Abstract
Objective:
To report the feasibility and effectiveness of a newly developed clinical exercise program for improving maximal cardiorespiratory fitness in Spanish cancer patients receiving adjuvant chemotherapy or radiation. We also examined the effectiveness of the exercise program for improving maximal muscular strength, body composition, fatigue, and quality of life, and explored if the effectiveness varied based on selected patient characteristics.
Design:
The study was a single group implementation feasibility study using a pre-posttest design.
Methods:
Participants performed a 12-week, twice-weekly, supervised, multi-component exercise program during adjuvant therapy. Paired t-tests were used to assess pre-post changes, and analyses of covariance were used to compare effectiveness based on selected patient characteristics.
Results:
We had 100 cancer patients referred to the clinical exercise program of which 85 (85%) initiated the exercise program and 76 (89%) completed the post-intervention fitness assessment. Exercise significantly improved VO2max by 4.8 mL/kg/minutes (P < .001, d = 0.74). Exercise also significantly improved chest strength (P < .001, d = 0.82), leg strength (P < .001, d = 1.27), lean body mass (P < .001, d = 0.11), skeletal muscle mass (P < .001; d = 0.09), fat mass (P < .001; d = 0.10), % body fat (P < .001; d = 0.17), quality of life (P = .0017; d = 0.41), and fatigue (P = .007; d = 0.46). Treatment modality, cancer type, and age affected some exercise responses, especially related to body composition changes.
Conclusions:
A 12-week, supervised, multi-component exercise program was effective for improving health-related fitness and quality of life in Spanish cancer patients receiving adjuvant therapy. Our results show the benefits of incorporating clinical exercise programming into the supportive care of cancer patients receiving treatments.
Registration:
The study protocol is registered at ClinicalTrials.gov (NCT05078216).
Introduction
Chemotherapy and radiotherapy are 2 widely used therapeutic approaches for the treatment of cancer. Despite their survival benefits, chemotherapy and radiotherapy cause several side effects which interfere with the patients’ functional capacity and quality of life.1,2 Among the most common side effects of these treatments are nausea, fatigue, diarrhea, neutropenia, muscle weakness, cardiotoxicity, and pain.3,4 Moreover, there are 2 main side effects that are directly related to a worse prognosis of the disease and an increase in mortality from a variety of causes: reduced cardiovascular capacity and changes in body composition 5 including loss of muscle mass and gain of fat mass. 6 Related to cardiorespiratory fitness (CRF), it has been observed that cancer treatments reduce it up to 30%7,8 which is associated with a worse prognosis of the disease and an increase of mortality. 9 Moreover, changes in CRF are associated with changes in quality of life (QOL) and fatigue. 10
Since there are no established pharmacological treatments for these side effects, much attention has been paid to other strategies and therapies to reduce these comorbidities. More specifically, exercise has been shown to improve these outcomes in cancer patients receiving adjuvant treatments such as chemotherapy and radiation therapy11-14 thereby improving fatigue and QOL.15-17 Exercise improves CRF through a variety of mechanisms including the capacity to transport oxygen (eg, cardiac output), oxygen diffusion to working muscles (eg, capillary density, membrane permeability, and muscle myoglobin content), and adenosine triphosphate (ATP) generation (eg, mitochondrial density and protein concentrations). 18 Moreover, improvements in CRF may lead to improved physical functioning and a greater capacity to perform activities of daily living which, in turn, may reduce perceptions of fatigue and improve QOL. 19
Based on this evidence, the Exercise Oncology Unit of the Spanish Cancer Association in Madrid was created to support integration of exercise programming into the usual care of patients with cancer. Our program targets patients with cancer receiving treatment who are experiencing at least 1 physical side effect (eg, weakness, fatigue, pain, muscle loss, and fat gain). To ensure patient safety, a clinical nurse evaluates and triages patients to appropriate services, including physical exercise. Research activities are integrated into the Unit’s programming to formalize data collection and facilitate timely evaluation of program effectiveness.
The primary purpose of this study was to report the feasibility and effectiveness of this newly developed clinical exercise program for improving maximal CRF in Spanish cancer patients receiving adjuvant chemotherapy or radiation therapy. Considering that multiple randomized trials have verified the importance of physical exercise during cancer treatment, we believe an important next step is demonstrating the implementation of such an exercise program in clinical practice in a community setting.
A secondary purpose was to examine the effectiveness of the exercise program for improving maximal muscular strength, body composition, fatigue, and QOL. We also explored if the effectiveness of the exercise program varied based on selected patient characteristics. We hypothesized that a 12-week, supervised, combined aerobic and strength exercise program would improve CRF, muscular strength, body composition, fatigue, and QOL in Spanish cancer patients receiving adjuvant cancer therapy regardless of patient characteristics.
