Abstract
Cancer is a major cause of morbidity and mortality worldwide and therefore there has been interest in discovering the phytoconstituents of medicinal plants exhibiting anticancer activities. Morinda citrifolia L., commonly known as Noni, has shown anticancer properties in in vitro, in vivo, and in clinical studies. A systematic review was conducted to collate scientific evidence on the anticancer properties of M. citrifolia using pre-determined keywords on 5 electronic databases: MEDLINE, CENTRAL, LILACS, Web of Science, and EBSCOHost. A total of 51 clinical and preclinical studies comprising 41 efficacy and 10 safety studies were included in this review. Our findings showed that M. citrifolia demonstrated various anticancer properties in different cancer models, via multiple mechanisms including antitumor, antiproliferative, pro-apoptotic, antiangiogenesis, antimigratory, anti-inflammatory, and immunomodulatory activities. M. citrifolia is deemed to be a potentially valuable medicinal plant in the treatment of cancer through its many intrinsic pathways. More well-designed and reported preclinical efficacy and safety studies are needed to allow for better translation into future clinical studies which could further substantiate the role of M. citriflolia in cancer treatment.
Introduction
Cancer is a major cause of morbidity and mortality worldwide, accounting for nearly 10 million deaths in 2020. The highest incidence (in terms of new cases of cancer) in 2020 was seen in breast (2.26 million cases) followed by lung, colon and rectum, and prostate cancer. Among these, lung cancer contributed the most mortality (1.8 million deaths).1,2 Meanwhile in Malaysia, according to the Malaysia Cancer Registry Report, a total of 115 238 new cancer cases were diagnosed for the period of 2012 to 2016 with breast cancer (19.0%) reported as the most common cancer among all Malaysians, as well as the most prevalent cancer among women, followed by colorectal cancer (13.5%), the most prevalent cancer among males.3,4 Despite the various cancer treatment modalities, efficacy, and safety remains a concern. Therefore, there is a need to explore novel strategies for the treatment of cancer.
There has been long-standing interest in the use of plant materials for cancer treatment due to their scalability and sustainability, as well as potential therapeutic benefits.5,6 Many in vivo and in vitro studies have shown naturally occurring phytoconstituents found in medicinal plants exhibiting anticancer activities via antiproliferative, pro-apoptotic, antimetastatic, antiangiogenic, autophagy regulation, multidrug resistance reversal, and immunomodulatory properties, as well as the potential to enhance chemotherapy as adjuvants. The bioactive compounds that have been popularly studied in cancer models include curcumin (polyphenol compound extracted mainly from the rhizomes of eg, Curcuma longa), epigallocatechin gallate (main polyphenol in green tea [Camellia sinensis], berberine [isoquinoline alkaloid mainly extracted from medicinal plants such as Coptidis chinensis Franch], artemisinin [sesquiterpene peroxide derived from annual wormwood (Artemisia annua L.)], and ursolic acid [ursane-type pentacyclic triterpenic acid found in the berries and leaves of a series of natural medicinal plants, including cranberry (Vaccinium macrocarpon Ait)]). 7
Morinda citrifolia L. (Noni), a member of the Rubiaceae family, is a small evergreen tree or shrub. It is native in regions of Southeastern Asia to Australia and currently has a pantropical distribution. 8 In Malaysia, M. citrifolia is commonly known as noni or mengkudu with other common names such as Indian mulberry, hai ba ji (China), and nuna (India). 9 M. citrifolia has been traditionally used to treat various ailments where the fruits, leaves, root, stem, and bark can be applied externally as a poultice or consumed orally as a decoction or in the form of fermented fruits.10,11 Some of the reported traditional uses are to relieve sore throat, carbuncle, peeling or cracking of the toes and feet, treating stomach ulcer, hypertension,12,13 enlarged spleen, nausea, colic and fever, diabetes, liver disease, hemorrhage, and coughs. 11 In pharmacological studies, M. citrifolia has showed anticancer activities on various cell lines such as lung, cervical, and breast cancer cells. 14 Modern studies also showed that M. citrifolia possess antioxidant, antimicrobial, antifungal, antiangiogenic, antidyslipidemic, hypoglycemic, hepatoprotective activity, and immunomodulatory properties. 15
The ongoing interest in the anticancer effect by M. citrifolia is evidenced through many published articles either from in vitro, in vivo, or clinical studies as well as several narrative reviews of its general potential pharmacological properties.14,16-18 However, there is no recent review focusing specifically on the anticancer activity of M. citrifolia since in the past decade, with the most recent narrative review published by Brown. 14 Therefore, the aim of this study is to produce an updated, extensive and systematic review on the anticancer properties of M. citrifolia. This review presents the current anticancer evidence for M. citrifolia and its bioactive phytoconstituents at all levels in humans, animals, and cells.
