Abstract
Objective:
It is not well determined whether liver metastasis is a prognostic factor for survival of metastatic non-small cell lung cancer (NSCLC) patients who received immune checkpoint inhibitors (ICIs). We compared the efficacy of ICIs in patients with NSCLC with or without liver metastases, aiming to evaluate the impact of liver metastasis on survival of NSCLC.
Methods:
We systematically searched Pubmed, Embase, and the Cochrane library databases for randomized controlled trials (RCTs) on the efficacy of ICIs in the treatment of NSCLC patients with or without liver metastases. The duration of this search was from January 1, 2000 to June 1, 2022. The reviewers screened the literature, extracted data and conducted quality assessment, and used RevMan 5.4 software and Stata 14 to perform analyses.
Results:
A total of 17 RCTs were included, published from 2019 to 2022. For NSCLC patients with liver metastases, the risk of disease progression decreased by 36% (HR = 0.64; 95% CI: 0.55-0.75; P < .01) when treated with ICIs, and the death risk (HR = 0.82; 95% CI: 0.72-0.94; P < .01) was also reduced after ICIs treatment. For those without liver metastases, they had significantly improved PFS (HR = 0.56; 95% CI: 0.52-0.60; P < .01) and OS (HR = 0.73; 95% CI: 0.67-0.80; P < .01), compared to those of the control group. Subgroup analysis of OS in liver metastases patients suggested that OS benefit was associated with treatment strategy (for anti-PD-L1 plus chemotherapy versus chemotherapy, HR=1.04; 95% CI: 0.81-1.34; P =.75).
Conclusion:
For NSCLC patients with or without liver metastases, ICIs administration could improve both PFS and OS, especially for those without liver metastases. More RCTs are essential to verify these findings.
Introduction
Lung cancer is one of the most common malignancies worldwide. Among them, non-small cell lung cancer (NSCLC) accounts for 82% with the purposes of treatment. 1 More than 55% of cases with stage I or II NSCLC receive surgery. 1 However, most patients are diagnosed with advanced disease because of its asymptomatic features, and undergo systemic therapy. 2 In recent years, the advances in improved treatment options provide novel strategies for treating advanced NSCLC, such as immune checkpoint inhibitors (ICIs).1,3-5 Although the survival of these patients has been dramatically improved with the application of ICIs in first or second-line settings,4,5 there is still a large proportion of patients who fail to gain benefit from these treatments.
For stage IV NSCLC patients, several biomarkers for ICIs have been proposed, such as PD-L1, 6 tumor mutation burden (TMB), 7 performance status (PS), 5 and body mass index (BMI). 8 In addition to those mentioned above, metastasis is suggested to be another important prognostic factor for survival of cancer patients after ICIs treatments. And the brain metastasis and lung metastasis are well studied in this population.9,10 Recently, the liver metastasis is regarded as another factor that may be associated with efficacy of ICIs.
The liver is one of the most common metastatic organs of lung cancer. In metastatic lung cancer, the incidence of isolated liver metastases in 54 697 patients can reach 10.4%, 11 and patients with liver metastases have a significantly poor prognosis. 11 In a large sample study of NSCLC, the correlation analysis between metastasis sites and prognosis suggests that liver metastasis is associated with the worst survival for lung adenocarcinoma and small-cell lung cancer. 12 The median overall survival (OS) of patients with liver metastasis was 3 months, while the median OS were 5 to 7 months for lung adenocarcinoma and small-cell lung cancer patients involving brain or bone metastases. 12 For patients with multiple metastases, shorter survival was observed in liver metastasis patients compared to those without liver lesions. 12 This is in accordance with the above study about the prognostic value of site-specific metastasis. 11
In NSCLC patients receiving immunotherapy, liver metastases from lung cancer have not gained enough attention compared to brain and bone metastases.2,4,13,14 As indicated from the current evidence, the relationship between the efficacy of immunotherapy and liver metastasis in this population is still under investigation.15,16 ICIs treated patients with liver metastases had significantly shorter OS and PFS than patients without liver metastases, 17 suggesting that patients with liver metastases have limited benefit from immunotherapy. 15 Recently, several studies indicate that a few factors can affect the efficacy of immunotherapy in cancer patients with liver metastases, among which the performance status and combination therapy may be the common ones.16,18,19 Targeting these factors could facilitate improving the efficacy of immunotherapy and prognosis in patients with liver metastases from lung cancer. However, it is not well known whether specific factors, such as pathological types or treatment regimen, could affect the outcomes of NSCLC patients who receiving ICIs treatment.