Methods
All eligible participants provided written informed consent before their inclusion in the exercise program. After collecting the program data, the study was submitted to the Instituto de Salud Carlos III Committee for Ethical Research for approval to analyze and publish the data (date of approval June 17, 2021, Expte: PI23_2021). The study was conducted following the principles of the Declaration of Helsinki. The study protocol is registered at ClinicalTrials.gov (NCT05078216).
Study Design, Participants, and Procedures
The study was an uncontrolled trial using a pre-post design with assessments at baseline and post-intervention (12 weeks). The study was conducted between September 2018 and March 2020 at the Exercise Oncology Unit of the Spanish Cancer Association in Madrid, Spain. The Exercise Oncology Unit is a cancer-specific community-based facility funded by the Spanish Cancer Association that provides free clinical exercise services to approximately 350 individuals with cancer each year.
Eligible patients with cancer from any hospital in Madrid were informed about the study by their oncologist or by other healthcare providers during checkup visits. Potentially eligible participants were referred to the Spanish Cancer Association where a clinical nurse was responsible for triaging the patients to the different supportive care services, including physical exercise. Patients were eligible for the present study if they were: (1) 18 years or older, (2) diagnosed with any local or regionally advanced primary cancer (stage I-IIIa), (3) receiving adjuvant chemotherapy or adjuvant radiotherapy after chemotherapy, (4) had an Eastern Cooperative Oncology Group (ECOG) ≤1, (5) able to walk 500 m without resting (6) presented with any physical side effect related to cancer treatments such as weakness, fatigue, changes in body composition, and/or pain, (7) did not present with any physical or psychological disability, (8) had an ejection fraction above 50%, and (9) fulfilled the American Thoracic Society criteria for performing a cardiopulmonary exercise test (CPET). Final determination of eligibility occurred at the time of the baseline fitness test.
Exercise Program
The exercise program was designed and conducted by a qualified exercise physiologist with oncologic training. The exercise program was individualized based on baseline fitness testing including cardiopulmonary fitness, muscular strength, and body composition. Participants were asked to complete a 12-week, twice-weekly, progressive, supervised multi-component exercise program. Each exercise session consisted of a warm-up at 50% of peak oxygen consumption (VO2peak) for 5 minutes, followed by 40 to 50 minutes of the main exercise phase, and then a cool-down for 5 minutes and stretching for 5 minutes. Cardiovascular exercise could be performed on an elliptical, stationary bicycle, treadmill, or rowing machine. Intensity was monitored by a PolarH10 heart rate control device (Polar Electro, 2021) and gradually increased over the 12-week program based on stablished exercise protocols. 20 Strength exercise was performed on Technogym machines including chest press, leg press, and a multifunctional crossover machine (Via Calcinaro, 2861, 47521 CESENA (FC)). Resistance intensity was gradually increased over the 12-week program and included a re-test of maximal strength in the sixth week of the program. Apart from the twice-weekly supervised sessions, as early as the fifth week, 1 individualized home-based session was encouraged for each patient based on a mid-program assessment of body composition.
Although the exercise program was individualized, 2 main goal-based exercise programs were developed based on the results of the body composition test. For patients with low or normal muscle mass and normal fat mass, the program focused on building muscle mass. For patients with normal muscle mass and high fat mass, the program focused on fat loss. The program design was the same for both groups in terms of structure; however, subtle differences in the starting points of exercise intensity for resistance and aerobic components were prescribed to align with the patient’s goal.
For those patients focused on gaining muscle mass, the progression of the resistance intensity was every 3 weeks beginning with 2 sets of 12 repetitions at 70% RM and progressing to 3 sets of 10 repetitions at 75% RM (week 3), 4 sets of 8 repetitions at 80% RM (week 6), and 4 sets of 8 repetitions at 85% RM (week 9). Regarding the cardiovascular exercise program, the intensity progressed every 4 weeks from 45% to 65% MHR (weeks 1-4), from 65% to 85% MHR (weeks 5-8), and from 85 to 100% MHR (weeks 9-12).
For those patients focused on fat loss, the progression of the resistance intensity was every 3 weeks beginning with 2 sets of 12 repetitions at 65% RM and progressing to 3 sets of 10 repetitions at 70% RM (week 3), 4 sets of 8 repetitions at 75% RM (week 6), and 4 sets of 8 repetitions at 80% RM (week 9). Regarding the cardiovascular exercise program, the intensity progressed every 4 weeks from 60% to 70% MHR (weeks 1-4), from 70% to 85% MHR (weeks 5-8), and from 85% to 100% MHR (weeks 9-12).
Outcome Measures
Health-related fitness assessments and patients reported outcomes were completed at baseline and post-intervention. Baseline testing was completed during adjuvant therapy whereas post-intervention testing was completed either during or after adjuvant therapy.
