Abstract
Background:
Cancer-cachexia is associated with chronic inflammation, impaired muscle metabolism and body mass loss, all of which are classical targets of physical exercise.
Objectives:
This systematic review and meta-analysis aimed to determine the effects of exercise on body and muscle mass in cachectic cancer hosts.
Data Sources:
PubMed/Medline, EMBASE, CINHAL, ISI Web of Science, and Cochrane Library were searched until July 2019.
Study Selection:
Trials had to be randomized controlled trials or controlled trials including cancer patients or animal models with cachexia-inducing tumors. Only sole exercise interventions over at least 7 days performed in a controlled environment were included.
Data Extraction:
Risk of bias was assessed and a random-effects model was used to pool effect sizes by standardized mean differences (SMD).
Results:
All eligible 20 studies were performed in rodents. Studies prescribed aerobic (n = 15), strength (n = 3) or combined training (n = 2). No statistical differences were observed for body mass and muscle weight of the gastrocnemius, soleus, and tibialis muscles between the exercise and control conditions (SMD = ‒0.05, 95%CI-0.64-0.55, P = 0.87). Exercise duration prior to tumor inoculation was a statistical moderator for changes in body mass under tumor presence (P = 0.04).
Limitations:
No human trials were identified. A large study heterogeneity was present, probably due to different exercise modalities and outcome reporting.
Conclusion:
Exercise does not seem to affect cancer-cachexia in rodents. However, the linear regression revealed that exercise duration prior to tumor inoculation led to reduced cachexia-severity, possibly strengthening the rationale for the use of exercise in cancer patients at cachexia risk.
Keywords
Introduction
Despite tremendous improvements in cancer treatment, cancer patients are often faced with severe cancer-related and treatment-induced side effects, such as fatigue or chemotherapy-induced peripheral neuropathy. 1 Cancer cachexia is among the most severe side effects and is characterized as a multifactorial disturbance of metabolism and the immune system, leading to progressive loss of total body mass and muscle mass. 2 According to previous estimates, almost 50% of all cancer patients develop a cachectic condition, while this concerns even 80% of hospitalized or advanced staged cancer patients.3,4
Although cachexia may occur in all types of cancer, especially gastrointestinal and lung cancer patients are disproportionally affected. 5 Moreover, chemotherapeutic drugs, such as doxorubicin, 6 may further exaggerate cachexia symptoms. In light of this, previous research has provided evidence that cachexia may reduce the patients’ tolerance to the medical treatment. 7 Furthermore, cachexia may induce perturbations of hormonal and hemorheological homeostasis and, thus, may lead to insulin resistance, anemia, hypogonadism, or edema as well as asthenia and fatigue, eventually reducing the patients’ quality of life.2,8-10 As a consequence of rapid weight loss, cancer cachexia also dramatically increases morbidity and mortality rates. 7
Although most of the pathophysiologic origin of cachexia is still unknown, chronic systemic inflammation is considered a main mediator.9,11 Thus, especially increased levels of tumor necrosis factor-α, interleukin-1 (IL-1), and IL-6 are often observed, all of which promote alterations in the protein metabolism, such as protein degradation signaling and reduced muscular protein synthesis.12,13
Considering the severity of cancer cachexia, it is somewhat surprising that treatment options remain limited, mostly reporting an inconsistent or inadequate efficacy. 14 Pharmacological treatments typically aim for reductions of inflammation and concomitant appetite stimulation, whereas nutritional treatment provides energy- and protein-rich supplementation and diet counselling. 14 However, from a mechanistic point of view, exercise training also appears to be a promising approach for the treatment of cancer cachexia. For example, aerobic exercise training has been shown to reduce low-grade systemic inflammation, while strength exercise is considered a crucial stimulus of muscle synthesis even under catabolic conditions.15-17 In fact, exercise training is commonly recommended to patients with cachexia of other origins, such as heart failure or rheumatoid arthritis.18,19 However, studies examining the efficacy of exercise training in cachectic cancer patients are still limited. 20
Therefore, the purpose of this systematic literature review and meta-analysis was to elucidate the effects of exercise training as a countermeasure for cancer cachexia in both human and animal models. Special consideration was given to the effects of different exercise training interventions on total body mass (BM) as the primary outcome and muscle mass and muscle cross-sectional area (CSA) as secondary outcomes.
