Abstract
Background:
People living with a cancer diagnosis often experience cancer-related fatigue (CRF). Between 9% and 45% of people report CRF as moderate to severe, negatively impacting their quality-of-life (QOL). The evidence-base for managing CRF recommends exercise-related therapies over pharmaceutical interventions. One such exercise-like therapy is Baduanjin mind-body exercise (MBE), which has additional benefits. A remotely delivered program may further benefit people with CRF. The primary objective of this pilot will test study feasibility of a remotely delivered Baduanjin MBE exercise program for people living with CRF.
Methods:
This is a randomized wait-list controlled pilot study and will take place in Sydney, Australia. Subject to informed consent, 40 adults with moderate CRF levels and receiving or previously received adjuvant chemotherapy, will undertake a home-based 8-week Baduanjin MBE program supported by online resources and instructors. The primary feasibility outcomes are recruitment, enrollment, retention, and adherence rates; and safety as measured by tolerance and adverse-event frequency. Clinical outcomes (eg, changes in CRF, QOL, and participant perceptions) are assessed at pre-intervention, week 1, week 4, week 8, and post-intervention. Analyses follows the Intent-to-Treat (all participants as per randomization) and per-protocol (participants adhering to the protocol). Missing data will be imputed from previous data entries and regression models may be tested to predict missing outcomes.
Discussion:
To our knowledge, this is the first study evaluating the feasibility and effects of Baduanjin MBE on CRF using a remote delivery method. These feasibility data will inform a fully powered future trial investigating evidence of effect on CRF and QOL.
Trial registration: Australian and New Zealand Clinical Trials Registry (ANZCTR 12623000177651).
Ringgold ID: 651498 Chinese Medicine Centre
Keywords
Introduction
Cancer-related fatigue (CRF) is a commonly reported cancer or cancer-related treatment side effect. This was estimated at 18.1 million new cancer diagnoses worldwide in 2018, 1 with CRF prevalence ranging as high as 60% to 96% in the affected population. 2 Between 9% and 45% of people report CRF as moderate to severe, 3 with younger people affected more than older people. 4
The National Comprehensive Cancer Network defines CRF as “a distressing, persistent, subjective sense of physical, emotional and/or cognitive tiredness or exhaustion related to cancer or cancer treatment” that is “not proportional to recent activity, and significantly interferes with usual functioning.” 5 While most severe in the first 6-months following a cancer diagnosis or following chemotherapy and/or radiotherapy treatment, CRF can persist for years, is challenging to manage, and can remain present throughout the disease trajectory, affecting quality of life (QOL).5 -8
Given this, CRF is described as a multidimensional construct integrating physical, psychological, and behavioral factors. There are several possible biological and genetic mechanisms underpinning the clinical presentation of CRF. 9 Peripherally, this may include changes to muscle metabolism, innervation, or adenosine triphosphate (ATP) dysregulation,10,11 while central factors may involve (as examples) dysregulation to neural-transmitters such as 5-hydroxytryptamine (5-HT). 12 and release of pro-inflammatory cytokines affecting the hypothalamus-pituitary-adrenal axis (HPA axis).10,13,14
Current CRF treatment guidelines tend toward recommending exercise-based activities over pharmaceutical interventions. 15 A meta-analysis of 26 randomized clinical trials (RCT) concluded aerobic exercise is effective for managing CRF in patients who had completed adjuvant therapy. 16 Cochrane reviews report aerobic exercise reduced CRF both during and after treatment, specifically in people with solid tumors,17,18 and help alleviate the cognitive and physiological effects of fatigue.19,20 Exercise and psychological treatments are additionally reported better than available pharmaceutical treatments in reducing CRF, in both the treatment and post-treatment stages.15,20 A meta-analysis of non-pharmaceutical cognitive, manual, and mind-body therapies (including tai chi and yoga) reported mild to moderate effects on alleviating CRF. 21
Amongst different forms of exercises, there is a historical type of exercise still widely practiced called Baduanjin mind-body exercise (MBE), which is a practice akin to Tai chi (Tai ji) originating from China. A systematic review concluded that Baduanjin MBE is beneficial for QOL, sleep, general life balance, and physical parameters (hand grip strength, flexibility, blood pressure, and heart rate) among healthy individuals,20,21 and participants with a variety of diseases.22 -25 Clinical studies showed that Baduanjin MBE significantly reduced fatigue and improved perceptions of QOL of people diagnosed with colorectal cancer and undergoing chemotherapy, and breast cancer survivors who completed chemotherapy.2,7 Baduanjin MBE is also reported to effectively alleviate depression and anxiety and increase health-related QOL in patients with lung cancer. 26