Methodology
Research Questions
This review was conducted based on the primary research question “What are the anticancer properties of Morinda citrifolia?” This primary question was further expanded to secondary research questions including the following:
i. What is the scientific evidence available with regards to the anticancer properties of M. citrifolia?
ii. What is the effective dose of different plant parts of M. citrifolia in treating different types of cancer?
iii. What are the possible anticancer mechanisms of action of M. citrifolia?
The following Population, Intervention, Comparison, and Outcomes (PICO) framework was applied to address the review’s research questions as shown in Table 1. Three main population categories were targeted to answer the 3 secondary research questions.
Population, Intervention, Comparison, and Outcomes (PICO) Framework.

Search Strategy
A systematic search was conducted by 2 independent investigators for published literature and ongoing trials with predetermined keywords. In general, a combination of keywords consisting of “Morinda citrifolia,” “noni,” “cancer,” “tumor,” “neoplasm,” “apoptosis,” and “chemotherapy” was used, catered, and adapted to each search engine. An example of the keywords search used for MEDLINE is presented in the Supplemental Appendix S1. A total of 5 electronic databases including MEDLINE, CENTRAL, LILACS (Latin American and Caribbean Health Sciences Literature), Web of Science (WoS), and EBSCOHost were searched since inception until September 2021. Additional relevant studies were also identified from the reference list of related review papers found during the initial search. All searches were performed and matched by 2 independent investigators. Search results were managed using bibliographic software (EndNote X9), and duplicates were removed. Investigators of ongoing clinical trials were contacted to obtain relevant interim information if necessary.
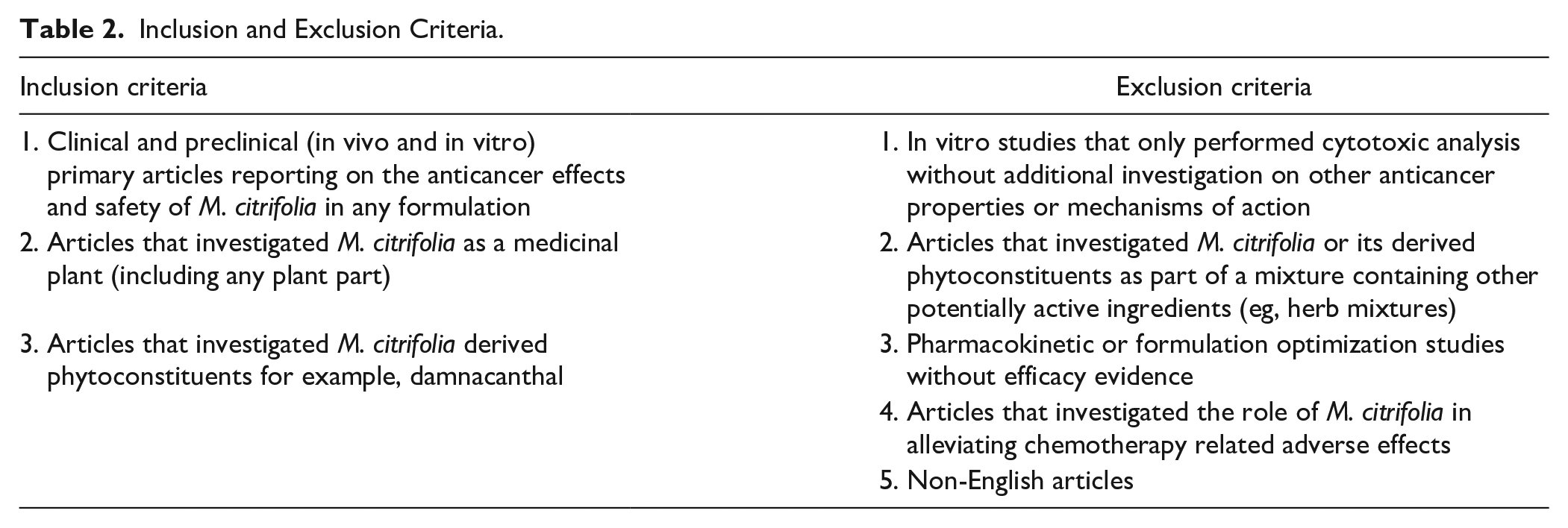
Article Inclusion
Title, abstract screening, and full-text paper inclusion were performed by 2 independent investigators. A third investigator was involved in cases of disagreements. Studies were selected based on the inclusion and exclusion criteria with reference to the research questions identified and PICO elements as depicted in Table 2. Only English language articles were included. This paper reviewed both M. citrifolia as a medicinal plant and M. citrifolia derived phytoconstituents, adhering to the study objectives. Articles investigating M. citrifolia in combination (as mixtures) with other interventions were excluded to facilitate causal relationship analysis between reported effects and anticancer efficacy of M. citrifolia or its derived phytoconstituents. Specifically for in vitro studies, we excluded studies that reported solely on cytotoxicity screening or evaluation of the intervention without additional information on potential anticancer mechanisms. This is to ensure that this review includes studies of more robust models to allow for better translation of anticancer efficacy from the scientific evidence.