Primary outcome
The primary outcome was CRF. CRF refers to the capacity of the circulatory and respiratory systems to supply oxygen to skeletal muscle mitochondria for energy production needed during physical activity. 21 A single-stage submaximal treadmill test was used to estimate maximal oxygen uptake (VO2max) and a difference of 3.39 ml/kg/minutes was used to indicate a clinically important difference. 22 Oxygen volume was recorded by a gas analyzer (FitMate MED; Tecnomed 2000, S.L) and used to predict maximal oxygen consumption in milliliters per kilogram per minute. VO2 ventilation, heart rate, and related parameters had a 15 second sampling rate. The protocol was carried out in accordance with the CPET criteria of ATS. Patients started with a 2-minute warm-up (3.5 km/hours) and there was a gradual increase in the speed of 0.1 km/hours every 15 seconds and an increase in the incline of 0.5% every 30 seconds. The Bruce protocol was used for patients that were not able to walk at 4 km/hours. Perceived effort was measured by the Borg scale every minute in both protocols.
Secondary outcomes
Maximal strength, which is the ability to exert maximal force against an external resistance and requires a maximal voluntary contraction, was evaluated by chest and leg press using a 3 to 5 repetition maximum (RM) protocol 23 following the National Strength and Conditioning Association guidelines. We applied the Mayhew formula to predict 1 RM. 24 The tests were performed on Technogym machines (Via Calcinaro, 2861, 47521 CESENA (FC)). Body composition, a measure of energy-nutrient balance, functionality, and health which is widely used in clinical research and field studies, 25 was assessed by the Inbody 770 (Microcaya, 2016 S.L), a multifrequency and direct segmental bioelectrical impedance device (6 frequencies that generate a total of 30 impedance values) which takes 60 seconds to obtain measures of weight, body mass index, lean body mass, skeletal muscle mass, fat mass, body fat, skeletal muscle index, waist-hip ratio, visceral fat area, and phase angle. In order to standardize body composition measurements, patients were asked to follow some nutritional recommendations.
In terms of patient-reported outcomes, QOL was measured by the Functional Assessment of Cancer Therapy-Fatigue (FACT-F) scale 26 which includes the FACT-General (FACT-G) plus the Fatigue subscale. FACT-F consists of 40 items including 27 items for the FACT-G scale and 13 items for Fatigue subscale. The FACT-G includes the 4 subscales of physical well-being, functional well-being, emotional well-being, and social well-being. Higher scores on the FACT-F, FACT-G, and fatigue subscale indicate better QOL/lower fatigue. The minimal important difference for the FACT-F, FACT-G, and fatigue subscale are 7, 4, and 3 points, respectively. 27
Health status was measured by the Euro-Quality of Life-5 (EQ-5) questionnaire. EQ-5 is an instrument which evaluates the generic QOL developed in Europe and widely used in cancer patients. The EQ-5 consists of 1 question for each of the 5 dimensions that include mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. Higher scores on the EQ-5 indicate better health-related quality of life. The minimal important difference in EQ-5 is 4 points. 28
Patient Characteristics
Demographic and behavioral variables were assessed by self-report. Medical variables were abstracted from medical reports. Patient characteristics evaluated to predict exercise response were treatment modality (chemotherapy vs radiation therapy), cancer type (breast vs other), comorbidities (≤1 vs 2+), BMI (≤25 vs 25+), age (<50 vs 50+), and exercise program goal (fat loss vs. muscle gain).
Sample Size
We needed to enroll 36 patients to detect a clinically important change in VO2max from baseline to post-intervention of 3.39 (±6.7) mL/kg/minutes with 80% power, assuming a 15% drop out and a 2-tailed alpha of .05. The estimated change of 3.39 (±6.7) was based on a meta-analysis of the effects of exercise on CRF in breast cancer patients and survivors. 22
Statistical Analyses
Continuous data are reported as mean and standard deviation (mean ± SD). Categorical data are reported as frequency and percentage. Changes from baseline to post-intervention were analyzed using a paired t-test and Cohens’ d was used to describe the amount of change from pre-to post by dividing the mean change by the baseline standard deviation. Analyses of covariance were used to compare between group differences based on the selected patient characteristics adjusting for baseline value of the outcome. All analyses were performed using STATA (15.0). Statistical significance was set at P < .05.