Baduanjin MBE involves a set of 8 gentle aerobic exercises coordinating physical movement with mindfulness and controlled breathing patterns. According to the American Sports Medicine Association and the American Centers for Disease Control and Prevention, Baduanjin MBE is a low-intensity exercise. The metabolic equivalent of Baduanjin MBE is <3.0 and 54% of the maximum heart rate. 27 As a low-intensity exercise, Baduanjin MBE training has been shown to reduce fatigue, and increase maximum oxygen uptake and muscle strength without causing significant adverse events.28 -33 In a healthy population, it is an effective and safe exercise to promote health-related physical fitness.34,35
Distinctly, Baduanjin MBE whilst providing exercise-like benefits has additional advantages as a low impact and low intensity intervention to assist people who cannot otherwise participate in standardized exercise interventions. Baduanjin MBE may therefore be an appropriate form of exercise in lieu of other prescribed exercises for people living with CRF who have difficulty performing aerobic exercises and/or strength-based training due to physical (muscle weakness), social (isolation), and psychological (pain, self-perception) barriers.36,37 We additionally propose that technology-supported remote programs offer additional opportunities for people to participate in cancer support services including for people living in regional and rural locations. Critically, remote technology-assisted delivery benefits include immediate access to support care services and convenience, 38 while supporting patient’s self-management strategies and health-related self-determination. 39
Baduanjin MBE has a local relevance to many countries’ health care systems. In Australia, (the location of this pilot study), it is a practice prescribed by registered Chinese Medicine health care practitioners. 40 However, further study is required to investigate its receptiveness in an Australian-based cancer-care context and population. Consequently, in this study we will test whether remote delivery of Baduanjin MBE is a feasible home-based program for people with CRF. To our knowledge, this will be the first study evaluating the feasibility and effects of Baduanjin MBE on CRF using a remote delivery method. To test the hypothesis, an 8-week remotely delivered Baduanjin Qigong MBE program is assessed on recruitment, tolerance, adherence, safety, and completion of self-reported CRF and QOL outcomes in adults who have had a confirmed medical diagnosis of a solid tumor and are receiving or have received adjuvant therapy.
Methods
Study Design
The study is designed as a randomized waitlist-controlled feasibility trial using mind-body exercise intervention with remote delivery. Participants with moderate levels of CRF as reported on the Brief Fatigue Inventory (BFI) will be randomized into the waitlist control or Baduanjin MBE intervention group for a period of 10-weeks, consisting of week 0 pre-testing, weeks 1 to 8 treatment period and week 9 post-testing. The data collection occurs 1 week prior to treatment period commencement (week 0), week 4, week 8, and a final time week later at follow-up (week 9). The study is proposed to run for approximately 9-months.
Participants
Inclusion criteria
Eligible participants are adults 18 years of age or older who have had a medical diagnosis of a cancer and are receiving adjuvant chemotherapy or have completed adjuvant chemotherapy within 6 months of study recruitment, and are suffering from moderate CRF as assessed by the BFI simple fatigue scale (a score of 4-6). Participation additionally requires people to have access to a personal computer/device that is Wi-Fi or hotspot connected and be able to use smart phones, email, and Zoom conferencing. Finally, eligible participants must understand the study requirements and be able to communicate and provide informed consent in English.
Exclusion criteria
People unable to participate safely or fully will be excluded from the study. Exclusion criteria include any of the following: uncontrolled cardiopulmonary disease, angina, uncontrolled hypertension or uncontrolled postural hypotension; uncontrolled nerve, muscle, or joint disease or injury affecting coordinated movement; psychiatric illness or serious cognitive impairment and communication or language barriers that effect informed consent; postoperative heart, cerebral vessel, or other serious complications; those not fully recovered from breast surgery, axillary surgery or transabdominal rectus abdominus muscle reconstructive surgery or any other surgery that would preclude full participation in the program; or if an individual is otherwise not approved by their oncologist as being potentially eligible. Additionally, people enrolled in other investigational studies which would impact CRF or who have a recent history (within the previous 3 months) of regularly participating in Baduanjin MBE will be excluded.
