Abstract
Background:
When it comes to chemotherapy, maintaining the dose and schedule of treatment are of vital importance, as clinical evidence suggests that dose intensity is associated with optimal treatment outcomes for various tumors. Yet, reduced dose intensity is a common method of mitigating the chemotherapy-induced side effects. Exercise has been shown to attenuate chemotherapy-related symptoms that frequently cluster together. Understanding this, we conducted a retrospective analysis in patients with advanced disease treated with adjuvant or neoadjuvant chemotherapy regimens and who completed exercise training during treatment.
Methods:
Data were collected retrospectively in a chart review of 184 patients, aged 18 years or older and treated for Stage IIIA-IV cancer. Data collection included baseline patient demographics and clinical characteristics, including age at diagnosis, cancer stage at initial diagnosis, chemotherapy regimen, and planned dose and schedule. Cancer types included brain (6.5%), breast (35.9%), colorectal (8.7%), non-Hodgkin’s lymphoma (7.6%), Hodgkin’s lymphoma (11.4%), non-small cell lung (16.8%), ovarian (10.9%), and pancreatic (2.2%). All patients completed at least 12 weeks of prescribed, individualized exercise. Each program included cardiovascular, resistance training, and flexibility components, under the supervision of a certified exercise oncology trainer once a week.
Results:
RDI was measured for each myelosuppressive agent in a regimen over the entire chemotherapy course and then averaged across the myelosuppressive agents in a regimen. An RDI of less than 85% was designated as the clinically meaningful threshold for reduction in RDI based on previously published studies.
Conclusions:
A considerable proportion of patients across regimens had dose delays (18.3%-74.3%) and dose reductions (18.1%-84.6%). Between 12% and 83.9% of patients missed at least 1 dose of a myelosuppressive agent that was part of their standard regimen. Overall, 50.8% of patients received less than 85% of the RDI. In short, patients with advanced cancer and an exercise adherence above 84.3% saw fewer chemotherapy dose delays and dose reductions. These delays and reductions occurred significantly less frequently compared to the published norms in the sedentary population (P < .05).
Introduction
When it comes to chemotherapy, maintaining the dose and schedule of treatment are of vital importance, as clinical evidence suggests that this is associated with optimal treatment outcomes. This includes survival and cure rates, when possible, palliation and quality of life when cure is not possible. 1 The occurrence of severe to life-threatening conditions, including thrombocytopenia, neutropenia, and anemia, often complicates chemotherapy regimens, leadings to dose reductions and delays. 2 Even in populations where the treatment of metastatic cancer is mostly palliative in nature, such as metastatic colon cancer, complications arising from late disease stage and associated treatments often lead to lower quality of life. In these instances, the objectives of treatment focus on prolongation of survival, symptom management, and maintenance of quality of life. 3
One recent meta-analysis reported the impact of treatment delay in curative treatment on the risk of death in breast, bladder, colon, lung, cervix, and head and neck cancers. Researchers found a 13% increase in risk of death for every treatment delay of 4 weeks in these tumor sites. 4 Similarly, delays in treatment have been linked to all-cause mortality in patients with endometrial, 5 liver, 6 breast, 7 and oral cancers. 8
Relative dose intensity (RDI) is defined as the ratio of the delivered dose intensity (per unit body surface area per unit time [mg/m2 per week]) to the planned dose intensity for a chemotherapy regimen. 9 RDI is used as a summary measure to describe dose delays and/or reductions occurring within a chemotherapy regimen. Clinical evidence suggests that improved outcomes are achieved with standard chemotherapy regimens in a dose-dependent manner. Those patients who receive higher dose intensities tend to experience improved overall survival, progression-free survival, and disease-free survival than those patients who receive lower dose intensities than planned. 10 In fact, an RDI less than 85% is considered to be a clinically significant reduction from standard or planned therapy.11,12 Yet, dose delays and reductions are common methods of mitigating the chemotherapy-induced side effects of treatment. Despite the fact that maintaining RDI is important to achieve improved outcome, a substantial proportion of patients are given less than 85% of the suggested dose. In advanced disease, research indicates that less than half of patients receive the 85% suggested dose.13,14
The impact of dose delays and reductions on patient outcomes has been evaluated in single center retrospective studies, with results suggesting reduced RDI confers inferior outcome,15,16 raising the question of how this may be improved. The Goldie-Coldman hypothesis addresses this issue by asserting that the delivery of higher doses of chemotherapy reduces the survival probability of chemotherapy-resistant cells. 17 The Norton-Simon hypothesis advances this by incorporating the concept of chemotherapy schedule. These hypotheses have significantly influenced oncology practice 18 and have led to an effort to maintain the full-dose chemotherapy on the planned schedule as a means to improve disease outcomes and overall quality of life. 15
Decades of well-designed studies have highlighted the role of exercise in mitigating the symptom severity of chemotherapy. Importantly, exercise has been successfully shown to attenuate chemotherapy-related symptoms that frequently cluster together.19-23 Specifically, this includes chemotherapy-induced peripheral neuropathy,24-26 sleep disturbances,27,28 fatigue,29,30 pain, 30 nausea, 30 and neutropenia. 31 Therefore, in light of the importance of maintaining dose intensity, we conducted a retrospective analysis in patients with advanced disease treated with adjuvant or neoadjuvant chemotherapy regimens and who completed exercise training during treatment.
