Abstract
Objective
To investigate the anti-metastatic effects of Babao Dan (BBD) on gastric cancer (GC) cells (AGS and MGC80-3) and explore the underlying molecular mechanisms by which it inhibits epithelial–mesenchymal transition (EMT).
Methods
AGS and MGC80-3 cells were treated with BBD. In addition, cells were treated with the EMT inducer transforming growth factor-β1 (TGF-β1). Cell viability was determined using the MTT assay, and the live cell ratio was calculated via cell counting. Cell invasion and migration were evaluated using the Transwell assay. Western blotting was performed to measure the protein expression of EMT biomarkers and related genes.
Results
BBD inhibited the viability, migration, and invasion of AGS and MGC80-3 cells, but it did not reduce the live cell ratio. Furthermore, BBD inhibited the expression of N-cadherin, vimentin, zinc finger E-box binding homeobox (ZEB)1, ZEB2, Twist1, matrix metalloproteinase (MMP)2, MMP9, TGF-β1, and p-Smad2/3, whereas E-cadherin expression was increased in AGS and MGC80-3 cells to different degrees. Using a GC cell model of EMT induced by TGF-β1, we proved that BBD inhibited p-Smad2/3 and N-cadherin expression, cell migration, and cell invasion.
Conclusion
BBD suppressed cell migration and invasion by inhibiting TGF-β–induced EMT and inactivating TGF-β/Smad signaling in GC cells.
Keywords
Introduction
Gastric cancer (GC) is a common malignant tumor with a poor prognosis, making it a serious threat to human health. According to the latest statistics of GLOBOCAN, approximately 1.033 million new cases of GC were diagnosed globally in 2018, and the disease caused 783,000 deaths, thereby ranking fifth in incidence and second in mortality among malignant tumors. 1 Among all cancers in China, GC ranks second in incidence and mortality. 2 Currently, tumor resection remains the primary therapeutic option for GC. However, many patients with GC are diagnosed with advanced disease, thereby precluding surgery. Simultaneously, because of the high risk of metastasis and recurrence, advanced GC carries a 5-year survival rate of 20% to 30%.3,4 Therefore, the high metastatic rate of GC requires us to clarify its mechanism and identify new drugs to improve clinical treatment.
Epithelial–mesenchymal transition (EMT) is an initiating factor of tumor cell metastasis. EMT is defined as a process in which epithelial cells are transformed into mesenchymal cells through the action of stimulating factors. Through the EMT process, the polarity of epithelial cells is lost, the connections between cells are broken, the cell framework is recombined, and the shape of cells is changed from flat to fusiform. 5 Cancer cells undergoing EMT can invade the blood system and metastasize to other organs. 6 The absence of epithelial cell differentiation characteristics and the appearance of mesenchymal markers are characteristics of the EMT process. Some factors involved in EMT can also be used as biomarkers to describe the program of a cell. For example, E-cadherin and other epithelial markers are absent following EMT, whereas N-cadherin and vimentin, which are mesenchymal markers, are overexpressed.7,8 In the EMT process, pleiotropic EMT-related transcription factors, such as zinc finger E-box binding homeobox (ZEB)1 and ZEB2 modulate the EMT phenotype. 9 Twist1, a transcription factor that promotes EMT, represses E-cadherin expression by binding to a specific regulatory region of the E-cadherin promoter.10,11 Matrix metalloproteinases (MMPs) are also related to metastasis, playing a role in EMT. 12
The specific signaling pathways that contribute to EMT in cancer cells remain unknown. However, a large number of studies have confirmed that, in many cancers, EMT-inducing signals emanating from the tumor-associated stroma, such as transforming growth factor (TGF)-β, hepatocyte growth factor, and epidermal growth factor, induce or functionally activate cancer cells. The TGF-β family plays a pivotal role in providing key signaling (TGF-β/Smad pathway) to stimulate epithelial cells to initiate EMT.13,14 TGF-β family members regulate many cellular functions, including embryonic development and carcinogenesis. TGF-β1 is an important member of the TGF-β family because it is a potent inducer of EMT and plays an important role in cancer metastasis. 15 TGF-β1 increases the invasive and metastatic capacity of epithelial cells in the EMT process through Smad-dependent or non-Smad-dependent signaling and drives the exosmosis of malignant cells to distal sites. 16 In the TGF-β/Smad pathway, TGF-β binds to the tumor cell membrane high-affinity receptors TGF-βR I and TGF-βR II and induces the phosphorylation and activation of Smad2 and Smad3, which form a trimer with Smad4 and translocate to the nucleus to interact with other transcription factors. Following translocation, the Smad trimer reduces the expression of E-cadherin and increases the expression of N-cadherin and vimentin.17,18 Thus, activation of the TGF-β/Smad pathway, which inhibits EMT, could be a potential treatment strategy for improving the prognosis and survival of patients with GC.