Ethics and ANZCTR
The study will adhere to the National Health and Medical Research Council’s (NHMRC), Australian National Statement on the Ethical Conduct of Human Research), further to the Australian Code for the Responsible Conduct of Research.41,42 This research was reviewed and approved by the institutional review board of the South Western Sydney Local Health District (SWSLHD) Human Research Ethics Committee (2022/ETH02353). The Principal Investigator remains responsible for communicating important protocol changes.
Recruitment and Screening
The SWSLHD catchment area includes a population of 1.2 million people. The whole population estimate of new cancer diagnoses per year is 5300, with approximately 65% of people diagnosed with cancer reaching 5-years of survivorship. 43
Recruitment
We will use a combination of clinical and community recruitment. People interested in participating may be referred by their treating oncologist at the Cancer Therapy Centre, Liverpool Hospital (SWSLHD), through other health care practitioners or through self-referral after seeing HREC approved advertising brochures on noticeboards, website, or participating institutions’ social media pages.
Screening and consent
Having read the participant information sheet, potential eligible study participants are screened by the trial manager from all people who have expressed an interest to the study or who have given permission to their oncologist to be contacted by telephone.
Participants are enrolled once the informed consent process has been completed and the participant has met all inclusion criteria and none of the exclusion criteria. The trial manager will record each new participant on the participant list sheet and assign them an anonymous study number after enrollment (Refer to Figure 1).

Recruitment and screening flow.
Participants may request to be withdrawn from the study at any time. At withdrawal, participants will be asked to specify whether (a) if they do not wish to contribute any further to the research and (b) if they also wish all their existing data to be deleted.
Randomization
A randomization list will be created using an online randomization tool. The list is generated by a research team member independent from the trial manager. The list will allocate 40 participants to one of two (2) equal sized groups (treatment and waitlist) in blocks of four (4) to assist with equitable participant allocation between groups throughout the recruitment process and avoid unequal groups if the study fails to meet enrollment targets. Forty (40) envelopes will be numbered 1 to 40 on the outside and the corresponding random allocation will be written on a card sealed inside the envelope. As soon as possible after the participant signs the consent form, the trial manager will select and open the next envelope in order. The participant’s group allocation will be recorded next to their study number.
Confidentiality and data security
Participants identifying details will be stored separately away from the main data sets, on a password protected computer behind the Western Sydney University firewall by the trial manager. The list will be maintained to allow confirmation of informed consent, allow withdrawal of participant data from the study and to assist in the data checking, auditing, and cleaning processes. Data files for analyses will only contain study numbers. Data is only accessible by members of the research team involved in data collection, auditing, and analysis.
Baduanjin Intervention
An online video (Baduanjin MBE Video) and an accompanying handbook with images as a secondary reference for participants (Baduanjin Mind-Body Exercise Instruction Handbook) have been developed to support people’s participation and reliability of instructions provided by the Baduanjin MBE practitioners. The 2 resources describe each of the 8 coordinated Baduanjin MBE movements, including postural positioning, breathing technique, and attentional focus.
The construct logic and validating context of Baduanjin sits within the Chinese medicine framework of health and wellbeing. Consequently, the movements and instructions authentically remain anchored in this construct and participants are introduced to the associated terminology and concepts in the online video and handbook. The intervention with participants is undertaken by trained Baduanjin MBE practitioners who are part of the research team, and not involved in trial administration, data collection, or the consenting process. All trainers have a minimum of 5-years Baduanjin MBE practice experience additional to formal university education in Chinese medicine mind-body exercises.
Baduanjin MBE includes 3 phases: warm-up, exercise, and cool-down which together comprise the intervention in this trial. During the warm-up and cool-down phases, the participants will perform deep breathing and stretching exercises to assist with commencing and finishing the routines while focusing their intent. The warm-up and cool down phase help ameliorate any potential muscle soreness arising from unfamiliar exercise. During the exercise phase, the participants perform 8 sets of movements led by the online video, cross-referenced to the handbook and/or with the instructor via Zoom.
Safety and Risks
People living with a cancer diagnosis and undergoing or who have undergone adjuvant chemotherapy are susceptible to adverse events. Adverse reactions to professionally administered Baduanjin MBE have been reported and may include mild dizziness or drowsiness after treatment (rare); and mild chest pain, shortness of breath, cold sweats, nausea, and vomiting (very rare). 44 However, our eligibility is designed to ensure participants with a reasonable functional status at baseline are included to support a good adherence of the study.
