Abstract
Background:
A statistical model is essential in determining the appropriate predictive indicators for therapies in many types of cancers. Predictors have been compared favorably to the traditional systems for many cancers. Thus, this study has been proposed as a new standard approach. A recent study on the clinical efficacy of Atractylodes lancea (Thunb) DC. (AL) revealed the higher clinical benefits in patients with advanced-stage intrahepatic cholangiocarcinoma (ICC) treated with AL compared with standard supportive care. We investigated the relationships between clinical efficacy and pharmacokinetic parameters of serum bioactivity of AL and its active constituent atractylodin and determined therapeutic ranges.
Methods:
Group 1 of advanced-stage ICC patients received daily doses of 1000 mg of standardized extract of the capsule formulation of AL (CMC-AL) for 90 days. Group 2 received daily doses of 1000 mg of CMC-AL for 14 days, followed by 1500 mg for 14 days, and 2000 mg for 62 days. Group 3 (control group) received palliative care. Cox proportional hazard model and Receiver Operating Characteristic (ROC) were applied to determine the cut-off values of AUC0-inf, Cmax, and Cavg associated with therapeutic outcomes. Number needed to treat (NNT) and relative risk (RR) were also applied to determine potential predictors.
Results:
The AUC0-inf of total AL bioactivity of >96.71 µg hour/ml was identified as a promising predictor of disease prognosis, that is, progression-free survival (PFS) and disease control rate (DCR). Cmax of total AL bioactivity of >21.42 was identified as a predictor of the prognosis of survival. The therapeutic range of total AL bioactivity for PFS and DCR is 14.48 to 65.8 µg/ml, and for overall survival is 10.97 to 65.8 µg/ml.
Trial Registration:
TCTR20210129007 (TCTR: www.clinicaltrials.in.th).
Keywords
Background
Intrahepatic cholangiocarcinoma (ICC), a biliary tract cancer, accounts for 20% of primary liver cancers. 1 It is recognized as a deadly gastrointestinal cancer with a poor disease prognosis due to the lack of effective biomarkers for early diagnosis and effective treatment. With advanced disease, patients receiving first-line chemotherapy have a 5-year overall survival (OS) rate of lower than 20%. 1 High rate resistance of ICC to first-line chemotherapy results in disease progression among the treated patients. 2 Research and development of effective alternative therapies are needed to tackle this fatal disease and improve the quality of life in patients with advanced-stage ICC.
A recent phase 2A clinical study of capsule formulation of the standardized extract of Atractylodes lancea (Thunb) DC. (CMC-AL), in conjunction with standard supportive care, revealed the clinical benefits in the treatment of advanced-stage ICC without toxicities 3 by promoting peripheral immunostimulant cells and suppression of pro-inflammatory cytokines. 4 As an immunomodulator, AL, combined with other drugs, is a new paradigm for treating ICC in promoting the recruitment of immune cells in the tumor microenvironment5-10 based on the new classification of ICC therapy. A prognostic model has been used to identify the essential prognostic factors (ie, host and tumor factors) that play a critical role in patient management and decision-making. Besides the host factors and tumor factors, the success of therapy depends on the levels of plasma/serum drug concentration (a surrogate for drug concentration at the target site). The findings of essential prognostic factors and the therapeutic range of the study drug to improve clinical efficacy and safety before conducting the phase 2B clinical trial are needed. The present study aimed to determine (i) the prognostic factors for improving clinical efficacy (ie, disease control rate, tumor size progression, progression-free survival, and overall survival) following treatment with CMC-AL in patients with advanced-stage ICC, and (ii) the therapeutic ranges of CMC-AL.
Methods
Study Design
This retrospective study is part of a single-center, open-label, randomized controlled, phase 2A trial which was conducted at Sakhon Na-Kon Hospital, Sakhon Na-Kon province, Thailand (n = 48). 3 The clinical trial is registered at the Thai Clinical Trials Registry (No. TCTR20210129007) (https://www.thaiclinicaltrials.org; WHO ICTRP search). The study was approved by the Sakhon Na-Kon Hospital (No. 043/2020, 2 February 2021). The study was conducted in accordance with Good Clinical Practice (GCP) guidelines and the Declaration of Helsinki. 11
Patients with advanced-stage ICC were randomized to 3 groups. Groups 1 (n = 16) and 2 (n = 16) to receive CMC-AL with different dosage regimens in conjunction with standard supportive care, while group 3 patients (n = 16) received only standard supportive care. Block randomization was applied to patients for enrollment in each group. The total study period was 4 months. It is noted that this study is a phase 2A clinical trial, the lowest number of participants for a pharmacokinetic analysis for a model prediction is at least 12 individuals for the proof of concept.