Babao Dan (BBD) was originally described in the Ming Dynasty, and it has been used for more than 400 years, mainly in combination with natural bezoar, snake gall, antelope horn, pearl, musk, and Panax notoginseng. According to Chinese medicine theory, BBD functions mainly by dissipating mass, relieving pain, removing blood stasis, clearing heat, and resolving dampness. 19 Modern clinicians believe that BBD can protect the liver and regulate the immune system, and it is used to treat infectious viral hepatitis, acute cholecystitis, and acute urinary infection. Recent studies revealed that BBD can inhibit tumor growth, and it is used in the adjuvant treatment of GC, non-small cell lung cancer, and liver cancer. 20 However, whether BBD exerts specific effects on EMT in GC remains unknown. In this study, we used the MTT assay, cell counting, Transwell assays, adhesion assays, and western blotting to investigate the effects of BBD on EMT in GC cells and the responsible mechanisms.
Materials and methods
Preparation of BBD solution
BBD powder was formulated into a 25-mg/mL solution using phosphate-buffered saline (PBS, Cat: SH30256.01, Hyclone, South Logan, UT, USA), sonicated for 15 minutes, autoclaved for 20 minutes, and stored at 4°C until use.
Cell culture and treatment
The human GC cell lines AGS and MGC80-3 were obtained from the Cell Bank of Chinese Academy of Sciences (Shanghai, China). Cells were cultured in RPMI-1640 medium (Cat: 11875500BT, Thermo Fisher Scientific, Waltham, MA, USA), supplemented with 10% fetal bovine serum (FBS, Cat: 10099141, Thermo Fisher Scientific) and antibiotics (1% penicillin and 1% streptomycin, Cat: SV30010, Hyclone) at 37°C in 5% CO2. AGS and MGC80-3 cells were treated with different concentrations of BBD (0, 0.25, 0.5, and 0.75 mg/mL, Lot: 180101, Xiamen Traditional Chinese Medicine Co. Ltd, Xiamen, China) for various periods. For some experiments, we treated the cells with 10 ng/mL TGF-β1 (Cat: 100-B-001, R&D Systems, Minneapolis, MN, USA) for 24 hours.
MTT assay
Cells were seeded in 96-well plates (1 × 104 cells/well) and incubated for 12 hours. Then, cells were treated with different concentrations of BBD for the indicated times (12, 24, 36, 48, and 72 hours). The medium was replaced with new medium containing 0.5 mg/mL MTT (100 μL/well, Cat: M8180, Solarbio, Beijing, China) and incubated for 4 hours. The medium was subsequently replaced with 100 μL of dimethyl sulfoxide (Cat: V900090, Solarbio, Beijing, China) containing dissolved formazan crystals. At a wavelength of 570 nm, the absorbance was measured using a microplate reader (Thermo Fisher Scientific).
Live cell ratio analysis
After mixing cells with 0.4% trypan blue in a 1:1 ratio, living cells were stained and analyzed using a Countstar Automated Cell Counter (Inno-Alliance Biotech, Wilmington, DE, USA).
Transwell assay
Transwell cell culture chambers were coated without or with Matrigel to detect cell migration and invasion, respectively. Cells were seeded into 6-well plates (3 × 105 cells/well) and treated with different concentrations of BBD for 24 hours. After treatment, the surviving cells were counted. Then, 5 × 104 cells were seeded into the apical chamber containing FBS-free medium, and 10% FBS-containing medium was added into the basolateral chamber. After 12 hours of incubation, cells were fixed with 4% paraformaldehyde and stained with crystal violet. Cells remaining in the apical chamber were removed using wet cotton swabs. Each bottom layer of Transwell membranes was photographed in five fields using a light microscope for cell counting.
