Abstract
Introduction:
As pharmacological strategies remain limited for relieving fatigue and associated cognitive symptoms, integrative modalities such as traditional Chinese medicine (TCM) could be explored as therapeutic strategies in cancer survivors. Here, we evaluate and report the efficacy and safety of a TCM concoction, modified Xiang Bei Yang Rong Tang (XBYRT), on quality of life (QOL), cancer-related fatigue (CRF), and cognitive symptoms, compared to placebo.
Methods:
In a single-centered, randomized, double-blinded, placebo-controlled pilot trial conducted from 2019 to 2022, fatigued cancer survivors ≥21 years old were recruited to receive the XBYRT intervention or placebo (5% diluted) once daily for the duration of 8 weeks. Patient-reported outcomes for QOL, CRF, cognition, blood samples for biomarker testing, and adverse events were collected at baseline (T0), 4 weeks (T1), 8 weeks (T2), and 10 weeks (T3) after baseline. Linear regression was performed to evaluate differences between groups at T2 and T3.
Results:
A total of 1502 patients were screened, with 672 patients considered eligible. Of the eligible, 15 XBYRT and 13 placebo subjects with similar mean ages (58.5 vs 58.4) were recruited. Both groups were predominantly Chinese (93% vs 62%), breast cancer patients (87% vs 62%), and diagnosed with stage 2 cancer (60% vs 46%). Although no significant difference was found in QOL between groups, the XBYRT group exhibited improved emotional fatigue at T3 (
Conclusion:
In this pilot study, XBYRT as an integrative therapy is safe and generates encouraging improvements in cognitive and fatigue symptoms. Difficulties with recruitment limited the generalizability of trial findings, thus findings should be verified through a larger, multi-centered trial.
Keywords
Introduction
Cancer patients are surviving longer due to advances in diagnosis and treatment. However, they are now faced with wide-ranging debilitating toxicities as a consequence of these treatments, some lasting years after treatment cessation. Cancer-related fatigue (CRF), occurring in up to 85% of cancer survivors, 1 is characterized by a distressing, persistent, subjective sense of physical, emotional, and/or cognitive tiredness that negatively impacts quality of life (QOL). The lasting impact of CRF was evident in up to 52% of cancer survivors 3-years post diagnosis, 1 and 42% of working cancer survivors. 2 Despite these consequences, there is limited understanding of the underlying pathogenesis and pharmacological treatments remain investigational with limited efficacy.
Consequently, many cancer survivors seek complementary and alternative medicine to address general health and symptoms that persist despite conventional treatments, of which traditional Chinese medicine (TCM) is commonly used in Asia.3
-5 We have designed a concoction in collaboration with certified TCM practitioners, namely Xiang Bei Yang Rong Tang (香贝养荣汤, XBYRT), which contains 15 herbal components (Table 1) aimed at improving patients’ QOL and reducing CRF and associated symptoms. Notably, the concoction contains
Formula and Components of the Modified Xiang Bei Yang Rong Tang Decoction.

To evaluate the efficacy of XBYRT in cancer survivors, we have conducted a randomized controlled pilot trial on patients’ QOL and CRF, with the goal to evaluate its role as an integrative oncology modality. Additionally, to evaluate its impact on physiological function, symptoms and safety, we also aim to compare the perceived cognitive function, plasma biomarkers and safety outcomes to comprehensively quantify the impact of XBYRT on cancer survivors.
Methods
This manuscript was prepared following the CONSORT 2010 statement (Supplemental Table S1).