Therefore, in order to summarize the efficacy of ICIs in NSCLC patients with or without liver metastases, and to evaluate whether the efficacy in this population is influenced by treatment regimen or pathology type, we conducted this meta-analysis to assess the association between liver metastases and efficacy of ICIs in NSCLC patients.
Methods
This study was conducted in compliance with PRISMA requirements.
Search Strategy
Two researchers independently searched Embase, PubMed, and the Cochrane Library databases until June 1, 2022. The key search terms were “non-small cell lung cancer,” “NSCLC,” “lung adenocarcinoma,” “lung squamous carcinoma,” “liver metastases,” “liver metastasis,” “immune checkpoint inhibitor,” “nivolumab,” “ atezolizumab,” “pembrolizumab,” “Tislelizumab,” “Camrelizumab,” “ Sintilimab,” “ Toripalimab,” “ Durvalumab,” “Avelumab,” “ Tremelimumab,” and “ Ipilimumab .”
Inclusion and Exclusion Criteria
Inclusion criteria
Included studies should meet PICOs criteria. Patients (P): Patients with non-small cell lung cancer diagnosed by pathology or cytology, with or without liver metastasis diagnosed according to imaging exams. Interventions (I): There were experimental and control groups. The experimental group received treatment strategies based on immune checkpoint inhibitors with or without other medications, such as chemotherapy-based anti-tumor regimen. The control group received either chemotherapy-based anti-tumor regimen or other medications that could also be ICIs. Outcome (O): the main outcome indicators of the efficacy evaluation were PFS and OS and their related HRs. Study Design (S): All studies included in this meta-analysis were randomized controlled trials (RCTs), and the full-text was published in peer-reviewed journals.
Exclusion criteria
Studies were excluded if they were: (1) Reviews, abstracts, and animal experiments; (2) Repeated publications; (3) Incomplete survival data and unclear outcome effects; (4) Errors in statistical methods, which cannot be provided or cannot be converted into HR and its 95% CI.
Data extraction
We assigned 2 reviewers to screen the literature and extract data, and discuss with each other or decide by the third evaluator in case of disputes. The reasons for exclusion were marked for articles that did not meet the inclusion criteria. Extracted data items included: first author, publication time, region, general information of patients, intervention methods, outcome indicators, etc.
Literature quality evaluation
Literature quality assessment was performed using the Cochrane Risk of Bias Assessment Tool. 20 The risk of bias was assessed in 6 aspects, mainly from selection (including random sequence generation and allocation concealment), implementation (including blinding investigators and subjects), measurement (blinded evaluation of research outcomes), follow-up (completeness of outcome data), reporting (optional reporting), and other (other sources of bias). Each item was judged as “low risk of bias,” “high risk of bias” and “unclear” according to the risk of bias assessment criteria.
Statistical analysis
According to the Cochrane Handbook, 20 statistical-based data analysis was made through RevMan 5.4 software, and the results were displayed using forest plots. All PFS and OS analyses were presented using HR and its 95% confidence interval. Heterogeneity analysis was performed using the I 2 test, with I 2 ≤ 50% representing low heterogeneity, and a fixed-effects model method was used for meta-analysis. I 2 > 50% represented significant heterogeneity, and the random effects model method was used for analysis. According to the number of articles analyzed by PFS and OS, funnel plots were made to assess publication bias. Subgroup analysis was performed according to the pathological type of patients (squamous cell carcinoma, adenocarcinoma) and the treatment regimen (ICIs plus chemotherapy, dual ICIs, ICIs plus chemotherapy plus anti-angiogenic therapy). Sensitivity analysis was performed to assess the reliability of the results. P < .05 was considered statistically significant.
Results
Search Results and Baseline Characteristics of Included Studies
A total of 491 relevant publications were retrieved, and 17 reports21-37 from 14 RCTs were finally included according to the exclusion and inclusion criteria (Figure 1). The diagnosis of patients enrolled in each study was based on the TNM diagnostic criteria for NSCLC, and the patients were at advanced stage. The interventions for the experimental group were ICIs-based anti-tumor therapy, and the control group was treated with chemotherapy-based, non-ICIs strategy. ICIs were applied in first- or second-line settings. Five RCTs included patients with non-squamous NSCLC, 3 studies included patients with lung squamous cell carcinoma, and 9 studies included patients with non-small cell lung cancer regardless of pathology types. Included studies reported the association of liver metastases with efficacy and/or survival outcomes, and some of them also reported results of efficacy and/or survival prognosis of ICIs in NSCLC patients without liver metastases (Table 1).

