Abstract
Background:
Cancer cachexia is a common but severe condition that causes muscle wasting, body weight loss, and progressive functional impairment, affecting over 50% of cancer patients. Currently, there are no effective treatments that can alleviate cachexia, and hence the discovery of new therapeutics that can effectively prevent or even reverse cancer cachexia is crucial. Babao Dan (BBD) is a Traditional Chinese Medicine (TCM) formula that has been used clinically in combating various cancers, however, its therapeutic potential in alleviating cancer cachexia remains unexplored. Our current study aims to determine the anti-cachectic effects of BBD treatment in alleviating cancer cachexia, as well as determining the underlying mechanisms involved.
Methods:
Mouse models of cancer cachexia were induced via implantation of CT26 colon adenocarcinoma cells, and the anti-cachectic effects and mechanisms of BBD were determined via examinations of body weight and muscle mass, as well as serum and muscle markers of cachexia and muscle atrophy.
Results:
CT26 tumor implantation reduced in the rapid occurrence of cancer cachexia characterized by marked reductions in body weight and muscle mass, functional decrease in muscle function and accelerated deaths. BBD administration not only demonstrated robust anti-cachectic ability via preventing decreases in body weight, muscle mass, and muscle atrophy, but also markedly prolonged survival. The effects of BBD in alleviating cancer cachexia and its associated adverse effects were due to its ability in preventing the activation of IL-6/STAT3 signaling post-CT26 tumor implantation.
Conclusion:
Our findings demonstrated the robust ability of BBD in preventing cancer cachexia and alleviating the main cachexia-induced symptoms as well as prolonging survival via inhibiting activation of IL-6/STAT3 signaling pathway. Therefore, our study demonstrating the strong anti-cachectic effects of BBD in mice may provide a theoretical basis for the use of BBD as a safe and effective drug in the treatment of cancer cachexia.
Introduction
Cancer remains the second most common cause of deaths, and an estimated 600 000 people will die of cancer in the United States in 2022. 1 The most common and grave condition accompanying cancer is the occurrence of cachexia, which causes progressive functional impairment, loss of body weight and decreased muscle mass, affecting approximately 50% to 80% of all cancer patients. 2 Patients with cachexia often have decreased sensitivity to anti-cancer therapeutics and more rapidly succumb to death resulting from respiratory and cardiac failure due to a weakened diaphragm and cardiac muscles. 3 However, currently there are still no effective therapeutic drugs for the prevention or treatment of cancer cachexia, and hence the provision of adequate nutritional support remains the mainstay of cancer cachexia treatment.
Cancer cachexia occurrence involves the action of diverse mediators derived from cancer cells and cells within the tumor microenvironment, including inflammatory and immune cells. 4 Numerous studies have demonstrated that the cytokine interleukin-6 (IL-6) plays a key role in the progression of cancer cachexia.5-9 Elevated levels of serum IL-6 levels have been shown to induce muscle and fat depletion, resulting in accelerated death in experimental models of cancer cachexia, whereas decreasing expression of IL-6 slows the progression of cachexia. 6
Murine colon adenocarcinoma CT26 cell line is widely used for cachexia studies in mice, resulting in distinct characteristics of cancer cachexia upon implantation, including reductions in body weight, muscle weight, and fat mass, as well as increased IL-6 expression.10,11 Skeletal muscle atrophy is one of the main features of cancer cachexia, and studies have shown that IL-6 induces STAT3 signaling pathway activation and promotes cachexia progression and skeletal muscle atrophy.5,12 The loss of skeletal muscle mass is the result of an imbalance between protein synthesis and degradation, which is mainly regulated by the lysosomal and ubiquitin-proteasome system (UPS) pathways.13,14 The most well-known targets of muscle atrophy are muscle RING-finger protein-1 (MuRF1) and muscle atrophy Fbox protein (Atrogin-1), belonging to the family of muscle-specific E3 ubiquitin ligases, which are highly expressed in disease states such as cancer cachexia and heart failure, causing muscle protein degradation via the proteasome.15,16 Myostatin is a member of the transforming growth factor β (TGF-β) family produced by skeletal muscle, and its overexpression leads to loss of skeletal muscle via inhibiting muscle synthesis. 17 Mechanistic studies have demonstrated that the activation of STAT3 signaling allows for the increased transcription of these 3 key genes encoding E3 ubiquitin ligases, MuRF1, Atrogin-1, and Myostatin. 18 Therefore, drugs that can directly target IL-6/STAT3 signaling pathway and inhibit the activation of its downstream muscle atrophy targets may have potential for the prevention and treatment of cancer cachexia.