Patients
Screening and recruitment of study patients were performed by an oncologist and the investigator team. Eligible patients were aged ≥18 years with advanced-stage (unresectable ICC or metastatic ICC), with Eastern Cooperative Oncology Group (ECOG) Performance Status of 0 to 2, serum carcinoembryonic antigen (CEA) of >5.2 ng/ml, tumor lesion size of >20 mm (at least 1 lesion), no history of treatment with chemotherapy or radiotherapy, normal cardiac function and electrocardiogram (ECGs), adequate bone marrow functions (polymorphonuclear cells of ≥1500 cells/mm3, platelets of ≥100 000 cells/mm3, hemoglobin of ≥8.0 g/dl, and normal blood coagulation or bleeding), normal liver functions (bilirubin of <1.5 of the upper limit of normal (ULN), alkaline phosphatase, alanine aminotransferase, or aspartate aminotransferase of <5 of ULN), and effective communication ability. Exclusion criteria were pregnancy or lactation, hypersensitivity or idiosyncratic reaction to herbal products or medicines, current or previous diagnosis of other cancers within 5 years, gastrointestinal abnormality, immune deficiency, and participation in other studies before study in the last 3 months. All patients provided informed consent before enrollment.
Treatment
Group 1: daily dose of 1000 mg CMC-AL (9 capsules) for 90 days, in conjunction with standard supportive care (n = 16). The standard supportive care consisted of the provision of general knowledge on cholangiocarcinoma, usage of necessary drugs, particularly analgesic drugs to relieve pain, assessment of quality of life, and follow-up of laboratory parameters related to cholangiocarcinoma.
Group 2: daily dose of 1000 mg CMC-AL (9 capsules) for 14 days, followed by 1500 mg (14 capsules) for 14 days, and 2000 mg (18 capsules) for 62 days, in conjunction with standard supportive care (n = 16).
Group 3: standard supportive care alone (n = 16).
A single CMC-AL capsule contains 2.45 and 4.06 mg atractylodin and β-eudesmol, respectively. The starting dose of CMC-AL of 1000 mg was about 50% of the maximum recommended safe dose (MRSD) extrapolated from previous animal studies, after proving the treatment safe in a Phase I clinical trial. 11 The drug was given in the morning, 2 hours before the meal.
Pharmacokinetic Study
The pharmacokinetic study was conducted in 32 patients. Blood samples (5 ml each) were collected from each patient at 0 (before dosing), 0.25, 0.5, 1, 1.5, 2, 2.5, 3, 4, 5, 6, and 8 hours on day 1 for group 1, and day 14 and day 28 for group 2.
Bioanalysis of Atractylodin and Serum Total Bioactivity of AL
HPLC-UV for determination of atractylodin
Atractylodin concentrations in plasma samples were determined using high performance liquid chromatography (HPLC) according to the previously reported method. 11
Bioassay for determination of total bioactivity of AL
The in vitro bioassay method for the determination of serum total AL bioactivity was according to the method of Cheoymang et al. 12 Total AL bioactivity was determined in human serum samples based on inhibitory effects on the growth of cholangiocarcinoma cell line (CL6) and Staphylococcus aureus (ATCC 25923), using MTT assay. The calibration curve (0, 0.39, 0.78, 1.56, 3.13, 2.56, and 50 ng/µl) was linear with correlation coefficients ≥.990. The quantification limit (LOQ) was 1.66 µg/ml using 20 µl serum sample.
Outcomes
The area under the curve (AUC0-t) (from zero to time t), AUC0-inf (from zero to infinity), maximum concentration (Cmax), average concentration (Cavg), volume of distribution (Vz/F), clearance (CL/F), terminal half-life (t1/2) of atractylodin and total AL bioactivity were calculated using non-compartmental analysis based on a linear trapezoidal rule (PKanalix version 2021R2, Antony, France: Lixoft SAS, 2021). Dose-dependent pharmacokinetic parameters were normalized with dose for evaluation of dose linearity. Data are reported as median (±ranges) values. Clinical efficacy at 4 months, including progression-free survival (PFS), overall survival (OS), and disease control rate (DCR) were assessed according to immune-related Response Evaluation Criteria in Solid tumor version. 13 The relationships between clinical responses and the pharmacokinetic parameters (AUC0-inf, Cmax, and Cavg) were determined.
Statistical Analysis
Comparisons of 2 independent and 2 dependent quantitative variables which were not normally distributed, were performed using Mann-Whitney U test and Wilcoxon Signed Rank test, respectively. Paired-t test and unpaired-t test were respectively applied for 2 dependent and 2 independent quantitative variables which were normally distributed. Chi-square analysis was applied for 2 independent qualitative variables. Relative risk (RR) and number needed to treat (NNT) were determined following chi-square analysis. Cox proportional hazard regression model was used in the multivariate analysis to identify the predictor of disease prognosis. Akaike Information Criterion (AIC) for the 4 selected model parameters (ie, AUC0-inf, Cmax, Cavg, and group), −2 Log-likelihood (−2LL) and Harrell’s C index were calculated for model performance and the Goodness of fit. Receiver Operating Characteristic (ROC) analysis based on the effect of the predictor of disease prognosis was applied to determine the cut-off values of the pharmacokinetic parameters. An area Under Curve (AUC) of ≥0.71 indicates more predictive accuracy. The sensitivity and specificity of each cut-off value were determined. RR, sensitivity, and specificity were reported as mean ± 95% confidence interval (CI). Statistical significance level was set at α = .05. Cross-validation was applied to assess the internal validity of the model. All computations and analyses were performed using GraphPad Prism version 9.5 for Windows (GraphPad Software, San Diego, California, USA, https://www.graphpad.com). Single imputation was applied for missing data.