The study screening criteria exclude people with a known and uncontrolled health condition that might increase their risk of experiencing a rare or very rare adverse event. If fatigue begins to worsen (BFI is more than 8/10), the participant is advised to seek medical advice and may stop study participation. All patients have access to standard care for symptom management. Adverse events will be recorded and reported as per the protocol and reviewed by the principal investigator, who will also liaise with the participant’s medical team.
Data Collection Process and Intervention Procedures
Baduanjin mind-body exercise intervention group
Participants commencing the Baduanjin MBE program meet with an instructor member of the research team (in week 0), who will organize delivery of the information pack and resources and provide technical assistance. The information pack includes the handbook, and the Exercise Logbook.
Participants receive the online video link in week 1 and a Baduanjin MBE instructor will organize a Zoom meeting. Participants will be invited to watch the online video, where the instructor will explain and demonstrate the Baduanjin MBE intervention, including the warming up and cooling down movements. An initial face-to-face practice session is also available on request.
In addition to self-guided daily practice, the participants will be asked to join in a coached Zoom exercise program with an instructor, either in the morning or afternoon or both sessions at their own choice throughout the intervention period (8 weeks). Participants will be requested to practice Baduanjin MBE at least 2 sessions per day, 6 days per week with each session lasting about 17 to 20 minutes.
Participants record their daily Baduanjin MBE schedule in the Exercise Logbook. The trial manager will make weekly telephone calls with each participant, checking on their progress and engagement with the Baduanjin MBE program and/or check in on unexpected side effects or adverse events that might be related to the exercise.
Finally, participants are asked to complete the BFI and Participant-Reported Outcomes Measurement Information System v2.0 (PROMIS-29v2.0) at baseline (week 0) as part of the study intake process, and again at the end of week 4 and week 8 of the exercise program. There is also a modified Credibility and Expectancy Questionnaire (CEQ): two questions are completed before entry into the study (week 0), and 2 at the completion (week 9).
One week after completing the Baduanjin MBE program (in week 9), the trial manager will contact participants a final time to check for participant reports of adverse experiences and prompt them to complete the final BFI and the PROMIS-29v2.0 measures, along with the final 2 questions from the CEQ (See Figure 2: Intervention group and control group study flow.).
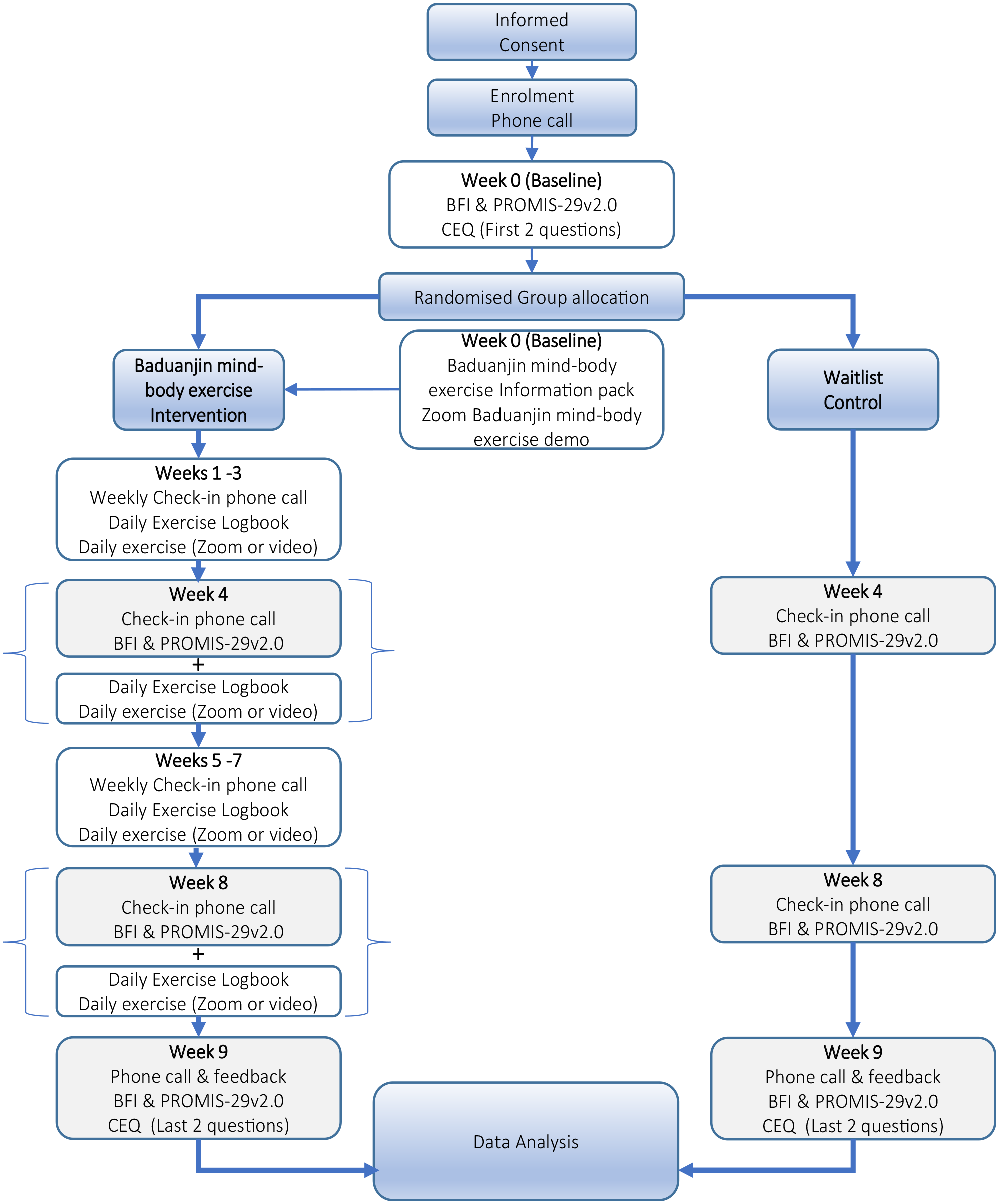
Intervention group and control group study flow.
Wait list control group