Results
Flow of participants through the study is reported in Figure 1. Briefly, 100 cancer patients receiving either chemotherapy or radiation therapy after chemotherapy were referred to the clinical exercise program between September 2018 and December 2019. At that time, recruitment was halted because of the COVID-19 pandemic. Of the 100 referrals, 11 (11%) patients were deemed ineligible for the present study because they were receiving neoadjuvant chemotherapy (n = 7) or did not receive adjuvant chemotherapy before adjuvant radiation therapy (n = 4). Of the 89 patients who were eligible and completed the baseline assessment, 85 (96%) patients initiated the clinical exercise program. After the 12-week exercise program, 9 (11%) patients did not return for the post-intervention assessment, leaving 76 (89%) patients with complete data for our primary outcome of CRF. Body composition data were available for 71 patients because of a device problem for 1 week in June 2019. Patient-reported outcomes were available for 43 patients because until January 2019 the virtual platform was not ready for patients to complete the questionnaires confidentially and securely. Median adherence to the supervised exercise program was 19.3/24 (80.3%) with a range of 14 (58.3%) to 24 (100%).

CONSORT diagram showing flow of participants through the study.
Descriptive statistics for the 85 cancer patients who initiated the clinical exercise program are reported in Table 1. Briefly, participants had a mean age of 49.3 ± 10.9 years, 91.8% were women, and 87.1% were Spanish. In terms of medical data, 63.5% had breast cancer, 68.2% had surgery prior to chemotherapy, 81.2% were receiving chemotherapy, and 18.8% were receiving radiation therapy after chemotherapy. The mean time since diagnosis was 10.2 ± 22.1 months. Apart from cancer, 32.9% of patients had 1 comorbidity and 40.9% had 2 or more comorbidities.
Baseline Characteristics of Cancer Patients Participating in a Clinical Exercise Program During Treatment, Overall, and by Treatment Modality.

Abbreviations: TSD, time since diagnosis; AC, adriamycin and cyclophosphamide; ABVD, doxorubicin hydrochloride (Adriamycin), bleomycin sulfate, vinblastine sulfate, and dacarbazine; FOLFOX, folinic acid, fluorouracil, and oxaliplatin.
Effectiveness of Exercise for Cardiorespiratory Fitness
The effectiveness of exercise for maximal CRF, overall, and by selected patient characteristics, is presented in Table 2. From baseline to post-intervention, VO2max increased by 4.8 mL/kg/minutes overall (P < .001, d = 0.74). Age had a significant effect on the VO2max response with younger patients experiencing a larger improvement in VO2max compared to older patients (adjusted between group difference = 3.6 mL/kg/minutes; P = .003; d = 1.03). There were no differences in VO2max response based on treatment modality, cancer type, comorbidities, body mass index, or exercise program goal.
Effects of Exercise During Cancer Treatment on Maximal Cardiorespiratory Fitness (VO2max), Overall, and by Selected Patient Characteristics.

Adjusted for baseline value of the outcome.
Effectiveness of Exercise for Secondary Outcomes
The effectiveness of exercise for secondary outcomes overall are presented in Table 3. Related to muscular strength, exercise had significant positive effects on chest strength (P < .001; d = 0.82) and leg strength (P < .001; d = 1.27). In terms of body composition, exercise significantly improved lean body mass (P < .001; d = 0.11), skeletal muscle mass (P < .001; d = 0.09), fat mass (P < .001; d = 0.10), % body fat (P < .001; d = 0.17), waist-hip ratio (P = .009; d = 0.14), and visceral fat area (P < .001; d = 0.14). In terms of patient-reported outcomes, exercise significantly improved the FACT-F (P = .017; d = 0.41) and fatigue subscale (P = .007; d = 0.46), however, no significant changes were found in the FACT-G and EQ-5 scales.
Effects of Exercise During Cancer Treatment on Secondary Health-Related Fitness and Patient-Reported Outcomes.