Babao Dan (BBD) is a Traditional Chinese Medicine (TCM) formulation that has been approved by the China Food and Drug Administration (CFDA) as a TCM alternative-based treatment for various cancers due to its anti-inflammatory and anti-tumor effects.19,20 However, the anti-cachectic potential of BBD remains entirely unexplored. Due to the key involvement of IL-6/STAT3 signaling in cancer cachexia occurrence and progression, we hypothesized that the therapeutic anti-cancer effects of BBD may be due to the regulation of IL-6/STAT3 signaling pathway, and furthermore, that BBD may be an effective drug for the alleviation of cancer cachexia.
Materials and methods
Preparation of Babao Dan (BBD)
BBD (Batch number: 190530) was obtained and authenticated by Xiamen Traditional Chinese Medicine Factory (Xiamen, China), and manufactured via special processes (Patent No. CN106483260 A). The components of Babao Dan consisted of: Panax notoginseng (Burk.) F.H. Chen., Forest Musk Abelmosk (Abelmoschus moschatus (Medicus)), Pearl (Pteria martensii (Dunker)), Antelope horn (Cornu Saigae Tataricae), Cultivated Bezoar (Bos taurus domesticus Gmelin), and Snake gallbladder of families (Elapidae, Colubridae, and Viperidae). The ratio of ingredients in the multiherbal preparation is patented by Xiamen Traditional Chinese Medicine Factory (Xiamen, China), and has not been publicly revealed. Stock solutions of BBD were prepared prior to use by dissolving grounded BBD powder (nominal mesh aperture of 180 μm) in PBS at a concentration of 24 mg/mL, filtered through a 0.22 μm filter and stored at −20°C for further use.
Component Analysis of BBD Using HPLC-MS/MS
HPLC-MS/MS analysis of Babao Dan extract including the supplier, contents, extraction and preparation procedures were detailed in our previous study. 20 Briefly, 0.3 g of BBD powder extract (1 mg/mL) and standards were respectively injected into the HPLC-MS/MS system (LC-30A, Shimadzu, Japan) and separated on a C18 ODS column (1.8 μm, 2.1 mm × 100 mm) with gradient elution. About 0.2% 2-sulfobenzoic acid hydrate (A) and acetonitrile (B) was used as mobile phase and the gradient elution procedure was as follows: 0 minute, A:B = 97:5; 0.01 minutes, A:B = 75:30; 37 minutes, A:B = 95:5; 37.1 minutes. The flow rate was 0.5 mL/min.
Animal Experimentation
All animal protocols were approved by the Animal Care and Use Committee of Fujian University of Traditional Chinese Medicine, Fujian, China (Approval No. FJTCM IACUC, 2020041), and performed according to the ARRIVE guidelines. A total of 40 healthy male Balb/c mice were purchased from Shanghai SLAC Laboratory Animal Company (Shanghai, China). Mouse models of tumor implantation were performed in 10 to 12-week-old mice, including: (1) survival experiment (n = 22) up to day 53 post-implantation; (2) mechanistic analysis of BBD (n = 18), mice sacrificed at day 36 post-implantation. Murine CT26 colon cancer-cell line was purchased from the Shanghai Cell Bank of the Chinese Academy of Sciences (Shanghai, China). CT26 cells were washed twice with PBS and resuspended in saline, then CT26 cells (2 × 107 cells/mL) were injected into the right axillary subcutis of Balb/c mice. After injection, the mice were randomly divided into three groups: control group without tumor implantation, model + PBS group, model + BBD (0.12 g/kg) group. Mice were given 0.12 g/kg of BBD orally every second day for 53 days starting 2 days after tumor implantation. The control group and tumor group mice were administered with the same volume of PBS. BBD optimal dosage was selected according to a previous study. 20 To study the potential anti-tumor mechanisms of BBD, a separate experiment was performed and mice were sacrificed at 36 days post implantation of CT26 cells. The body weight and grip strength were measured on the indicated day.