Inclusion and Exclusion Criteria.
Data Charting
Three different data extraction tables were specifically designed for: (1) clinical studies, (2) preclinical studies (in vivo), and (3) preclinical studies (in vitro), to comprehensively capture the required information from the included articles. In general, the categories of main data extracted include the following:
(i) Article identifier: designated number; title; and author
(ii) Article characteristics: year; country; type of study (randomized controlled trials, case series, in vivo, in vitro, etc.); and objectives
(iii) Study subjects: sample size; animal model (age, gender, species)
(iv) Intervention: plant part used; formulation; dose; route; duration
(v) Comparator: formulation; dose; route; duration
(vi) Outcome measures: efficacy, safety, mechanism of action
Data extraction was performed independently by 2 investigators with disparities addressed by a third investigator. All investigators were briefed and trained on using the data extraction tables prior to initiation of data extraction to ensure consistency.
Data Analysis
Qualitative analysis was presented descriptively and numerically based on the type of study (clinical, preclinical in vivo, and preclinical in vitro), intervention (plant-based or phytoconstituent-based), cancer type, overall efficacy summary by cancer type, and mechanism of action.
In the absence of randomized controlled clinical trials, pooled quantitative analysis was only performed on preclinical in vivo studies. Quantitative analysis was performed using Cochrane Review Manager (RevMan, version 5.4) software 19 to generate pooled effect analysis involving 3 or more in vivo studies investigating the same quantifiable outcome. The reported mean ± standard deviation (SD) or standard errors of mean (SEM) values and the number of subjects per group comparing M. citrifolia (in any formulation or isolated phytochemical constituents) against comparator (control or standard treatment) were extracted for analysis. Pooled outcome estimates for continuous data were reported as standardized mean differences (SMD) and 95% confidence intervals (CI), with random effects model applied for all outcomes analyzed in view of the different species and methodologies used to induce the same experimental cancer model. All SEM values were converted into SD values using the in-built calculator of the RevMan software. The I 2 statistic was used to assess heterogeneity among pooled studies. Further subgroup analysis was conducted for studies that investigated between compound (phytoconstituent) or plant extract/juice, high or low dose, and comparator type (negative control and other treatments) where appropriate.
Risk of bias assessment was only conducted on in vivo animal studies since there were no randomized controlled clinical trials identified. The risk of bias assessments was conducted by 2 independent investigators, with disparities addressed by a third investigator. The RevMan 5.4 software and Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) tool for animal interventions studies were used for this purpose. 20 This review was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses checklist 21 (Supplemental Appendix S2 and S3).
Results
Summary of Findings
A total of 2602 articles were identified based on the search criteria described in the methodology. Upon removal of duplicates and record screening by the abstract and title, 2207 records were excluded. Of the 106 full-text articles that were screened for eligibility, 55 records were excluded for having no outcome measures, different plant species, drug-herb interactions and for being in a foreign language. A final count of 51 papers met all the inclusion criteria and hence were included for analysis. Figure 1 depicts the selection process and Table 3 presents the details of the included studies.

PRISMA flow chart.
Efficacy
Clinical evidence
A Phase I clinical trial 22 and 2 case reports of gastric cancer 23 that utilized noni as part of the treatment for cancer were included in this review. A summary of the clinical findings is described in Table 4.
Clinical Findings Related to Use of Noni in Cancer Patients.

Plant part used was not reported.
Preclinical Evidence
Quantitative analysis
Experimental breast cancer, lung cancer, and leukemia met the pre-specified criteria for meta-analysis. Subgroup analysis was performed where appropriate based on the outcome measures stated in Table 5 to address heterogeneity potentially due to differences in intervention or dose.
Outcome Measures for Meta-Analyses.