Trial Design
The study was a randomized, double-blinded, placebo-controlled, parallel trial, conducted from October 2019 to October 2022 (ClinicalTrials.gov: NCT04104113, September 26, 2019). The study received SingHealth Centralized Institutional Review Board (IRB) approval (CIRB No.: 2019/2135) and the protocol was previously published. 14
Participants
We recruited cancer survivors through oncologist referral at the National Cancer Centre Singapore (NCCS). Eligible participants were ≥21 years old, reported a fatigue screen score of ≥4 in the past 7 days (0, no fatigue; 10, worst fatigue) as recommended by ASCO cancer-related fatigue guidelines, 15 had completed surgery, chemotherapy, or radiotherapy and were not planned on receiving adjuvant therapy during the study period, except for aromatase inhibitors or ovarian suppression for breast cancer survivors. Patients with metastases, cancer recurrence, untreated fatigue-causing co-morbidities, on fatigue-inducing medications, taking warfarin, receiving or planning to receive TCM treatment, or breastfeeding or intending to conceive were excluded. Certified TCM physicians also screened the potential participants to ensure patient eligibility to receive the concoction based on the TCM syndrome differentiation. According to TCM principles, satisfaction of the TCM syndrome differentiation involves the deficiency of qi and blood by experiencing either: (1) two major symptoms with typical pulse and tongue conditions or (2) one major symptom and two possible symptoms with pulse and tongue conditions (Supplemental Table S2). The National Standard in People’s Republic of China and the Clinical Practice Guidelines of Chinese Medicine in Oncology currently endorses this method of condition identification. 16
Intervention
Study participants were randomized to either the XBYRT or placebo arms, and participants were given investigational products prepared as granules designed to be dissolved in hot water for once daily consumption for 8 weeks. In the XBYRT arm, participants received a 24 g daily dosage, selected based on the safety and efficacy guidelines from the
To advise on safe concomitant administration with medications, we had previously evaluated the potential interaction of XBYRT with the activities of CYP3A4 and CYP2D6 and found that the herbal components did not inhibit the CYP enzymes. Further, resulting liver cell viability demonstrated that XBYRT is not likely to cause hepatotoxicity. 13
Assessments
Before treatment initiation, participants’ demographics, cancer diagnosis, medical history, and concomitant medications were recorded. Questionnaires, blood draws and safety monitoring were completed at four time points: baseline (T0) and 4 weeks (T1), 8 weeks (T2), and 10 weeks (T3) after baseline. Pulse conditions were also assessed at four time points to ensure patient safety. Due to the COVID-19 pandemic, subsequent pulse assessments were conducted through teleconsultations (phone or video call) or to be omitted. Based on Singapore’s TCM Practitioners Board guidelines, while the first visit must be in person, follow ups can be virtual.
Quality of life (QOL)
Health-related QOL was measured using the global health status (GHS) domain of the EORTC Core Quality of Life questionnaire (EORTC QLQ-C30) questionnaire. A higher score indicated higher QOL.
Cancer-related fatigue (CRF)
The Multidimensional Fatigue Symptom Inventory-Short Form (MFSI-SF) measures CRF across five subscales: general, physical, emotional fatigue, and mental fatigue, and vigor. Except for the vigor subscale, higher scores in each domain represented more fatigue. An overall score was obtained by combining subscale scores (subtracting vigor scores), with higher scores indicating worse fatigue symptoms. We have previously performed a study to evaluate the psychometric properties and measurement equivalence of the English and Chinese versions of MFSI-SF in breast cancer and lymphoma patients in Singapore. 19
Perceived cognition
Perceived cognitive function was assessed using the FACT-Cog v3, which produced four subscales’ scores: perceived cognitive impairment (CogPCI), perceived cognitive abilities (CogPCA), impact of perceived cognitive impairment on QOL (CogQOL), and comment from others on cognitive function (CogOTH). Totaling the subscale scores generated the total score, with better perceived cognition indicated by higher scores. We have previously evaluated the psychometric property and measurement equivalence of the English and Chinese versions of FACT-Cog v3 among breast cancer patients in Singapore. 20
Safety monitoring
Safety was assessed through patient reports and blood tests evaluating toxicities to organ functions (eg, renal and liver function tests, full blood count, and electrolyte level). Adverse events (AEs) were recorded and graded by research nurses using the Common Terminology Criteria for Adverse Events (CTCAE) version 5.