Western blot
Total cellular protein was extracted using RIPA Lysis Buffer (Cat: AR0105-100, Boster, Wuhan, China) and a protease inhibitor cocktail (Cat: CW2200, CWBIO, Beijing, China). Before gel electrophoresis, protein samples were heated at 95°C. Equivalent amounts of protein samples were loaded into each well of a 10% SDS-PAGE gel (Cat: CW0022M, CWBIO, Beijing, China) for electrophoresis, followed by transfer to a nitrocellulose filter membrane (Cat: HATF00010, Merck Millipore, Darmstadt, Germany). Then, the membrane was blocked with 5% skim milk powder at room temperature for 1 hour. Subsequently, the membrane was incubated with primary antibodies (all 1:1000) against E-cadherin (Cat: 14472, Cell Signaling Technology, Danvers, MA, USA), N-cadherin (Cat: 14215, Cell Signaling Technology), vimentin (Cat: 3390, Cell Signaling Technology), MMP2 (Cat: 10373-2-AP, ProteinTech Group, Chicago, IL, USA), MMP9 (Cat: 10375-2-AP, ProteinTech), ZEB1 (Cat: 21544-1-AP, ProteinTech), ZEB2 (Cat: 14026-1-AP, ProteinTech), Twist1 (Cat: 25465-1-AP, ProteinTech), TGF-β1 (Cat: 21898-1-AP, ProteinTech), Smad2/3 (Cat: 8685, Cell Signaling Technology), p-Smad2/3 (Cat: 8828, Cell Signaling Technology), and β-actin (Cat: 60008-1-Ig, ProteinTech Group, Chicago, IL, USA) on a shaker at 4°C overnight. After incubation, membranes were washed with TBST and incubated with secondary horseradish peroxidase-conjugated antibodies (HRP-conjugated Affinipure Goat Anti-Mouse IgG [H+L], Cat: SA00001-1, ProteinTech; HRP-conjugated Affinipure Goat Anti-Rabbit IgG [H+L], Cat: SA00001-2, ProteinTech; both 1:5000) for 1 hour on a shaker. Finally, protein bands were detected via enhanced chemiluminescence (Cat: S60009, Yuheng Bio, Suzhou, China) and captured using the ChemiDoc™ MP System (Bio-Rad, Hercules, CA, USA).
Statistical analysis
All data were presented as the mean ± SD. At least three independent experiments were performed. SPSS version 21.0 software (IBM, Armonk, NY, USA) was used for statistical analyses. First, the normality of the data was checked. If the distribution was normal and the variance was homogeneous, then we used one-way ANOVA to assess significance among three or more groups. If the variance was not identical, then the rank-sum test was used. If the data did not conform to the normal distribution, then the non-parametric test was used. P < 0.05 and P < 0.01 were considered statistically significant.
Ethics and consent
The submitted manuscript is mainly an in vitro study. There were no studies involving animals or humans in our report and thus ethics approval was not obtained, and a consent statement was not required.
Results
BBD inhibited the viability of GC cells
To detect the effect of BBD on cell viability, GC cells were treated with different concentrations of BBD (0, 0.25, 0.5, and 0.75 mg/mL) for 12, 24, 36, 48, or 72 hours. As shown in Figure 1a and 1b, BBD significantly inhibited the viability of AGS and MGC80-3 cells in a concentration- and time-dependent manner (P < 0.05). However, BBD treatment did not reduce the live cell ratio (Figure 1c and 1d) after 24 hours. These results indicated that BBD could inhibit the proliferation of AGS and MGC80-3 cells without directly killing cells.

Babao Dan (BBD) inhibited the viability of gastric cancer cells but did not reduce the live cell ratio. (a, b) AGS and MGC80-3 cells were incubated with different concentrations of BBD (0, 0.25, 0.5, and 0.75 mg/mL) for 12, 24, 36, 48, or 72 hours. (c–d) AGS and MGC80-3 cells were incubated with different concentrations of BBD (0, 0.25, 0.5, and 0.75 mg/mL) for 24 hours. *P < 0.05 and **P < 0.01 versus the control (0 mg/mL).
BBD inhibited the migration and invasion of GC cells
The Transwell assay was used to measure cell migration and invasion. As shown in Figure 2a and 2b, treatment with different concentrations of BBD decreased the numbers of migratory and invasive cells (P < 0.05 or P < 0.01). At a BBD concentration of 0.75 mg/mL, the migration rate was 9.00% in AGS cells and 12.11% in MGC80-3, whereas the invasion rates were 13.00% and 56.34%, respectively. These results indicated that BBD could inhibit the migration and invasion of GC cells.

Babao Dan (BBD) inhibited the migration and invasion of GC cells. AGS and MGC80-3 cells were incubated with different concentrations of BBD for 24 hours. (a) Migration assay (×200), (b) invasion assay (×200). *P < 0.05 and **P < 0.01 versus the control (0 mg/mL).