i. Experimental breast cancer
Pooled estimates from 3 studies24-26 showed that noni administration did not result in significantly different effects on tumor volume in experimental breast cancer (SMD −1.67, 95% CI −3.70-0.35) when compared to controls. Subgroup analysis according to intervention type (noni fruit juice; 2 studies and phytoconstituent; 1 study) also did not show any significant difference in effects (Figure 2). Abu et al 24 used 4T1-bearing BALB/C mice, Ali et al 25 used Swiss albino mice inoculated with Ehrlich cancer cells while Clafshenkel et al 26 used female mouse mammary tumor virus (MMTV)-neu transgenic mice as experimental breast cancer models. Two studies (Ali et al and Clafshenkel et al) compared noni treatment against a negative control group while 1 study (Abu et al) did not report details of the intervention used in the control group.
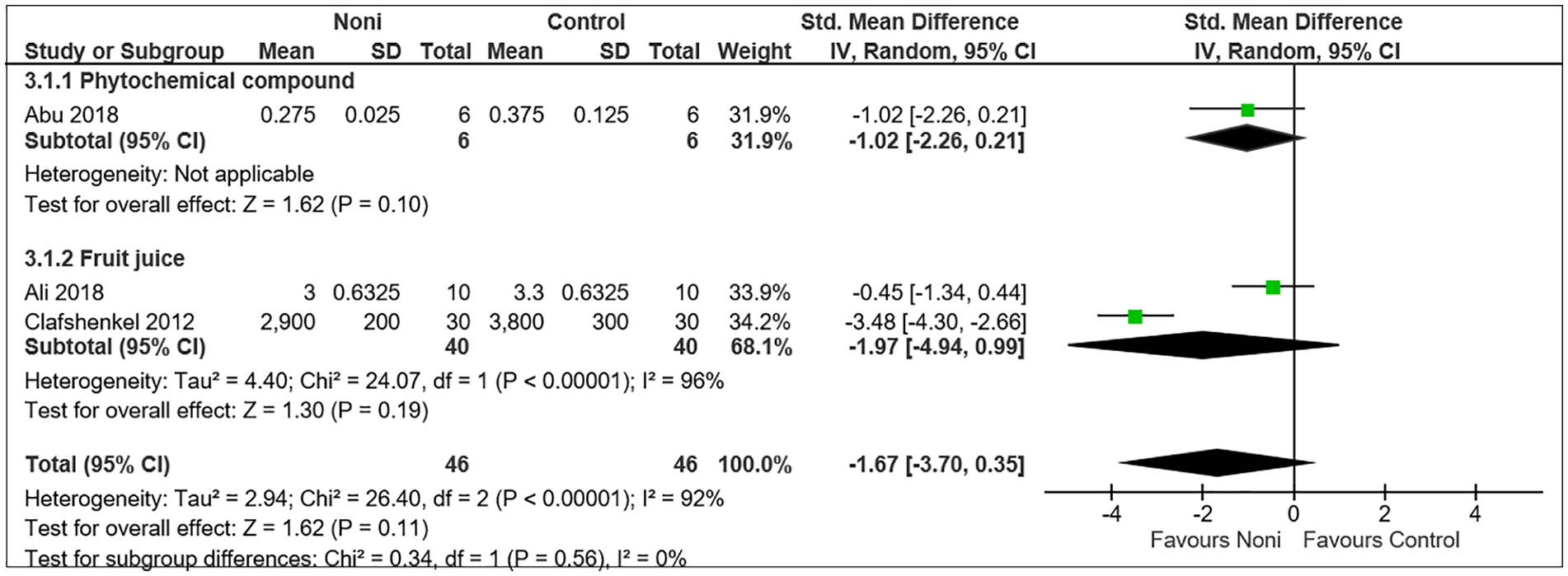
Pooled effect size of noni treatment on tumor volume in experimental breast cancer.
ii. Experimental lung cancer
Pooled estimates from 3 studies27-29 showed that noni administration significantly reduced experimental lung cancer tumor volume compared to overall control (SMD −1.31, 95% CI −2.50-−0.13) (Figure 3). Subgroup analysis revealed significant effects of noni administration in reducing tumor volume when compared to negative controls that is, saline or vehicle (SMD −2.70, 95% CI −3.50-−1.90) but no significant difference when compared to other treatments including oxaliplatin and erlotinib (SMD −0.39, 95% CI −2.11-1.33). These findings are shown in Figure 4. However, all 3 studies, Lim et al27,28 and Ma et al 29 reported vastly different values for tumor volume for high and low dose noni treated groups which may have contributed toward high heterogeneity I 2 = 91%. Further sub-analysis did not show any significant difference between high dose noni versus other treatment (SMD −1.73, 95% CI −3.65-0.19) and low dose noni vs other treatment (SMD 0.21, 95% CI −2.10-2.52) (Figure 5). Two articles, Lim et al27,28 used standardized ethanolic leaf extract (300 and 150 mg/kg) while 1 study, Ma et al 29 reported on fermented noni fruit juice (0.2 mL/10 g and 0.4 mL/10 g). All 3 studies used male BALB/c mice inoculated with A549 cells as the experimental lung cancer model.