Blood plasma processing and storage
A 9 mL blood sample was collected in ethylenediaminetetraacetic acid tubes. Subsequently, it was then centrifuged for 10 minutes at 1069 ×
Inflammatory cytokines
Plasma levels of interleukin (IL)-2, IL-4, IL-6, IL-8, IL-10, granulocyte-macrophage colony-stimulating factor (GM-CSF), interferon (IFN)-γ, and tumor necrosis factor (TNF)-α, were quantified using 50 µL of each sample with a highly sensitive multiplex immunoassay (Bioplex Human Cytokine 9-Plex Panel, Bio-Rad, USA).
Plasma brain-derived neurotrophic factor (BDNF) levels
We quantified BDNF levels with an enzyme-linked immunosorbent assay (ELISA) kit (Biosensis BEK-2211-1P/2P, Australia) using 100 µL of sample diluted 100-fold.
BDNF Val66Met genotyping (rs6265)
Genomic DNA was isolated from the buffy coat with QIAmp DNA Blood Mini Kit (Qiagen, Germany) and was polymerase chain reaction (PCR) amplified with forward primers (5′-GGACTCTGGAGAGCGTGAA-3′) and reverse primers (5′-CGTGTACAAGTCTGCGTCCT-3′). PCR products genotyping was completed by automated Sanger sequencing with a 3730xl DNA Analyzer (Applied Biosystems, USA).
Outcomes
Our primary endpoint was the difference of QOL scores between treatment and control groups, hence the primary analysis involved comparing the T2 and T3 GHS scores between the treatment and control groups. Secondary analyses included comparing MFSI-SF and FACT-Cog total and subscale scores, plasma BDNF and cytokine levels, at T2 and T3, as well as prevalence of AEs between the two arms. Exploratory outcomes included the association of the biomarkers with patient-reported outcomes (PROs).
Sample Size
To ensure an adequate sample size to inform a future phase III trial design, we aimed to recruit 80 subjects (40 per arm) as recommended by Teare et al. 21 after accounting for a 10% dropout rate.
Randomization
Block randomization with a block size of 10 was performed by a third-party clinical trial service provider using the sealed envelope method. The physicians, trial pharmacists, study team, and study participants were blinded to the block size, randomization sequence and treatment assignment.
Statistical Methods
Baseline characteristics were descriptively summarized using counts and percentages for categorical variables, and means and standard deviations (SD) for continuous variables. Differences in all outcomes were tested at T2 and T3 using linear regression with or without baseline adjustment. Further sensitivity analysis was performed for significant outcomes by adjusting for rs6265 genotypes which are known to influence patient outcomes. 22 All analyses were two-tailed, tested at 5% significance level, and conducted on R version 4.3.2.
Results
Participants
A total of 1502 cancer survivors were screened for eligibility between October 2019 to October 2022, of which 830 were ineligible as they were either not fatigued (n = 678) or met exclusion criteria (n = 152). Of the 672 that were eligible, 28 survivors entered the trial. The rest of the survivors did not enroll for the following reasons: not interested (n = 512), too busy or had procedure concerns (n = 87), were considering participation but did not get back to research personnel (n = 33), and other reasons (n = 12; Figure 1).