Methods
Search Process
The databases PubMed/Medline, EMBASE, CINHAL, ISI Web of Science, and Cochrane Library were systematically searched for relevant literature until July 4, 2019. The search procedure followed the guidelines provided by PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses). The original protocol was registered with the international database for prospectively registered systematic reviews in health and social care (PROSPERO: CRD42019137964). However, the protocol was later changed in the following domains: (1) the screened electronic databases were extended from PubMed to PubMed/Medline, EMBASE, CINHAL, ISI Web of Science, and Cochrane Library and (2) the systematic review was extended to a meta-analysis. The search was carried out using both medical subject headings as well as keywords adapted according to the requirements of the database (Table 1). The results of the search and medical subject headings terms were gathered, duplicates were removed, and 2 reviewers screened the remaining articles for title and abstract independently. If the title and abstract met the inclusion criteria, the articles were evaluated for eligibility in a subsequent full-text analysis. Furthermore, references and citation reports of the included studies were checked for additional eligible literature. Disagreements between the reviewers were resolved by consensus or further consultation of a third author. Finally, studies eligible for the systematic review were screened for inclusion into the pooled analysis. If data were missing or could not be determined, corresponding authors were contacted to provide the missing data.
MeSH and Search Terms.

Abbreviation: MeSH, medical subject heading.
Eligibility Criteria
Study eligibility was assessed using the PICOS (population, intervention, comparison, outcomes, and study design) method (Table 2). Studies identified in the systematic review were eligible for the meta-analysis if they reported mean values and standard deviations of at least one relevant outcome for both exercise and control conditions.
Screening Criteria for Study Inclusion Into the Review and Meta-Analysis.

Data Extraction
The following data were extracted: (1) name of authors, (2) year of publication, (3) study design and population, (4) animal and tumor model, and (5) characteristics of the intervention, such as type, duration, intensity, volume, and frequency. Furthermore, objective measures of BM as well as muscle mass and muscle CSA were extracted for both intervention and control groups. Because of inconsistencies in total BM assessment, we summarized the changes in total BM, carcass mass, and BM gain, unless differences were present within individual studies.
Data Synthesis and Analysis
The number of parameters considered for pooled analysis had to be present in at least 3 studies. The analysis was carried out using the standardized mean difference (SMD) as the outcome measure and a random-effects model was used to pool effect sizes using R (3.6.1), 22 RStudio (1.2.1335), 23 and the metafor package (version 2.2.1). 24 The amount of heterogeneity (ie, τ2), was estimated using the restricted maximum-likelihood estimator. 25 In addition to the estimate of τ2, the Q test for heterogeneity 26 and the I2 statistic 27 were reported. Cook’s distances were used to examine whether study results may be influential in the context of the model. Studies with a Cook’s distance larger than the median plus 6 times the interquartile range of the Cook’s distances were considered to be influential. 28 Additionally, linear regression to account for heterogeneity using a mixed-effects model were conducted to test the following moderator variables: (1) type of exercise, (2) duration of intervention prior or (3) post tumor inoculation, (4) frequency of training, and (5) frequency × total duration of exercise intervention. A trim-and-fill-contour funnel plot was provided to estimate the number of studies potentially missing from the meta-analysis. 29 The rank correlation test 30 and the regression test 31 using the standard error of the observed outcomes as predictor were used to check for funnel plot asymmetry. The model was initially calculated using reported post-values only. Due to the design of a majority of eligible studies in which training was commenced weeks before tumor injections, pooled effects sizes were additionally calculated for BM, using relative changes from pre-tumor injection to killing.