The flow chart of search and study collection.
Baseline Characteristics of the Included Studies.

Abbreviations: NSCLC, non-small cell lung cancer; aNSCLC, advanced NSCLC; mNSCLC, metastatic NSCLC; CT, chemotherapy; OS, overall survival; PFS, progression-free survival; ICI, immune checkpoint inhibitor.
Quality Assessment Results of Included Studies
The included RCTs were evaluated using the Cochrane Handbook risk assessment tool, including 6 aspects: selection, implementation, measurement, follow-up, reporting, and other sources of bias. All studies used randomization methods, but blinding for assessment of interventions and outcome measures was not used in all studies. Overall, the quality of the included studies was at low risk of bias (Figure 2).

Quality assessment of the included studies. (A) The details of bias assessment. (B) The summary of bias assessment.
The Effect of Liver Metastasis on the Correlation Between PFS and OS
Correlation analysis of PFS and OS in NSCLC patients
We analyzed the correlation between PFS and OS in patients with or without liver metastases after ICIs based treatment (Figure 3A). The HRs of PFS and OS showed a good correlation result (Pearson r = .89, P < .01). For PFS and OS (Figure 3B), the correlation analysis was also significant (Pearson r = .84, P < .01).
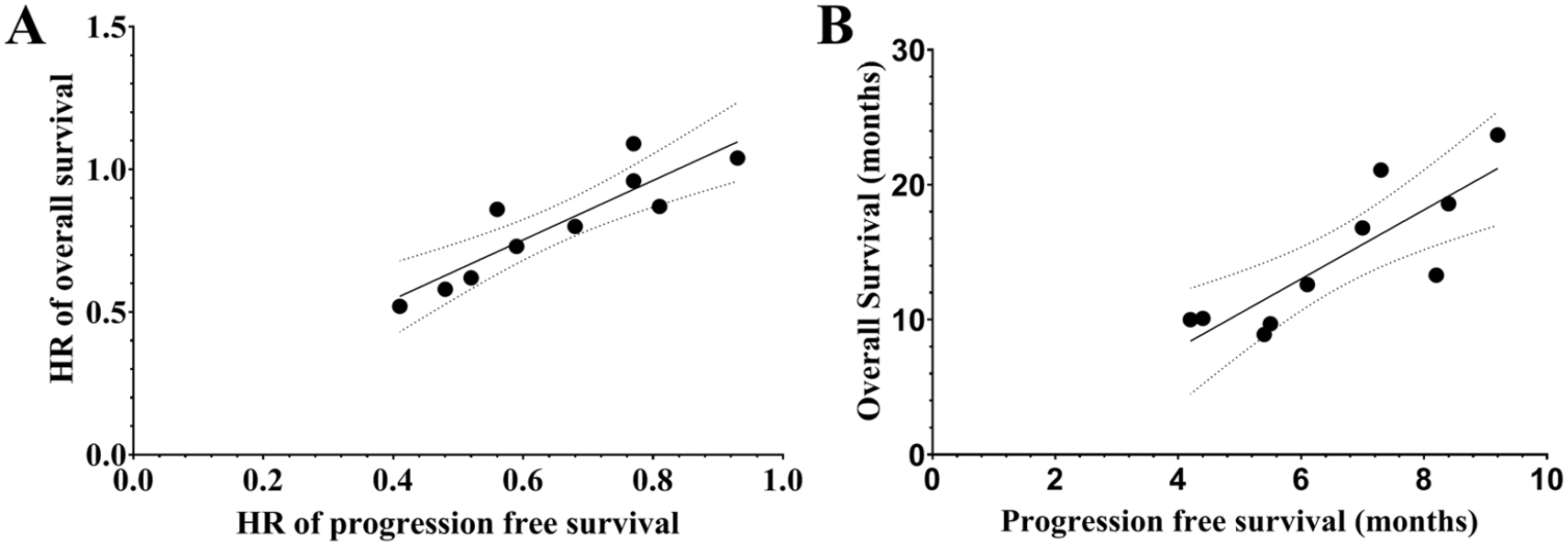
The correlation analysis of PFS and OS in NSCLC patients treated with immunotherapy based treatments. (A) The correlation analysis between HRs of OS and PFS in NSCLC patients treated with immunotherapy-based treatment versus chemotherapy-based treatments. (B) The correlation analysis between OS and PFS in NSCLC patients treated with immunotherapy-based treatment versus chemotherapy-based treatments.