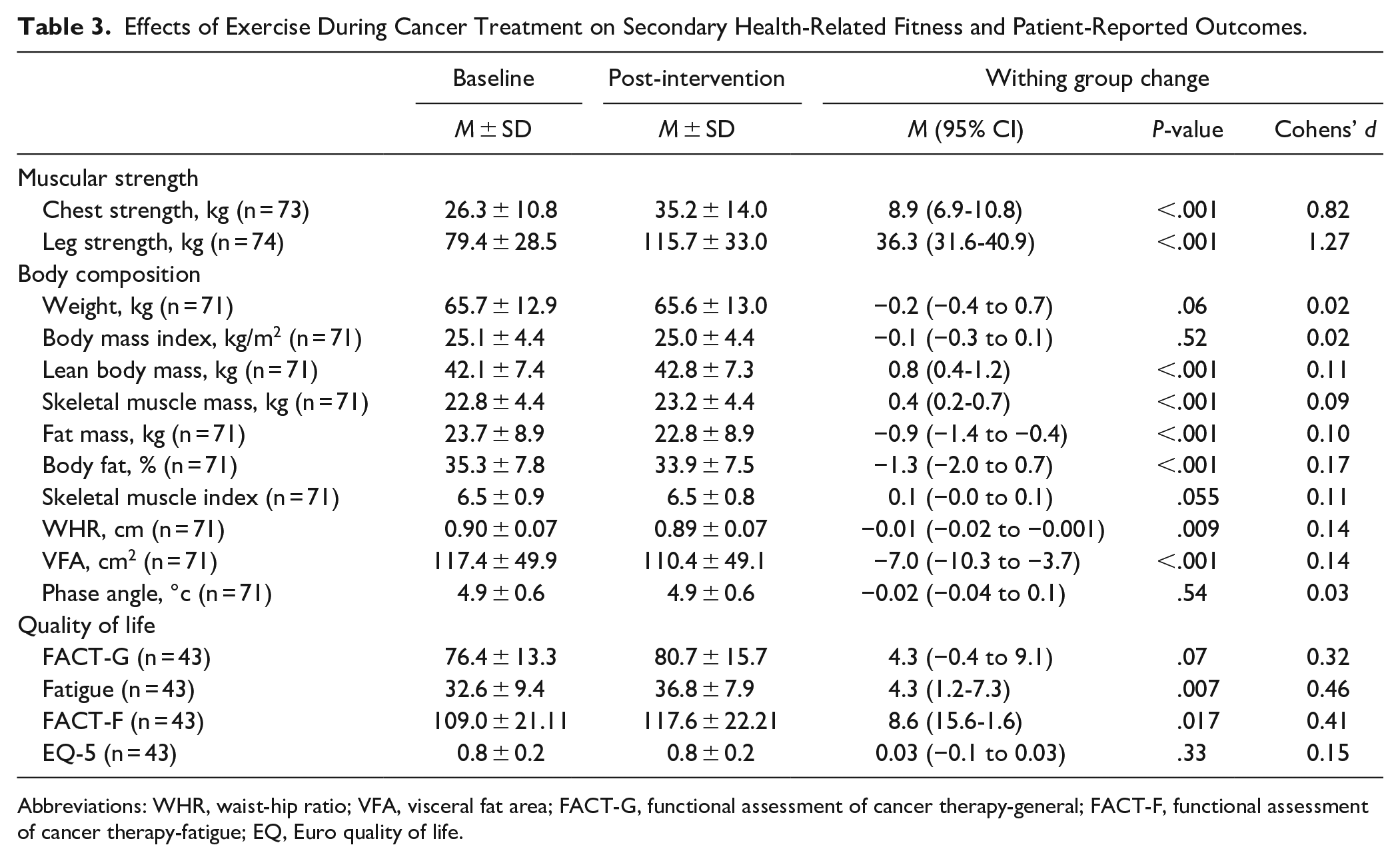
Abbreviations: WHR, waist-hip ratio; VFA, visceral fat area; FACT-G, functional assessment of cancer therapy-general; FACT-F, functional assessment of cancer therapy-fatigue; EQ, Euro quality of life.
Associations of Patient Characteristics With Secondary Exercise Responses
Considering patient characteristics, there were several significant associations with exercise responses for treatment modality (Table 4), cancer type (Table 5), and age (Table 6). For treatment modality, patients receiving radiation therapy compared to chemotherapy lost more weight (P = .023; d = .43), reduced body mass index more (P = .018; d = 0.01), and improved fatigue more (P = .028; d = 0.99). Related to cancer type, breast cancer patients lost more weight (P = .004; d = 0.53) and reduced body mass index more (P = .005; d = .47) than other cancer types; however, other cancer patients added more lean body mass (P = .004; d = 0.60) and skeletal muscle mass (P = .003; d = 0.63) than breast cancer patients. In terms of age, in addition to VO2max, younger patients improved their waist-hip ratio (P = .010; d = 0.58) more than older patients; however, older patients lost more weight (P = .024; d = 0.26) and reduced their body mass index more (P = .047; d = 0.53) than younger patients. The only other significant association involved exercise program goal, where patients whose objective was to gain muscle mass lost more visceral fat (P = .032; d = 1.23) than patients whose objective was fat loss.
Significant Effects of Cancer Treatment Modality on the Exercise Training Responses of Spanish Cancer Patients Participating in a Clinical Exercise Program.

Adjusted for baseline value of the outcome.
Significant Effects of Cancer Type on the Exercise Training Responses of Spanish Cancer Patients Participating in a Clinical Exercise Program.