Results
Patients’ Follow-Up
Thirteen patients in group 1 discontinued treatment due to adverse event (2 cases on days 28 and 42), disease progression (5 cases on days 43, 51, 54, 62, and 66), withdrawal of consent (1 case on day 56), and death (5 cases on days 40, 43, 52, 59, and 59). Nine patients in group 2 discontinued treatment due to adverse event (1 case on day 49), disease progression (4 cases on days 62, 64, 70, and 90), withdrawal of consent (1 case on day 80), and death (3 cases on days 59, 63, and 73). Fifteen patients in group 3 discontinued treatment due to withdrawal of consent (3 cases on day 30, 38, and 45), and death (12 cases on days 31, 31, 38, 39, 40, 42, 45, 47, 49, 50, 52, and 73).
Pharmacokinetic Study
Data from 12 patients in group 1, 15 patients in group 2 on day 14, and 12 patients in group 2 on day 28 were available for pharmacokinetic analysis (Figure 1). AUC0-inf, AUC0-6h, Cmax, Cavg, Vz/F, CL/F, and t1/2 of total AL bioactivity and atractylodin in all groups are summarized in Table 1.

Flow chart diagram of participants enrollment in the phase 2A study.
Comparisons of Pharmacokinetic Parameters Between Atractylodin and Total AL Bioactivity.

Data are presented as median (±ranges). Abbreviations: AUC0-inf, area under curve from 0 h to infinity; AUC0-6h, area under curve from 0 to 6 h; Cmax, maximum concentration; Cavg, average concentration; Df, degree of freedom; Vz/F, volume of distribution; CL/F, clearance; t1/2, Half-life; t, t-test; W, Wilcoxon-matched-pairs signed rank test (Sum of signed rank).
All pharmacokinetic parameters except t1/2 in all groups were significantly different between atractylodin and total AL bioactivity (Table 1). When the dose of CMC-AL was increased from 1000 to 1500 mg (group 1 day 1 vs group 2 day 14), AUC0-inf, Cmax, and Cavg of atractylodin were significantly different, while t1/2 remained unchanged. All dose-dependent and dose-independent pharmacokinetic parameters of total AL bioactivity were similar. When the dose of CMC-AL was further increased from 1500 to 2000 mg (group 2 day 14 vs day 28), the AUC0-inf, Cmax, and Cavg of both atractylodin and total AL bioactivity (normalized with dose) were similar (Table 2).
Statistical Analyses for Comparisons of the Pharmacokinetic Parameters of Atractylodin and Total AL Bioactivity Between (i) Group 1 on Day 1 and Group 2 on Day 14 (ii) Group 2 on Day 14 and Group 2 on Day 28.
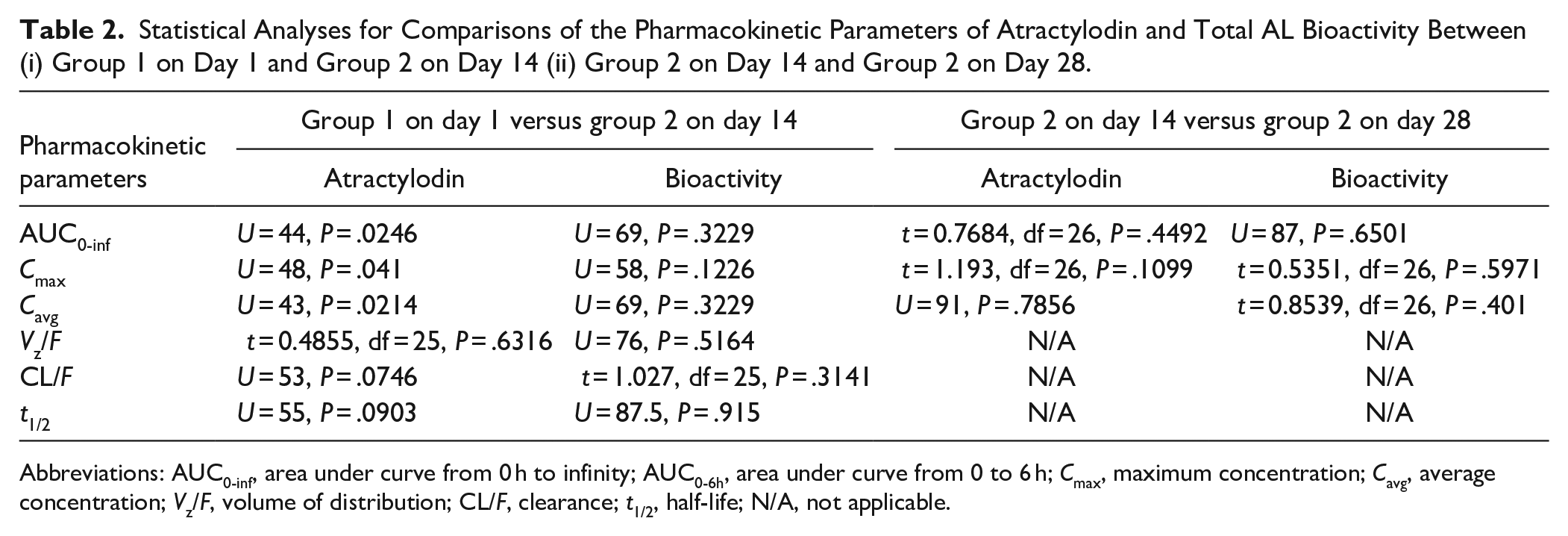
Abbreviations: AUC0-inf, area under curve from 0 h to infinity; AUC0-6h, area under curve from 0 to 6 h; Cmax, maximum concentration; Cavg, average concentration; Vz/F, volume of distribution; CL/F, clearance; t1/2, half-life; N/A, not applicable.