Risk of Bias Assessment
Risk of bias of the included studies was assessed independently by two reviewers, using the tool provided by the Office of Health Assessment and Translation. The Office of Health Assessment and Translation tool provides an approach to evaluate both human and animal model studies for their risk of bias. 32 All studies were screened for the following risk of bias domains (and subdomains): (1) selection bias (randomization and allocation concealment), (2) performance bias (identical experimental conditions and blinding), (3) attrition/exclusion bias (complete data, exposure characterization, and outcome assessment), (4) all measured outcomes reported, and (5) other bias (threats to internal validity). The risk of bias tool rates every domain and subdomain within the range of (1) definitely low risk of bias, (2) probably low risk of bias, (3) probably high risk of bias or not reported, and (4) definitely high risk of bias.
Results
A total of 2417 references were identified during the search process. Out of these hits, 24 studies met the inclusion criteria for the review and thereof 20 studies were included in the meta-analysis (Figure 1). We contacted 14 corresponding authors to provide missing data. Subsequently, six authors provided the missing data,33-38 four authors did not respond but their studies contained partial data to be considered in the analysis,39-42 and an additional four authors did not respond and were excluded because of a lack of considerable data.43-46

Flowchart of the search process.
All eligible studies were performed with animal models and, thus, no human trials were included. The included studies used the following tumor models: (1) Walker-256 breast carcinoma,3,34,36,42,43,45,47-50 (2) Colon-26 carcinoma,33,38,40,41,51 (3) the MC4-L2 breast cancer, 52 (4) the Yoshida sarcoma, 39 (5) 4T1-breast tumor, 35 (6) the Lewis Lung carcinoma, 38 (7) Morris hepatoma 7777, 53 (8) ApcMin/+ with IL-6 overexpression for intestinal neoplasia,8,46 and (9) N-methyl-N-nitrosourea–induced breast cancer.37,54 A detailed overview including the study description and individual results of all eligible studies is provided in Table 3.
Summary of Relevant Outcomes in all 24 Included Studies a .

Abbreviations: 1 RM, one repetition maximum; C26, colon-26; CSA, cross-sectional area; CT, controlled trial; EDL, extensor digitorum longus; EMS, electrical muscle stimulation; GSN, gastrocnemius; IL, interleukin; LLC, Lewis lung carcinoma; max HR, maximum heart rate; MNU, N-methyl-N-nitrosourea; N, statistical population; RCT, randomized-controlled trial; TAE, tumor aerobic exercise; TC, tumor control; TE, tumor exercise; TIE, tumor intense-aerobic exercise; TME, tumor moderate-aerobic exercise; TSE, tumor strength exercise, VO2 max, maximum oxygen uptake; ↑, statistical increase compared with controls; ↔, no effects; ↓, statistical reduction compared to controls.
Trials are sorted by (1) inclusion in meta-analysis or review, (2) study design, (3) year of publication, and (4) alphabetical order.
Risk of Bias
All studies showed a probably high risk of bias within the domain of blinding, which is known to be a persistent difficulty of exercise interventions (Table 4). In addition, several studies reported incomplete data due to missing reports of results.3,8,33-35,38,39,44,46,47,50,52-54 Allocation concealment appeared to be a frequent risk of bias.33,35,39,46,47,49-51,53 Furthermore, particularly the studies of White et al 46 and Lima et al 50 were rated with a probably high risk of bias in the categories randomization and all measured outcome reported. While we acknowledge that deviations may have not been thoroughly reported in the included studies, we were not able to find other sources of bias (i.e. threats of intervanl validity).
Risk of Bias Scoring of Included Studies Following the OHAT Risk of Bias Assessment Tool.

Abbreviation: OHAT, Office of Health Assessment and Translation.
, definitely low risk of bias; + probably low risk of bias; − probably high risk of bias; −− definitely high risk of bias.
