Abstract
Purpose:
Sipjeondaebo-tang (SDT) is a widely used traditional herbal medicine for relieving fatigue. This randomized, placebo-controlled, preliminary study evaluated SDT for cancer-related fatigue, which is the most common symptom experienced by patients with cancer.
Patients and Methods:
Patients with a Brief Fatigue Inventory (BFI) score of at least 4 were randomly assigned in a double-blinded manner to receive SDT (3 g 3 times daily) or placebo orally for 3 weeks. The BFI was the primary outcome measure and secondary outcome measures included the Hospital Anxiety and Depression Scale (HADS), the European Organization for Research and Treatment of Cancer-Quality of Life Questionnaire (EORTC QLQ-C30), immunoregulatory tests, and safety.
Results:
A total of 50 participants were randomly assigned and 48 patients completed the trial. Based on intention-to-treat analysis, fatigue, which was the primary outcome, was improved in both arms compared with the baseline, and was significantly better in the SDT group than in the placebo group at week 3 (3.56 ± 1.18 vs 4.63 ± 1.83, P = .019). Secondary outcomes, including anxiety, depression, and immunoregulatory tests, did not improve significantly in either group. However, quality of life measured using the EORTC QLQ-C30 improved in both arms compared with the baseline, and the global health subscale was significantly better in the SDT group than in the placebo group (P = .02). No significant toxicities were observed.
Conclusion:
SDT may improve cancer-related fatigue and quality of life in patients with cancer. A further randomized clinical trial with large sample size is warranted.
Keywords
Introduction
Cancer-related fatigue (CRF) is a distressing, persistent, subjective sense of physical, emotional, and/or cognitive tiredness or exhaustion related to cancer or cancer treatment that is not proportional to recent activity and interferes with usual functioning. It is also the most prevalent symptom experienced by patients with cancer that affects quality of life.1-4 CRF is estimated to occur in up to 90% of patients during active cancer treatment 5 and 27% to 82% of patients after treatment. 6 Multidimensional factors of physical and psychosocial mechanisms can cause CRF, which starts before diagnosis and persists for months or years after treatment completion.4,7-9 CRF may also be a predictor of shorter survival. 10
The National Comprehensive Cancer Network 4 guidelines recommend non-pharmacological and pharmacological interventions for CRF after managing concurrent symptoms and treatable contributing factors including pain, emotional distress, anemia, sleep disturbance, nutritional deficit, and comorbidities. Despite a variety of interventions that have been beneficial to patients with CRF, there is currently no gold standard in its treatment. 11
Sipjeondaebo-tang (Juzentaihoto in Japanese, Shi-Quan-Da-Bu-Tang in Chinese, SDT) is a traditional herbal medicine comprised of 10 herbs and has been widely used for relieving fatigue in East Asia. 12 In previous studies, SDT was shown to be an immune-augmentative drug that induced natural killer cell activity and decreased regulatory T cells.13,14 In human study, SDT has an adjuvant effect on maintenance of antibody on influenza vaccination in elderly. 15 SDT has also been shown to improve chronic fatigue syndrome and the quality of life and anorexia in patients with cancer.16-18 To the best of our knowledge, no double-blind, randomized, placebo-controlled study of the efficacy of SDT in CRF has been published.
With the high prevalence of CRF and the limited number of therapeutic options, the purpose of this double-blind, randomized, placebo-controlled, preliminary trial was to evaluate the efficacy and safety of SDT on CRF.
Patients and Methods
Study Design
This randomized, double-blind, placebo-controlled, preliminary study was conducted to investigate the efficacy of 3 weeks of SDT treatment on CRF. The accrual occurred from May 2018 to June 2020 at Kyung Hee University Hospital in Gangdong, Republic of Korea. Participants who met the eligibility criteria and voluntarily signed the informed consent were randomly assigned in a 1:1 ratio into the SDT and placebo groups. A permuted-block randomization sequence using a 2 × 2 block based on a computer-generated table of random digits was used for group assignment. Random assignment was implemented via another statistician who was not associated with the current study. Only pharmacies had access to the randomization sequence. The random list ensured full allocation concealment, which was concealed in an opaque envelope prior to the first visit of the subject. The professional clinical research coordinator enrolled the participants and recorded the case report form from every patient-reported questionnaire. Both the researchers and the participants were blinded, and the analysis was performed by a separate statistician, who belongs to the Kyung Hee Medical Science Research Institute.