Meta-Analysis Results
The relationship between liver metastasis and PFS
PFS in NSCLC patients with liver metastases and subgroup analysis based on pathological types and treatment strategy
Ten articles analyzed the relationship between liver metastasis and PFS. According to the heterogeneity analysis, there was no significant heterogeneity among the studies (P = 0. 30, I 2 = 14%), so a fixed effect model was used for analysis. The results of meta-analysis showed that the PFS of patients with liver metastases who received treatment in the experimental group was significantly longer than that in the control group (HR = 0.64, 95% CI: 0.55-0.75, P < .01).
The studies were classified according to the pathological features of patients, and then subgroup analysis was performed (Figure 4A). For lung squamous cell carcinoma, treatment with ICIs could reduce the risk of disease progression, and there was a statistically significant difference compared to the control group (I 2 = 0%; HR = 0.68; 95% CI: 0.51-0.91, P =.01). The PFS benefit of ICIs in non-squamous cell carcinomas was also significant (I 2 = 29 %; HR = 0.66, 95% CI: 0.52-0.84, P < .01). Regardless of pathological types, ICIs-based therapy significantly reduced the risk of disease progression (I 2 = 61%; under fixed effects model, HR=0.60, 95% CI: 0.46-0.78, P < .01; under random effects model, HR = 0.57, 95% CI: 0.37-0.89, P = .01).

Forest plots for the association between pathological types, treatment strategy, and PFS of NSCLC patients with or without liver metastasis. (A) Pathology-guided subgroup analysis of PFS in liver metastasis patients treated with immunotherapy-based treatment versus chemotherapy-based treatments. (B) Treatment combination-guided subgroup analysis of PFS in liver metastasis patients treated with immunotherapy-based treatment versus chemotherapy-based treatments. (C) Pathology-guided subgroup analysis of PFS in patients without liver metastasis treated with immunotherapy-based treatment versus chemotherapy-based treatments. (D) Treatment combination-guided subgroup analysis of PFS in patients without liver metastasis after immunotherapy-based treatment versus chemotherapy-based treatments.
The studies were grouped based on treatment strategy, such as PD-1 inhibitor plus chemotherapy, and PD-L1 inhibitor plus chemotherapy (Figure 4B). The results of the meta-analysis showed that compared with chemotherapy, PD-1 inhibitor combined with chemotherapy (I 2 = 0%; HR = 0.50, 95% CI: 0.37-0.68, P < .01), and PD-L1 inhibitor combined with chemotherapy (I 2 = 0%; HR = 0.77, 95% CI: 0.61-0.97, P = .02) significantly reduced the risk of disease progression. While PD-L1 inhibitor combined with chemotherapy versus bevacizumab combined with chemotherapy (BCT), PD-1 inhibitor combined with BCT versus BCT showed a trend to reduce the risk of disease progression, but there were no statistically significant differences (P > .05 for all).
PFS in patients with NSCLC without liver metastases and subgroup analysis based on pathological types and treatment strategy
Eight articles analyzed the relationship between absence of liver metastasis and PFS. According to the heterogeneity analysis, there was no significant heterogeneity among the studies (P = .19, I 2 = 29 %), so a fixed effect model was used for analysis. The results of meta-analysis showed that in patients without liver metastases, the PFS of the experimental group was significantly prolonged compared to the control group (HR = 0.56, 95% CI: 0.52-0.60, P < .01).
Subgroup analysis was performed according to the pathological types of patients (Figure 4C). The combined results showed that the benefits of ICIs treatment were statistically significant for lung squamous cell carcinoma (P = .17, I 2 = 41%; HR = 0.58, 95% CI: 0.51-0.67, P < .01) and non-squamous cell carcinoma (P = .22, I 2 = 32%; HR = 0.56, 95% CI: 0.50-0.62, P < .01). Likewise, in patients with NSCLC including the above pathological types, ICIs -based therapy also significantly reduced the risk of disease progression (HR = 0.49, 95% CI: 0.39-0.61, P < .01).