Risk of Bias domains: (1) selection bias (randomization and allocation concealment), (2) performance bias (identical experimental conditions and blinding), (3) attrition/exclusion bias (complete data, exposure characterization, and outcome assessment), (4) all measured outcomes reported, and (5) other bias (threats to internal validity).
Pooled Analysis
In the meta-analysis, 18 RCTs3,8,33-42,47,48,51-54 and two CTs49,50 were included. In the study of Pin et al, 38 three different exercise experiments with rodents were performed, all of which were deemed eligible and consequently included in the pooled analysis. The overall count of included rodents into the meta-analysis was n = 416, out of which 215 rodents were exercised and 201 rodents served as controls. One study provided only a range for the included population and, thus, the median of the range was used for analysis. 34
The observed effects of postintervention comparisons for BM (SMD = −1.05, 95% confidence interval [CI] = −2.20 to 0.11, P = .08) showed no statistical difference between the conditions (Figure 2). A large heterogeneity was observed (Q(18) = 165.8, P < .01, τ2 = 6.1, I2 = 95.5%), with two studies being highly influential.48,49 None of the moderators explained any heterogeneity (all P > .05; Table 5).

Body mass and mass of gastrocnemius, soleus, and tibialis muscles comparing tumor-bearing exercise training interventions (EX) and tumor-bearing control (CON), using absolute values of endpoint comparisons.
Linear Regression Analysis Using a Mixed-Effect Model.

Abbreviations: BM, body mass; ΔBM, change in body mass from pre- to postintervention; GSN, gastrocnemius muscle; SOL, soleus muscle; TIB, tibialis muscle.
When considering only the training period with tumor presence (Δ), also no statistical between-condition effects were observed for BM (SMD = 0.11, 95% CI = −0.24 to 0.45, P = .11; Figure 3), but study heterogeneity was reduced (Q(12) = 21.9, P = .04, τ2 = 0.2, I2 = 44.1%). Testing for moderators indicated that the duration of exercise training prior to tumor inoculation accounted for 48.9% of the heterogeneity (P = .04), while no effect was observed for the remaining moderators (Table 5).

Changes in body mass comparing tumor-bearing exercise training interventions and tumor-bearing control.
The observed effects of postintervention comparisons for gastrocnemius (GSN) muscle mass (SMD = 0.61, 95% CI = −0.10 to 1.32, P = .09) showed no statistical difference between conditions (Figure 2). A large heterogeneity was observed (Q(15) = 77.9, P < .01, τ2 = 1.8, I2 = 86.1%), but no study was identified as influential. None of the moderators explained any heterogeneity (all P > .05; Table 5).
Similarly, no statistical between-group effect was observed for postintervention comparisons of soleus (SOL) muscle mass (SMD = 0.99, 95% CI = −0.45 to 2.43, P = .18; Figure 2). A large heterogeneity was observed (Q(3) = 16.8, P < .01, τ2 = 1.8, I2 = 86.6%), but no study was identified as being influential. Testing for moderators revealed that the duration of the exercise intervention following tumor inoculation as well as the training frequency accounted for 64.1% (P = .04) and 70.8% (P = .02) of the heterogeneity, respectively (Table 5).
For postintervention comparisons of tibialis (TIB) muscle mass, no statistical between-group difference was observed (SMD = 0.30, 95% CI = −0.85 to 1.46, P = .61; Figure 2). A large heterogeneity was observed (Q(3) = 11.3, P = .01, τ2 = 1.1, I2 = 76.8%), but no study was identified as influential. Both the type of exercise and the training frequency each accounted for 80.3% (P = .02) of heterogeneity, respectively (Table 5).
Publication Bias
The funnel plot did not show a clear funnel-shape across all assessed and pooled effect sizes (Figure 4). The regression test indicated funnel plot asymmetry (P < .01) but not the rank correlation test (P = .93). The visual observation provided by the trim-and-fill function confirmed study heterogeneity, while potential publication bias and methodological heterogeneity are likely, as indicated by a large cluster in the top-center of the plot with no values in the bottom right and left corners, respectively.