Methods
Study Population
After obtaining IRB approval (approval number 1713991, approval date April 30, 2021), data were collected retrospectively in a chart review of 184 patients, aged 18 years or older and treated for Stage IIIA-IV cancer. All patients initiated adjuvant or neoadjuvant chemotherapy at Kettering Medical Center in Dayton, OH between January 1, 2019 and March 31, 2021.
Inclusion criteria for the study included patients who had received at least 1 intravenously administered myelosuppressive agent in the first chemotherapy cycle, did not receive any oral myelosuppressive agents, and did not participate in any clinical trials during the chemotherapy course. Patients were excluded from the study if they had evidence of disease progression during the 12-week exercise intervention, unplanned regimen changes, or death beyond the first event date. All patients were referred to the exercise program by their oncologist and consented to the exercise program.
Study design
The medical record of each patient constituted the data source for this study. Data collection included baseline patient demographics and clinical characteristics, including age at diagnosis, cancer stage at initial diagnosis, chemotherapy regimen, and planned dose and schedule. Specific variables that were collected include: cardiovascular changes (through echocardiogram, resting heart rate, blood pressure response), treatment-related side effects, treatment compliance, and medications. In addition, demographic characteristics of subjects, including age, gender, type of cancer, BMI, comorbid conditions, performance status, and ethnicity were collected.
Using the patient medical record, we retrospectively identified the first course of adjuvant or neoadjuvant chemotherapy received by patients with advanced-stage cancer. Patients were grouped according to chemotherapy agents received in cycle 1, planned number of cycles, and planned duration of each cycle for all tumor types. The treatments received by patients in each group were compared with standard tumor regimens, as described by NCCN Guidelines.32-37 Adjuvant or neoadjuvant chemotherapy was determined based on the value of the “Line of therapy” variable in the electronic medical record: “adjuvant” or “neoadjuvant” for patients with breast cancer, ovarian cancer, colorectal cancer, and non-small cell lung cancer, and “first line” for patients with non-Hodgkin or Hodgkin lymphoma.
Exercise protocol
All patients completed at least 12 weeks of prescribed, individualized exercise in accordance with ACSM guidelines. 38 Exercise was offered in-person, on-site at the cancer center, due to COVID precautions. For patients who had a compromised immune function, lived far away from the cancer center, or wished to exercise in their home, a virtual supervised exercise program was made available to them. 39 Each program included cardiovascular, resistance training, and flexibility components, and were completed under the supervision of a certified exercise oncology trainer once a week. Exercise sessions were not performed within 24-hour of a chemotherapy treatment. Cardiovascular exercise was performed on a treadmill, recumbent bike, or using no equipment inside the patient’s home (ie, walking). The intensity level for the aerobic exercise ranged from 30% to 45% of the individual’s predicted VO2max, controlled by heart monitors (model S810i; Polar Electro) and lasted 30 minutes. During the first week of exercise training, the intensity level for aerobic training began at 30% of the individual’s predicted VO2max. Each week, intensity was gradually progressed until the participant reached 45%. Strength training involved a full body workout, with emphasis on all major muscle groups and employed machines, free weights, and tubing. Each exercise program was individualized for each patient, but included the following resistance exercises: chest press, seated row, overhead press (when the patient was able), leg extension, leg curl, leg press. Resistance intensity was set at a minimum of 30% of the individual’s 1RM, or when the patient felt sufficiently fatigued after 10 repetitions completed with proper form. As the patient’s RPE values decreased with each exercise, intensity was progressively increased, accordingly. Patients strived to complete 3 sets of 10 repetitions for each exercise. Flexibility training involved static stretching of all major muscle groups for 15 to 20 seconds at the completion of each workout. Each session lasted approximately 60 minutes. Patients were also given exercises to perform at home in between sessions, and activity levels were monitored by their trainer by patient self-report or data from wearable activity monitor.