From week 0 to week 9 (10 weeks in total), participants will continue their usual daily care, medications, and other lifestyle routines without modification, and undergo no Baduanjin MBE intervention. Participants will complete the BFI and the PROMIS-29v2.0 questionnaires at the same time-points as the intervention group (Refer to Figure 2 for study flow process.).
Participants in the waitlist control will be offered entry into the Baduanjin MBE program at completion of their waitlist control period. Entry is as a non-study participant.
Outcomes and Measures
Primary outcome and measures
The primary feasibility outcomes include recruitment and enrollment (as measured by the number of inquiries per month of active recruitment and conversion rate); retention (the number of participants completing the study); and adherence rates in the Baduanjin MBE exercise intervention group (the frequency of participation in exercise sessions each week as reported in the Exercise Logbook).
The intervention group participants will be required to complete relevant entries relating to exercise and/or safety in the Exercise Logbook for each day during the 8-week intervention period. Exercise will be recorded as completion of exercise or not. Adverse events will be recorded descriptively and reported by frequency. Adverse events will be further recorded during weekly telephone check-ins with the trial manager.
Secondary outcomes and measures
Secondary outcomes include participant reported changes in CRF and QOL (ie, effectiveness), and participants perceptions of the credibility of the intervention.
Changes in CRF are measured using the BFI. The BFI comprises 9 items measured on a ten-point scale that assesses severity and impact of fatigue on the participant’s activities of daily living.45,46 A global fatigue score is calculated by averaging the scored items.
Changes in health-related QOL (including fatigue) are measured by the PROMIS-29v2.0 profile, which uses a single 0 to 10 numeric rating across 7 health domains (physical function, fatigue, pain interference, depressive symptoms, anxiety, ability to participate in social roles and activities, and sleep disturbance), with 4 items per domain. 47 It assesses health intervention impacts and tracks changes in health overtime. 48
Participants’ perception and credibility of the Baduanjin MBE program is measured using a modified CEQ. 49 Two questions are completed before randomization about participants expectations, and 2 questions after the final Baduanjin MBE session on the credibility of their experiences. 50 Each question is measured on a Likert scale of 1 to 9 where 9 is the most credible.
Data quality and compliance checks
A research team member independent of the trial manager will undertake fortnightly audits of collected data and study documentation to ensure compliance with study protocol and procedures, cross-referencing with data entry logs.