Cell Culture
Mouse colon adenocarcinoma CT26 cells were cultured in RPMI-1640 medium (Thermo Fisher Scientific, USA) with l-glutamine supplemented with 10% FBS (Gibco, MA, USA) and penicillin-streptomycin (100 µg/mL) in a 37°C humidified incubator with 5% CO2.
Grip Strength
Forelimb strength was assessed by grip strength meter (mouse gripper type KW-ZL). Forelimb grip strength was tested weekly after tumor implantation. Briefly, mice were acclimated in the test chamber for at least 10 minutes, then allowed to grasp a rectangular metal grid attached to a force transducer on their forelimbs. Mice were lifted by their hind limbs and gently pulled by their tails (avoiding hind limb contact with the gripper), and the peak grip strength generated was recorded by the transducer. The test was performed 8 times in 3-minute period and the average value was calculated.
ELISA Measurement of Serum IL-6
Briefly, mouse serum was collected at day 36 post-tumor implantation and centrifuged at 3000× rpm for 10 minutes. Serum IL-6 level was measured using ELISA according to the manufacturer’s instructions (Meimian Biotechnology, MM-0163M1, China).
Histological Examination
Quadriceps muscles were fixed with 4% paraformaldehyde overnight, paraffin-embedded, and sectioned (6 µm). The myofiber sizes were measured in sections stained with hematoxylin and eosin (H&E) using standard techniques. Mean myofiber size was determined by measuring the cross-sectional areas of at least 50 individual muscle fibers/mice from the same cross-sectional plane, expressed in units of µm 2 .
Western Blot Analysis
Total muscle proteins were extracted with RIPA Lysis buffer containing phosphatase/protease inhibitors, and WB was performed according to standard protocol. Briefly, proteins were separated on 10% SDS-PAGE gels and transferred to 0.22 μm PVDF membranes, blocked with 5% non-fat milk for 1 hour and washed with TBST. The membranes were then incubated with primary antibodies overnight at 4°C. Subsequently, membranes were incubated with the corresponding secondary antibodies for 2 hours at room temperature, then the membranes were washed in TBS-T buffer for 30 minutes and the resulting protein bands were detected by chemiluminescence. The intensity of the bands was quantified using ImageJ software via the mean gray value parameter, normalized to the loading standard. Antibodies for MuRF1 (ab77577), Myostatin (ab71808), Atrogin-1 (ab168372), were obtained from Abcam (USA). Antibodies for GAPDH (60004-1-Ig), STAT3 (60199-1-Ig) were obtained from Proteintech (USA). Antibody for p-STAT3 (Tyr705) (#9145) was obtained from Cell Signaling Technology (USA). Antibodies for Goat anti-Mouse-IgG (L3032), Goat anti-Rabbit-IgG (L3012) were obtained from Signalway Antibody (USA).
Statistical Analysis
Data were expressed as the mean ± standard deviation (SD). All statistical analyses were performed using SPSS 23.0 software. One-way ANOVA followed by LSD multiple comparisons tests were used to analyze data. P < .05 was considered statistically significant.
Results
HPLC Analysis of BBD Extract
The peak retention times of BBD extract were determined by HPLC analysis and quantified using the calibration curves of the corresponding chemical standards. The major components contained in the BBD extract were determined to be Ginsenoside Rk3 (main peak at 10.48 minutes), Lupenone (main peak at 14.40 minutes), Ginsenoside F4 (main peak at 16.23 minutes), Diosgenin (main peak at 27.27 minutes), Notoginsenoside R2 (main peak at 14.24 minutes), and Isofraxidin (main peak at 7.41 minutes), as illustrated in Figure 1.