Outcome measures
A dose delay was defined as a delay of 7 or more days in the administration of at least 1 myelosuppressive agent in any chemotherapy cycle relative to the standard day of administration.11,12 A dose reduction was defined as any reduction of 15% or more in chemotherapy doe for at least 1 myelosuppressive agent in any chemotherapy cycle relative to the standard dose.11,12 Missing doses were identified if the patient did not receive at least 1 myelosuppressive agent that was part of the standard regimen in any cycle. A patient with a missing dose was assumed to have both a dose delay and dose reduction in that cycle, as was consistent with previous studies.11,12,40 RDI was identified as the ratio of delivered dose intensity to the dose intensity of the standard tumor regimen, as described in NCCN Guidelines. RDI was measured for each myelosuppressive agent in a regimen over the entire chemotherapy course and then averaged across the myelosuppressive agents in a regimen. An RDI of less than 85% was designated as the clinically meaningful threshold for reduction in RDI based on previously published studies.11,12,40
Statistical Analyses
Data consisted of numerical and categorical variables corresponding to patient characteristics. Numerical variables were expressed in the form of the mean ± standard error, whereas the categorical variables were expressed as frequency.
For each standard tumor regimen, RDI was calculated during the first chemotherapy course and dose delays and dose reductions were identified in any cycle of the course. Descriptive statistics were reported by standard tumor regimen for the patient baseline characteristics, measured at the initiation of chemotherapy. The incidence of dose delays (≥7 days) was calculated in any cycle of the course, incidence of dose reductions (≥15%) in any cycle of the course, percentage of patients with any missing dose for any myelosuppressive agent, RDI, and incidence of reduced RDI (<85%) over the course. Once determined, a binomial test was completed on the percent of patients who had dose delays and reductions, to compare these outcomes to the published norms in the sedentary population.
Sensitivity analyses were conducted using planned regimen (planned dose, planned cycle length, and planned number of cycles) as the benchmark for calculation of dose delays, dose reductions, and reduced RDI. Planned dose for myelosuppressive agent was assumed to be the maximum of all doses received across cycles, which was the first administered dose for all patients included in the study.
Results
Study Population
This study included 184 patients with 23 different tumor types who received 1 of 20 chemotherapy regimens. Characteristics of the study population are displayed in Table 1. Table 2 presents the standard chemotherapy regimens according to cancer type.
Characteristics of Study Population.

Values are means ± SE.
Standard Chemotherapy Regimens According to Cancer Type.*

Abbreviations: TC, docetaxel, cyclophosphamide; TAC, docetaxel, doxorubicin, cyclophosphamide; DD, Dose Dense; AC, doxorubicin, cyclophosphamide; TCH, docetaxel, carboplatin, trastuzumab; BEP, bleomycin, etoposide, cisplatin; FOLFOX/mFOLFOX, folinic acid, 5-fluorouracil; 5-fu, 5-fluorouracil and oxaliplatin; ABVD, doxorubicin hydrochloride, bleomycin sulfate, vinblastine sulfate, dacarbazine; R-CHOP/CHOP, rituximab, cyclophosphamide, hydroxydaunorubicin hydrochloride, vincristine, prednisone; RCVP/CVP, rituximab, cyclophosphamide, vincristine sulfate, prednisone; FOLFIRINOX, folinic acid, fluorouracil, irinotecan hydrochloride, oxaliplatin.
Exercise Adherence
Overall, exercise adherence was 84.3%. In general, as ECOG scores worsened, exercise adherence dropped. For those who had a baseline ECOG score of 1 or 0, exercise adherence was 92%. Conversely, those with a baseline ECOG score of 2 or greater had an exercise adherence of 72.8%.