Efficacy
The DCR in groups 1, 2, and 3 were 5 (31.25%), 9 (56.25%), and 5 (31.23%), respectively. DCR was significantly higher in group 2 (high-dose AL) compared with group 1 (low-dose AL) [Odd ratio (OR): 0.35 (0.19-0.64, z = 3.566 (12.71, 1), P < .001) and group 3 [OR: 0.35 (0.19-0.64), z = 3.566 (12.71, 1), P < .001]. The PFSR in groups 1, 2, and 3 were 6 (37.5%), 8 (50%), and 4 (25%), respectively. PFSR was significantly higher in group 2 compared with group 3 (palliative care) [OR: 0.33 (0.18-0.59), z = 3.65 (13.33, 1), P < .001]. No significant differences were found for other comparisons as follows: group 1 versus group 2 [OR: 0.587 (0.34, 1), z = 1.85 (3.44, 1), P = .06], group 1 versus group 3 [OR: 0.57 (0.31-1.04), z = 1.84 (3.36,1), P = .07]. The OSR in groups 1, 2, and 3 were 10 (62.5%), 13 (81.25%), and 4 (25%), respectively. OSR was significantly higher in group 2 compared with group 3 [OR: 0.07 (0.018-0.47), z = 3.188 (10.16, 1), P = .001], but not group 1 [OR: 0.46 (0.1-2.65), z = 0.93 (0.86, 1), P = .35]. In addition, OSR was significantly higher in group 1 compared with group 3 [OR: 0.17 (0.04-0.89), z = 2.33 (5.43, 1), P = .02].
Identification of Predictors of Disease Prognosis
PFS
Multivariate cox analysis revealed no significant associations between ICC disease progression and the pharmacokinetic parameters of atractylodin or total AL bioactivity in all groups (Supplemental Table 1). When the data from total AL bioactivity in groups 1 (day 1) and 2 (day 28) were combined, however, significant differences in the AUC0-inf (t = 2.78, df = 20, P = .01), Cmax (t = 3.249, df = 20, P = .004), and Cavg (t = 2.761, df = 20, P = .01) were found between patients with the progressive and non-progressive disease. In addition, the dose-independent pharmacokinetic parameters—CL/F (U = 21, P = .016) was significantly higher in patients with progressive compared with non-progressive disease (1.18 L/h vs 0.72 L/h).
The ROC analysis of AUC revealed AUC0-inf and cut-off value of 0.81 (0.6-1.0, P = .02) and 96.71, respectively. The corresponding values for Cmax were 0.85 (0.69-1.00, P = .007) and 32.39, respectively, and for Cavg were 0.80 (0.59-1.00, P = .02) and 14.48, respectively. The sensitivity and specificity of AUC0-inf were 85.71 (60-97.46)% and 75 (40.93-95.56)%, respectively. The sensitivity [78.57 (52.41-92.43)%] and specificity [75 (40.93-95.56)%] of Cmax and Cavg were equal. The RR and NNT for AUC0-inf (z = 2.848, P = .004) were 3.43 (1.36-12.14) and 1.65 (1.18-8.74), respectively. Both values were similar for Cmax (z = 2.458, P = .01) and Cavg (z = 2.458, P = .01) [RR = 2.54 (1.18-7.15) and NNT = 1.95 (1.27-25.03)].
Inhibitory Activity on Tumor Size Progression
Multivariate cox analysis revealed no significant correlation between the increase in tumor size of ICC and the pharmacokinetic parameters of atractylodin or total AL bioactivity in all groups (Supplemental Table 2). The dose-dependent pharmacokinetic parameters (AUC0-inf, Cmax, and Cavg) of both atractylodin and total AL bioactivity were comparable in all groups (Supplemental Table 3).