HPLC profiles of the top bioactive compounds contained within BBD extract. Chromatograms corresponding to Ginsenoside Rk3 (orange), Lupenone (gray), Ginsenoside F4 (yellow), Diosgenin (blue), Notoginsenoside R2 (green), and Isofraxidin (pink), as shown according to their intensity peaks at different retention times.
BBD Prevents Cancer Cachexia and Prolongs Survival in CT26 Tumor-Bearing Mice
We first evaluated the anti-cachexia potential of BBD in a murine model of colon cancer using CT26 colorectal adenocarcinoma. There was a significant reduction in tumor-free body weight in CT26 tumor-bearing mice from day 20 following tumor implantation (Figure 2A) compared to the control group without tumor implantation (CTR). Surprisingly, mice administered with BBD markedly attenuated this decrease in tumor-free body weight, with a distinctively decreased trend of body weight loss from day 20 following CT26 tumor implantation (Figure 2A). Upon sacrifice on day 36 post-tumor implantation, tumor-free body weight of mice treated with BBD treatment was significantly greater than CT26 tumor-bearing mice treated with an equivalent volume of PBS (Figure 2B). We further performed an additional model to examine whether BBD can also enhance survival following CT26 tumor implantation. Kaplan-Meier analysis showed that while mice implanted with CT26 tumors all perished by day 53 following tumor implantation, mice that were administered with BBD starting from day 2 post-implantation, had significantly prolonged life span and actually had zero deaths by day 53 post-tumor implantation (Figure 2C). These results demonstrate the robust effect of BBD in not only preventing cancer cachexia occurrence, but also prolonging survival following CT26 tumor implantation.

BBD prevents cancer cachexia and prolongs survival in CT26 tumor-bearing mice. (A) Changes in tumor-free weight over time following CT26 tumor implantation. (B) Tumor-free body weight at time of sacrifice on day 36 post-tumor implantation. n = 6 for control, n = 8 for model, n = 9 for BBD group. (C) Kaplan-Meier survival analysis, n = 6 for each group. * P < .05 and **P < .01 versus healthy controls without tumor implantation; #P < .05 versus for BBD tumor-bearing mice versus model group treated with an equivalent volume of PBS. Abbreviation: CTR, control group without tumor implantation.
BBD Prevents Cachexia-Induced Muscle Atrophy
Next, we examined whether BBD administration had any effect on muscle atrophy as a result of cancer cachexia by measurements of skeletal muscle mass and muscle function. Forelimb grip strength was determined using a grip strength device weekly post-tumor implantation, which showed a steady and significant decline in forelimb grip strength over time following CT26 tumor-implantation (Figure 3A). Notably, mice administered with BBD had significantly improved forelimb grip strength at weeks 4 and 5 post-tumor implantation compared to CT26 tumor-bearing mice treated with an equivalent volume of PBS (Figure 3A). Upon sacrifice on day 36 post-tumor implantation, the quadriceps muscle weight was also significantly reduced in mice implanted with CT26 tumors, verifying the occurrence of cancer cachexia-induced muscle atrophy, but this was significantly attenuated in mice administered with BBD (Figure 3B).

BBD attenuates cachexia-induced muscle atrophy in CT26 tumor-bearing mice. (A) Forelimb grip strength over time following CT26 tumor implantation. (B) Quadriceps muscle weight at the time of sacrifice on day 36 post-tumor implantation. n = 6 for control, n = 8 for model, n = 9 for BBD group. (C and D) Representative images of H&E staining (C) and quantification (D) of quadriceps muscles cross-sectional areas at day 36 post-CT26 tumor implantation. n = 6 for each group. Scale bar, 50 μm. *P < .05 and **P < .01 versus healthy controls without tumor implantation; #P < .05 and ##P < .01 versus for BBD tumor-bearing mice versus model group treated with an equivalent volume of PBS. Abbreviation: CTR, control group without tumor implantation.
We further performed Hematoxylin & Eosin (HE) staining to determine the degree of muscle atrophy as a result of CT26 tumor-induced cachexia. The cross-sectional area of quadricep muscle fibers were significantly smaller in CT26 bearing mice indicative of muscle atrophy, whereas mice administered with BBD had significantly larger quadricep muscle fibers (Figure 3C and D). Taken together, these results demonstrated that BBD robustly attenuated skeletal muscle mass loss as a result of cancer cachexia.