Sample Size Calculations
Group sizes of 12 particpants, 51 and 15 to 20 participants per group are reported to give reasonable bias-corrected estimation of medium effect sizes in repeat-measure designed studies. 52 For this study, a sample size of n = 29 is necessary to have a 95% confidence of detecting an event which has a true probability of π = .10. To overcome the probable 15% to 25% study participant attrition rate and to ensure availability of sufficient participants, a sample size of n = 40 participants will be recruited. Therefore, 20 participants each will be allocated to the Baduanjin MBE intervention and control groups. This number is sufficient to maximize chances of identifying feasibility issues that might arise with the intervention. 52
Analysis
Key feasibility endpoints are recruitment rates and the participation and completion of the study interventions (from Exercise Logbook data) within the stated study periods. Primary outcomes are binary and will be reported as percentages with associated 95% confidence intervals.
Both an interim and end-of-study analysis of safety data will be undertaken. Analysis of safety data will include categorization by event type (pre-specified, emerging/unexpected), frequency counts, time to event including exercise sessions completed before event onset.
Secondary outcome measure endpoints will include changes in reported fatigue and QOL as measured respectively by the BFI and PROMIS-29v2.0 scores at baseline (week 0), compared with at 4 weeks (mid-point), 8 weeks (final week) and 9 weeks (1 week follow-up). Participant’s CEQ ratings are compared prior to study group randomization with their credibility scores on completing the Baduanjin MBE program.
Analyses of the endpoints will follow the Intent-to-Treat (all participants as per randomization), and per-protocol (participants adhering to the protocol). These analyses will include, as appropriate, participant demographic (co-variates) and Baduanjin MBE participatory frequency data. The secondary outcome data includes testing the analysis methods and assumptions of normality in the data with the full expectation these will be non-significant. Histograms and descriptive statistics will be used to investigate the distributions of these numeric measures. Departures from normality will be addressed using transformations (for skewness) or trimming or Winsorizing (for unresolvable outliers). Comparison between treatment and waitlist groups will be pilot tested using independent samples t-tests, repeated measures ANOVA and/or linear mixed models.
Missed weekly training sessions will not construe a protocol deviation and may be imputed from previous data entries. Pilot tests of possible regression based and/or multiple imputation techniques will be undertaken. This includes using these data to formulate future trial hypotheses.
All data will be entered and managed using the Research Electronic Data Capture (REDCap) management system and all analyses performed using SPSS (V 24.0; IBM Corp; Armonk, NY, USA) or descriptive analyses.
Discussion
Cancer-related fatigue is a critical QOL research area given its persistence into the post-cancer treatment period. 53 Improvements in survivorship rates mean more people are living with a cancer-related fatigue issue. There are many features and strengths in the design of the study.
Baduanjin MBE program is easy to learn. The movements are gentle, slow, and simple, and often reported as relaxing. 33 As a low intensity activity, Baduanjin MBE may benefit the unmet needs of patients with CRF who have difficulty performing prescribed aerobic and/or strength training exercises such as walking. Baduanjin MBE is also relatively safe. A systematic review identified 22 of 47 clinical trials (involving a total of 3877 participants) with adverse events reporting protocols, with only 2 trials subsequently reporting adverse events, underscoring the potential safety of the exercise. 46 Additionally, qigong interventions are reported to have biological and psychological effects reflecting CRF as a multidimensional construct. 54 Given the complex nature of fatigue and limited CRF pharmaceutical treatment options, Baduanjin MBE therefore presents a potentially useful intervention with additional benefits for patients with special needs.
Designing a remote Baduanjin MBE program provides a potentially flexible, self-management strategy. It engages commonly available communication technologies and services such as Zoom video conferencing, telephone, email, and post, instead of in person site visits, (although the latter is available in this study upon request), to deliver the benefits of traditionally based medical practices to groups or individuals who may not be able to attend class or locate a suitably trained practitioner nearby. 38 This also supports ease of user interaction with the program’s instructors and the research team. Importantly, there are no special material costs for performing the Baduanjin MBE program or the use of these technologies to participants; any study related Wi-Fi or telephone usage costs will be covered.
The program has been specifically designed to support participant adherence. Low participant adherence to MBEs in RCTs (such as yoga and Qigong) have been reported. A Qigong RCT investigating cancer symptom clusters inclusive of fatigue, reported significant declines in participation and adherence rates, from 84.6% (n = 66) at 3 weeks to 61.5% (n = 48) at the conclusion of the 6-week program. 55 The current study proposes a longer 8-week intervention but supported by self-practicing strategies. Self-practicing strategies reportedly increase adherence through supporting participant’s locational agency and convenience with when to practice (compared to an on-site study).38,56 High participant adherence and minimal loss to follow-up in Qigong and Taichi based MBE RCTs are grounded in maintaining participants motivation. 57 Therefore, the addition of regular weekly telephone check-ins and availability of instructors via Zoom are designed to support study procedure compliance. Additionally, the Exercise Logbook has a dual purpose of documenting participants’ progress while also reporting frequency and compliance with the Baduanjin MBE, which may assist with motivating participants.