Overall pooled effect size of noni treatment on tumor volume in experimental lung cancer.
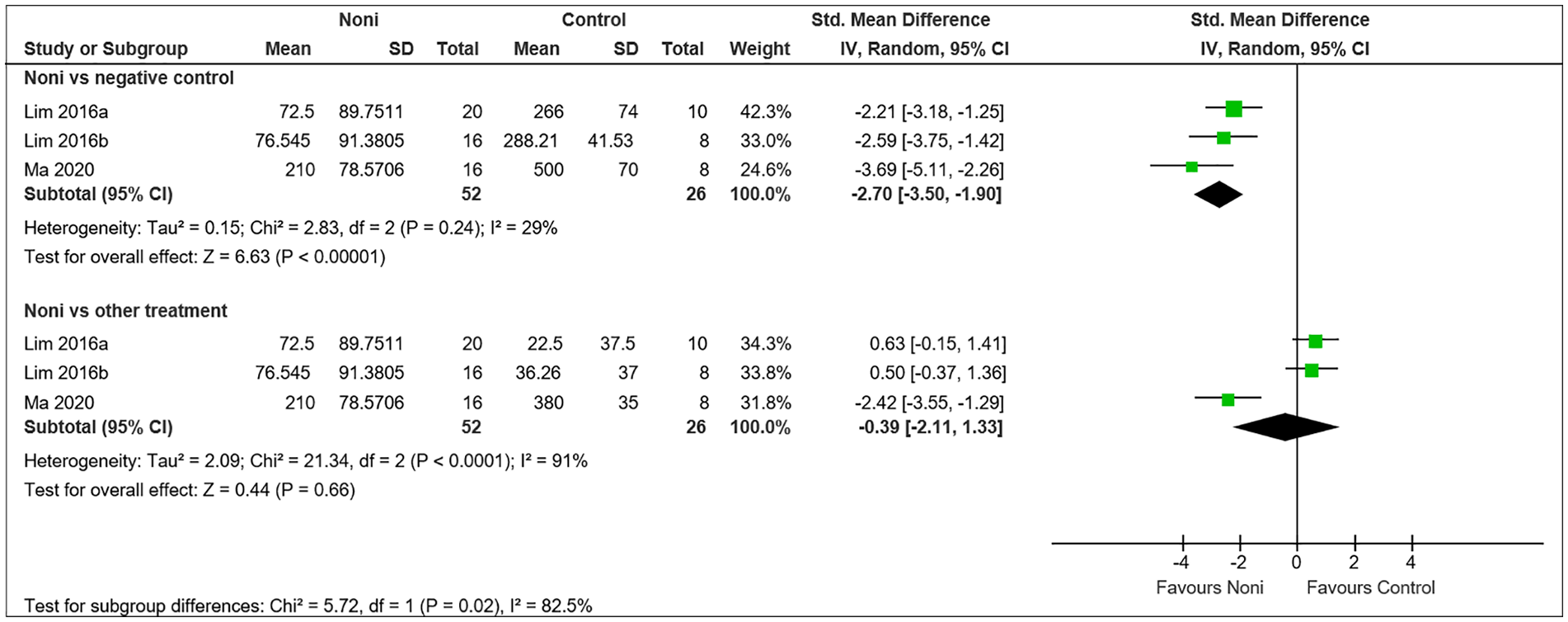
Subgroup analysis on pooled effect size of noni treatment on tumor volume in experimental lung cancer based on comparator (negative control and other treatment).

Subgroup analysis of high or low dose noni treatment vs other treatment on tumor volume in experimental lung cancer.
iii. Experimental leukemia
Pooled estimates showed that noni administration did not exert significantly different effects when compared to controls on neutrophil (SMD −1.59, 95% CI −3.59-0.41), lymphocyte (SMD −0.29, 95% CI −0.88-0.30), erythrocyte (SMD 0.65, 95% CI −0.11-1.40), and leukocyte (SMD −1.01, 95% CI −2.67-0.66) counts in experimental leukemia (total studies = 3 for each parameter). Hazilawati et al30,31 used 8-week-old male Sprague Dawley rats in both studies (leukemia model induced by N-methyl-N-nitrosourea) while Ahmadi et al 32 used 1.5-month-old male BALB/c mice injected with WEHI-3B cells as experimental leukemia models. Both studies by Hazilawati et al investigated on dried fruit while the study by Ahmadi et al investigated noni leaf extract. As shown in Figure 6, subgroup analysis according to intervention type (dried fruit, 2 studies; leaf extract, 1 study) showed no significant difference in all outcomes (data not shown) although Hazilawati et al explained that the significant differences in effect reported in both studies could be explained by a dose-dependent effect of the dried fruit administration (5000 vs 3000 mg/kg as part of food ration) in the individual studies. The intervention details in the control group were unclear for both studies by Hazilawati et al while Ahmadi et al compared noni against All-Trans-Retinoic-Acid (ATRA).