Relevant subgroup analyses were performed according to immunotherapy strategies (Figure 4D). The results of the meta-analysis showed that compared with chemotherapy, PD-1 inhibitor combined with chemotherapy (P = .37, I 2 = 6%; HR = 0.52, 95% CI: 0.46-0.59, P < .01), and PD-L1 inhibitors and chemotherapy (P = .17, I 2 = 40%; HR = 0.58, 95% CI: 0.53-0.64, P < .01) significantly reduced the risk of disease progression in patients without liver metastases.
The Relationship Between Liver Metastases and OS
OS in NSCLC patients with liver metastases and subgroup analysis based on pathological types and treatment strategy
Eight articles analyzed the relationship between liver metastasis and OS. According to the heterogeneity analysis, no significant heterogeneity was found among the studies (P = .12, I 2 = 37%), so a fixed effect model was used for analysis. The results of meta-analysis showed that the OS of patients without liver metastases was significantly longer than that of the control group (HR = 0.82, 95% CI: 0.72-0.94, P = .004).
Subgroup analysis by pathological type (Figure 5A) showed that the survival benefits did not show statistically significant differences among lung squamous cell carcinoma (HR = 1.09, 95% CI: 0.76-1.57, P = .65) and lung non-squamous cell carcinoma (P = .27, I 2 = 24%; HR = 0.84, 95% CI: 0.63-1.11, P = .21). In the subgroup analysis of treatment strategies, the results of the meta-analysis showed that compared with chemotherapy, PD-1 inhibitor monotherapy (HR = 0.67, 95% CI: 0.50-0.89, P = .007), and combination of PD-L1 inhibitor and chemotherapy (HR = 0.62, 95% CI: 0.39-0.98, P = .04) significantly reduced the risk of death. However, the combination of PD-L1 inhibitor and chemotherapy (I 2 = 0%; HR = 1.04, 95% CI: 0.81-1.34, P = .75), or PD-1 inhibitor combined with CTLA-4 inhibitor (HR = 0.94, 95% CI: 0.73-1.21, P = .64) failed to significantly reduced the risk of death (Figure 5B). PD-L1 inhibitor combined with chemotherapy was superior to bevacizumab combined with chemotherapy (BCT) in reducing the risk of death (HR = 0.87), while PD-L1 inhibitor combined with BCT versus BCT showed a significantly lower risk of death (HR = 0.52, P < .05). For liver metastasis-free patients who were treated with Durvalumab + Tremelimumab+CT versus Durvalumab + Tremelimumab, the OS were 7.9 versus 8.9 months (HR = 0.81, P > .05), respectively. 36
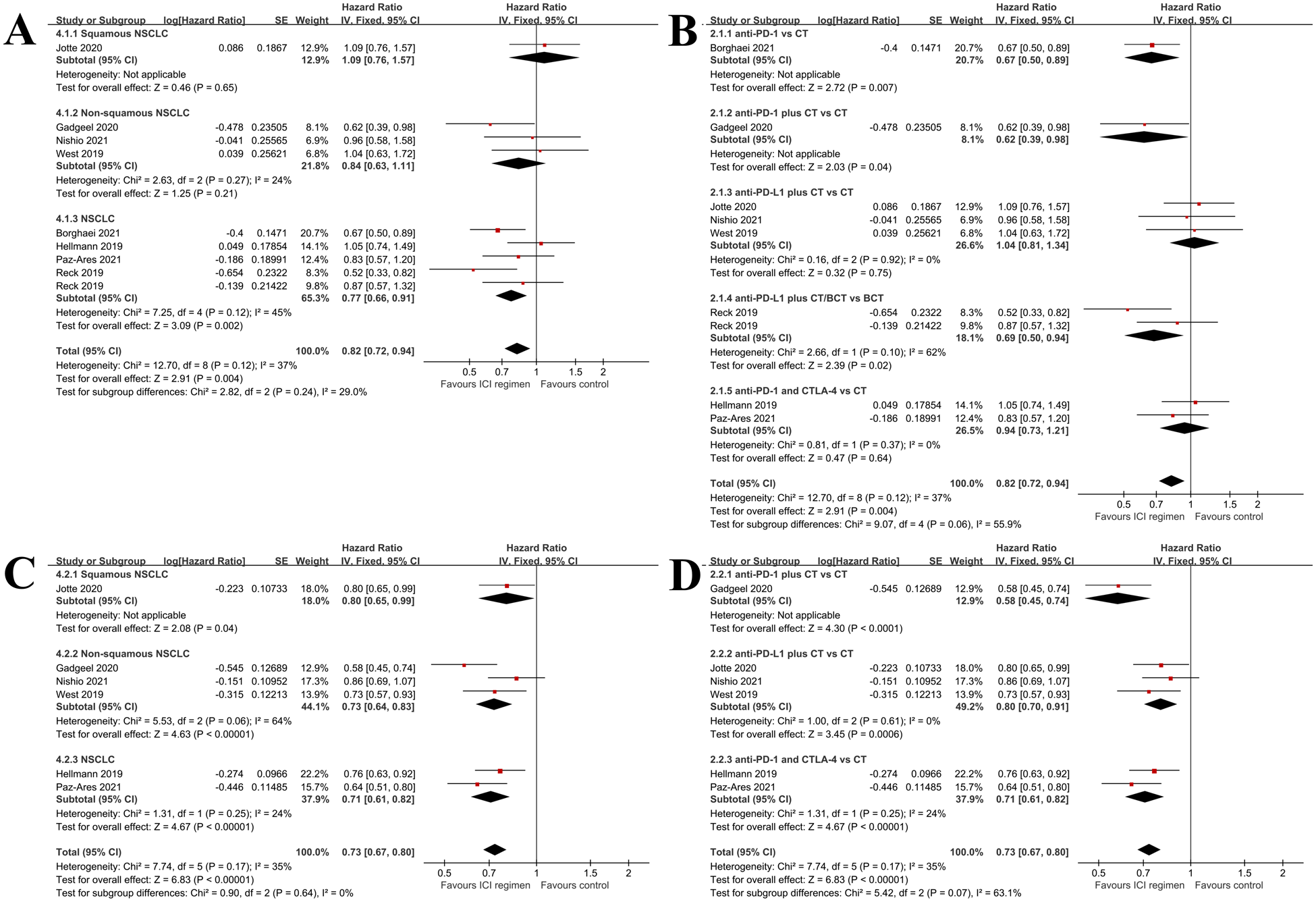