This study was reviewed and approved by the Institutional Review Board of Kyung Hee University Hospital at Gangdong (KHNMC-OH-IRB 2017-08-006). The protocol was registered in the Clinical Research Information Service (KCT0003442). All research adhered to the tenets of the Declaration of Helsinki, and the study was performed according to the Good Clinical Practice guidelines. The study proposal has not changed since the allocation started.
Eligibility
Patients were eligible to participate in the trial if they had a confirmed malignant solid tumor, were older than 19 years; were at least 1 month past their last major treatment (ie, surgery, radiotherapy, or chemotherapy); complained of moderate or severe fatigue, defined as a Brief Fatigue Inventory (BFI) score of 4 or more; had fatigue that started or got worse according to cancer progression or cancer-related treatment and lasted more than 1 month; and had an Eastern Cooperative Oncology Group (ECOG) performance status of below 2. Patients were excluded if they had moderate to severe pain (Numeric Rating Scale (NRS) of 4 or more); had at least one of the following: pleural effusion, ascites, or edema on the extremities with a severity of grade 3 or more, according to Common Terminology Criteria for Adverse Events (CTCAE) ver. 4.03; had anemia that required a transfusion (hemoglobin ≤ 7 g/dL); were diagnosed with hypothyroidism, a psychological or mental disorder, nutritional dystrophy, or paralytic or atrophic myopathy including myasthenia gravis; had a history of alcoholic or psychotropic drug abuse; were pregnant or breast-feeding; planned to receive surgery, chemotherapy, or radiotherapy during the study period; were 2 times higher or more than normal serum levels of aspartate transaminase (AST) or alanine transferase (ALT); or were one and a half times higher or more for their serum creatinine level. The eligibility criteria did not change from the registration of the study proposal to the end of the study. All participants signed an informed consent form before participating in the study.
Any kind of concurrent use of medication or dietary supplements that could have potentially affected fatigue was allowed only when the participants maintained the same type and dosage of medication for more than 2 weeks before the study initiation.
At baseline, the following data were recorded: demographic data, information on cancer and treatment history for cancer, comorbidities, BFI score, Hospital Anxiety and Depression Scale (HADS), and European Organization for Research and Treatment of Cancer-Quality of Life Questionnaire (EORTC QLQ-C30) score.
Intervention
Patients enrolled in the study were randomly assigned to receive either SDT (3 g) or matched placebo orally 3 times per day for 3 weeks. All participants took fatigue-relieving education by a professional nurse.
SDT (Desiten, Hanpoong Pharm & Foods Co., Seoul, Korea) was an already approved product for chronic fatigue or remnant fatigue after disease by the Ministry of Food and Drug Safety (MFDS) in Korea. SDT granules were a dark brown mixture of spray-dried hot water extracts of 10 medicinal plants consisting of Astragali Radix (10.5%), Panax ginseng radix (10.5%), Atractylodes Rhizoma Alba (10.5%), Poria sclerotium (10.5%), Rehmanniae Radix (10.5%), Angelicae Gigantis Radix (10.5%), Paeonia Radix (10.5%), Cnidii Rhizoma (10.5%), Glycyrrhizae Radix et Rhizoma (5.3%), and Cinnamomi Ramulus (10.5%). Each herb in SDT was quality controlled from their places of origin to the final product by the manufacturing company. A placebo consisting of lactose, corn starch, and caramel coloring was prepared by the same pharmaceutical company, and it has a similar appearance, dose, and scent as SDT.
An education in fatigue relief consisted of 7 domains: knowing reasons for fatigue, gaining insight into fatigue state, energy conservation, nutrition coaching, sleep hygiene education, exercise coaching, and additional methods the patient can try. Participants were educated by a professional nurse and recommended to maintain the education throughout the study period.
Participants in both groups were recommended to maintain the dosage of SDT or placebo throughout the trial period. Dose modification was not scheduled. If participants failed to take more than 70% of SDT or placebo during the trial, the participant was dropped from the study out due to low compliance. If fatigue was completely resolved within the study period, early cessation of the study was also permitted.