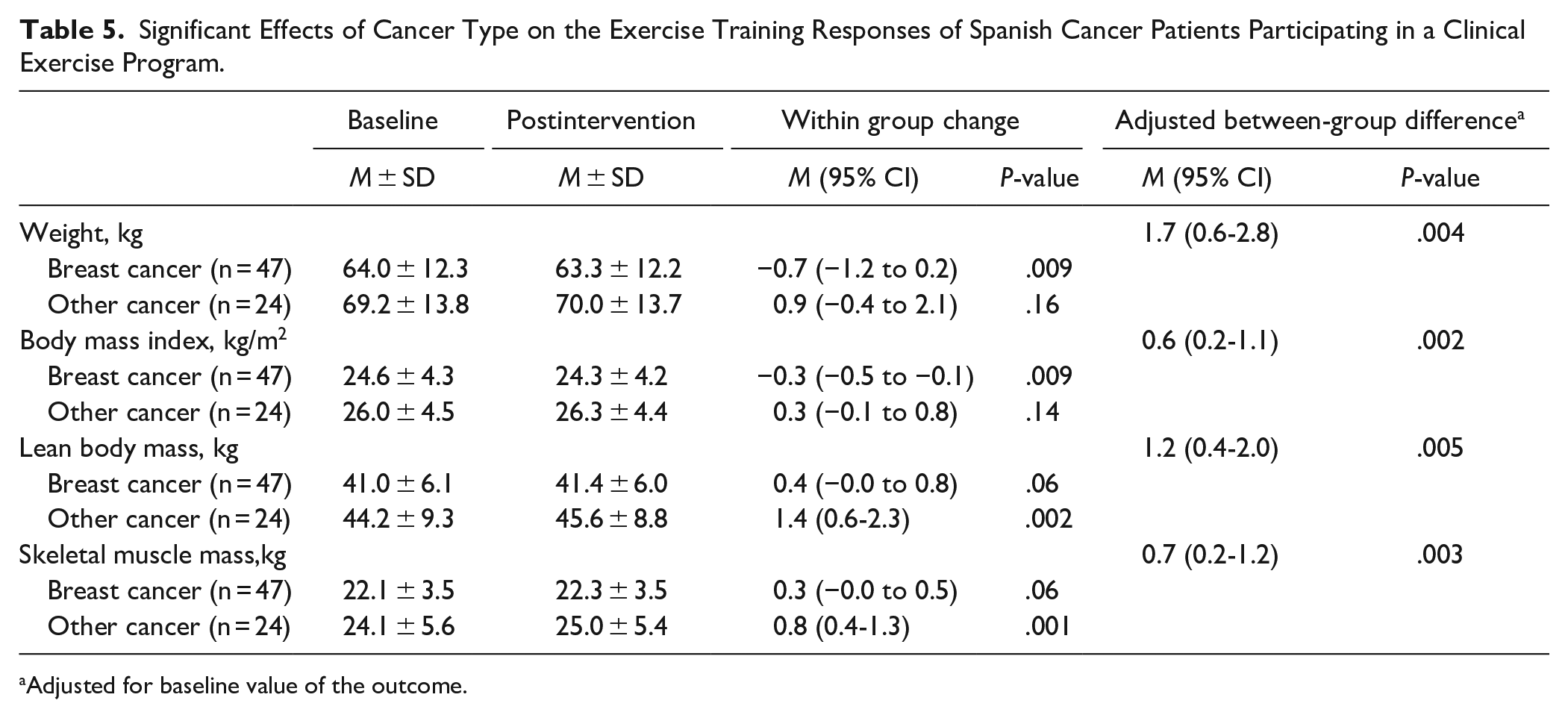
Adjusted for baseline value of the outcome.
Significant Effects of Age on the Exercise Training Responses of Spanish Cancer Patients Participating in a Clinical Exercise Program.