Forest plots for the association between pathological types, treatment strategy, and PFS of NSCLC patients with or without liver metastasis. (A) Pathology-guided subgroup analysis of OS in liver metastasis patients treated with immunotherapy-based treatment versus chemotherapy-based treatments. (B) Treatment combination guided-subgroup analysis of OS in liver metastasis patients treated with immunotherapy-based treatment versus chemotherapy-based treatments. (C) Pathology-guided subgroup analysis of OS in patients without liver metastasis treated with immunotherapy-based treatment versus chemotherapy-based treatments. (D) Treatment combination-guided subgroup analysis of OS in patients without liver metastasis after immunotherapy-based treatment versus chemotherapy-based treatments.
OS in NSCLC patients without liver metastases and subgroup analysis based on pathological types and treatment strategy
Six publications analyzed the relationship between absence of liver metastases and OS. The heterogeneity analysis did not find there was significant heterogeneity among the studies (P = .17, I 2 =35%), so the fixed-effects model was used. The results of meta-analysis showed that the OS of patients without liver metastases was significantly improved relative to the control group (HR = 0.73, 95% CI: 0.67-0.80, P < .01).
Subgroup analysis by pathological type showed that squamous cell carcinoma (HR = 0.80, 95% CI: 0.65-0.99, P = .04) and non-squamous cell carcinoma (I 2 = 64%; under fixed effects model, HR=0.73, 95% CI: 0.64-0.83, P < .01; under random effects model, HR= 0.72, 95% CI: 0.57-0.90, P = .004) patients had significantly better OS than those of the control group (Figure 5C). Subgroup analyses were performed according to the immunotherapy strategy that patients received. Compared with chemotherapy, PD-1 inhibitor combined with chemotherapy (HR = 0.58, 95% CI: 0.45-0.74, P < .01), PD − L1 inhibitor combined with chemotherapy (I 2 = 0% ; HR = 0.80, 95% CI: 0.70-0.91, P < .01), and PD-L1 inhibitor combined with CTLA-4 inhibitor (I 2 = 0%; HR = 0.71, 95% CI: 0.61-0.82, P < .01) were all associated with significantly reduced risk of death (Figure 5D). For liver metastasis patients who were treated with Durvalumab + Tremelimumab+CT versus Durvalumab + Tremelimumab, the OS were 18.7 versus 16.8 months (HR = 0.81, P > .05), respectively. 36
Sensitivity Analysis
For the analysis of PFS and OS, we assessed the effect of each study on the results using a sensitivity analysis that removed a single study. The results showed that the removal of each study did not have a significant influence on the pooled effect results (Supplemental Figure 1).