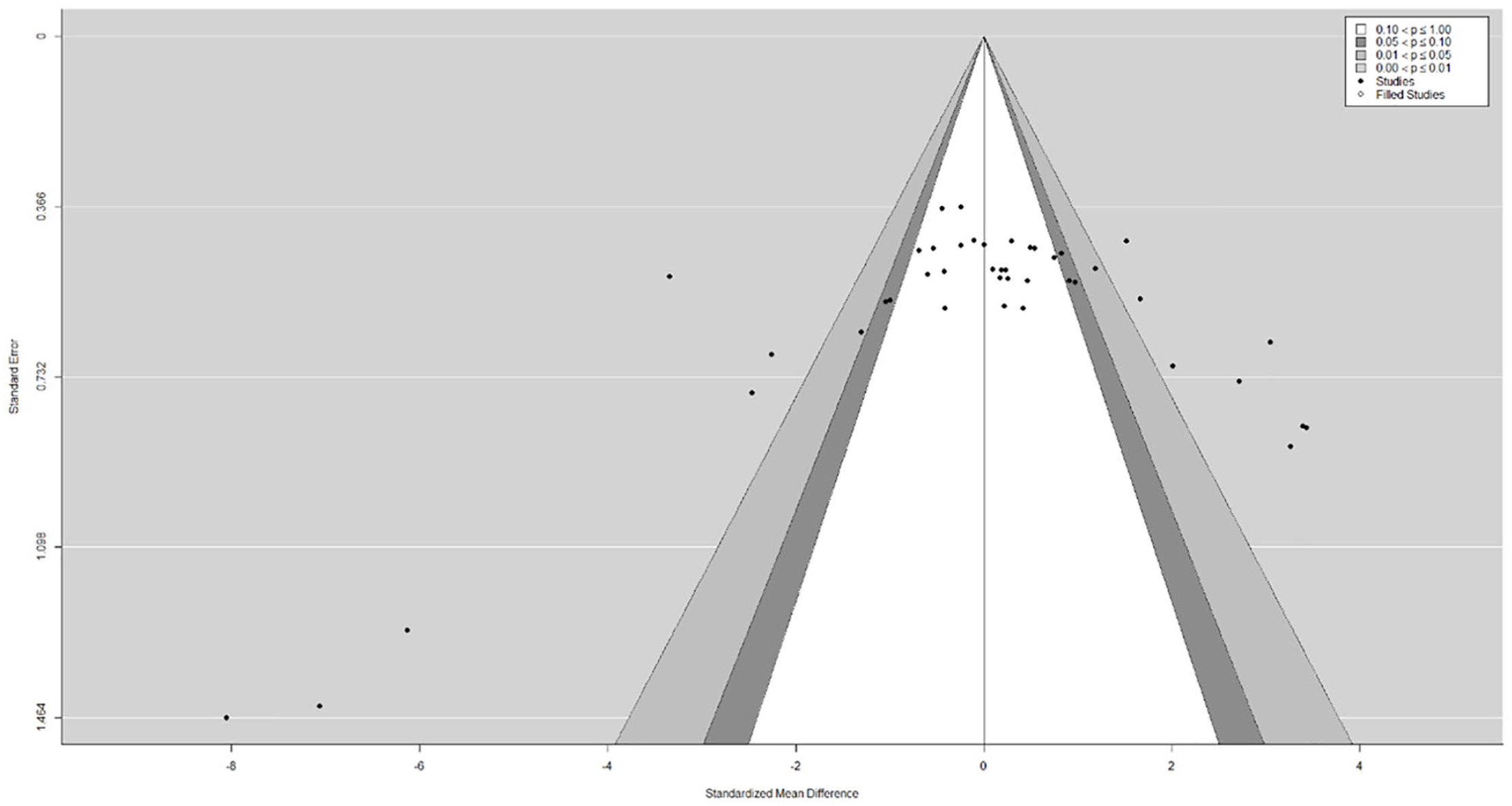
Funnel plot for publication bias assessment including the trim-and-fill function to plot potentially missing publications as well as the contour function to visualize a significance threshold.