Pooled effect size of noni treatment on (a) neutrophil, (b) lymphocyte, (c) erythrocyte, and (d) leukocyte count in experimental leukemia.
Descriptive Analysis
Noni had been studied in breast cancer, lung cancer, leukemia, and general cancer pathogenesis mainly by utilizing mice and rat models. There were 4 studies on breast cancer which used plant extract and 1 study which used nordamnacanthal, a naturally occurring anthraquinone in noni. Lung cancer and leukemia studies used plant extracts in 4 and 3 studies, respectively. Three studies on general cancer pathogenesis made use of plant extract. A summary of these findings is shown in Tables 6a and b. The efficacy summary reported in the table is generalized to Noni exceeding the activity of comparator or adjuvant, unless stated otherwise. Detailed outcome measures are presented in the Supplemental Appendix S4 and S5.
(a) Summary of In Vivo Preclinical Evidence (Plant Extract).

Abbreviations: CP: cisplatin; bw: body weight; UV: ultraviolet; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells; NK: natural killer; IFN-ɣ: interferon gamma; ATRA: all-trans-retinoic-acid; fNE: fermented noni exudate; LPS: lipopolysaccharide; PBS: phosphate buffered saline.
Plant part used was not reported.
Noni given as combination or adjuvant to conventional treatment.
(b) Summary of In Vivo Preclinical Evidence (Phytochemical).

Quality of Other In Vivo Studies: Risk of Bias (ROB) Assessment Summary
Risk of bias assessment (ROB) was performed on 15 in vivo studies using the RevMan 5.4 software to establish transparency of evidence synthesis on results and their findings. The summary of the ROB analysis is depicted in Figure 7.

(a) Risk of bias assessment of each included study and (b) risk of bias assessment summary of all studies.
All studies reported unclear risk for selection bias in terms of sequence generation and allocation concealment. The procedure on allocation sequence for the selection of animals in respective treatment group were not generated. Information on allocation concealment was not reported by personnel involved in the studies. All studies had unclear risk of performance bias as the information on random housing of animals and blinding of intervention groups was not reported. Detection bias for random outcome assessment of animals in different treatment groups and blinding of the accessor was reported as unclear for all the animal studies due to an absence of information provided in the individual papers included.
The baseline characteristics of animals for the selection bias category, that is, age, weight, gender, breed, and number of animals, were reported in 4 studies (26.7%). Other studies had not provided at least one of the required parameters. A majority of the studies (73.3%) were reported as high risk for attrition bias due to missing or incomplete outcome data reported in the results section. Most of these studies lacked pivotal information on either the initial number of animals included in the study or the final number of animals included in the analysis. Besides, the reasons for the number of animals excluded for result analysis also was not clearly justified. The pattern of analysis of reporting bias for selective outcome reporting is similar to the attrition bias for incomplete data except for an additional study by Abu et al. 24 Although all the animals were accounted for in this study, reporting bias was present as the creatinine and albumin levels were monitored but unreported. Consequently, 12 out of 15 studies (80%) exhibited reporting bias. Financial conflict of interest constitutes other bias reported in studies. About 60% of the studies were sponsored by either the government, university, or non-profitable organization; hence were categorized as low risk. Two of the studies were reported as high risk due to being sponsored by industry, namely Alnoni Ltd, Antalya, Turkey 32 and Morinda, Inc., Utah, USA. 36 Four studies did not provide any funding information.
In Vitro Findings
The in vitro studies were performed by utilizing both the plant extracts and also the phytoconstituents on the respective cancer cell lines such as for breast, lung, cervical, oral, and colon cancers. A summary of the included studies is shown in Tables 7a and b. The efficacy summary reported in the table is generalized to Noni exceeding the activity of comparator or adjunct, unless stated otherwise. Detailed outcome measures are presented in the Supplemental Appendix S6 and S7.
(a) Summary of In Vitro Preclinical Evidence (Plant Extract).