DCR
Multivariate cox analysis revealed no significant associations between DCR and the pharmacokinetic parameters of atractylodin or total AL bioactivity in all groups (Supplemental Table 4). When the data from group 1 (day 1) and group 2 (day 28) were combined, however, the AUC0-inf (t = 3.12, df = 23, P = .005), Cmax (t = 3.506, df = 23, P = .002), and Cavg (t = 3.21, df = 23, P = .004) of total AL bioactivity were found to be a significant predictor of DCR between patients who did not respond and those who responded to treatment. Interestingly, CL/F (U = 21, P = .001) and Vz/F (U = 34, P = .018), but not t1/2, were significantly higher in responders compared with non-responders (CL/F 1.18 L/h vs 0.69 L/h and Vz/F 2.21 L vs 1.47 L).
The AUC of ROC analysis revealed AUC0-inf, Cmax, and Cavg of 0.83 (0.65-0.99, P = .006), 0.84 (0.71-1.00, P = .003), and 0.83 (0.65-0.998, P = .006), respectively. The cut-off values for AUC0-inf, Cmax, and Cavg were 96.71, 32.39, and 14.48, respectively. The sensitivity and specificity of AUC0-inf were 85.71 (60.06-97.46)% and 72.73 (43.44-90.25)%, respectively. Both parameters for Cmax and Cavg were equal to AUC0-inf. RR and NNT for AUC0-inf (z = 2.961, P = .003) were 4.0 (1.46-14.36) and 1.67 (1.22-7.05), respectively. The corresponding values for Cmax (z = 2.961, P = .003) were close to AUC0-inf, and that for Cavg (z = 2.565, P = .01) were 2.88 (1.25-8.72) and 1.95 (1.30-15.25), respectively.
OSR
Multivariate cox analysis revealed no significant associations between OS and the pharmacokinetic parameters of atractylodin or total AL bioactivity in all groups (Supplemental Table 5). When data from group 1 (day 1) and group 2 (day 28) were combined, significant differences in OS and the AUC0-inf (t = 2.361, df = 14, P = .033), Cmax (U = 10, P = .031), and Cavg (t = 2.372, df = 14, P = .033) of total AL bioactivity were found. In addition, CL/F was significantly higher (t = 2.176, df = 13, P = .04) in the deaths than in the survivors (1.22 L/h vs 0.84 L/h).
The AUC of ROC analysis revealed AUC0-inf, Cmax, and Cavg of 0.78 (0.55-1.00, P = .06), 0.83 (0.62-1.00, P = .03), and 0.82 (0.60-1.00, P = .04), respectively. The cut-off values for AUC0-inf, Cmax, and Cavg were 70.46, 21.42, and 10.97, respectively. The sensitivity and specificity for AUC0-inf were 83.33 (43.65-99.15)%, and 80 (49.02-96.45)%, respectively. All parameters were equal to AUC0-inf for Cmax and Cavg. The RR and NNT for AUC0-inf (z = 2.472, P = .01) were 6.43 (1.37-37.21) and 1.66 (1.16-24.15), respectively. The corresponding values for Cmax (z = 2.933, P = .003) and Cavg (z = 2.472, P = .01) were 8.33 (1.79-47.58), 1.36 (1.09-6.984), 6.45 (1.36-37.21), and 1.66 (1.16-24.15), respectively.
When the data from groups 1 (day 1) and 2 (day 28) were combined, the AUC0-inf and Cmax of atractylodin were not significantly different. Cavg, on the other hand, was considered to be a significant predictor of OS at 4 months (U = 10, P = .0312). The AUC of ROC analysis and cut-off value of atractylodin for Cavg were 0.83 (0.62-1.00, P = .03) and 22.10, respectively. The sensitivity and specificity were 83.33 (43.65-99.15)%, and 80 (49.02-96.45)% respectively. RR and NNT (z = 2.472, P = .01) were 6.45 (1.36-37.21) and 1.66 (1.16-24.15), respectively. CL/F was significantly lower (t = 2.257, df = 14, P = .04) in the survivors than in the deaths (302.45 L/h vs 433 L/h). AUC0-inf, Cmax, and Cavg of atractylodin or total AL bioactivity in other groups were comparable.
Cross-Validation
Since there were no significant associations between clinical responses (PFS, DCR, and OS) and the pharmacokinetics of total AL bioactivity (AUC0-inf, Cmax, and Cavg) in group 2 patients on day 14 (internal validation), cross-validation was applied for internal validity of the model to confirm that the prognostic factors can be applied to other scenarios.
Progressive Disease
The sensitivity and specificity for AUC0-inf were 66.67 (30-94.08)%, and 83.33 (43.65-99.15)%, respectively. The corresponding values for Cmax were 50 (18.76-81.24)%, and 83.33 (43.65-99.15)%, respectively. All values for Cavg were equal to Cmax.
DCR
The sensitivity and specificity for AUC0-inf were 66.67 (30-94.08)% and 85.71 (48.69-99.27)%, respectively. The corresponding values for Cmax were 50 (18.76-81.24)% and 85.71 (48.69-99.27)%, respectively. These values for Cavg were equal to Cmax.