Discussion
The purpose of this systematic literature review and meta-analysis was to evaluate the current evidence of the effects of exercise interventions on cancer cachexia. A total of 24 animal models were eligible for the systematic review, while thereof 20 studies were included in the meta-analysis. No statistical differences were observed for BM and muscle mass between the control and the exercise conditions. However, a large study heterogeneity was observed for all outcomes. Moreover, exercise duration prior to tumor inoculation was identified as a significant moderator on the BM under tumor presence.
Considering the high prevalence and clinical relevance of cancer cachexia, it was surprising that at the time of screening no human RCT or CT that specifically screened for cachectic symptoms has been published. In fact, this lack of human trials was previously identified by a Cochrane review that was published six years ago 20 and appears not having improved ever since. The reasons for this paucity may be related to the criteria of cancer cachexia, which have been established as late as 2012 and, thus, a framework of precise classification and treatment was missing. 21 Another major reason might be related to the pathogenesis of cancer cachexia, often developing only in the late stages of the disease, sometimes shortly before demise. 3 Therefore, the late but rapid progression of cachexia makes it difficult to conduct well-designed and controlled studies as well as to recruit eligible patients and to complete comprehensive exercise interventions.
All included animal-based studies were conducted with rodents using tumor models well known for the development of cancer cachexia.37,55-59 Our pooled analysis revealed no statistical effects of sole exercise on characteristics of cancer cachexia. However, also a large study heterogeneity was observed for all outcomes. This was attributed to the number of different animal and tumor models used as well as to the profound differences in characteristics of the exercise interventions, such as exercise type, duration, frequency, and intensity. In addition, the assessment methods, the timing of measurements, and eventually the final data reporting varied across the included studies. For example, several studies included in our postintervention comparison commenced exercise prior to tumor inoculation, while BM was assessed or at least reported only immediately prior to the start of the exercise period and after completion.3,36,48,53 These tumor-free exercise periods may strongly affect BM, as was, for example, shown in the study by Salomão et al. 49 When calculating relative changes for BM from pretumor injection to killing, it was shown that 60 days of training prior to tumor inoculation led to a much smaller weight gain when compared with inactive controls (~220 g vs ~330 g). Consequently, also BM at killing significantly differed between trained and nontrained rats (~293 g vs ~401 g), but was dramatically affected by the pretumor training rather than the exercise training after tumor inoculation. In fact, this phenomenon was also observed in other studies,34,48,50 indicating potential limitations of a sole comparison based on reported postintervention values. Indeed, this might be one explanation for the observed discrepancies in our calculated effect sizes for exercise training when comparing the pooled analysis based on postintervention values and those retrieved from the relative changes.
The duration of exercise prior to tumor inoculation was identified as the only statistical moderator, explaining study heterogeneity for changes in BM. Our findings, therefore, indicate that a greater level of fitness prior to tumor injection could reduce the severity of cancer cachexia symptoms. Exercise has previously been shown to condition and prime the immune system for the tumor burden and may, therefore, reduce the cancer cachexia impact in rodents, as, for example, discussed in the study of Pedersen et al. 60 Therefore, we suggest that an increased overall fitness may provide a preventive measure to reduce cancer-induced BM loss, at least in rodents.
