3 stages of lymphedema with the help of the Kruskal–Wallis test, the findings pointed to certain noteworthy statistical differences between the 3 stages of lymphedema (P < .05). The stage III lymphedema patients showed the lowest quality of life values when compared those of the stage I and stage II lymphedema patients in all the scales. As for PSQI scores, the stage III lymphedema patients showed worse values than the stage I and stage II lymphedema patients (P < .05) as detailed in Table 2.
Analyzing the correlation between lymphedema stages and the domains of quality of life and quality of sleep using the Spearman coefficient correlation displayed a strong correlation between the quality of life and quality of sleep domains for the lymphedema stages as shown in Table 3.
Correlation of QLQ-C30 and PSQI Items With Lymphedema Stages.

Abbreviations: EORTC QLQ-C30, European organization for research and treatment of quality of life cancer 30; PSQI, Pittsburgh sleep quality index.
Discussion
This study was designed to evaluate the quality of life and sleep quality among Saudi women with secondary lymphedema subsequent to having undergone breast cancer surgery. The main findings exhibited that both quality of life and quality of sleep significantly decreased in Saudi women in all the 3 stages of breast cancer-related lymphedema. Most of the study participants (51.54%) had been diagnosed with stage II lymphedema. However, the worst mean values for both quality of life and quality of sleep were observed in the case of in stage III lymphedema patients.
The results of this study indicate that the mean values of the physical scales were the lowest in the 3 lymphedema stages for the functional status scales of EORTC QLQ-C30. This finding may be linked to definite aspects that may be affecting the study participants such as age, time elapsed after mastectomy, and different treatment methods. 29 This particular finding is in agreement with that of Almutairi et al 29 and Jassim and Whitford 30 who had found in their studies that the emotional domain showed the highest values in all of the lymphedema stages when compared with the other domains of the functional status scales, and this could be linked to the Saudi culture and the emotional and social support that the Saudi people who are cancer survivors get due to their strong family ties, including the social support particularly during the treatment period. Contrary to these results, however, Hassan et al 17 ended up with the lowest emotional status values among Egyptian females with gynecologic and breast cancers.
Also, our study’s findings show that the scores on the global health scales ranged between 32.4 and 38.7 for the Saudi women in different stages of breast cancer-related lymphedema. These findings agreed with those of an earlier study by Almutairi et al 29 that showed a mean value of 31.15, however this result disagreed with some other previous studies designed for different populations as the mean value on the same scales was 66.8 in the United Kingdom, 66.5 in South Korea, 65.5 in Germany, and 63.9 in Bahrain.30-32 The different treatment modalities that were prescribed for the different women patients included in this study may have affected the mean values of the global health quality of life scales.
As regards, the symptoms scales, it was found that insomnia got the highest score followed by appetite loss, dyspnea with lastly diarrhea having the lowest score. These results agreed with a previous study which had found that insomnia was highly incident among breast cancer survivors whilst diarrhea had received low scores indicating that its incidence was rather low. 33
Regarding quality of sleep, all participants in different the lymphedema stages suffered from sleep disturbances as found with the use of PSQI, while stage III lymphedema showed the worst mean values of PSQI when compared with the mean values for stages I and II of lymphedema. The findings also showed that the sleep duration overnight was less than 7 hours/night for stage II and stage III lymphedema patients. This finding may be related to age, body weight, chemotherapy, or radiotherapy interventions. The percentage of sleep efficiency was reported to be less than 85% in all participants, and this confirmed that they had clinically significant sleep disturbances. Further, the percentage in this regard was found to be greater than the percentage identified by Savard et al, 34 for breast cancer patients in Canada. This difference in the 2 findings could be related to the use of the semi-structured questionnaire to evaluate the quality of sleep,34,35 which could lead to lesser percentages than self-reported questionnaire utilized in this current cross-sectional study.
The present study has some strengths. It has included the assessment of quality of life and quality of sleep as clinically significant assessment factors and utilized valid and reliable Arabic versions of the 2 questionnaires that were administered to the study participants—EORTC QLQ-C30 to assess quality of life and PSQI to assess quality of sleep among breast cancer survivors. Moreover, all data have been collected with the help of these self-structured questionnaires in the form of interviews. Last but not the least, this study is the first such study that assesses the differences in quality of life and quality of sleep among patients suffering from different stages of breast cancer-related lymphedema.
However, this study also has some limitations. The design of this study as a cross-sectional study has no cause and no effect and could not recognize what variable can cause another, even as the design of this study helped detect the factors that further affect the health of lymphedema patients. As such it provides leads for further research even though it could not cover all the relevant and related aspects. The sample size of 163 was small, and this may affect the generalizability of this study’s findings as it cannot be said to be representative of all Saudi women with breast cancer-related lymphedema. Further, this study did not assess the effect of the different interventional methods on the quality of life and/or quality of sleep of lymphedema patients. Finally, this study only used subjective assessment tools and did not use any objective assessment modalities. Further studies on this subject should include large sample sizes and employ a combination of subjective and objective assessment modalities.
Conclusions and Recommendations
Both quality of life and quality of sleep significantly decreased in Saudi women suffering from different stages of breast cancer-related lymphedema, and those suffering from stage III lymphedema exhibited the worst mean values of quality of life and quality of sleep. Future research should be designed to replicate and expand the findings of this study in addition to assessing the risk factors that affect the quality of life and quality of sleep among Saudi women suffering from breast cancer-related lymphedema.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University through the Fast-track Research Funding Program to support publication in the top journal (Grant no. 42-FTTJ-71).