Physical Fitness
Fitness outcomes pre- and post-exercise intervention are presented in Figure 1. The physical fitness measures of cardiorespiratory fitness, muscular endurance, and muscular strength significantly improved compared to baseline (P < .001). Specifically, cardiorespiratory fitness improved by 18.78%, muscular endurance increased by 50.08%, and muscular strength by 9.92%. Flexibility also improved by 1.18%, but this was not found to be significant (P > .05).
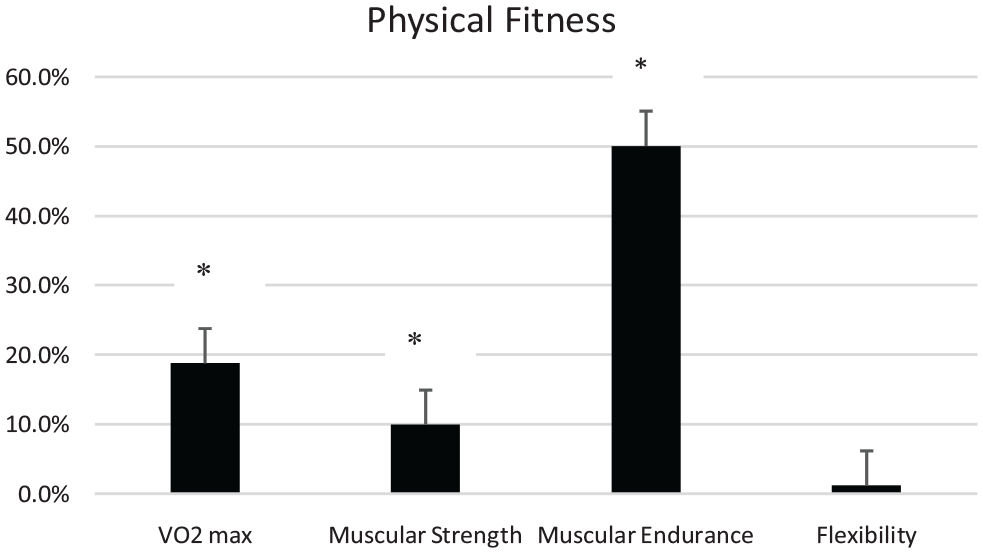
Pre- to post-changes in fitness parameters. Values are mean percent change ± standard error. *P < .05.
Dose Delays, Dose Reductions, and RDI
As shown in Table 3, a considerable proportion of patients across regimens had dose delays (18.3%-74.3%) and dose reductions (18.1%-84.6%). Further, between 12% and 83.9% of patients missed at least 1 dose of a myelosuppressive agent that was part of their standard regimen.
Incidence of Dose Delays, Dose Reductions, and Missing Doses. Values are means.

Abbreviations: TC, docetaxel, cyclophosphamide; TAC, docetaxel, doxorubicin, cyclophosphamide; DD, Dose Dense; AC, doxorubicin, cyclophosphamide; TCH, docetaxel, carboplatin, trastuzumab; BEP, bleomycin, etoposide, cisplatin; FOLFOX/mFOLFOX, folinic acid, 5-fluorouracil; 5-fu, 5-fluorouracil; ABVD, doxorubicin hydrochloride, bleomycin sulfate, vinblastine sulfate, dacarbazine; R-CHOP/CHOP, rituximab, cyclophosphamide, hydroxydaunorubicin hydrochloride, vincristine, prednisone; RCVP/CVP, rituximab, cyclophosphamide, vincristine sulfate, prednisone; FOLFIRINOX, folinic acid, fluorouracil, irinotecan hydrochloride, oxaliplatin.
Table 4 presents RDI, which ranged from 12.8% for patients with breast cancer treated with dose-dense AC followed by paclitaxel every 2 weeks to 79.3 for patients with pancreatic cancer treated with folinic acid, 5-fluorouracil, oxaliplatin, followed by gemcitabine and paclitaxel. Overall, 50.8% of patients received less than 85% of the RDI. The incidence of reduced RDI was higher among patients with worse physical performance scores, as measured by ECOG scores. These data are presented in Table 5.
Incidence of Reduced RDI. Values are means. Parentheses indicate 95% Confidence Interval.