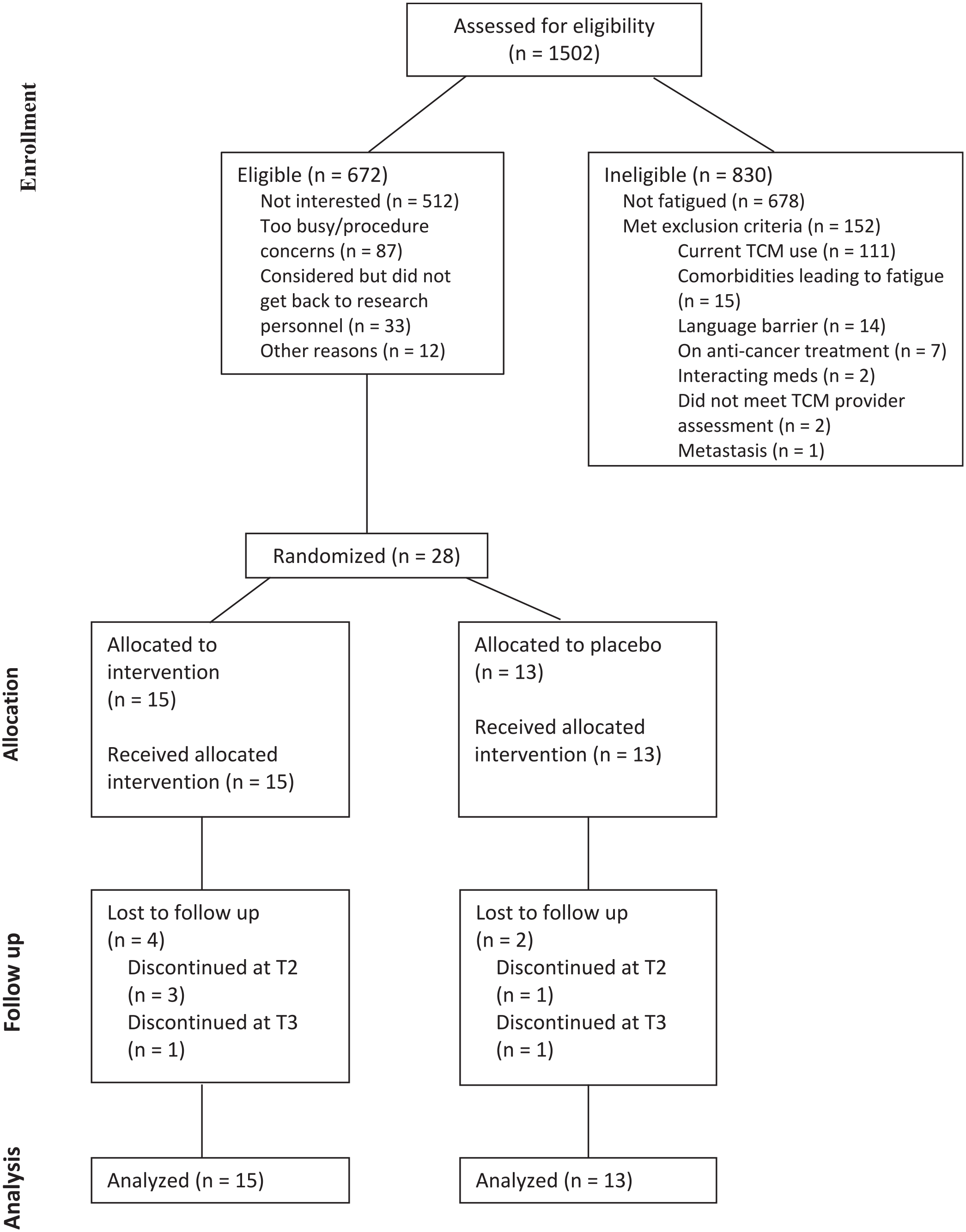
Subject CONSORT diagram.
Fifteen XBYRT and 13 placebo subjects with comparable mean ages (58.5 vs 58.4) were recruited (Table 2). Both groups mainly consisted of Chinese (93% vs 62%), breast cancer patients (87% vs 62%) and stage II cancer diagnoses (60% vs 46%). Surgery, chemotherapy, and radiotherapy were the most common treatment modalities in both groups. There was a lower proportion of rs6265 Met carriers among the XBYRT arm compared to placebo (47% vs 77%). We observed no difference between the two groups for all PROs and continuous biomarkers at baseline (all
Baseline Characteristics of XBYRT and Placebo Groups.

Abbreviations: BDNF, brain-derived neurotrophic factor; BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; EORTC QLQ-C30, European Organization for Research and Treatment of Cancer Quality of Life Questionnaire—C30; FACT-Cog, fatigue assessment of cancer therapy—cognitive function; GHS, global health score; GMCSF, granulocyte-macrophage colony-stimulating factor; IFNg, interferon gamma; IL, interleukin; MFSI-SF, multidimensional fatigue symptom inventory—short form; PCA, perceived cognitive abilities; PCI, perceived cognitive impairments; Q, quartile; QOL, quality of life; SD, standard deviation; SNP, single nucleotide polymorphism; T0, timepoint 0; T1, timepoint 1; T2, timepoint 2; T3, timepoint 3; TNFα, tumor necrosis factor alpha; XBYRT, Xiang Bei Yang Rong Tang.