Equipoise is apparent in the study’s design, and this has been undertaken to minimize the risk and time burden on participants. We have calibrated the Baduanjin MBE intervention dosage as two 17 to 20-minute sessions twice a day for 6 days a week. Fewer times practicing may not have a sufficient impact on CRF or QOL changes, while more frequent and extended times might aggravate CRF. Therefore, we have noted a minimum 8-week program to maximize the accumulative benefits of repeated dosage while minimizing the potential risk of adverse events. Collecting clinical study data can be burdensome for participants, (and it also affects the adherence). In addition to the Exercise Logbook the outcome measure questionnaires (BFI and PROMIS-29v2.0) are estimated to take less than 10 minutes to complete.
Quality control has been central to the design of the study as part of the feasibility. For example, a critical design feature of the program is standardization which differentiates it from other Qigong studies. Unlike acupuncture, there are no standardized guidances for Baduanjin MBE RCTs, compounding the current paucity of reported research, and to be addressed in future research.54,58 Therefore, to address the potential confounder of practice variation, we commenced an iterative co-design process comprising an international, multi-disciplinary team of medical and allied health care investigators. This included experienced Chinese medicine practitioners, oncology physiotherapist, and medical cancer specialists along with professionally trained MBE practitioners, combining disciplinary perspectives to develop an agreed set of operationalized Baduanjin MBE instructions detailed in the online video and handbook resources. A trainer resource for Baduanjin MBE instructors was developed for the same reason. The participant resources additionally provide information for conceptual insight about committing to an intervention potentially different from their previous experiences of cancer-related healthcare, supporting their health-related autonomy and competence. 39
While potentially meaningful, we have excluded people from eligibility on neoadjuvant therapy as it would be difficult to interpret the findings at this stage. 59 Patients with poor functioning status, (for example, oncology practice defined as ECOG PS 3-4), might not tolerate even minor exercise despite Baduanjin MBE having a lower cardiac load and few side effects.46,59 While the intervention is not expected to affect anemia-related fatigue levels independent of low hemoglobin (Hgb < 12 g/dl), given the complex nature of CRF the feasibility eligibility does not exclude on this basis. Also, while there are potential benefits for people living with severe CRF to respond to the Baduanjin MBE intervention, 3 we have maintained a moderate CRF BFI rating as an eligibility criterion for the feasibility.
Finally, the data from the feasibility study will be used to identify an effect size, along with standard errors and standard deviations. These will contribute to powering future trials with the aim to attract research funding and contribute evidence to inform clinical and patient oncology-related decision making for prescribing Baduanjin MBE programs.
Footnotes
Acknowledgements
The authors would like to thank and acknowledge contributions from Jie Hao (Western Sydney University), Kelly Mok (Liverpool Hospital), Wei Chua (Liverpool Hospital), Weng Ng (Liverpool Hospital), Meera Agar (Liverpool Hospital), Danise Berger Robertson (Western Sydney University), and Yoann Birling (Western Sydney University) for their expertise, time, and assistance contributing the initial work at an early stage. The authors would like to thank Yanmei Liu (Shaanxi University of Chinese Medicine and Western Sydney University) for translation and editing to the study resources (video and handbook).
Author Contributions
Conceptualization: XSZ, SW, SHD, KW, AL. Methodology: XSZ, SW, AL, SHD, PF, KW, YBH, EM. Resources: KW, YBH, AL, XSZ. Writing—Original Draft: SW, XSZ. Writing—Review & Editing: SW, XSZ, KW, AL, SHD, YBH, YTS, ET, EN, GW, EM, SDF, PF, XSW, MC. Visualization: SW, AL. Supervision: XSZ, SW, SDF, EM. Project administration: XSZ, SW.
Data Availability Statement
The data that support the findings of this study will be available in a published, aggregated form. Reasonable access requests to the data, the handbook and video supporting the trial, can be sent to the corresponding author. Participants also have access to a copy of the finalized study report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All involved authors are employed by the respective affiliations noted on the title page, with the Western Sydney University as the leading institution in which philanthropic funding for researchers’ time was also used.