Abbreviations: TC, docetaxel, cyclophosphamide; TAC, docetaxel, doxorubicin, cyclophosphamide; DD, Dose Dense; AC, doxorubicin, cyclophosphamide; TCH, docetaxel, carboplatin, trastuzumab; BEP, bleomycin, etoposide, cisplatin; FOLFOX/mFOLFOX, folinic acid, 5-fluorouracil; 5-fu, 5-fluorouracil; ABVD, doxorubicin hydrochloride, bleomycin sulfate, vinblastine sulfate, dacarbazine; R-CHOP/CHOP, rituximab, cyclophosphamide, hydroxydaunorubicin hydrochloride, vincristine, prednisone; RCVP/CVP, rituximab, cyclophosphamide, vincristine sulfate, prednisone; FOLFIRINOX, folinic acid, fluorouracil, irinotecan hydrochloride, oxaliplatin.
Performance Status and Reduced RDI. Values are Means.

Abbreviations: TC, docetaxel, cyclophosphamide; TAC, docetaxel, doxorubicin, cyclophosphamide; DD, Dose Dense; AC, doxorubicin, cyclophosphamide; TCH, docetaxel, carboplatin, trastuzumab; BEP, bleomycin, etoposide, cisplatin; FOLFOX/mFOLFOX, folinic acid, 5-fluorouracil; 5-fu, 5-fluorouracil.
Comparison to Published Norms
The measured dose delays and reductions in our population occurred significantly less frequently than previously published data.11,39 Specifically, our dose delays measured between 18.3% and 74.3%, compared to published delays of 22.9%-88.4%. Table 6 presents a comparison of these data. Table 7 presents a comparison of Reduced RDI values between our exercise population and published data. In previously published data, RDI less than 85% ranged from 15.6% for patients with breast cancer treated with dose-dense AC followed by paclitaxel every 2 weeks to 87.6% for patients with colorectal cancer treated with 5-FU, whereas in our study RDI less than 85% ranged from 12.8% for patients with breast cancer treated with dose-dense AC followed by paclitaxel every 2 weeks to 79.3 for patients with pancreatic cancer treated with folinic acid, 5-fluorouracil, oxaliplatin, followed by gemcitabine and paclitaxel. Our values were significantly lower in breast cancer patients treated with a combination of docetaxel and cyclophosphamide, as well as NSCLC patients treated with carboplatin and paclitaxel regimens. In addition, CRC patients treated with FOLFOX4 and non-Hodgkin’s lymphoma patients treated with R-CHOP/CHOP had significantly fewer RDIs than their sedentary counterparts. Finally, breast, colorectal, and ovarian cancers were subdivided into stage III and stage IV, respectively and data regarding dose delays and RDI for each subgroup are presented in Table 8.
Comparisons of Dose Delays, Dose Reductions, and Missing Doses Between Exercise Population and Previously Published Norms. Values are Means.*

Abbreviations: TC, docetaxel, cyclophosphamide; TAC, docetaxel, doxorubicin, cyclophosphamide; DD, Dose Dense; AC, doxorubicin, cyclophosphamide; TCH, docetaxel, carboplatin, trastuzumab; BEP, bleomycin, etoposide, cisplatin; FOLFOX/mFOLFOX, folinic acid, 5-fluorouracil; 5-fu, 5-fluorouracil; ABVD, doxorubicin hydrochloride, bleomycin sulfate, vinblastine sulfate, dacarbazine; R-CHOP/CHOP, rituximab, cyclophosphamide, hydroxydaunorubicin hydrochloride, vincristine, prednisone; RCVP/CVP, rituximab, cyclophosphamide, vincristine sulfate, prednisone.
P < .05.
Comparisons of Reduced RDI. Values are Means. Parentheses Indicate 95% Confidence Interval.*

Abbreviations: TC, docetaxel, cyclophosphamide; TAC, docetaxel, doxorubicin, cyclophosphamide; DD, Dose Dense; AC, doxorubicin, cyclophosphamide; TCH, docetaxel, carboplatin, trastuzumab; BEP, bleomycin, etoposide, cisplatin; FOLFOX/mFOLFOX, folinic acid, 5-fluorouracil; 5-fu, 5-fluorouracil; ABVD, doxorubicin hydrochloride, bleomycin sulfate, vinblastine sulfate, dacarbazine; R-CHOP/CHOP, rituximab, cyclophosphamide, hydroxydaunorubicin hydrochloride, vincristine, prednisone; RCVP/CVP, rituximab, cyclophosphamide, vincristine sulfate, prednisone.