BBD inhibited EMT and regulated related gene expression in GC cells
In the EMT process, E-cadherin is downregulated and N-cadherin and vimentin are upregulated. As shown in Figure 3a and 3b, different concentrations of BBD improved the expression of E-cadherin and reduced the expression of N-cadherin (P < 0.05 or P < 0.01). Vimentin was reduced by BBD treatment in AGS cells but not in MGC80-3 cells. ZEB1, ZEB2, Twist1, MMP2, and MMP9 are essential EMT-related transcription factors that regulate metastasis-related genes. ZEB1, ZEB2, Twist1, MMP2, and MMP9 expression was detected using western blotting. The results in Figure 3a and 3b revealed that all of these proteins were downregulated by treatment with BBD (P < 0.05 or P < 0.01). These findings suggested that BBD can inhibit EMT by regulating E-cadherin, N-cadherin, vimentin, ZEB1, ZEB2, Twist1, MMP2, and MMP9 expression.

Babao Dan (BBD) inhibited epithelial-mesenchymal transition and regulated related gene expression in gastric cancer cells. AGS and MGC80-3 cells were incubated with different concentrations of BBD for 24 hours. (a, b) Protein expression was measured in AGS and MGC80-3 cells using western blotting. (c, d) Protein expression was expressed as fold changes relative to the control in AGS and MGC80-3 cells. *P < 0.05 and **P < 0.01 versus the control (0 mg/mL).
BBD blocked the activation of TGF-β/Smad signaling
As shown in Figure 4a and 4b, different concentrations of BBD reduced the expression of TGF-β1 and p-Smad2/3 (P < 0.05 or P < 0.01), whereas Smad2/3 expression was unchanged. These results clarified that BBD blocked the activation of the TGF-β/Smad signaling pathway.

Babao Dan (BBD) inhibited the activation of the transforming growth factor-β (TGF-β)/Smad signaling pathway. AGS and MGC80-3 cells were incubated with different concentrations of BBD for 24 hours. (a, b) Protein expression of TGF-β1, Smad2/3, and p-Smad2/3 in AGS and MGC80-3 was assessed using western blotting. (c, d) Protein expression was expressed as fold changes relative to the control in AGS and MGC80-3 cells.
BBD inhibited TGF-β1-induced EMT by inactivating TGF-β/Smad signaling
TGF-β plays a vital role in the EMT process by activating the Smad signaling pathway. Thus, we treated GC cells with 10 ng/mL TGF-β1 (Smad2/3 cascade agonist) to induce EMT progression. As shown in Figure 5a and 5b, p-Smad2/3 and N-cadherin expression was increased by BBD compared with that in untreated cells (P < 0.05 or P < 0.01), whereas E-cadherin and Smad2/3 expression was not altered. However, treatment with 0.5 mg/mL BBD reversed the effects of TGF-β1 on N-cadherin and p-Smad2/3 expression (P < 0.05 or P < 0.01). Then, we performed the Transwell assay to verity the migration and invasion of GC cells. As shown in Figure 5e, stimulation with TGF-β1 increased the numbers of migratory and invasive cells (P < 0.05 or P < 0.01), and these effects were blocked by BBD (P < 0.05 or P < 0.01). These results suggested that BBD inhibited TGF-β1-induced EMT by inactivating the TGF-β/Smad signaling pathway.