Publication Bias
Publication bias was assessed for meta-analyses with a number of included studies greater than or equal to 6. This study mainly assessed the publication bias of studies on the relationship between liver metastases and PFS and OS.
As shown in Figure 6, funnel plots of PFS and OS were visually symmetrical distributed, suggesting low risk of publication bias. Publication bias of PFS in liver metastasis-free patients was assessed using Begg’s test and Egger’s test. The results of Egger’s test showed no significant publication bias (P = .11), and the results of Begg’s test showed no significant publication bias (P = .19).

Funnel plots for PFS and OS in NSCLC patients with or without liver metastasis. (A) Funnel plot of PFS for NSCLC patients with liver metastasis. (B) Funnel plot of PFS for NSCLC patients without liver metastasis. (C) Funnel plot of OS for NSCLC patients with liver metastasis. (D) Funnel plot of OS for NSCLC patients without liver metastasis.
Discussion
Liver is one of the most common organs for cancer metastasis in lung cancer patients. Lung cancer patients with liver metastases usually have a poor prognosis. Although the rapid development of novel agents, such as immunotherapy, providing more options for treating cancers, the outcomes are still not satisfactory. Therefore, it is necessary to analyze the efficacy of immunotherapy and its influencing factors in NSCLC patients with liver metastases. This study found that NSCLC patients with or without liver metastases could benefit from immunotherapy in terms of PFS and OS. However, among NSCLC patients with liver metastases, subgroup analyses showed that pathological type and treatment regimen could be factors affecting the PFS and OS of these patients. For patients without liver metastases, significant survival benefits in terms of PFS and OS were independent of pathological types or treatment regimens.
Multiple clinical studies have shown that liver metastasis is a negative predictor of response to ICIs. In a clinical trial of the PD-L1 antibody durvalumab in metastatic NSCLC, patients with liver metastasis had shorter survival times compared with patients without liver metastasis. 36 In the treatment of patients with advanced NSCLC, Vokes et al observed a subgroup of patients with liver metastasis and reported 3-year follow-up results, suggesting that the prognosis of patients with liver metastasis was poor. 21 Similar results were also reported by Liu et al; they found that decreased immunotherapy response and shortened PFS were observed in NSCLC patients with liver metastasis. 38 Our findings are in accordance with the above results. For patients with liver metastasis, the risk of death is reduced by 18%, while it is 27% for those without liver metastasis. These suggest that liver metastasis is a clinical feature associated with poor survival outcomes.
In recent years, a few meta-analyses also evaluated the connection between liver metastasis and the efficacy of ICIs in cancer patients. A meta-analysis in 2020 explored this issue and found that the pooled HR of death in the liver metastasis subgroup was 0.82 (P < .05), while it was 0.72 (P < .05) for those without liver metastasis. 39 They concluded that in patients with advanced or metastatic cancers there was no statistically significant association between liver metastasis and the efficacy of ICIs. 39 These are consistent with our findings that patients could have a better survival outcome regardless of liver metastasis. However, it was performed based on various solid cancer types, and we only included RCTs of NSCLC patients. Furthermore, the impact of other clinical factors on the efficacy of immunotherapy in liver metastasis patients are not included in their analyses. In our analysis, the combination of different anti-tumor agents could be a factor that affecting the efficacy of ICIs. In another meta-analysis, the authors compared the efficacy of different immunotherapy agents in advanced NSCLC patients with liver metastasis, and found that the efficacy of ICIs depends on the combination strategy. 40 Pembrolizumab plus chemotherapy, and atezolizumab plus bevacizumab plus chemotherapy were superior to other regimens for liver metastasis patients. 40 These are consistent with our findings, again suggesting that treatment combination options are one of the factors affecting the efficacy of immunotherapy. Additional studies41,42 also assessed the relationship between liver metastasis and efficacy of immunotherapy. While these 2 studies were focusing on both NSCLC and small cell lung cancer, we only included and analyzed the data from RCTs conducted in NSCLC patients. Other differences are the statistical methods, the types of the included studies, and the introduced subgroup sets. Of note, our study updated the PFS and OS by incorporating recently published RCTs, and analyzed the factors that may influence the efficacy of immunotherapy in patients with liver metastases. Besides treatment strategy, the results of the subgroup analyses suggest that the pathological type received by the patients may be an important factor associated with the efficacy of immunotherapy.