Outcome Measures
The primary outcome measure was the BFI to evaluate the efficacy of 3 weeks of SDT in improving CRF. Patients completed the BFI at baseline, and after week 1, week 2, week 3 (treatment completion), and week 6. The BFI is a 9-item scale validated in the cancer population for the assessment of fatigue severity and impact on function.19,20 The secondary outcome measures were depression and anxiety using the HADS, quality of life using the EORTC QLQ-C30, immunoregulatory tests of cytokines, including interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α), and lymphocyte subsets (CD3+, CD4+, CD8+, CD4+/CD8+, and CD16+56+), and safety. The HADS consists of a 14-item questionnaire measuring depression and anxiety validated in a number of clinical situations and has been widely used for patients with advanced diseases.21,22 The EORTC QLQ-C30 is a 30-item measurement that evaluates the overall quality of life in patients with cancer and consists of functional, symptom, and global health status subscales.23,24 Peripheral blood samples for lymphocyte subset and cytokine tests were taken from each patient at baseline and after week 3. Cell preparation and assay procedures were conducted by Green Cross Laboratories©.
Safety Evaluation
Any adverse events were assessed using the Common Terminology Criteria for Adverse Events (CTCAE, version 4.1). Laboratory tests, including those for hemoglobin, white blood cell count, platelet count, aspartate aminotransferase, alanine aminotransferase, total bilirubin, blood urea nitrogen, and serum creatinine, were performed at the baseline and end of the study. We also followed the World Health Organization-Uppsala Monitoring Center (WHO-UMC) causality assessment system.
Sample Size
As a reference study using the BFI score for SDT was absent when we built the study structure, we calculated the sample size based on the study conducted by Chen 25 which used a similar traditional herbal medicine, Bojungikki-tang, for CRF. Both herbal medications share several major ingredients, especially Panax ginseng Radix and Astragalli Radix, and are traditionally used for fatigue. The standard deviation in the former study was 1.95. We estimated that a total of 50 evaluable participants were needed for the 0.05 alpha, 80% power, and 20% dropout rate to detect a difference between groups of 1.8 score in this 1:1 study.
Statistical Analysis
An independent statistician performed the statistical analyses. The intention-to-treat (ITT) principle was used with the last-observation-carried-forward (LOCF) method. All values are presented as mean and standard deviation or N (%) unless stated otherwise. Baseline variables were compared between groups using the independent t-test, chi-square test, Wilcoxon rank sum test, or Fisher’s exact test. The duration of illness was analyzed using the Kaplan–Meier method. The primary outcome was analyzed with the corresponding one-way analysis of covariance (ANCOVA), considering the baseline scores as covariates. Repeated measures of ANOVA were used to analyze the serial change of BFI to detect inter-group and inter-time differences and group-by-time interactions for ITT analysis. An independent t-test was used to compare changes in scores after performing the Shapiro-Wilk test to confirm the normality of the distribution. If a normal distribution was not achieved, the Wilcoxon signed rank test was used. All statistical analyses were 2-sided, and statistical significance was set at 0.05. All statistical analyses were performed using Statistical Analysis System (SAS), version 9.4 (SAS Institute, Cary, NC, USA).
Results
Patient Characteristics
A total of 50 patients were recruited for the study and randomly allocated to the SDT and placebo groups in a 1:1 ratio. Three participants in the placebo group discontinued the intervention, as detailed in Figure 1. In total, 47 patients completed the trial and 50 were included in the ITT analysis for the primary endpoint. Baseline patient characteristics are provided in Table 1, illustrating that there were no statistically significant differences between the arms in baseline assessed variables.

CONSORT diagram.
Baseline Demographic and Clinical Characteristics.