Abbreviations: CYP2C19: cytochrome P450 2C19; TLR: toll-like receptor; ROS: reactive oxygen species; HIF: hypoxia-inducible factor; AKT/PKB: protein kinase B; ERK: extracellular-regulated protein kinase; JNK: c-Jun N-terminal kinase; IL: interleukin; STAT: signal transducer and activator of transcription.
Plant part used was not reported.
Noni given as combination or adjuvant to conventional treatment.
(b) Summary of In Vitro Preclinical Evidence (Phytochemical).

Abbreviations: TPA, 12-O-tedtradecanoylphorbol-13-acetate; EGF, epidermal growth factor; AP1, inducible eukaryotic transcription factor.
Noni given as combination or adjuvant to conventional treatment.
Possible Mechanisms of Action
All the included in vivo and in vitro studies were analyzed for reporting on possible mechanisms of action to better illustrate the potential of noni as an anticancer agent. The majority of the studies displayed pro-apoptotic, antiangiogenesis, antimigratory, antitumor, and antiproliferative effects which facilitate the elimination of cancer cells. The proposed mechanisms of action and supporting evidence are summarized in Table 8.
Summary of Preclinical Evidence (Potential Mechanistic Studies).

Numbers in superscript indicate the cited reference.
Safety and Toxicity Assessment
Ten studies24,63-71 reported on the safety and toxicological assessment of M. citrifolia. Table 9 describes the safety implications of Noni investigated on animal toxicity, chronic, and subchronic toxicities as well as cell toxicity studies.
Safety and Toxicity Assessment of Morinda citrifolia.

Abbreviations: SOD, superoxide dismutase; NOAEL, no-observed-adverse-effect-level; bw, body weight; MMC, mitomycin C; DMBA, 7,12-dimethylbenz[a]anthracene.
Discussion
Overall, M. citrifolia demonstrated various anticancer properties in different experimental cancers, via several mechanisms including antitumor, antiproliferative, pro-apoptotic, antiangiogenesis, antimigratory, anti-inflammatory, and immunomodulatory activities. The scientific evidence gathered were mostly confined to preclinical studies. This review provides a comprehensive evaluation including subgroup analysis and risk of bias assessment of available evidence and it is one of the first systematically conducted reviews on the efficacy and safety of M. citrifolia as an anticancer agent.
Focusing on the included preclinical studies, the role of M. citrifolia was mostly studied in experimental breast cancer, lung cancer, and leukemia. Various mechanisms of action have been found to modulate the anticancer properties of M. citrifolia in animal and cell cancer models most notably through pro-apoptotic, anti-migratory, and cell proliferation disruption properties. The proliferation of cancer cells may be inhibited through the suppression of the AKT/NF-κB signaling pathway leading to apoptosis. 29 In addition, downregulation of cell proliferation Ki67 and PCNA proteins, inhibition of anti-apoptotic protein Bcl-2 expression, and the upregulation of apoptotic caspase-3 protein enhances the apoptotic pathway to eliminate the tumor cells. 29 Furthermore, Noni has demonstrated the ability to disrupt cell migration to inhibit metastasis, halting the progression of tumor cells.43,45 Despite positive outcomes reported in individual studies, most of the selected studies for pooled and subgroup analysis showed no significant differences between treatment and comparator groups, with the exception for experimental lung cancer whereby the administration of M. citrifolia resulted in significant reduction in lung tumor volume. However, the heterogeneity was very high (I2 = 97%). Further subgroup analysis revealed significant difference in reducing tumor volume between the M. citrifolia treated group and negative control/untreated group (with acceptable heterogeneity of I 2 = 29%), while this significant effect was not identified when compared to groups treated with other treatment intervention (erlotinib and oxaliplatin).29-31 Different formulation of M. citrifolia, comparator (drug), and treatment duration could have contributed to the high heterogeneity being observed.