These assumptions are in line with current perspectives of cancer cachexia prevention. 61 While specific evidence in humans is still lacking, first results of human trials with cancer cachexia-relevant outcomes indicated that multimodal approaches including especially strength exercise might contribute to BM and muscle mass maintenance in patients susceptible for cachexia.62-64 However, our meta-analysis did not identify the type of exercise as a significant moderator, but at the same time it also revealed a dramatic underrepresentation of trials including strength exercise. Indeed, sole strength exercise was deployed in only 5 studies, reporting either a prevention of BM loss,36,50 a mitigated muscle mass loss,3,36 or even an increases in BM. 3 This was surprising, considering that strength training is well known as an anabolic stimulus, promoting muscle hypertrophy. In fact, the trials incorporating strength training reported reduced tumor-induced muscle-catabolic factors such as proteolysis-inducing factor (PIF), 49 increased testosterone levels, 3 and improved muscle CSA, 36 all of which suggest promising mechanisms of muscle mass maintenance in cachectic cancer hosts. However, whether the effects of strength exercise for cancer cachexia are superior to that of aerobic training remains unclear. In fact, only one study has directly compared both types of training but failed to show reductions in tumor-induced BM loss in either of the conditions, while providing some evidence for different mechanistic-pathways to counteract cancer cachexia. 40
In line with body mass, our pooled analysis did not show a statistically significant benefit of exercise interventions for changes of muscle mass in GSN, TIB, or SOL. However, our analysis was limited by the insufficient reporting of outcome variables, combined with missing response of the contacted authors.40-42 Thus, the results of these analyses need to be interpreted with caution, due to the heterogeneity and potential publication bias as well as the individual risk of bias in some studies. However, our moderator analysis identified exercise variables such as training duration, frequency, and type of exercise as statistical moderators for SOL and TIB muscle mass. In fact, the effects of these training variables may be related to the mechanisms by which exercise may attenuate the loss of muscle mass. Especially aerobic exercise is well known as a potential anti-inflammatory stimulus and previous research has shown that these effects are highly related to the exercise intensity, duration, and muscle mass involvement.65,66 Interestingly, the only 2 trials using high-intensity aerobic exercise and, therefore, a higher metabolic rate found positive effects on total BM41,45 and muscle mass 41 compared with both controls and moderate aerobic exercise. However, due to insufficient reporting of outcome values, these studies could not be included in the pooled analysis of BM.41,45 Therefore, the appropriate dosage of exercise remains an additional important factor of exercise planning and should be considered in future studies.
When interpreting the present findings, one has to bear in mind that our interpretations are solely based on animal models and, thus, the translation of the findings into cancer care is currently limited. Nonetheless, our findings can provide preliminary but relevant data and future directions in the conception of human trials incorporating exercise training. Therefore, well-designed and controlled trials assessing not only the safety and feasibility but also the underlying pathophysiology and potential exercise-dependent dose-response relationships in patients with manifested cancer cachexia are warranted. Currently, first evidence is emerging that exercise appears to be safe and feasible in pancreatic cancer patients with cachexia. 67 In fact, the 6 months progressive strength training led to significant increases in muscle mass, while total BM remained unchanged. In addition, first clinical trials deploying multimodal interventions including exercise with defined cachectic cancer patients are currently planned 68 or even recruiting patients, 69 suggesting more insights of into the effects of exercise in cachectic cancer patients in the near future.
Conclusions
Our systematic review revealed a clear lack of human exercise trials including cancer patients specifically screened for cachexia. Moreover, since our meta-analysis of 20 animal models did not reveal statistically significant effects of exercise interventions on total BM or muscle mass, the role of exercise in the treatment of cancer cachexia remains questionable. However, the duration of exercise prior to tumor inoculation was associated with an attenuated loss of BM, suggesting that overall fitness of rodents may affect the cancer cachexia progression. Furthermore, the results of our analyses were affected by a large heterogeneity, somewhat hindering the interpretation of the pooled data. Based on this, we encourage the implementation of human trials in order to develop dose-response relationships of different types of exercise with related targeted cellular pathways. In these trials, a universal cachexia sensitive outcome, such the cachexia index,34,36 could be a useful assessment to standardize clinical outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