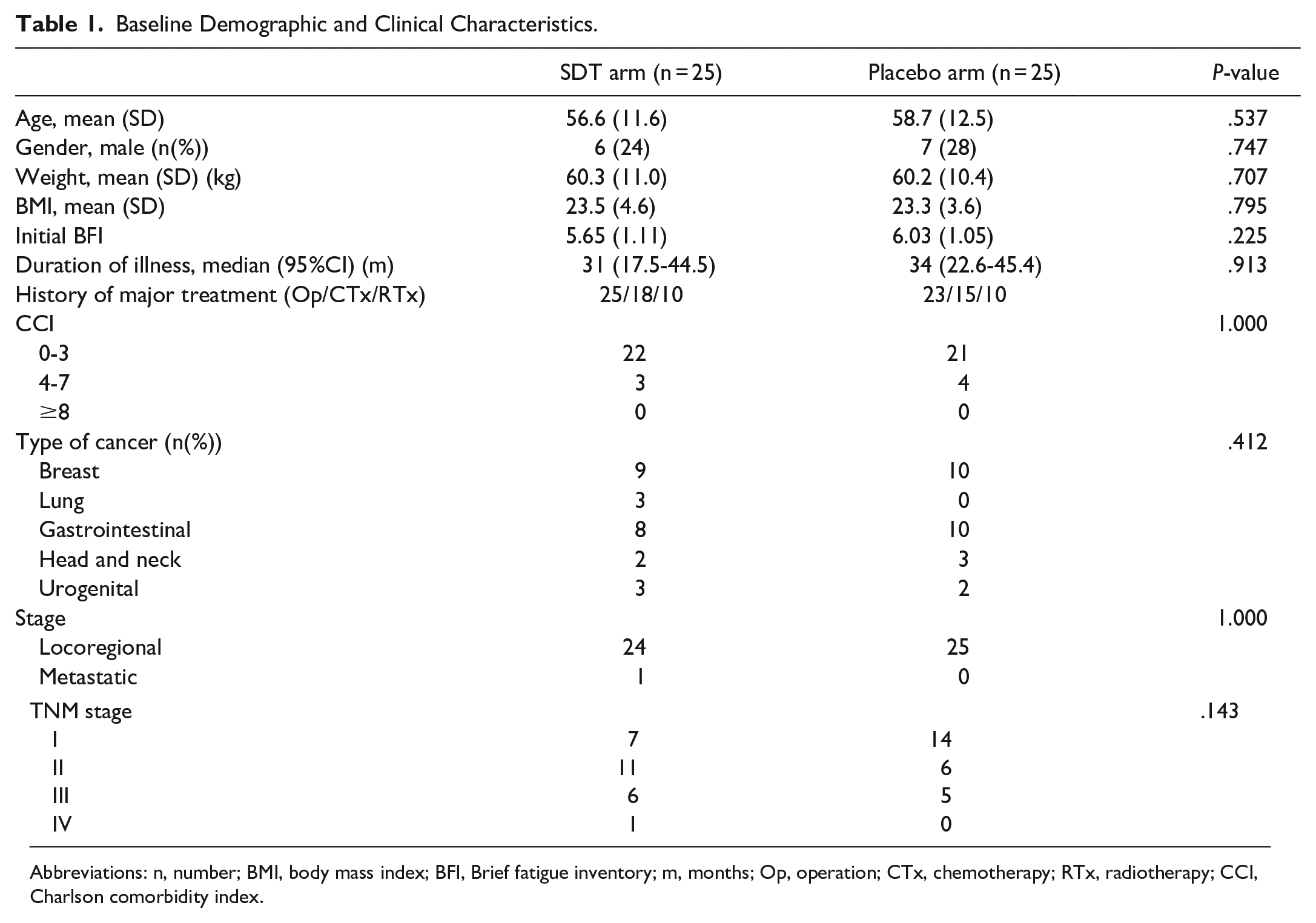
Abbreviations: n, number; BMI, body mass index; BFI, Brief fatigue inventory; m, months; Op, operation; CTx, chemotherapy; RTx, radiotherapy; CCI, Charlson comorbidity index.
Efficacy
After 3 weeks of treatment, mean BFI scores decreased significantly in both arms compared with the baseline, and decreased significantly in the SDT arm compared with the placebo arm adjusted for baseline fatigue (P = .04; Table 2). Anxiety and depression did not improve significantly in both arms compared with the baseline, and there was no significant difference between the arms. Patient quality of life, as assessed through the EORTC QLC-C30, improved significantly in both arms compared with the baseline in terms of the functional and symptom scales, but there was no significant difference between the arms. The global health status scale improved significantly only in the SDT arm compared with the baseline, and showed a significant difference between the arms (P = .02).
Outcomes at Baseline and Week 3 for Fatigue, Anxiety, Depression and Quality of Life.