P < .05.
Incidence of Dose Delays, Dose Reductions, and RDI for Breast, Colorectal, and Ovarian Subgroups According to Stage. Incidence of Dose Delays, Dose Reductions. Values are Number of Patients in Each Category.

Discussion
In this investigation, we analyzed data for the incidence of chemotherapy dose delays, dose reductions, missing doses, and reduced RDI relative to standard chemotherapy regimens among patients who exercised during treatment. It is unknown whether the current published data on these variables take into consideration the exercise status of patients. However, being that less than 2% of cancer patients currently meeting the published guidelines for exercise during treatment, 11 we can assume that these variables were measured on patients who were sedentary.
In the present study, we sub-analyzed reduced RDI scores according to patient performance status. We noted that for patients with higher (worse) ECOG scores, the likelihood of reduced RDI increased. This is in line with previously published data. 11 In our cohort, patients with ECOG scores equal to zero had an overall reduced RDI of 52.11%. This is lower than published values for individuals with advanced cancer, where exercise status was not measured. In that group, published values were 58.23%. Similarly, RDI for patients with ECOG scores ≥1 for our group was 59.6%, which was lower than the previously published data of 64.3%. We can assume that exercise status was not measured in this study, as authors of the report suggested that daily activities were restricted in those patients with ECOG scores ≥1. 11 It is possible that doses may have been reduced in patients with higher ECOG scores by physicians to minimize further toxicity. In previous investigations, we have found reductions in symptom severity in patients who exercised during treatment. A reduction of symptoms could explain the greater likelihood of receiving at least 85% RDI in the present study. 40
Further, we found that for patients with advanced cancer who adhered to their exercise regimen, chemotherapy dose delays and dose reductions were relatively common among tumor types and treatment regimens. However, these delays and reductions occurred less frequently than previously published data.11,12 Specifically, our dose delays measured between 18.3% and 74.3%, compared to published delays of 22.9%-88.4%.40-42 In our data, these dose delays were lowest among breast cancer patients and highest among patients with pancreatic cancer. Comparatively, we did not find published data examining dose delays among patients with pancreatic cancer.
In clinical practice, patients often receive chemotherapy at dose intensities lower than NCCN Guidelines for standard regimens. Traditionally, patient age,43,44 performance status, 12 and neutropenia 2 are common causes of dose delays and reductions. Further toxicities include anemia, 45 fatigue, 46 nausea and vomiting, 45 neuropathy, 46 patient request, 47 and weight change. 46 However, our research and that of previous investigators have found that exercise attenuates treatment related toxicity.41,48,49 This could explain the difference in dose delays and reduced dose observed in the present study, as compared to previously published studies.
Limitations
We acknowledge the presence of limitations in this study due to its complexity. These include a lack of controls, due to the retrospective nature of the study. In addition, there is a potential presence of bias due to patient self-selection into the exercise program. Future studies should include the use of prospective analysis whereby patients are randomized into an exercise group or a sedentary control group. Further, this investigation examined changes in physical fitness, but not the physical function of the patients. Assessing the patient’s physical function during treatment would be a positive added endpoint for clinical trials for clinicians. To measure physical function, clinicians have traditionally relied on instruments focusing on the capacity of the individual to accomplish activities of daily living (ADL) or instrumental ADL scales. In addition, a growing number of physical performance tests have been developed to assess the biology of the function through standardized tasks that mirror specific functional domains. These include the gait speed test, the Short Physical Performance Battery, the Time Up-and-Go test, and the 6-minute walk test. Beyond simply providing data on mobility or strength, these assessments have been shown to indicate markers of wellbeing linked to a variety of chronic conditions.
Conclusions
In this retrospective investigation, we observed that patients who undergo an individualized, supervised, prescriptive exercise program during cancer treatment in advanced cancer experience fewer dose delays, dose reductions and incidence of reduced RDI than those who do not. If randomized controlled trial data becomes available to support the observations herein, exercise should become a part of the cancer care plan for all individuals who battle cancer.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Karen Wonders and Kathryn Schmitz. The first draft of the manuscript was written by Karen Wonders and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The Kettering Health Network Research Ethics Committee approved all study methods prior to the onset of any data collection.
Consent to Participate
Written informed consent was obtained from all individual participants included in the study.
Consent to Publish
The authors affirm that human research participants provided informed consent for publication of all data.