BBD Attenuates Cancer Cachexia via Inhibiting IL-6/STAT3 Signaling Pathway
Due to the important involvement of IL-6/STAT3 signaling in the development of cancer cachexia, we further examined whether the effect of BBD was via regulation of IL-6/STAT3 signaling. We examined serum expression of the IL-6 cytokine using ELISA, which showed that mice bearing CT26 tumors had significantly increased levels of IL-6 compared to the control group (Figure 4A). In contrast, mice administered with BBD had significantly decreased levels of serum IL-6 in CT26-tumor bearing mice, which was essentially at the level of control group (Figure 4A). Furthermore, western blot analysis of the excised quadriceps muscles in CT26-tumor bearing mice at day 36 post-implantation showed significant upregulation of p-STAT3 expression, with the ratio of p-STAT3/total STAT3 of greater than fourfold higher compared to control mice (Figure 4B and C). Notably, the level of p-STAT3 upregulation following CT26 tumor-implantation was significantly reduced in the quadriceps muscles of mice administered with BBD, demonstrating that BBD can effectively alleviate cancer cachexia via inhibiting the activation of IL-6/STAT3 signaling pathway (Figure 4B and C).

BBD attenuates cancer cachexia via inhibiting IL-6/STAT3 signaling pathway. (A) ELISA analysis of serum IL-6 levels at day 36 post-CT26 tumor implantation. (B and C) Quantification (B) and representative western blot analysis (C) showing the expression of p-STAT3 in the quadriceps muscles of mice post tumor-implantation. Phosphorylated level of pSTAT3 expression was normalized to the level of total STAT3 expression. GAPDH was used as the loading control. n = 4 for each group. * P < .05 and **P < .01 versus healthy controls without tumor implantation; #P < .05 for BBD tumor-bearing mice versus model group treated with an equivalent volume of PBS. Abbreviation: CTR, control group without tumor implantation.
BBD Inhibits Expression of IL-6/STAT3 Downstream Targets Atrogin-1, MuRF1, and Myostatin in Cancer Cachexia
The activation of IL-6/STAT3 signaling results in the downstream activation of 3 key muscle atrophy targets, namely Atrogin-1, MuRF1, and Myostatin. Western blot analysis of the excised quadriceps muscles in CT26-tumor bearing mice at day 36 post-implantation showed significant upregulation of all 3 muscle degradation factors, in which the protein expression of Atrogin-1 was upregulated almost eightfold, while MuRF1 and Myostatin expression were also upregulated approximately twofold compared to control mice (Figure 5A and B). Of note, the levels of Atrogin-1, MuRF1, and Myostatin were significantly reduced in the quadriceps muscles of CT26-tumor bearing mice administered with BBD, demonstrating that BBD can robustly prevent cancer cachexia-induced muscle atrophy via inhibiting IL-6/STAT3 signaling downstream targets of muscle atrophy (Figure 5A and B).