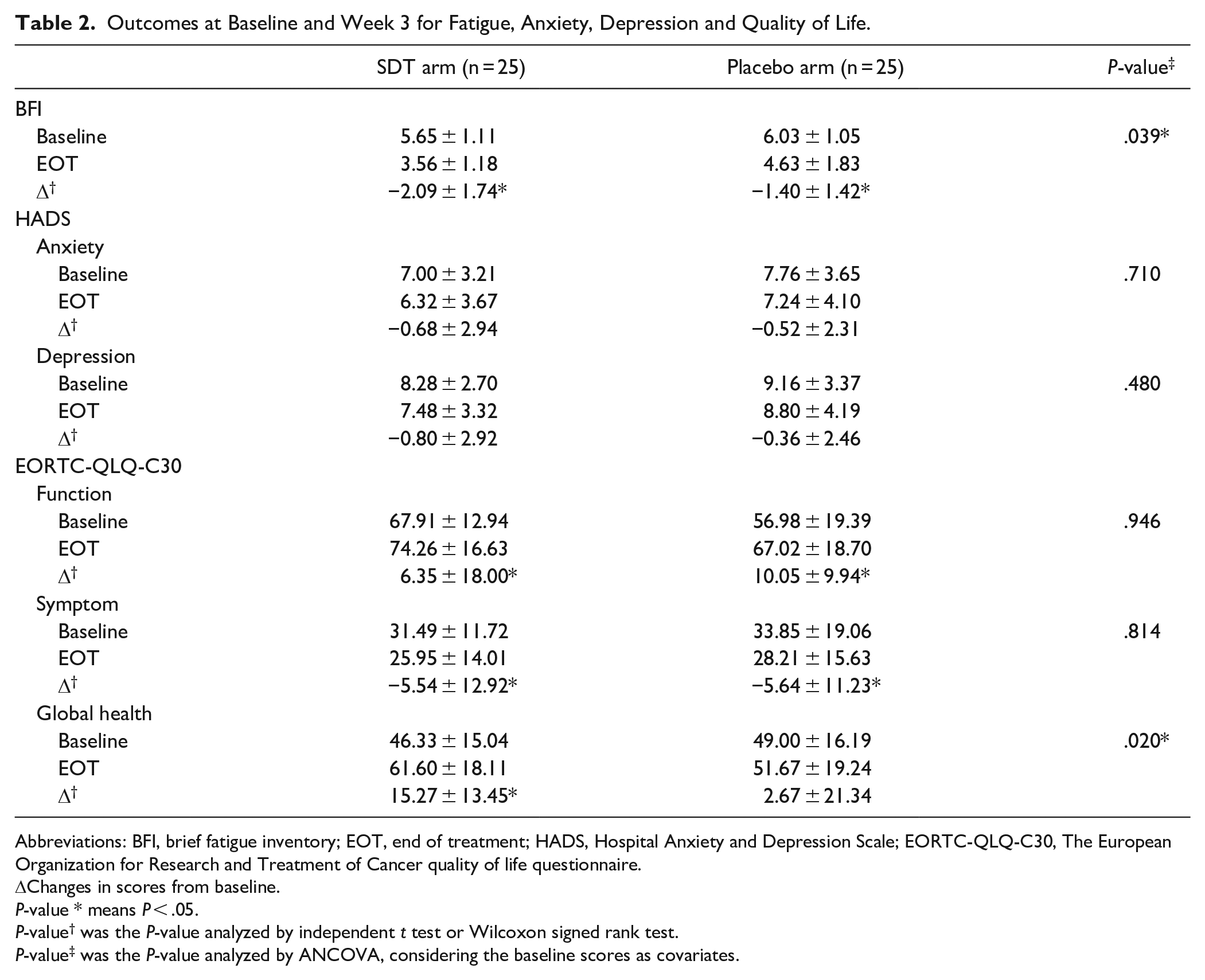
Abbreviations: BFI, brief fatigue inventory; EOT, end of treatment; HADS, Hospital Anxiety and Depression Scale; EORTC-QLQ-C30, The European Organization for Research and Treatment of Cancer quality of life questionnaire.
ΔChanges in scores from baseline.
P-value * means P < .05.
P-value† was the P-value analyzed by independent t test or Wilcoxon signed rank test.
P-value‡ was the P-value analyzed by ANCOVA, considering the baseline scores as covariates.
The fatigue level in both arms gradually decreased from the baseline and was at a minimum after week 3, and then rebounded after week 6 (Figure 2). After performing a repeated measures ANOVA, inter-group and inter-time differences of the BFI score from the baseline to week 6 showed significant differences (P = .007 and P < .001, respectively). However, the group-by-time interaction difference did not show a significant difference (P = .487).

Changes in brief fatigue inventory scores from baseline.