OSR
There were 3 patients who survived at 4 months of treatment. The sensitivity and specificity for AUC0-inf were 100 (56.55-100)% and 0%, respectively. The corresponding values for Cmax were 100 (56.55-100)% and 33.33 (1.7-88.15)%, respectively. These values for Cavg were equal to Cmax.
Discussion
The prognosis models for the PFS, DCR, and OS associated with CMC-AL therapy in patients with advanced-stage ICC were built with cross-validation. The models were presented as independent predictors of disease prognosis with cut-off values. The therapeutic ranges of atractylodin and total AL bioactivity were also determined. The study was the first that applied total bioactivity, instead of a single active constituent, as a marker of pharmacokinetic profiles following treatment with herbal medicine. This approach is convincing as herbal medicine may consist of a mixture of various active constituents, some of which are unidentified.
Pharmacokinetic Study
The pharmacokinetics of atractylodin and total AL bioactivity on days 14 and 28 were similar, suggesting no accumulation of atractylodin and AL after daily dosing. This could be due to the short half-life of atractylodin and total AL bioactivity. More frequent dosing, that is, twice/thrice/fourth daily dosage regimens, would be a favorable choice to maintain plasma/tissue concentrations of AL at the target sites of AL action. Further phase 2B clinical trial is required to confirm the optimal dose regimens of CMC-AL in patients with advanced-stage ICC to avoid an excessive number of capsules administered for each dose. The pharmacokinetics of atractylodin following the once-daily dose of 1000 mg of CMC-AL observed in advanced-stage ICC patients was in agreement with the previous reports in the phase 1 study in healthy volunteers, 11 suggesting no influence of ICC disease on the pharmacokinetics of atractylodin. Therefore, the determined dosage regimen of CMC-AL in patients with advanced-stage ICC can be applied to all stages of the patients. Total AL bioactivity showed dose linearity over the dose range of 1000 to 2000 mg. On the other hand, atractylodin showed dose non-linearity over the range 1000 to 2000 mg. All the dose-independent pharmacokinetics of atractylodin and total AL bioactivity were similar over this dose range.
Predictors of Disease Prognosis
Progression-free survival (PFS)
Based on the ROC analysis for the PFS at 4 months, all of the dose-dependent pharmacokinetic parameters were promising predictors of disease progression (AUC ≥ 0.71), with high predictive accuracy. AUC0-inf provided the best sensitivity and specificity (AUC = 0.81). Although the AUC of Cmax was highest, the sensitivity and specificity of Cmax were lower than AUC0-inf. The AUC0-inf was, therefore, the preferable choice as a predictor of PFS at 4 months (Figure 2). The suggested AUC0-inf (cut-off) was >96.71 µg hour/ml to improve PFS in advanced-stage ICC patients with CMC-AL treatment. For cross-validation, the sensitivity of AUC0-inf was slightly decreased, while the specificity was increased, indicating high predictive accuracy. It was obvious that the sensitivity of Cmax and Cavg was decreased to 50%, while the specificity was increased to 83.33%. Cmax and Cavg could be optional predictors of ICC prognosis. In the previous reports, no association between Cmax of total AL bioactivity and the incidence of adverse reaction were found. 11 The therapeutic windows of total AL bioactivity for PFS range from 14.48 (cut-off value for Cavg) to 65.8 µg/ml. It was noted for the higher CL/F of total AL bioactivity in patients with disease progression than those without disease progression. Pharmacogenetics of drug-metabolizing enzymes and/or protein transporters may be involved. Further study focusing on genetic polymorphisms of drug-metabolizing enzymes and/or protein transporters would assist in determining the predefined criteria for CMC-AL treatment in ICC. Such high CL/F in patients with disease progression resulted in insufficient AUC0-inf, Cmax, and Cavg that could lead to treatment failure due to suboptimal drug concentrations in plasma and target tissues. Besides genetic polymorphisms, high CL/F in patients with progression disease may be due to induction of drug metabolizing enzymes during the on-going phase of disease progression. Significant influence of renal function is unlikely since there was no significant difference in estimated glomerular filtration rate (eGFR) between those with and without disease progression [11].The results from this study suggest that AUC0-inf is the best surrogate predictor of disease prognosis for disease progression in patients with advanced-stage ICC. Cmax and Cavg could be used as surrogates for treatment optimization. Patients with AUC0-inf of <96.71 µg hour/ml were associated with an increased risk of disease progression of up to 12.14-fold (RR: 3.43, 95%CI: 1.36-12.14) compared with those with AUC0-inf of >96.71 µg hour/ml. The risk of disease progression was also increased in patients with Cmax of <32.39 µg/ml and Cavg of <14. About 48 µg/ml, although the contribution was less pronounced