QOL, CRF, and Perceived Cognition
Descriptively, a widening gap between the XBYRT and placebo groups was observed for all PRO (Figure 2 and Supplemental Table S3) over the course of the trial. However, we found no difference in GHS scores (primary endpoint) at T2 and T3 between the groups (Supplemental Tables S3 and S4).

Assessment and biomarker measurements across all timepoints between the XBYRT and placebo groups.
Regarding CRF, the XBYRT arm demonstrated better emotional fatigue symptoms at T3 (
Regarding perceived cognition, no difference was detected in the FACT-Cog subscales across all time points between the two groups. However, after adjusting for baseline, the XBYRT group exhibited better perceived cognitive function, represented by CogPCI, at T2 (
Biomarkers
The XBYRT group demonstrated significantly higher BDNF levels (in pg/mL) at T2 (
Sensitivity Analysis: Adjusting for rs6265 Genotypes
In a sensitivity analysis adjusting for rs6265 genotypes (Val/Val vs Met carriers), FACT-Cog CogPCI and total scores at T3 (after baseline adjustment), and BDNF levels at T2 and T3 remained significantly higher among XBYRT (
Safety
In the XBYRT arm, grade 3 adverse events were reported in three patients (20%): one experienced insomnia at T1; the second patient experienced insomnia and headache at T1; the third experienced headache at T2. In the placebo arm, two subjects (15%) experienced grade 3 adverse events: one experienced constipation at T1; the second patient experienced insomnia (T1 and T2), dry mouth (T2 and T3), and flushing (T2 and T3). Safety findings are summarized in Supplemental Table S7.
Exploratory Analysis: BDNF-PRO Spearman Correlation Analysis
Because XBYRT treatment appears to influence emotional fatigue, perceived cognition and BDNF levels, an exploratory Spearman correlation analysis was conducted to explore the relationship between BDNF and the various PROs. Within the XBYRT group, increase in BDNF levels is significantly associated with the increase in FACT-Cog CogPCI and total scores, from T0 to T3 (
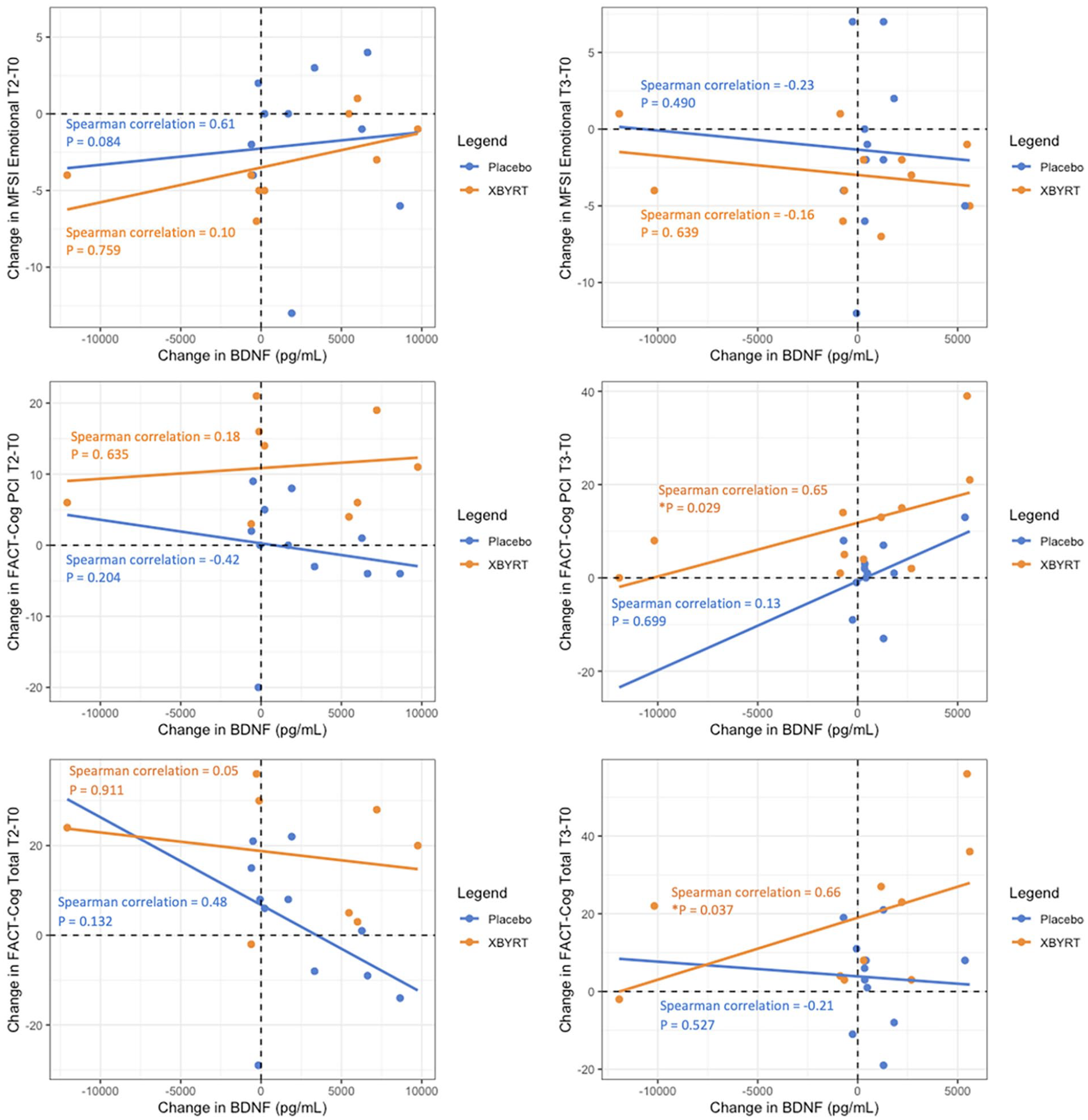
Spearman correlation coefficients for differences between T2 and baseline and T3 and baseline for PROs.
Discussion
In this randomized controlled pilot trial of herbal medicine for CRF, we found an improvement of fatigue and cognitive symptoms among those treated with XBYRT compared to placebo. We observed a significant sustained improvement after treatment for both emotional fatigue and perceived cognition. Our analysis of plasma biomarkers reported significant upregulation in BDNF levels after 8 weeks of XBYRT, although this increase was not sustained after treatment cessation. This increase in BDNF levels was associated with improved cognitive function after cessation of XBYRT therapy. We did not observe any significant safety concerns with the concoction. Taken together, we conclude that the XBYRT treatment demonstrated encouraging efficacy and safety data that warrant further investigation as a potential clinically feasible treatment for CRF and cancer-related cognitive impairment (CRCI).