Lymphocyte subset and cytokine tests showed no significant differences in both arms compared with the baseline, and between the arms adjusted for baseline, as shown in Table 3.
Lymphocyte and Cytokine Levels at Baseline and After 3 weeks of Study.

Abbreviations: IL, interleukin; EOT, end of trial; TNF-a, tumor necrosis factor-alpha; CD, cluster of differentiation.
Δ Changes in scores from baseline.
P-value† was the P-value analyzed by independent t test or Wilcoxon signed rank test.
P-value‡ was the P-value analyzed by ANCOVA, considering the baseline scores as covariates.
Safety
No serious adverse events related to SDT or placebo administration occurred during the study period. All laboratory results, including liver and renal function, were normal after SDT administration. However, 1 participant in the SDT group experienced grade 1 dyspepsia, which subsided within 3 days without any rescue treatment. One participant in the placebo group complained of pruritus, assessed as grade 1 according to the CTCAE. Although this was the reason why the participant in the placebo group ended the clinical trial, the pruritus also subsided within 3 days.
Discussion
In this randomized, double-blinded, placebo-controlled, preliminary study, we found that SDT administration for 3 weeks improved CRF as measured by the BFI scale. Every participant in the SDT group ended the trial successfully without any serious adverse events.
Fatigue is the most common cancer-related symptom, and persistent fatigue can present in cancer survivors, patients with geriatric cancer, patients with cancer who received surgery or chemotherapy, and patients in an advanced stage, regardless of tumor type. 26 As cancer progresses, the incidence of CRF typically increases. At the end of life, it is estimated that 90% of patients with cancer experience fatigue. 27 As pharmacological interventions for CRF, erythropoietin and methylphenidate have shown some efficacy in fatigue reduction; however, concerns about the safety of these agents, including tumor progression, sleep disruption, and decreased appetite, might limit their usefulness.28-30
SDT has been used to improve the following conditions; poor physical strength, fatigue/malaise, loss of appetite, night sweats, coldness of hands and feet, and anemia. 12 SDT alleviated hematotoxicity in patients with breast cancer receiving chemotherapy without counteracting the effect of their treatment, 31 and improved the progression-free survival of patients with postoperative recurrence of non-small cell lung cancer by preventing nutritional deterioration induced by adverse effects of anti-cancer drugs. 12
Our results support the findings of another clinical study that showed SDT to be effective in improving CRF in patients with lung cancer after chemotherapy. 32 However, in contrast to this prior study, in which the control group was patients that received the usual care, our study was conducted as a placebo-controlled study with validated measures for CRF assessment.
Other prior studies showed that SDT augmented immune function by inducing natural killer cell activity, decreasing regulatory T cells, and enhancing quality of life.13-17 However, our results showed that SDT did not improve quality of life as measured by the EORTC QLQ-C30, except for the global health status scale, and did not affect lymphocyte or cytokine levels. The median duration from cancer treatment including surgery, chemotherapy, and radiotherapy in all participants in this study was 32.5 months, which implied that fatigue was not related to cancer treatment, and the participants had good performance status with ECOG 0-1. Future studies should address the potential effects of SDT on quality of life or immune function as primary outcome measurements.
Panax ginseng and Astragalus included in SDT are considered the main herbs for alleviating CRF. The systematic review and meta-analysis of randomized controlled trials indicate that ginseng is safe and efficacious in reducing fatigue33,34; however, the efficacy of ginseng against CRF is controversial and requires further research. 35 Moreover, Astragalus polysaccharide injections relieve CRF among patients with advanced cancer,36,37 and have been reported as a promising edible immunomodulatory herbal medicine. 38 Additionally, Astragalus-based herbal medicines combined with platinum-based chemotherapies may improve survival, increase tumor response, and reduce chemotherapy toxicity compared to platinum-based chemotherapies alone. 39
There are some limitations to this study. First, the enrolled patients had heterogeneity in their cancer type. Although CRF is a common cancer-related symptom regardless of cancer type, the refining criteria for eligibility should be addressed in future research. Second, the duration of SDT administration and the follow-up period in this study were short owing to concerns about low compliance.
In conclusion, we found that SDT can be an effective therapeutic option in reducing CRF in patients with cancer. Larger long-term efficacy and safety studies should be conducted in the future.
Footnotes
Authors’ Note
This trial was registered with the Clinical Research Information Service (CRIS), Republic of Korea, ID: KCT0003442.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (HF20C0038).