when compared with AUC0-inf (RR: 2.54, 95%CI: 1.18-1.25). The NNT values (1.65, 1.95, 1.95 for AUC0-inf, Cmax, and Cavg, respectively) indicate a large effect size difference between the patients who have these pharmacokinetic parameters above or below the cut-off levels. This suggests that progression-free disease is expected to be observed in 1 of every 2 patients who received treatment with CMC-AL who have AUC0-inf, Cmax, and Cavg above the cut-off values; in other words, there is a 50% chance that those having AUC0-inf, Cmax, and Cavg above the cut-off point will prevent the disease progression. CMC-AL showed potential anti-CCA activity in reducing the risk of disease progression in advanced-stage ICC patients. First-line therapy with conventional drugs (gemcitabine/cisplatin combination) was reported to reduce the risk of disease progression by up to 1.88-fold (1.56, 95%CI: 1.31-1.88) compared with gemcitabine alone in patients with advanced-stage CCA. 14 Targeted therapy with ivosidenib (an isocitrate dehydrogenase-1 inhibitor) on the other hand, reduced the risk of disease progression by 4-fold (2.7, 95%CI: 1.85-4) compared with placebo. 15 A study of regorafenib in patients who failed gemcitabine/platinum-based therapy showed that the risk of disease progression was reduced by up to 3.44-fold (2.04, 95%CI: 1.23-3.44) compared with placebo. 16 In addition to targeted therapy, a recent study of immunotherapy reported that patients treated with durvalumab in combination with gemcitabine in patients with advanced-stage CCA had a significant decrease in the risk of disease progression by up to 1.59-fold (1.33, 95%CI: 1.12-1.59) compared with placebo. 17

A prognostic factor of progression free survival (PFS) and cut-off values.
DCR
Similarly to PFS, AUC0-inf was the best predictor of disease prognosis associated with DCR, although sensitivity and specificity were equal to Cmax and Cavg. Based on ROC analysis, the AUC of AUC0-inf (0.81) was lower than Cmax (0.84), but was still considered highly accurate (Figure 3). However, the sensitivity (66.67%) and specificity (85.71%) of the validated AUC0-inf was the highest. The sensitivity of Cmax and Cavg was on the other hand, was dramatically decreased to 50%. Furthermore, the RR for AUC0-inf was up to 14-fold (4, 95%CI: 1.46-14.36) to increase DCR, while the RR for Cavg was up to 8.72-fold (2.88, 95%CI: 1.25-8.72). Hence, the increased in DCR for AUC0-inf was 1.64-fold higher than Cavg. The NNT of AUC0-inf was lower than Cavg, (1.67 vs 1.95). The NNT for the success of treatment based on the prognostic predictor of AUC0-inf was also only 2, which indicates a large effect size difference between the 2 groups. Although Cavg may not be a favorable predictor of DCR, it could be used to identify therapeutic window of total AL bioactivity (14.48-65.8 µg/ml) (Figure 4).

A prognostic factor of DCR and cut-off values.

Therapeutic ranges of progression free survival (PFS) and disease control rate (DCR).
Similarly to PFS, CL/F, and Vz/F were significantly different between the responders and non-responders based on the DCR criteria. Apart from the contribution of pharmacogenetic factors that resulted in accelerated CL/F, physiologic factors such as the presence of ascites or obesity may have resulted in the expansion of the volume of distribution and thus, suboptimal systemic drug exposure. Patients with ascites or obesity may therefore, require a high dose regimen of CMC-AL.
OS
In contrast to PFS and DCR, the most promising predictor of prognosis for OS was Cmax (Figure 5). This was supported by the ROC analysis (AUC > 0.71), sensitivity, and specificity. In addition, the sensitivity and specificity obtained from the cross-validation of Cmax was higher than AUC0-inf. Alternatively, Cavg could be applied. With the highest RR, patients with Cmax of >21.42 µg/ml had an increased OS by up to 47.58-fold (8.33, 95%CI: 1.79-47.58) compared with those with Cmax of <21.42 µg/ml. Patients with AUC0-inf of >70.86 µg hour/ml, on the other hand, had an increased OS by up to 37.21 (6.43, 95%CI: 1.37-37.21) compared with those with AUC0-inf of <70.86 µg hour/ml. In addition, the NNT for Cmax was lower than AUC0-inf (1.36 vs 1.66). In case where the measurement of total AL bioactivity was not available, Cavg of atractylodin would be an optional choice as it provided comparable AUC to Cmax of total AL bioactivity, although the RR and NNT values were relatively lower (only up to 37.25 and 1.65, respectively). Interestingly, the cut-off value for OS was lower than PFS and DCR, indicating the requirement of lower drug concentrations to obtain desirable therapeutic outcome. Thus, the therapeutic window of total AL bioactivity for OS ranged from 10.97 (cut-off value of Cavg) to 65.8 µg/ml (Figure 6). CL/F was also higher in the non-survivors (deaths) compared with survivors, indicating the significant contribution of drug clearance in the non-survivors. In-depth analysis of the association between drug clearance in non-survivors is necessary to enhance the clinical efficacy of CMC-AL therapy in patients with advanced-stage ICC.