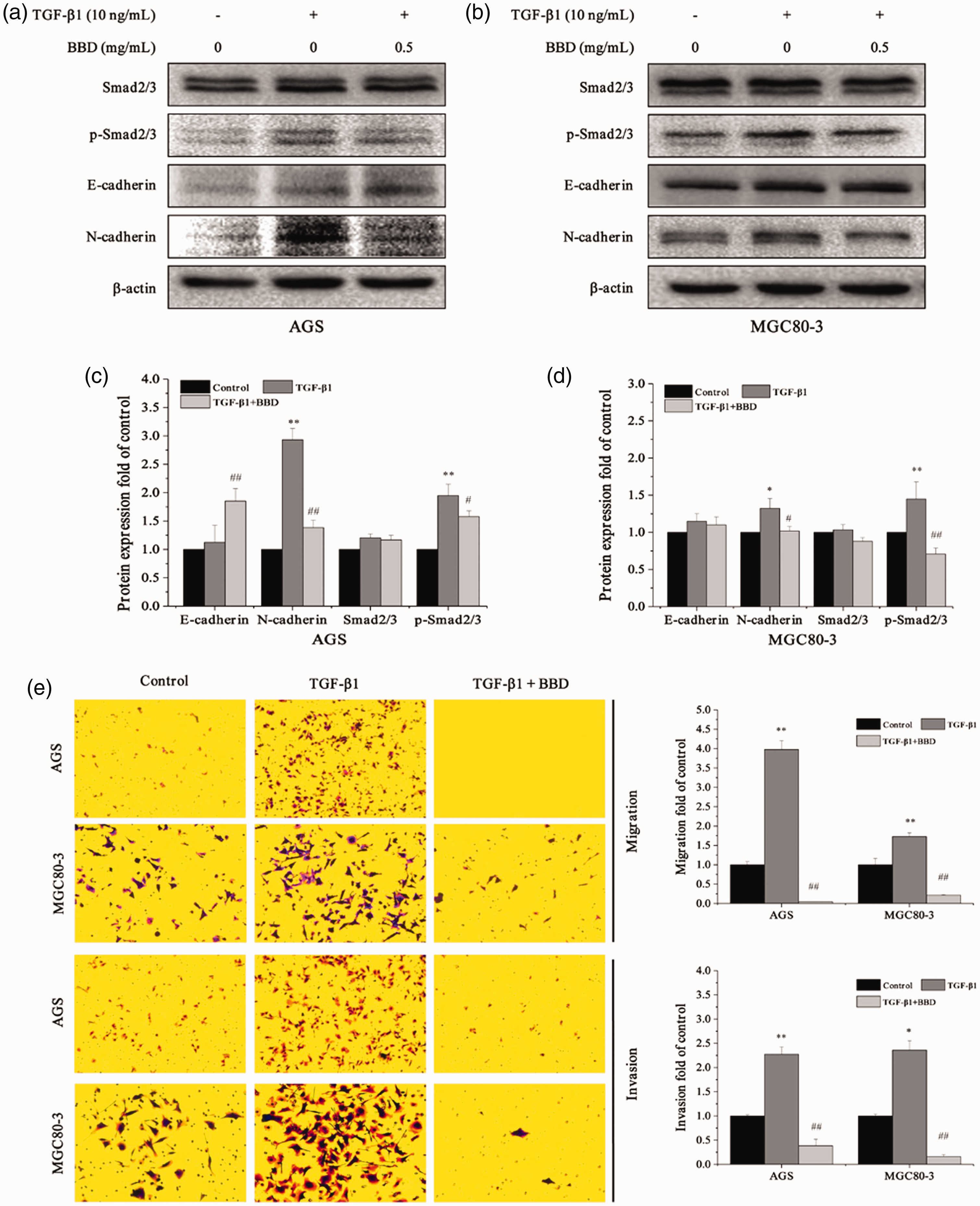
Babao Dan (BBD) inhibited transforming growth factor (TGF)-β1-induced epithelial-mesenchymal transition by inactivating the TGF-β/Smad signaling pathway. AGS and MGC80-3 cells were treated with TGF-β1 (10 ng/mL) and BBD (0.5 mg/mL) for 24 hours. (a, b) Protein expression was measured in AGS and MGC80-3 cells using western blotting. (c, d) Protein expression was expressed as fold changes relative to the control in AGS and MGC80-3 cells. (e) Transwell assay was used to detect the migration and invasion of gastric cancer cells. *P < 0.05 and **P < 0.01 versus the control (0 mg/mL) or #P < 0.05 and ##P < 0.01 versus TGF-β1 (10 ng/mL).
Discussion
Because of the lack of effective treatments for tumor metastasis, the mortality of GC has not obviously declined in the past decade. The major cause of treatment ineffectiveness and death in GC is metastasis. Although more than 200 anti-cancer drugs have been approved for clinical use, few are effective against metastasis. 22 Therefore, the need to find an effective anti-metastatic drug for use in GC treatment is urgent.
Recently, traditional Chinese medicine (TCM) has been increasingly favored by patients and clinicians because of its curative effect and few side effects compared with those of modern chemotherapeutic agents.22,23 Noticeably, many TCMs, such as ginseng, Astragalus membranaceus Bunge, and Viola odorata, have been reported to inhibit cancer metastasis through different biological mechanisms.24–26 BBD is a common formula used in TCM, and it is considered to dissipate mass, relieve pain, remove blood stasis, clear heat, and resolve dampness. Excitingly, several reports have described the use of BBD in cancer treatment to diminish the side effects of radiotherapy and chemotherapy.27,28 However, the mechanism of the anti-metastatic effect of BBD in GC remains unknown. In this study, we first verified the inhibitory effects of BBD on the viability and motility of AGS and MGC80-3 human GC cells. Our findings indicated that BBD inhibited the viability, migration, and invasion ability of GC cells instead of killing cells directly.