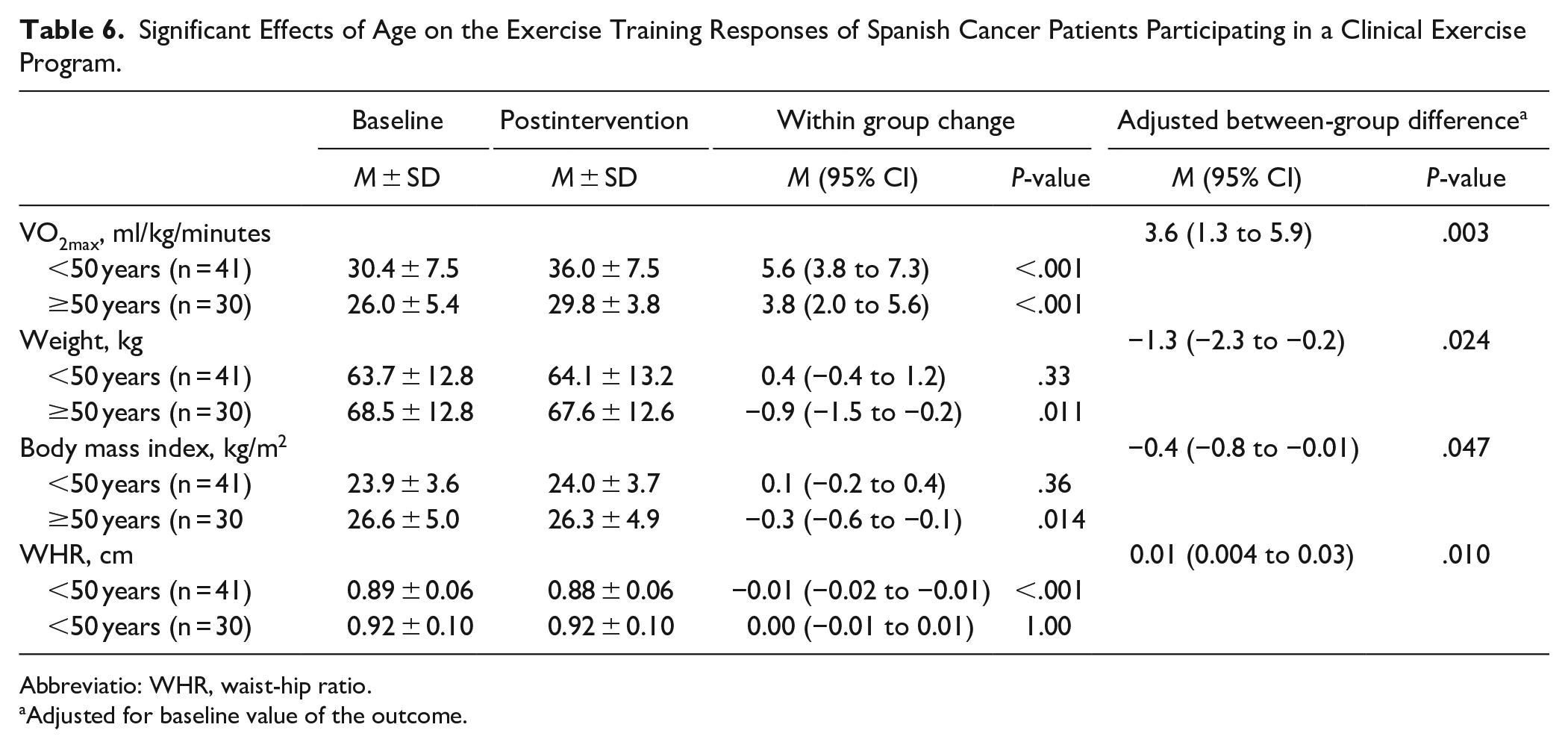
Abbreviatio: WHR, waist-hip ratio.
Adjusted for baseline value of the outcome.
Discussion
As hypothesized, a 12-week, supervised, combined aerobic and strength exercise program significantly improved aspects of health-related fitness, QOL, and fatigue across most patient subgroups, with only minor variations in response based on cancer treatment modality, cancer type, and age. Moreover, this intervention was facilitated by a clinical nurse and delivered in a friendly and supportive environment which promoted a social dimension where the patients felt safe and valued.
The primary finding of the study was that our clinical exercise program resulted in a significant improvement in CRF of 4.8 mL/kg/minutes or 17% from baseline. This magnitude of change is higher than the 2.80 mL/kg/minutes reported for exercise during cancer treatment in a meta-analysis of 48 studies. 29 The magnitude of improvement in our study may be larger than in previous research because of our higher intensity exercise program and the high adherence. The increase of 4.8 mL/kg/minutes in our study may be meaningful because every 3.39 mL/kg/minutes of cardiorespiratory fitness has been associated with a decreased risk of all-cause mortality by 13%. 30 Moreover, this magnitude of improvement in CRF in our study was achieved regardless of cancer treatment modality, cancer type, comorbidities, and BMI. We did note that younger patients (<50 years) experienced a substantially larger improvement in CRF of 5.6 mL/kg/minutes compared to 3.8 mL/kg/minutes in older patients (50+ years) consistent with previous research.31,32
Related to secondary outcomes, our clinical exercise program also improved chest strength by 8.9 kg (34%) and leg strength by 36.3 kg (46%). Moreover, these gains in strength were achieved regardless of cancer treatment modality, cancer type, comorbidities, age, and BMI. In a systematic review of 14 trials, Padilha et al, 33 observed that strength training during adjuvant therapy increased leg muscular strength by an average of 26.2 kg. Similarly, another review 34 observed an average increase of 14.6 kg for leg strength (9 trials) and 6.9 kg for chest strength (9 trials). The strength improvements in our study may be important as higher levels of muscular strength (10.7 kg for leg press and 7.7 kg for bench press) have been associated with a 35% reduced cancer mortality in men previously diagnosed with cancer. 35 Moreover, benefits in muscle mass and strength promote significant improvements in clinical and functional outcomes, which are important domains of QOL regardless of the type of cancer. 36
In terms of body composition, our clinical exercise program significantly improved lean body mass, skeletal muscle mass, fat mass, % body fat, waist-hip ratio, and visceral fat area. These improvements may be important considering that a loss of muscle mass in cancer patients predicts chemotherapy toxicity, worse outcomes, and poor QOL37-39 whereas a gain in fat mass in cancer patients may increase risk of secondary cardiovascular and metabolic diseases. 40
Interestingly, body composition responses to our clinical exercise program were the outcomes most influenced by patient characteristics. Specifically, weight loss (and BMI loss) was greater in patients who were receiving radiation therapy, had breast cancer, and were older. These subgroup effects may be important considering that women who gain weight after a breast cancer diagnosis are at a greater risk for breast cancer recurrence and death compared with lean women and women who do not gain weight after diagnosis. 41
Conversely, gains in lean body mass and skeletal muscle index were greater in patients with other types of cancer. It is possible that some of the other cancer types we included in our study (eg, ovarian, colorectal, and lymphoma) may have experienced more muscle loss during treatment and, therefore, benefit more from the clinical exercise program in terms of muscle gain. Moreover, 7 of 26 (26.9%) patients in the “other cancer” group were men, which may have influenced muscle mass gains. Integrating body composition measures into clinical exercise programs offers tremendous promise to help patients with cancer live longer and healthier lives. 42