In terms of safety, there have been several case reports on the potential association of M. citrifolia with adverse kidney 72 and liver injuries.73-75 However, these studies were not included in this review as the study population did not have cancer and furthermore had other underlying conditions. There were 2 compounds isolated from M. citrifolia which have been associated with hepatotoxicity; anthraquinones (dose-dependent) and coumarins (idiosyncratic), however such causality needs to be further evaluated. 74 Although some of these events have raised concerns on the potential hepatotoxic effect of M. citrifolia, many important confounding factors could not be accounted for including the contribution of contamination or adulterants, dose, and formulation related effects. As phytoconstituent profiles of finished or processed herbal formulations largely depend on the agroclimatic factors and processing methods (eg, different solvents and drying methods resulting in different phytochemical composition 76 ) of raw materials, it is inherently challenging to apply a blanket rule on M. citrifolia induced hepatoxicity based on case reports, unless a bioactive causative phytoconstituent is identified. A Phase 1 clinical study administered escalating doses of 500 mg of dehydrated noni fruit to advanced cancer patients and it was found that the acute toxicity was not dependent on the dose though liver function analysis was not reported in this study. 22 On the other hand, 9 preclinical studies conducted in animals showed normal hematological and biochemical parameters with no signs of mortality and toxicity except for a study by Mohamad Shalan et al 66 which utilized leaf and fruit extract. In fact, a study by Rosly et al 65 which utilized a comparably larger sample size and a higher dose of dried fruit also reported no mortality and liver injuries. Moreover, several safety studies in which the patient (s) consumed Tahitian Noni Juice® also did not report any adverse events. However, these studies were not reported in this review due to the intervention being a mixture of noni, grape and blueberry juice.77-79 Therefore, to address the uniqueness of herb-based products, regulators often require comprehensive preclinical toxicity data explicit to the product or test items of interest prior to approval for use in humans.80,81
The present review had identified an additional 22 studies as compared to a previous review by Brown. 14 In addition, our review paper enabled better translation of anticancer efficacy of M. citrifolia due to excluding studies that solely reported on in vitro cytotoxicity results without further exploration of other anticancer mechanisms or activities. From our findings, it can be observed that new preclinical studies have been consistently conducted on M. citrifolia, which remains a popular medicinal plant investigated for cancer. Although the anticancer properties of M. citrifolia were substantially studied in preclinical studies, it is vaguely translated into clinical trials as only a single Phase 1 study, conducted more than a decade ago, was identified. 22 There may be many reasons that could have contributed toward the slow progress made toward human clinical trials such as a lack of funding, scarcity of sufficient data on safety and efficacy, challenges in consistent raw material sourcing, among others. The assessment on reporting quality of included preclinical studies performed in this review raises significant concerns on the current reporting quality of published animal studies on this topic. As the translation to clinical research will depend on the quality of preclinical data available, there is a need to improve the awareness of guidelines on the internal validity of individual animal experiments, good reporting practices, as well as the potential risk of bias concerning animal studies among researchers.20,82
This review included English papers hence evidence from other languages may be excluded. Although we attempted to pool a few studies for quantitative analyses, the high heterogeneity and small number of studies suited for meta-analysis are inherent limitations of this review. Further improvements in study design such as baseline characteristics (ie, age, body weight, environmental factors), comparator group, and treatment duration should be considered to achieve homogeneity in order to reach a conclusive data. The purpose of excluding herbal products containing a mixture of M. citrifolia with other active herbal ingredients was to eliminate confounding factors, which resulted in the exclusion of several clinical and preclinical papers that reported on Tahitian Noni Juice®, which is made up of a combination of noni, grape, and blueberry juices.77-79 To enable a better understanding of the role of M. citrifolia in mixtures, future reviews can be conducted to assess the safety, herb-herb, and herb-drug interactions data available for M. citrifolia.
Conclusion
Based on currently available clinical and preclinical efficacy evidence, it is apparent that noni is a potentially valuable medicinal plant in the treatment of cancer. The anticancer activities of M. citrifolia is evidently shown in breast and lung cancer models in which the tumor volume is significantly decreased through apoptosis as well as disruption in cell migration and proliferation pathways. Although several hepatotoxicity cases were reported, there is insufficient evidence to adequately assess the causality of Noni as the causative agent. More well-designed and reported preclinical efficacy and safety studies are needed to allow for better translation into future clinical studies.
Supplemental Material
sj-docx-1-ict-10.1177_15347354221132848 – Supplemental material for Efficacy and Safety of Morinda citrifolia L. (Noni) as a Potential Anticancer Agent
Supplemental material, sj-docx-1-ict-10.1177_15347354221132848 for Efficacy and Safety of Morinda citrifolia L. (Noni) as a Potential Anticancer Agent by Hemahwathy Chanthira Kumar, Xin Yi Lim, Farah Huda Mohkiar, Shazlan Noor Suhaimi, Nurmaziah Mohammad Shafie and Terence Yew Chin Tan in Integrative Cancer Therapies
Footnotes
Acknowledgements
We would like to thank the Director General of Health Malaysia, Deputy General of Health Malaysia (Research and Technical Support), and the Director of Institute for Medical Research for their support and permission to publish this article.
Author Contributions
The first 2 authors contributed equally to this work. All authors provided critical feedback and helped shape the final version of the manuscript for publication.
Data Availability Statement
The authors declare that (the/all other) data supporting the findings of this study are available within the article (and its supplementary information files).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study did not require ethics approval.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