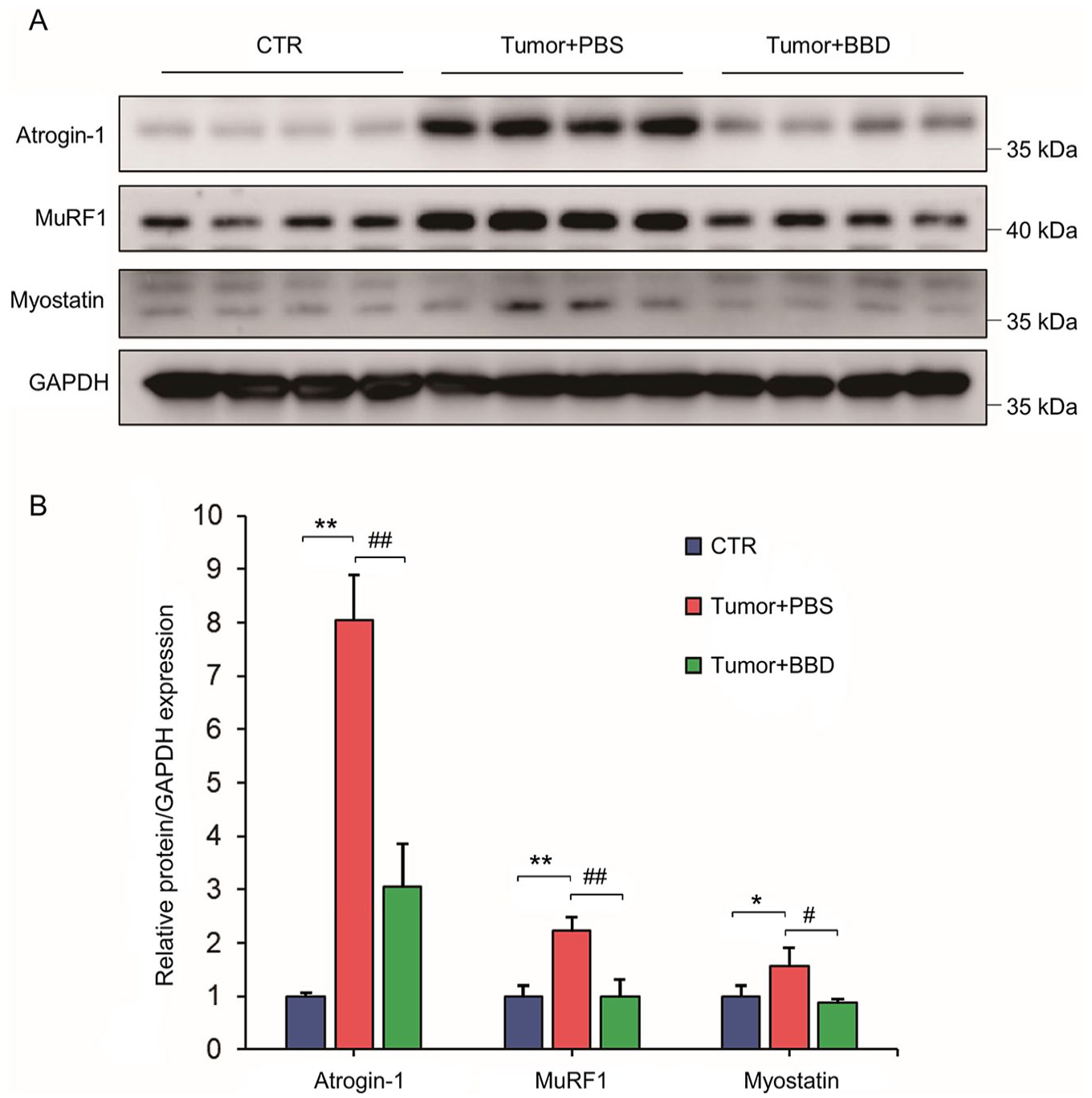
BBD inhibits expression of IL-6/STAT3 downstream targets Atrogin-1, MuRF1 and Myostatin in cancer cachexia. (A and B) Representative western blot analysis (A) and quantification (B) showing the expression of muscle atrophy proteins Atrogin-1, MuRF1, and Myostatin in mice quadriceps muscles at day 36 post-CT26 tumor implantation, where expression was normalized to the level of GAPDH expression as the loading control. n = 3 for each group. * P < .05 and **P < .01 versus healthy controls without tumor implantation; #P < .05 and ##P < .01 versus for BBD tumor-bearing mice versus model group treated with an equivalent volume of PBS. CTR, control group without tumor implantation.
Coupled with the decrease in tumor-free body weight, these results demonstrated that BBD could ameliorate the main features of cancer cachexia by significantly preventing the decrease in body weight and muscle mass and reduction in muscle atrophy, as well as greatly prolonging survival post-tumor implantation, via inhibiting IL-6/STAT3 signaling pathway.