Next, we evaluated the expression levels of key molecules of EMT. EMT significantly contributes to the occurrence and development of tumors. Via this special mechanism of transformation, tumor cells acquire greater migratory and invasive ability.29,30 Abundant evidence indicates that the dissemination of epithelial cancer cells occurs in the early phase via the invasion and metastasis of epithelial cancers, which are driven by EMT. Many reports have stated that EMT induces the metastatic cascade, which is evidenced by the deficiency of epithelial polarity and adhesion during the development of GC. The hallmarks of the EMT program are the loss of adherence junctions and apical–basal polarity, acquisition of a mesenchymal phenotype, and increased motility and invasion. Epithelial cells are characterized by intact cell–cell interactions through adhesion molecules such as E-cadherin and cytokeratin within tight junctions, adherens junctions, desmosomes, and gap junctions. Apical-basal polarity is also a key epithelial feature. In response to various extracellular cell- and tissue-specific EMT-inducing signals, a group of EMT-inducing transcription factors are upregulated in epithelial cells to orchestrate the morphological, cellular, and molecular changes occurring during EMT. 31 E-cadherin and N-cadherin are extremely important biomarkers for maintaining the EMT balance. It has been frequently reported that E-cadherin was absent and that N-cadherin was acquired in tumor cells following EMT. 32 These changes impaired cell–cell adhesion, leading to the detachment of cells and the migration and invasion of cancer cells. In this study, we also confirmed that BBD enhanced the expression of E-cadherin while reducing that of N-cadherin. ZEB1 and ZEB2 can regulate the progression of EMT, and Twist, a helix–loop–helix transcription factor, suppresses the expression of E-cadherin, reduces cellular adhesion, and increases motility. 12 In our study, BBD also reduced the expression of ZEB1, ZEB2, and Twist1. Some studies have reported that Twist and MMPs can facilitate EMT and induce metastasis.33,34 MMP2 and MMP9, two members of the MMP family, are required for tumor invasion and metastasis.35,36 BBD inhibited GC metastasis by downregulating MMP2 and MMP9, thereby balancing the extracellular matrix environment. Altogether, BBD had metastasis-suppressing effects on GC cells through the modulation of EMT.
EMT is regulated by multiple signaling pathways. The TGF-β/Smad pathway, which can be activated by TGF-β, plays a dominant role in the EMT process. TGF-β has been found to be responsible for tumor metastasis through the Smad signaling pathway. Smad2 and Smad4 are important proteins regulating transcription and the expression of downstream genes involved in tumor metastasis.37,38 Perera et al.39,40 reported that TGF-β1 significantly promoted the expression of metastasis-related genes, such as MMPs, by activating the Smad signaling pathway in highly invasive breast cancer cells. In addition, TGF-β can induce the invasion and metastasis of epithelial cells in the EMT process through the TGF-β/Smad signaling pathway. 16 Whether BBD exerts anti-metastatic effects on GC cells by inhibiting TGF-β-induced EMT through TGF-β/Smad pathway inactivation requires further verification. In this study, we reported that BBD reduced the protein expression of TGF-β1 and p-Smad2/3, indicating that this inhibition is mediated by blocking the activation the TGF-β/Smad signaling pathway. Then, we assessed Smad2/3, p-Smad2/3, E-cadherin, and N-cadherin expression following TGF-β1 stimulation, and the results illustrated that BBD further inhibited TGF-β1–induced EMT by inactivating the TGF-β/Smad signaling pathway. Further, we produced a TGF-β1–induced EMT model in GC cells and demonstrated that TGF-β1 promoted the migration and invasion of GC cells, and these effects were dramatically reversed by BBD. In addition, TGF-β1 stimulation did not significantly increase the live cell ratio (data not shown), suggesting that TGF-β1 does not promote cell proliferation but it instead increases migratory and invasive activity. Therefore, we provided evidence that BBD deactivated the TGF-β/Smad signaling pathway, thereby inhibiting TGF-β-induced EMT.
In conclusion, BBD inhibits GC cell migration and invasion via inhibiting TGF-β–induced EMT and deactivating the TGF-β/Smad signaling pathway. Elucidation of these mechanisms provides evidence to support the clinical application of BBD.