According to theories of TCM, CRF is characterized both by a deficiency in qi, the body’s vital energy, and in blood.23,24 Chemotherapy and radiotherapy can result in impairment and consumption of Qi and blood, as these anti-cancer treatments suppress bone marrow, are cytotoxic, and result in fatigue, weakness, lack of energy or decline in physical functioning, causing the body to be in a deficiency state. It has been found that a qi deficiency correlated with CRF and worse QOL among cancer patients.
5
Although there was no impact in overall QOL in our study, we observed an improvement in emotional fatigue and perceived cognitive function in the intervention arm. These clinical outcomes are consistent with the therapeutic intent for individual ingredients of XBYRT, which includes
Interestingly, Met carriers of
There have been previous interventional studies which have evaluated various TCM concoctions containing multiple herbs in ameliorating CRF. For a study performed in Korea, 40 patients with CRF were randomized into either the control or intervention (Bu-Zhong-Yi-Qi-Tang [BZYQT]). The main ingredients of this decoction include
Considering the shortcomings of these prior studies, our trial has taken multiple approaches, including a placebo-controlled randomized design to evaluate the efficacy of our concoction. The adoption of this trial design to generate evidence demonstrating efficacy of XBYRT was a major strength of our study. To establish the role of TCM as a viable option for supportive care in cancer, the same standards imposed on pharmacological trials should also apply. Our study incorporated several unique and essential features that future trials should consider when evaluating herbal concoctions. Firstly, given the variation in composition of herbal concoctions, we have previously conducted toxicology studies of XBYRT to identify potential pharmacokinetic interactions and ensure patients who were on medications at risk of interactions with XBYRT were excluded from the study. 13 Additionally, an active placebo at 5% strength of XBYRT was used. As the taste of herbal concoctions is widely known to be strong and typically bitter, the use of an active placebo was critical to prevent participants from recognizing that they were on the placebo arm. Maintaining blinding of participants is especially crucial given the use of patient-reported tools in our study.