Discussion
Cancer cachexia is a complex condition with multifactorial characteristics, and hence there is no current effective treatment for it. Current treatment options are mainly limited to treating the symptoms of cachexia, including providing adequate nutrition support, or appetite-enhancing drugs such as progestins, however these drugs are often associated with numerous side effects. 21 Therefore, there is an urgent need to develop targeted and effective and drugs for the treatment of cancer cachexia. In this regard, TCM formulations that involve ingredients derived from natural sources have received increasing attention as potential alternative therapies for the treatment of numerous diseases such as cancer due to their low toxicity coupled with high efficacy. In this study, we determined for the first time the robust anti-cachectic ability of one such TCM, BBD in a murine model of cancer cachexia, which prevented cancer cachexia and its associated symptoms.
Aberrant activation of IL-6/STAT3 signaling pathway is a key factor associated with weight loss and muscle atrophy and poorer outcomes in cancer cachexia, whereby in particular, CT26 tumor-bearing mice have been shown to exhibit especially high levels of serum IL-6.6,22,23 The binding of IL-6 to the STAT3 receptor mediates the phosphorylation of STAT3, resulting in its dimerization and nuclear accumulation, and subsequently causes muscle atrophy via increased expression of muscle atrophy targets MuRF1, Atrogin-1, and Myostatin.5,18 In contrast, inhibition of the IL-6/STAT3 axis can prevent muscle wasting and prolong the survival of mice following tumor implantation. 24 Therefore, targeting the IL-6/STAT3 signaling pathway may be a promising approach for the treatment of cancer cachexia as well as cachexia-related symptoms such as muscle atrophy. Our current study demonstrating the robust anti-cachectic effects of BBD via inhibiting the IL-6/STAT3 axis, suggest that BBD may be an effective therapeutic drug for the treatment of cancer cachexia.
The results of our study were consistent with previous reports that show increased levels of serum IL-6 and STAT3 phosphorylation in mouse skeletal muscle following CT26 tumor implantation, 25 indicative of STAT3 pathway activation in cancer cachexia occurrence. Notably, we demonstrated that BBD not only significantly inhibited the elevation of serum IL-6 levels and suppressed the activation of STAT3 in muscles, but also attenuated the occurrence of cachexia-induced muscle atrophy via inhibiting the upregulation of Atrogin-1, MuRF1 and Myostatin. Moreover, BBD treatment significantly prolonged the survival of mice post-tumor implantation and enhanced muscle function, thereby demonstrating its high potential in the treatment of cancer cachexia and its associated adverse effects.
There are several limitations to our study. Although we showed the combined efficacy of BBD formulation in alleviating cancer cachexia, the action of individual constituents contained within BBD were not separately explored. Future studies that examine the action of these constituents individually would be worthwhile in determining the exact activity of BBD’s anti-cachectic ability. Moreover, whether the robust ability of BBD in prolonging survival following CT26 tumor implantation can also be replicated in other larger animals was not examined and may require further investigation.
Conclusions
Our study elucidated the underlying mechanism of BBD’s anti-cachectic ability, via inhibiting the adverse activation of IL-6/STAT3 signaling axis. Taken together, these findings provide important insights into the role and mechanism of BBD and presents a crucial experimental basis for the use of BBD in the clinical treatment of cancer cachexia.
Footnotes
Author Contributions
Zhengliang Qiu: Writing – original draft, Investigation, Validation, Visualization, Formal analysis. Da Wo: Writing – review & editing, Visualization, Resources. Xiaomei Zhong: Validation, Resources. Jinxiao Chen: Investigation, Methodology, Formal analysis, Visualization, Validation. En Ma: Resources, Software. Jia He: Resources, Software. Jun Peng: Resources, Software. Weidong Zhu: Supervision, Resources, Project administration. Dan-ni Ren: Conceptualization, Supervision, Project administration, Funding acquisition, Methodology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Corporate-academic Collaboration Research Foundation, Fujian University of Traditional Chinese Medicine, China (HX2020008), Scientific research foundation for the top youth talents of Fujian University of Traditional Chinese Medicine (XQB202201), and Scientific Research Foundation for the High-level Talents, Fujian University of Traditional Chinese Medicine, China (X2019001-talent, X2021001-talent, X2021002-talent, and X2021003-talent).