A prognostic factor of OS and cut-off values.

Therapeutic ranges of OS.
It was evident for the potential of CMC-AL to increase the OS in ICC patients with advanced-stage ICC. Gemcitabine/cisplatin combination therapy was shown to reduce the risk of death by up to 1.92-fold (1.56, 95%CI: 1.25-1.92) (HR: 0.64; 95%CI: 0.52-0.80, P < .001). 18 The second-line therapy capecitabine on the other did not reduce the risk of death (hazard ratio (HR): 0.81, 95%CI: 0.63-1.04, P > .05) in patients with advanced-stage CCA. 19 The FOLFOX regimen containing folinic acid, fluorouracil, and oxaliplatin, on the other hand, was reported to reduce the risk of death by up to 2-fold (1.44, 95%CI: 1.03-2) compared with control. 20 Patients with immunotherapy—durvalumab in combination with gemcitabine was reported to increase the survival rate of patients with advanced-stage CCA by up to 1.5-fold (1.25, 95%CI: 1.03-1.5). 17
Conclusions
The predictors of ICC disease prognosis (ie, AUC0-inf, Cmax, and Cavg) were successfully established with different cut-off values to improve PFS, DCR, and OS in patients with advanced-stage ICC who were treated with CMC-AL. In addition, the therapeutic ranges of total AL bioactivity were determined. Notably, the study highlights the importance of the measurement of pharmacokinetic parameters of total AL bioactivity since these parameters were clinically correlated with clinical outcomes rather than a single active constituent—atractylodin. This approach should be applied to other herbal medicines to determine the pharmacokinetic-pharmacodynamic relationship and therapeutic window. The limitation of the study is the low number of participants recruited in each group, as well as the lack of external validation. Therefore, the prediction of ICC disease prognosis using the proposed surrogates may not be applied to all scenarios. In addition, the small sample size resulted in lower sensitivity and specificity in the cross-validation, which may have had a direct effect on model accuracy. Further study for an external validation with larger sample size is needed to confirm the model accuracy and applicability.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231223967 – Supplemental material for Pharmacokinetic Analysis of Prognostic Factors in Patients With Advanced-Stage Intrahepatic Cholangiocarcinoma Following the Administration of Capsule Formulation of the Standardized Extract of Atractylodes lancea (Thunb) DC
Supplemental material, sj-docx-1-ict-10.1177_15347354231223967 for Pharmacokinetic Analysis of Prognostic Factors in Patients With Advanced-Stage Intrahepatic Cholangiocarcinoma Following the Administration of Capsule Formulation of the Standardized Extract of Atractylodes lancea (Thunb) DC by Teerachat Saeheng, Juntra Karbwang, Anurak Cheomung, Nisit Tongsiri, Tullayakorn Plengsuriyakarn and Kesara Na-Bangchang in Integrative Cancer Therapies
Footnotes
Acknowledgements
Not applicable.
List of Abbreviations
AIC Akaike Information Criterion
AL Atractylodes lancea Thunb (DC)
ALP Alkaline phosphatase
AST Alanine aminotransferase
ATD Atractylodin
AUC Area Under Curve
Cavg Average concentration
Cmax Maximum concentration
CMC-AL Capsule formulation of standardized extract of Atractylodes lancea Thunb (DC)
CL/F Clearance
DCR Disease control rate
ECOG Eastern Cooperative Oncology Group
ECGs Electro cardiograms
HR Hazard ratio
ICC Intrahepatic cholangiocarcinoma
−2LL −2 Log-likelihood
NNT Number-need to treated.
OS Overall survival
PFS Progression-free survival
ROC Receive Operating Curve
RR Relative risk
T1/2 Terminal half-life
Vz/F Volume of distribution
Author Contributions
Conceptualization: T.S., J.K., and K.N.; Data curation: T.S.; Formal analysis: T.S.; Investigation: A.C., T.P. Methodology: T.S.; Supervision: J.K., K.N.; Validation: T.S.; Visualization: T.S.; Writing-original draft: T.S.; Writing-review & editing: A.C., T.P., J.K., K.N.
Availability of Data and Materials
The authors confirm that the data supporting the findings of this study are available within the article and/or its Supplementary Materials and methods. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Thammasat Postdoctoral Fellowship, the Thailand Science Research and Innovation Fundamental Fund. Also, it received funding from Thammasat University under the project Center of Excellence in Pharmacology, and Molecular Biology of Malaria, and Cholangiocarcinoma (No. 1/2556, dated 12 October 2013), and the National Research Council of Thailand (No. 45/2561, dated 10 September 2018). KN is supported by the National Research Council of Thailand under the Research Team Promotion grant. All funders have no roles for publication.
Ethic Approval and Consent to Participate
This project received approval from the Sakhon Na-Kon Hospital (No. 043/2020, 2 February 2021). All participants signed written informed consent forms prior to enrollment.
Consent to Participate
The consent to participate was waived due to the retrospective nature of the study.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