The following reasons may be e potential mechanisms by which liver metastasis is a prognostic factor for immunotherapy. Liver metastases can deplete CD8+ T cells, leading to acquired immunotherapy tolerance. The combination of radiotherapy and immunotherapy targeting liver metastases can improve the body’s antitumor immunity. 43 Liver metastasis restrains immunotherapy efficacy via macrophage-mediated T cell elimination. The immunosuppressive microenvironment of the liver, may help tumor cells to evade anti-tumor immune surveillance caused by immunotherapy therapy, thereby reducing immunotherapy efficacy. 44 Therefore, the addition of angiogenesis inhibitors may improve the tumor microenvironment of liver metastases, thereby enhancing the efficacy of immunotherapy. 23 Combined with the above results, in the future immunotherapy for NSCLC liver metastases, combined therapy for liver metastasis may be an improvement option that we can choose. For example, in patients with metastasis, the use of radiotherapy for liver metastasis can reduce the risk of disease progression.45,46 Indeed, many studies are working on this issue by targeting liver metastasis and it has achieved considerable results in hepatocellular carcinoma.44,47
This study has several limitations. First, although the included studies are all RCTs, the patients and their baseline characteristics (such as gender, age, tumor burden, number of liver metastases, etc.) are inevitably different, which may cause selection bias. Secondly, the included studies have different treatment regimens, which may increase the risk of bias in outcome comparison and reduce the strength of the pooled effects. Thirdly, the included studies lack sufficient blinding, which may lead to selection bias and reduce the quality of the analysis. Another limitation is that a few subgroups of the meta-analysis only included one study due to limited number of eligible studies discovered by the search. These subgroups with limited studies are part of the entire meta-analyses and thus not deleted. We used a descriptive analysis for these subgroups. This may under- or over-estimate the power of the subgroup.
In conclusion, the presence of liver metastasis is a negative predictor of efficacy in NSCLC receiving immunotherapy. Future studies targeting liver metastasis will facilitate improving the prognosis of NSCLC patients with liver metastases.
Supplemental Material
sj-docx-2-ict-10.1177_15347354231164584 – Supplemental material for Assessing the Relationship Between Liver Metastases and the Survival of Patients With Non-Small Cell Lung Cancer After Immune Checkpoint Inhibitors Treatment: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-ict-10.1177_15347354231164584 for Assessing the Relationship Between Liver Metastases and the Survival of Patients With Non-Small Cell Lung Cancer After Immune Checkpoint Inhibitors Treatment: A Systematic Review and Meta-Analysis by Huilin Xu, Pingpo Ming, Zhenyu Zhao, Nan Zhao, Dingjie Zhou, Xixian Tang and Dedong Cao in Integrative Cancer Therapies
Supplemental Material
sj-tif-1-ict-10.1177_15347354231164584 – Supplemental material for Assessing the Relationship Between Liver Metastases and the Survival of Patients With Non-Small Cell Lung Cancer After Immune Checkpoint Inhibitors Treatment: A Systematic Review and Meta-Analysis
Supplemental material, sj-tif-1-ict-10.1177_15347354231164584 for Assessing the Relationship Between Liver Metastases and the Survival of Patients With Non-Small Cell Lung Cancer After Immune Checkpoint Inhibitors Treatment: A Systematic Review and Meta-Analysis by Huilin Xu, Pingpo Ming, Zhenyu Zhao, Nan Zhao, Dingjie Zhou, Xixian Tang and Dedong Cao in Integrative Cancer Therapies
Footnotes
Acknowledgements
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Medical Scientific Research Project of Wuhan Municipal Health Commission Project (WX20D50).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