In terms of QOL and fatigue, our clinical exercise program significantly improved both outcomes by 8.6 and 4.3 points, respectively, exceeding the minimal important differences on these scales.43,44 However, it is important to consider that in our study, only 43% of the patients filled out the QOL questionnaires because until January 2019 the virtual platform was not ready for patients to fill out the questionnaires confidentially and securely. Despite the smaller sample size, exercise appeared to have an even more beneficial effect on fatigue for patients who were receiving radiation therapy after chemotherapy. Previous systematic reviews and meta-analyses have confirmed small, but significant, effects of exercise on QoL and fatigue in cancer patients during and after treatments. 44 This improvement is important considering that fatigue is one of the most common and devastating symptoms in cancer patients during and after treatments that reduces QOL. 45 Moreover, cancer patients continue to experience fatigue symptoms for months or years after successful treatment and high levels of fatigue may even predict shorter survival. 46 For these reasons, it is crucial to implement exercise programs during treatments as soon as possible to reduce or delay the onset of fatigue.
Our study has important strengths and limitations. Strengths of our study include the novel clinical exercise intervention that is integrated into the care of patients undergoing adjuvant cancer therapy, the selection of patients presenting with at least 1 physical side effect, the targeted exercise program based on body composition needs, the comparison of exercise responses based on clinically relevant patient characteristics, the adequate sample size, and the excellent adherence and completion rates considering the challenges experienced by patients on cancer treatments.
The primary limitation of our study is the absence of a control group. Given the focus on implementation into clinical care, however, a control group was not considered appropriate. Other limitations of our study include the lack of information on eligibility and recruitment rates, limited demographic information, few male participants, unequal comparison groups (eg, chemotherapy and radiation therapy), a submaximal cardiorespiratory fitness test, failure to assess home-based exercise adherence, the lack of mid-program assessments, and lack of a longer-term follow-up. Finally, as a future research direction, it could be interesting to do a complete intermediate assessment of outcomes to determine how quickly improvements are experienced in the program.
Conclusion
Our study tested a novel multimodal exercise intervention designed for cancer patients receiving adjuvant treatments and experiencing side effects. Our results showed that such a high-intensity exercise program, facilitated by a clinical nurse and supervised by a qualified exercise physiologist, was acceptable and substantially improved CRF, muscular strength, body composition, fatigue, and QOL. Therefore, we believe that with proper screening and supervision cancer patients on treatment can benefit from tailored exercise oncology support as part of the overall therapeutic care plan.
Footnotes
Acknowledgements
The study was carried out thanks to the Spanish Cancer Association, so it is essential to thank the work of all the people involved in this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was carried out thanks to the Spanish Cancer Association, but no financial assistance was needed to carry it out.
Ethical Approval and Clinical Trial Registration
All eligible participants provided written informed consent before their inclusion in the program. After collecting the program data, the study was submitted to the Instituto de Salud Carlos III Committee for Ethical Research for approval to analyze and publish the data (date of approval June 17, 2021, Expte: PI23_2021). The study was conducted following the principles of the Declaration of Helsinki. The study protocol is registered at ClinicalTrials.gov (NCT05078216).
Confirmation of Ethical Compliance
The study received ethical approval from the Carlos III Health Institute and was conducted following the principles of the Declaration of Helsinki. All eligible patients provided written informed consent for study participation. The study protocol is registered at ClinicalTrials.gov (NCT05078216).