However, we encountered significant challenges with recruitment, achieving 35% of the recruitment target, due to a variety of reasons including COVID-19. Nearly half (45%) of the approached patients were deemed ineligible due to low or absent fatigue levels, 34% cited a lack of interest to participate due to concerns with TCM usage, and 7% were excluded due to current TCM use. To address similar recruitment challenges in future trials of TCM, the expected duration of recruitment should be adjusted based on the prevailing acceptability of TCM within the target population. Moreover, aligning trial recruitment with ongoing symptom monitoring activities presents a promising strategy to ensure that the trial can be suitably offered to patients with reported symptom(s) of interest, such as fatigue.
Conclusion
This randomized controlled pilot trial found XBYRT as a potentially safe integrative therapy that produced encouraging improvements in cognitive and fatigue symptoms. It is, however, necessary to keep in mind the potential challenge of applying standardized TCM decoctions to patients when designing studies in the future, as individualized decoctions may be more practical or optimal for treatment. To verify our findings, a larger, multi-centered trial will inform whether XBYRT is an appropriate intervention to manage and improve symptoms in cancer patients and survivors.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251314514 – Supplemental material for HEalth-Related Quality of Life-Intervention in Survivors of Breast and Other Cancers Experiencing Cancer-Related Fatigue and Associated Cognitive Symptoms Using TraditionAL Chinese Medicine: The ‘HERBAL’ Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354251314514 for HEalth-Related Quality of Life-Intervention in Survivors of Breast and Other Cancers Experiencing Cancer-Related Fatigue and Associated Cognitive Symptoms Using TraditionAL Chinese Medicine: The ‘HERBAL’ Trial by Alexandre Chan, Daniella Chan, Ding Quan Ng, Huang Fang Zheng, Quan Ming Tan, Chia Jie Tan, Jolene Hui Min Toh, Ning Yi Yap, Yi Long Toh, Yu Ke, Edmund Chun Ann Wang, Queenie Pei Ni Lim, Han Kiat Ho, Lita Chew and Tira J. Tan in Integrative Cancer Therapies
Footnotes
Acknowledgements
We thank the Singapore Ministry of Health for providing research funding for this study. In addition, we acknowledge Dr Raymond Ng, Dr Veronique Kak Mien Tan, Seng Kok Ricky Ang, Tze Kiat Tan, Yan Peng Leona Quek, Kok Keong Teo, Si Yu Ong, Koh Kong Tan and Wei Sheng Loo for their assistance with this study.
Authors Contributions
Conceptualization: TJT and AC. Methodology: TJT, HFZ, QMT, CJT, NYY, and AC. Investigation: TJT, HFZ, QMT, ECAW and QPNL. Acquired and analyzed data: DQN, DC, JHMT, and AC. Interpretated data: DQN, DC, and AC. Visualization: DC. Drafted the manuscript: DQN, DC, CJT, YK, and AC. Revised and approved final version of manuscript: All authors. Project administration: TJT, LC, and AC. Funding acquisition: TJT, HFZ, QMT, CJT, NYY, and AC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial is funded by the Singapore Ministry of Health Traditional Chinese Medicine Research Grant (TCMRG-3-NUS-01).
Ethical Approval
Institutional Review Board (IRB) approval: CIRB No.: 2019/2135.
Trial Registration
ClinicalTrials.gov (NCT04104113).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
